- Submissions

Full Text
Research in Medical & Engineering Sciences
Heavy Metal Removal: Phytoremediation and Physicochemical Methods
Jayeeta Banerjee1 and Sudip Kumar Das2*
1Medical Laboratory Sciences, School of Allied Medical Sciences, Lovely Professional University Jalandhar- Delhi G. T. Road, Phagwara, India
2Department of Chemical Engineering, University of Calcutta, Kolkata, India
*Corresponding author:Sudip Kumar Das, Department of Chemical Engineering, University of Calcutta, 92 A. P. C. Road, Kolkata-700009, India
Submission: July 24, 2024;Published: February 09, 2026

ISSN: 2576-8816Volume12 Issue 2
Abstract
Industrial discharge with inappropriate disposal of heavy metal raises many ecological as well as health issues. Techniques such as physico-chemical methods and phytoremediation are actively used in heavy metal removal. This review is intended to highlight the mentioned methods’ positive impact and fundamental knowledge. Future scope regarding heavy metal removal also anticipated (Graphical Abstarct).
Keywords:Heavy metal; Physicochemical technology; Phytoremediation; Future scope
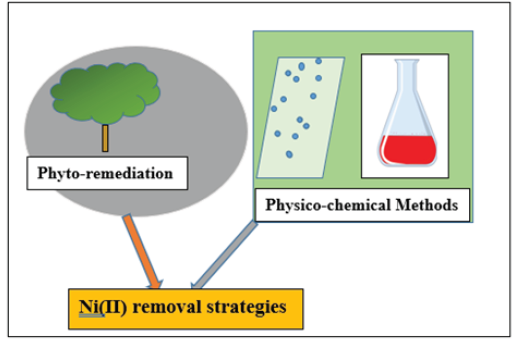
Figure 1:Graphical Abstract
Introduction
Heavy metals occur from natural and anthropogenic sources, leading to considerable apprehension for the entire ecosystem due to their non-biodegradable nature (Figure 1) [1].
Heavy metal toxicity consequences impaired physiological balance in humans, followed by the occurrence of acute and chronic health conditions (Figure 2) [2].
Figure 2:Different sources of heavy metal.
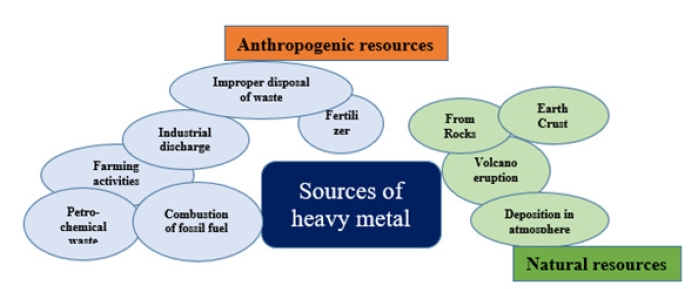
Figure 3:Occurrence of disease due to ill effect of heavy metal on human health.
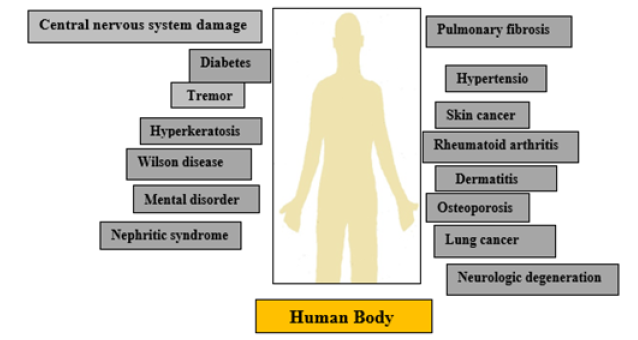
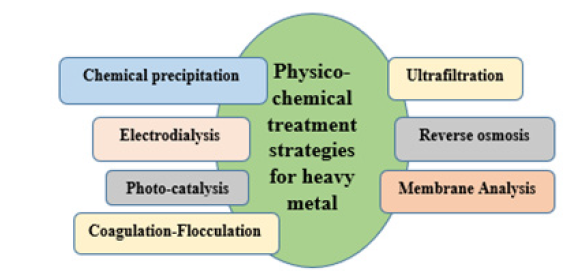
Figure 4:Schematic representation of Physico-chemical methods.
Therefore, possible negligible heavy metal exposure is necessary by using efficient decontamination techniques such as physicochemical methods and phytoremediation to avoid hazardous consequences [3]. Each application has its benefits and drawbacks [4]. Most commercial technologies offer diverse setbacks such as substantial operating costs, costs, efficiency, production of toxic sludge, etc. As a result, establishing a novel metal decontamination technique is necessary [5].
Physico-chemical methods are subdivided into different processes that are described below and pictorially represented (Figure 3).
Chemical precipitation
The chemical precipitation method involves the removal of heavy metals from inorganic effluents. Adding chemical reagents or precipitants to waste products facilitates the production of sulphides, carbonates, and hydroxides, which are then treated with filtration or sedimentation [6].
Membrane analysis
Membrane analysis involves metal ions, organic components, and suspended materials that can be removed using membrane filtration. The filtration process is eco-friendly and has separation specificity [7].
Reverse osmosis
Reverse osmosis is a pressure-driven process that forces the solution to pass from a semipermeable membrane from a higher concentration to a lower concentration to filter unwanted molecules. This method was highly used for the removal of Cu(II) and Ni(II) [8].
Ultrafiltration
Ultrafiltration especially removes colloidal particles. Ultrafiltration can be classified into- polymer-induced ultrafiltration and Micellar-induced ultrafiltration (PUEF, MUEF). In PUEF, where wastewater, soluble primer aids removal, whereas in MUEF, wastewater is treated with anionic surfactant, which forms micelles and heavy metal gets trapped in micelles [9].
Electrodialysis
Electrodialysis is a very effective method for industrial wastewater, which can separate charged metal ions by passing the ion exchange membrane from the solution after applying electrical potential. A series of studies have been performed with this method [10].
Photocatalysis
Photocatalysis methods use non-toxic semiconductors to destroy environmental pollutants. Especially the pollutants from the pharmaceutical industry degraded photo-catalytically, and selenium-doped ZnO nanocomposite semiconductors were used for removal [11].
Coagulation and flocculation
Coagulation and flocculation are effective methods to remove heavy metals/pollutants. It is based on Zeta potential measurements to define separation mechanisms such as electrostatic interaction between heavy metal and coagulation-flocculation agents [12].
The advantages of chemical methods involve a speedy metal elimination process, easy separation, the possibility of modification of chemical plant upon requirement, effective in a higher concentration of the metal solution, and production of toxic sludge [13,14].
The disadvantages of chemical methods are expensive and have many disadvantages, such as non-specificity, generation of toxic sludge, constant usage of toxic reagents, and incomplete removal of heavy metals [13,14].
Phytoremediation
Figure 5:Schematic representation of phytoremediation methods.
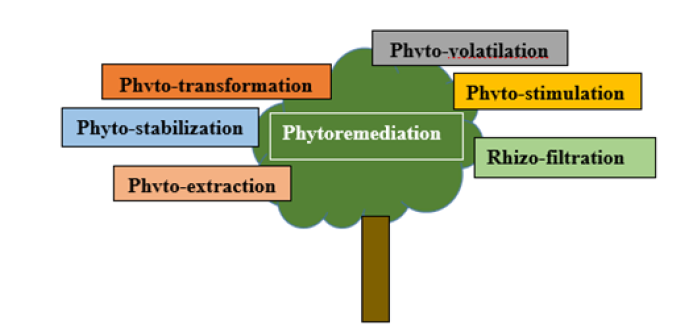
Phytoremediation involves remediation of soil, water, air, and pollutants using plants. It is an in-situ, eco-friendly, and natural clean-up method, which involves plant growth in the contaminated environment to decontaminate pollutants through uptake by plant roots. This mechanism has several advantages: cost-effectiveness, non-specific metal selectivity, and publicly accepted method. Phytoremediation is a collective process and can be classified into Phyto-extraction, phyto-stabilization, phyto-transformation, phytostimulation, phyto-volatilation and rhizo-filtration (Figure 4) [15- 17].
Phytoextraction
Phytoextraction is an in-situ clean-up and decontamination of soil and water through the active uptake of contaminants by plants, which involves a series of actions such as metal dissolution and its absorption and active transport. This process is hyperaccumulative regarding roots (secretion of metal-capturing agents) such as phytosiderophores, organic acids, and carboxylates [15].
Phytostabilization
process of phytostabilization includes active adsorption and accumulation of heavy metal using plant roots from soil, which involves a decrease in metal solubility and leaching prevention [17].
Phototransformation
Phototransformation is responsible for the state of transformation so that pollutants become sediments. This process involves the breakdown of pollutants by the action of different metabolic pathway enzymes [16].
Phytostimulation
Phytostimulation involves the microbial breakdown of organic pollutants in plants’ roots, or rhizosphere, and the entire process is aerobic [16].
Phytovolatilisation
Phytovolatilation involves the uptake of volatile pollutants such as (CCL4) through transpiration [15,18].
Rhizo-filtration
The rhizo-filtration process involves adsorption and accumulation of contaminants in the roots [15-17].
The advantages of phytoremediation are that it is an entirely nature-driven and eco-friendly cleaning method, which uses the natural inherent absorbing property of growing into the contaminated base to pollutant degradation at a meagre cost. It also offers product recycling of plants after phytoremediation [18].
The disadvantages of phytoremediation are the requirement of a more extended period for metal removal and difficulties of plant generation and growth for further metal removal. Considering the facts, including disadvantages regarding the inefficiency of the physicochemical and phytoremediation methods, biological methods are trending [15].
Future prospects
1. Maintaining the chemical and thermal stability of a novel
membrane (with heavy metal specificity) is found to be a novel
approach for wastewater bioremediation.
2. Electrochemical treatment is stability, recyclability, and cost
constrain.
3. The Photo-catalyst method has scope as it produces no toxic
sludge and uses no chemicals.
4. Production of ion-exchange resin as commercial sorbent is
highly encouraged.
Conclusion
Considerable implementation of physico-chemical removal techniques and phytoremediation for heavy metal removal offers numerous rewards over various setbacks of outdated methods. However, there needs to be a relevant research gap regarding the proper understanding of removal methods for cost analysis, sludge disposal management, etc. A significant study regarding proper evaluation and assessment of various effective treatment methods is necessary to employ it.
References
- Ruotolo R, Marchini G, Ottonello S (2008) Membrane transporters and protein traffic networks differentially affecting metal tolerance: A genomic phenotyping study in yeast. Genome Biol 9(4): R67.
- Hosiner D, Gerber S, Lichtenberg-Fraté H, Glaser W, Schüller C, et al. (2014) Impact of acute metal stress in saccharomyces cerevisiae. PLoS One 9(1): e83330.
- Sharma R, Jasrotia T, Sharma S, Sharma M, Kumar Rajeev Vats R, et al. (2021) Sustainable removal of Ni (II) from waste water by freshly isolated fungal strains. Chemosphere 282: 130871.
- Sławomir W (2017) Biosorption of nickel (II) and zinc (II) from aqueous solutions by the biomass of yeast Yarrowia lipolytica. Polish Journal of Chemical Technology 19(1): 1-10.
- Sharma R, Jasrotia T, Umar A, Sharma M, Sharma S, et al. (2022) Effective removal of Pb(II) and Ni(II) ions by Bacillus cereus and Bacillus pumilus: An experimental and mechanistic approach. Environmental Research 212(Pt B): 113337.
- Akinterinwa A, Adebayo I (2019) Chemical precipitation approach to the removal of heavy metals from wastewater for discharge into sanitary sewerage. Journal of Water Pollution & Purification Research 6(1).
- Zhu WP, Sun SP, Gao J, Fu FJ, Chung TS (2014) Dual-layer polybenzimidazole/polyethersulfone (PBI/PES) nanofiltration (NF) hollow fiber membranes for heavy metals removal from wastewater. Journal of Membrane Science 456: 117-127.
- Bakalár T, Milan B, Lucia G (2009) Heavy metal removal using reverse osmosis. Acta Montanistica Slovaca 14(3): 250-253.
- Chemat F, Tomao V, Virot M (2008) Ultrasound-assisted extraction in food analysis. Routledge Handbooks Online.
- Huang JH, Zeng GM, Zhou CF, Li X, Shi LJ, et al. (2010) Adsorption of surfactant micelles and Cd2+/Zn2+ in micellar-enhanced ultrafiltration. J Hazard Mater 183(1-3): 287-293.
- Cifuentes L, García I, Arriagada P, Casas J (2009) The use of electrodialysis for metal separation and water recovery from CuSO4-H2SO4-Fe solution. Separation and Purification Technology - Sep Purif Technol 68: 105-108.
- Shyni LS, Jagadish K, Srikantaswamy S, Abhilash MR (2016) Photocatalytic degradation and removal of heavy metals in pharmaceutical waste by selenium doped ZnO nano composite semiconductor. Journal for Research| Volume 2(5): 47-54.
- Shah BA, Shah AV, Singh RR (2009) Sorption isotherms and kinetics of chromium uptake from wastewater using natural sorbent material. International Journal of Environmental Science and Technology 6: 77-90.
- Krishnan KA, Sreejalekshmi KG, Baiju RS (2011) Nickel (II) adsorption onto biomass-based activated carbon obtained from sugarcane bagasse pith. Bioresource technology 102(22): 10239-10247.
- Abdur Razzak S, Faruque MO, Alsheikh Z, Alsheikhmohamad L, Alkuroud D, et al. (2022) A comprehensive review on conventional and biological-driven heavy metals removal from industrial wastewater. Environmental Advances 7: 100168.
- Johnson PD, Watson MA, Brown J, Jefcoat IA (2002) Peanut hull pellets as a single use sorbent for the capture of Cu(II) from wastewater. Waste Manag 22(5): 471-480.
- Pehlivan E, Altun T, Parlayıcı S (2009) Utilization of barley straws as biosorbents for Cu2+ and Pb2+ Journal of Hazardous Materials 164: 982-986.
- Sharma S, Rana S, Thakkar A, Baldi A, Murthy RSR, et al. (2016) Physical, chemical and phytoremediation technique for removal of heavy metals. Journal of Heavy Metal Toxicity and Diseases 1: 2.
© 2026 Sudip Kumar Das. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






