- Submissions

Full Text
Novel Research in Sciences
Leucovorin is a Better Folate Source for Pregnancy
George Ayoub*
Psychology Department, Santa Barbara City College, USA
*Corresponding author:George Ayoub, Psychology Department, Santa Barbara City College, Santa Barbara, CA 93109, USA
Submission: January 06, 2026;Published: February 02, 2026
.jpg)
Volume17 Issue 2February 02, 2026
Abstract
Folinic acid (leucovorin) supplementation in Folate Receptor Alpha Autoantibody (FRAA)-positive pregnancies offers a biologically targeted strategy to reduce Autism Spectrum Disorder (ASD) risk and is proving superior to folic acid across a broader range of reproductive and neurodevelopmental outcomes. A pilot randomized trial by Giorlandino et al. together with emerging clinical data on infertility and preclinical evidence on a negative impact of excess folic acid, suggests that leucovorin is the optimal choice of folate form for precision prenatal care. For pregnancies where FRAA is present, standard folic acid supplementation resulted in a majority of the births diagnosed as autistic within three years, while leucovorin supplementation resulted in a 10% autism rate. This striking difference is further evidence of the importance of choosing the appropriate form of folate for perinatal care, with leucovorin proving the safer choice, especially in the presence of FRAA [1-5].
Overview
Leucovorin (folinic acid) has emerged as a promising alternative to folic acid for pregnancy, particularly in women with Folate Receptor Alpha Autoantibodies (FRAA) and those with folate metabolism polymorphisms. This commentary examines the pilot randomized trial by Giorlandino et al. in FRAA‑positive pregnancies alongside clinical observations in infertility and preclinical data on excess folic acid exposure. In the Giorlandino trial, leucovorin supplementation, compared with folic acid, was associated with markedly lower Autism Spectrum Disorder (ASD) incidence, reduced ASD symptom severity, and higher Bayley-4 cognitive scores in offspring, despite a small sample size. Case series in women with MTHFR polymorphisms and infertility suggest that switching from folic acid to reduced folates (folinic acid or methylfolate) can restore fertility and lead to successful pregnancies, highlighting folate form as a modifiable determinant of reproductive outcome. Preclinical work further indicates that excess prenatal folic acid can alter cortical DNA methylation and gene expression networks, raising concern that high-dose folic acid may perturb brain development in susceptible populations. Together, these converging lines of evidence support a shift from a one‑size‑fits‑all folic acid paradigm toward precision folate strategies that incorporate FRAA status, genetic background, and folate form, and they motivate the central question of whether leucovorin is a better folate source for pregnancy in defined high‑risk groups [6-10].
FRAA, Folate Transport and ASD Risk
Giorlandino et al. intervened along a mechanistically coherent causal chain in which FRAA impair folate receptor‑mediated transport across the placenta and blood–brain barrier, causing cerebral folate deficiency despite normal systemic folate. FRAA block folate receptor alpha and diminish 5‑methyltetrahydrofolate (5‑MTHF) delivery to the fetal brain, a mechanism linked to ASD, infantile‑onset cerebral folate deficiency, and related neurodevelopmental syndromes. FRAA are substantially more prevalent in children with ASD and in mothers of ASD‑affected children, defining an immune‑mediated endophenotype within the broader ASD spectrum. In ASD children with cerebral folate deficiency, open‑label and case series data indicate that approximately two‑thirds improve with leucovorin, especially in communication and irritability, and experimental models show that maternal FRAA exposure induces offspring behavioral changes that leucovorin can prevent, supporting a causal role for impaired folate transport [1-3]. Within this framework, the Giorlandino trial is the first randomized human study to test whether correcting FRAA‑related folate transport failure during pregnancy can modify early ASD risk markers instead of treating established ASD postnatally.
Prevention Signal in FRAA‑Positive Pregnancies
In the trial, 210 women planning pregnancy were screened and 17.1% were FRAA‑positive, indicating that this immune risk factor is not rare in the general preconception population. Among the 29 FRAA‑positive women who conceived and were randomized, 18 completed follow‑up (10 received leucovorin, 8 received folic acid) with supplementation initiated after pregnancy confirmation and continued to delivery. Figure 1 provides a visual summary of their study. The neurodevelopmental signal, although generated from a small per‑protocol sample, is striking: ASD incidence at 24- 30 months was 10% in the leucovorin group versus 62.5% in the folic acid group (Fisher’s exact p=0.043), corresponding to an odds ratio around 0.07-0.08 and an estimated relative risk reduction of approximately 84%.
Figure 1:
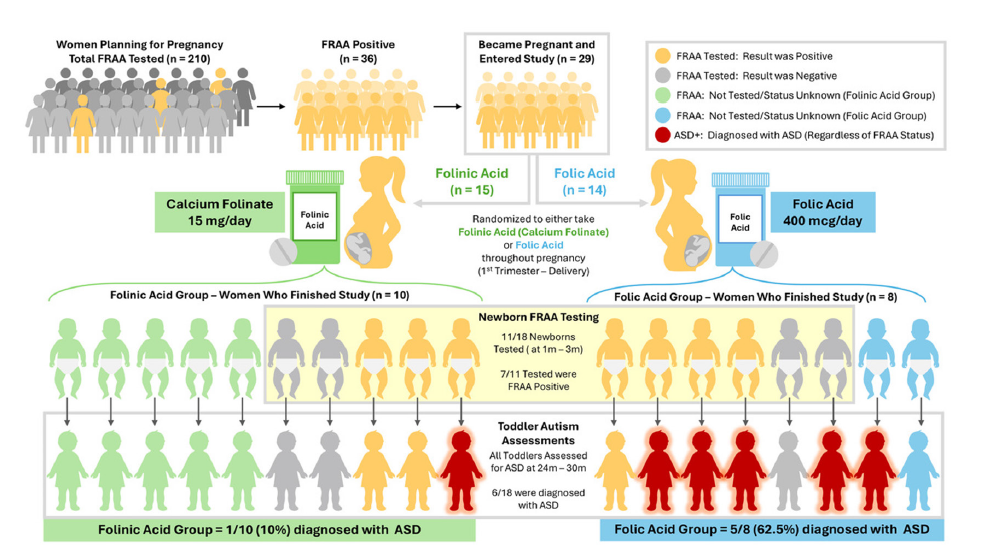
Learning disorders were less frequent in the leucovorin arm (10% vs. 50%; odds ratio 0.13), and ASD symptom severity (ADOS‑2) was significantly lower while Bayley‑4 cognitive scores were substantially higher, each with very large effect sizes (Cohen’s d>1.4-1.5). The single ASD case in the leucovorin arm carried a pathogenic SHANK2 variant, illustrating that folate‑immune mechanisms represent one etiological route among many and that pathway correction cannot prevent strongly genetic forms [4]. The clustering of greater ASD severity and lower cognition in the folic acid group, plus a strong negative correlation between ADOS‑2 scores and Bayley‑4 indices, reinforces a consistent pattern of folate‑mediated neuroprotection when FRAA‑positive pregnancies receive leucovorin.
Why Leucovorin May Outperform Folic Acid
Standard prenatal folic acid policy assumes intact folate receptor alpha‑mediated transport to deliver 5‑MTHF to the placenta and choroid plexus, an assumption that fails in FRAA‑positive women. In FRAA‑positive pregnancies, this receptor pathway is partially blocked or dysfunctional, creating cerebral folate deficiency despite apparently adequate circulating folate, and undermining the protective intent of folic acid [5,6]. Leucovorin differs from folic acid in two clinically relevant ways: it enters cells through alternative transport systems, notably the reduced folate carrier and proton‑coupled folate transporter, thereby bypassing antibody‑blocked folate receptor alpha, and it is already in a reduced, metabolically active form that more directly supports nucleotide synthesis and methylation, processes essential for neurogenesis and synaptogenesis [5,7,8]. Preclinical data indicate that high‑dose folic acid can, in some settings, be associated with adverse neurodevelopmental effects, whereas leucovorin shows neuroprotective benefits without the same concern profile, underscoring that the chemical form and transport route of folate are critical variables rather than interchangeable means of raising total folate [9]. Taken together, these observations suggest that leucovorin may be a more physiologically appropriate folate source in contexts of impaired receptor‑mediated transport or altered folate metabolism, including FRAA positivity and certain genetic variants.
Reversal of Infertility by Switching Folate Form
An underappreciated but highly relevant strand of evidence comes from clinical series in women with infertility and MTHFR polymorphisms, in whom changing the folate form alone appears to restore fertility and enable healthy pregnancies. In a case series by Ledowsky et al. [10], women with diagnosed infertility and MTHFR variants who had been using folic acid were transitioned to alternative folate formulations, leucovorin or methylfolate, resulting in successful conceptions and healthy births after prior unsuccessful attempts [10].
These infertility cases highlight several important principles for pregnancy folate policy: first, that folic acid is not universally benign nor uniformly effective across genotypes, and second, that correcting the form of folate, without altering the nominal dose, can reverse a clinically significant reproductive phenotype. When viewed alongside FRAA‑positive pregnancies in the Giorlandino trial, these data suggest that both immune‑mediated and genetically mediated impairments of folate handling may respond more favourably to reduced folate forms, including leucovorin.
From a translational perspective, the infertility experience strengthens the argument for individualized folate formulations, incorporating both autoantibody status and genetic polymorphisms (such as MTHFR) into preconception and early-pregnancy risk assessment. It also broadens the rationale for leucovorin and related reduced folates from neurodevelopmental prevention into the domain of reproductive success, positioning folate form as a modifiable determinant of both conception and subsequent fetal brain development [11].
Excess Folic Acid and Altered Brain Development
Preclinical work demonstrates that more folic acid is not necessarily better and that high-dose folic acid exposure during vulnerable developmental windows can perturb brain development at the epigenomic and transcriptomic levels. In a recent study, excess prenatal folic acid altered cortical DNA methylation and gene expression networks, indicating that supra‑physiologic levels of this synthetic vitamin can reprogram neurodevelopmental trajectories in ways that may not be benign [9]. A recent clinical study on folic acid use observed that of the 1200 women followed, those with folic acid supplementation in the second trimester had children with higher rates of behavioral problems at age four. Similarly, the women with folic acid supplementation during preconception and second trimester had children with poorer cognitive development [12].
These findings carry several implications for pregnancy folate policy: they challenge the assumption that increasing folic acid intake is uniformly protective and highlight the importance of physiological folate forms that align more closely with endogenous metabolism. They also raise the possibility that, in susceptible subgroups, including those with FRAA, MTHFR polymorphisms, or other metabolic bottlenecks, high folic acid exposure could create a mismatch between circulating folate and intracellular utilization, amplifying epigenetic perturbations in the developing brain.
Against this backdrop, leucovorin offers a mechanistically attractive alternative: it directly feeds into reduced folate pools, can bypass certain transport and metabolic constraints, and has not been associated with the same pattern of adverse preclinical neurodevelopmental changes at comparable doses. The convergence of preclinical data on excess folic acid with clinical observations of leucovorin’s benefits in ASD, FRAA‑positive pregnancies, and infertility underscores that the choice of folate form may help avoid iatrogenic risk while maximizing neuroprotection.
Toward Precision Folate Strategies in Pregnancy
The Giorlandino trial raises important translational questions for obstetrics, neurology, and nutrition regarding screening and targeted supplementation. FRAA are present in 60-70% of mothers of children with ASD in prior studies and in 17% of women planning pregnancy in this trial, suggesting that FRAA testing could function as a feasible biomarker for stratifying neurodevelopmental risk and guiding folate formulation.e general representation of the places taken into consideration.
In FRAA‑positive pregnancies, leucovorin was associated with lower ASD incidence, reduced ASD symptom severity, and improved cognitive outcomes, while folic acid failed to provide comparable protection, and leucovorin already carries a favorable safety profile in pregnancy when used for hematologic indications. Combined with the infertility case series and preclinical folic acid data, these results support a broader re‑examination of folate policy that differentiates by immune status, genotype, and metabolic context rather than relying solely on generic folic acid dosing targets.
Current guidelines, built primarily around neural tube defect prevention, remain anchored to folic acid and do not address the implications of FRAA, MTHFR polymorphisms, or potential neurodevelopmental effects of excess folic acid. A next‑generation framework would integrate FRAA screening, selected genetic testing, and careful consideration of folate form, favoring leucovorin or other reduced folates in defined high‑risk groups, while larger multicentre trials extend and validate the promising pilot signal observed by Giorlandino et al.
Integrating Immunology, Genetics and Nutrition
Perhaps the most significant conceptual advance from this set of findings is the integration of maternal autoimmunity and genetic variation into prenatal nutrition and ASD prevention paradigms. FRAA‑mediated cerebral folate deficiency bridges immunology, metabolism, and neurodevelopment, and the infertility and preclinical folic acid data add genetic and epigenetic dimensions, together defining a multi‑layered folate biology that is not addressed by one‑size‑fits‑all folic acid supplementation.
Future work should expand FRAA research beyond ASD to include language delay, learning disorders, and ADHD; harmonize FRAA assays, thresholds, and reporting standards to enable clinical laboratory implementation; and systematically explore timing, dosing, and co‑nutrient strategies for leucovorin, including preconception initiation. Against this evolving evidence base, the question “Is leucovorin a better folate source for pregnancy?” becomes not merely speculative but empirically grounded, with early data suggesting that, for specific immunologic and genetic subgroups, the answer may well be yes.
Acknowledgement
The author thanks Shana Anderson for generating the skilled rendition of the clinical trial and making it available for use.
References
- Frye RE, Slattery J, Delhey L, Sailey A, Rose S, et al. (2018) Folinic acid improves verbal communication in children with autism and language impairment: A randomized double-blind placebo-controlled trial. Mol Psychiatry 23(2): 247-256.
- Renard E, Bruno L, Rosa MG, Oussalah A, Edward VQ, et al. (2020) Folinic acid improves the score of Autism in the EFFET placebo-controlled randomized trial. Biochimie 173: 57-61.
- Panda PK, Sarama S, Gupta D, Indar KS, Kiran M, et al. (2024) Efficacy of oral folinic acid supplementation in children with autism spectrum disorder: A randomized double-blind, placebo-controlled trial. Eur J Pediatr 183(11): 4827-4835.
- Ayoub G (2025) Neurodevelopmental impact of maternal immune activation and autoimmune disorders, environmental toxicants and folate metabolism on autism spectrum disorder. Curr Issues Mol Biol 47(9): 721.
- Frye RE, Slattery JC, Quadros EV (2017) Folate metabolism abnormalities in autism: Potential biomarkers. Biomark Med 11(8): 687-699.
- Natasha BK, Ramaekers VT, Sequeira JM, Quadros EV (2021) Folate receptor alpha autoantibodies in autism spectrum disorders: Diagnosis, treatment and prevention. J Pers Med 11(8): 710.
- Frye RE, Daniel AR, Scahill L, Harris H, Edward VQ, et al. (2020) Treatment of folate metabolism abnormalities in autism spectrum disorder. Semin Pediatr Neurol 35: 100835.
- Ayoub G (2025) Vitamins, Vascular Health and Disease. Nutrients 17(18): 2955.
- Haghani V, Sara MA, Noemi C, Ralph G, Janine MS, et al. (2025) Excess prenatal folic acid supplementation alters cortical DNA methylation and gene expression networks. Front Nutr 12: 1699376.
- Ledowsky CJ, Schloss J, Steel A (2023) Variations in folate prescriptions for patients with the MTHFR genetic polymorphisms: A case series study. Explor Res Clin Soc Pharm 10: 100277.
- Ayoub G (2025) Autism spectrum disorder as a multifactorial disorder: The interplay of genetic factors and inflammation. Int J Mol Sci 26(13): 6483.
- Qiang W, Tuyan F, Yan S, Hui G, Zhu B, et al. (2025) A new perspective on the association between maternal folic acid supplementation across the perinatal period and cognitive and behavioral outcomes in children at age 4: Findings from the MABC birth cohort study. Eur J Nutr 65(1): 12.
© 2026 George Ayoub. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






