- Submissions

Full Text
Aspects in Mining & Mineral Science
Quantification of Free Cyanide (CN-) by Volumetric Analysis in Synthetic Solutions of NaCN
Esmeralda Y García A1, Ramiro Escudero G1*, Rosa E Pérez S2, Martín Reyes P3 and Roberto Guerra G4
1 Institute of Research in Metallurgy and Materials, Mexico
2 Pharmacobiology Faculty, Mexico
3 Academic Area of Earth Sciences and Materials, Autonomous University of the State of Hidalgo, Mexico
4 Chemical Engineering faculty, Michoacan University of San Nicolas de Hidalgo, Mexico
*Corresponding author:Ramiro Escudero G, Academic Area of Earth Sciences and Materials, Autonomous University of the State of Hidalgo, Mineral de la Reforma 42186, Pachuca, Hidalgo, Mexico
Submission: December 11, 2025: Published: December 19, 2025

ISSN 2578-0255Volume14 Issue 5
Abstract
There are at least five standardized methods to determine cyanides in water. These procedures have certain limitations and must be applied according to the type or components in the liquid medium. What is clear is that each step of the method applied must be well understood to avoid introducing errors that could lead to the incorrect quantification of free cyanide in solution. An important application of one of the procedures is in mineral processing, specifically in the quantification of free cyanide in residual solutions, for example, during the leaching of gold ores with cyanide. In this work, unreacted cyanide was quantified with known concentrations of cyanicide elements contained in synthetic solutions (simulating a pregnant residual solution). The free cyanide was quantified by the volumetric method based on the color change of the problem solution upon adding the titration reagent AgNO3. Experimental results show that CN and the metal form the complex Me(CN)x-, and the residual CN-, determined by the colorimetric technique, is satisfactorily quantified with an error of less than 3%; however, it is established that a limitation of this technique is the correct quantification of free cyanide if the CN- is less than 0.001g/l. The correct calculation of cyanide (without reaching saturation) that must be added to ensure the dissolution of all the gold contained in a mineral, avoids cyanide passivation and excessive contamination of groundwater and soils.
Keywords:Cyanidation; Leaching; Cyanicides; Cyanide passivation; Free cyanide
Theory
There is little literature on the subject of standardized procedures applied to determine with some accuracy the cyanide in solution, which does not react after a leaching process, even in mining practice procedures are not applied to establish the amount of cyanide necessary to cyanidate the cyanicide species and the gold contained in a gold-bearing ore. The addition of cyanide is based on empirical decisions. This fact is never associated with phenomena such as cyanide passivation and low gold recoveries [1]. NMX-AA-058-SCFI-2001 [2] and ASTM D2036-09 [3], are documents that describe standard test methods for cyanides in water. These include the quantification of cyanide species after distillation, cyanides amenable to chlorination by difference, weak acid dissociable cyanides and cyanides amenable to chlorination without distillation. Common interferences in the analysis for cyanide include oxidizing agents and sulfides, aldehydes, mainly. Aspects that are not considered in these communications are the sensitivity of the techniques, especially for the relatively high and low concentrations of cyanide in solution, in addition to human errors due to the visual assessment of the person applying the technique. In the case of the dissolution of gold and cyanicide metals contained in a gold-bearing ore, the technique applied to quantify the unreacted (or residual) cyanide is the one mentioned above as weak acid dissociable cyanides, which involves the quantification of free cyanide in solution, by colorimetry.
Procedure for quantifying free cyanide (CN-) by volumetry or colorimetry
The weak acid dissociable cyanides procedure consists of the
following steps:
a. Preparation of the titrating reagent (AgNO3) at 0.1 normal,
b. Preparation of the KI reagent at 5% (v/v),
c. Preparation of the solution with cyanide,
d. Titration with silver nitrate,
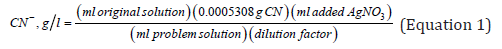
From (Equation 1), the constant 5.308 x 10-4 corresponds to the grams of cyanide that are associated with 1ml of AgNO3; in other words, one milliliter of silver nitrate reacts or identifies 5.308 x 10-4 grams of cyanide, according to the stoichiometric ratio of the following reaction.
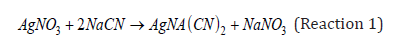
Certainly, cyanide in solution can be empirically quantified by constructing calibration curves that relate the silver nitrate “consumed” or added, and the known cyanide added to the solution; that is, the grams of silver nitrate that stoichiometrically correspond to react with one gram of cyanide (98g NaCN/169.86g AgNO3=0.576), for a certain dissolution factor of the sample with cyanide. Clearly, the limitation of this empirical quantification is that it does not consider the saturation factor of the solution with CN.
Difference between empirical and standardized methods
It is clear that the empirical method based on the development of calibration curves does not take into account the grams of silver nitrate contained in one liter of silver nitrate, nor does it consider the dilution factor.
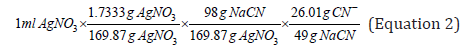
Where the result is the cyanide saturation factor, which is equal to 0.05308g of CN- (free cyanide). This constant is substituted into (Equation 1) and together with the dilution factor, generalizes the equation to determine the cyanide that did not react in solution. In this work, cyanide in solution is quantified using empirical calibration curves and applying the equation proposed by the standardized method, which considers the saturation factor of the solution with cyanide, regardless of the degree of dissolution of the analyzed sample.
Experimental Methodology
Materials and reagents
For the quantification of free cyanide by the volumetric technique, a 25ml burette, 100ml Erlenmeyer flasks, universal support, burette clamp, 500, 100 and 50 milliliters volumetric flasks and a 100 milliliters beaker were used. The chemical reagents used were Silver Nitrate (AgNO3), Sodium Cyanide (NaCN), Sodium Hydroxide (NaOH), Potassium Iodide (KI), distilled water, Au, Ca, Mg, Cu and Fe standards-all the above reagent grades.
Quantification of free cyanide in synthetic solutions without metals
Solutions with known concentrations of CN (0.05 to 0.25g) dissolved in distilled and deionized water were prepared. The pH of each sample was set at 11.5. To quantify the CN in solution, the procedure was applied according to the standards NMX-AA-058- SCFI-2001 and ASTM D2036-09.
Quantification of free cyanide in synthetic solutions with metals
Five cyanicidal elements (Au, Ca, Mg, Cu and Fe) were used to prepare synthetic solutions with known concentrations of these elements (Meadded). The quantification was carried out in unitary and binary synthetic solutions; each determination was carried out in triplicate. In the experiments with unitary solutions, the concentration of metals was set at 50ppm in 50ml of deionized water; in the case of binary solutions, the concentration of gold was maintained at 50ppm in 25ml of deionized water, and the rest of the metals were varied from 200 to 300ppm. The percentage of cyanide associated with metals varied from 0.002 to 0.13 (Metal/CNadded ratio from 0.04 to 5), depending on the cyanide concentration in the synthetic solutions. To prevent the formation of hydrocyanic acid, the pH of the synthetic solutions was kept at 11.5; this also ensures that the cyanide dissociated from NaCN will react with the cyanicide metal.
Identification of CN-metal complexes by UV-Vis technique
Each Metal-CN solution before free cyanide quantification was analyzed using the UV-Vis technique to identify the complex species formed for unitary and binary solutions. UV-Vis Spectroscopy equipment, Perkin Elmer brand, model Lambda BIO Series 24117, was used at 200 to 800nm wavelengths.
Result and Discussion
Results of free CN- quantification, empirically through calibration curves
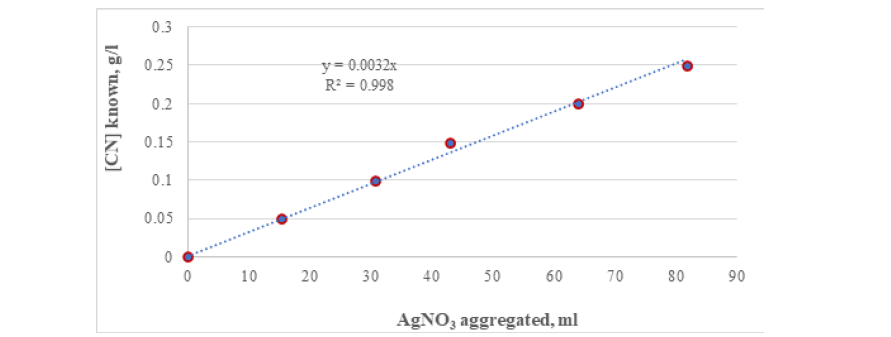
Figure 1 shows the results of CN- quantification by constructing calibration curves, for a dilution factor of 1.0. From the figure above, it is established that the concentration of cyanide in solution can be quantified if the milliliters of silver nitrate added during the colorimetric test are known; this applies to a dissolution factor of 1.0 and the CN range included in this test ([CN] much longer than 0.001g/l). It should be noted that some deviation from linearity is observed for the known minimum and maximum CN values. The equation shown in Figure 1 indicates that the concentration of cyanide in solution can be empirically determined by multiplying the milliliters of silver nitrate by the factor 0.0032. Applying the above relationship to the known values of CN, a certain deviation or error is observed, as the cyanide concentration decreases, as shown in Table 1. This deviation is due to the fact that the relationship between cyanide and silver nitrate is not always linear, this deviation is due to the fact that the relationship between cyanide and silver nitrate is not always linear, as shown in later paragraphs.
Table 1:[CN] known and calculated according to the equation included in Figure 1.
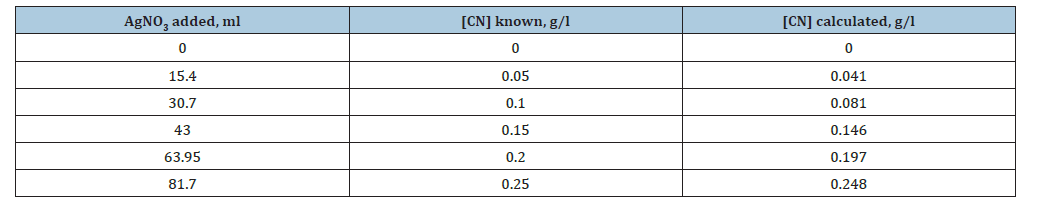
Figure 1:Calibration curve for the data with known concentration of CN in distilled water.
Results of free CN- quantification by volumetry. unitary and binary solutions
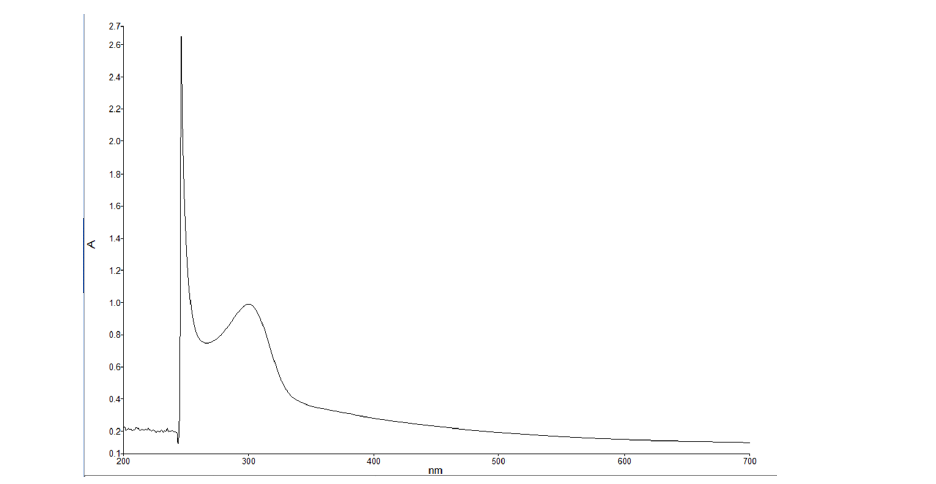
Identification of CN-metal complexes by UV-Vis technique: The solutions analyzed by this technique show evidence of the formation of the Me(CN)− 2 — complexes for the metals Au, Ca, Mg, and Cu, while for Iron, the species Fe(CN)− 6 — was identified. Its characteristic position at the corresponding wavelengths coincides with those reported in the literature [4-10]. An example of the spectra obtained is shown in Figure 2, which corresponds to one of the solutions with Mg. From the identification of the formation of metal complexes with cyanide, it is established that the CN determined by the volumetric technique is the residual or free cyanide, which is the one that did not react with the metals (CN−).
Figure 2:UV-Vis spectrum of the solution with the mixture of CN and Mg in distilled water.
Results of free CN- quantification by volumetry. All solutions
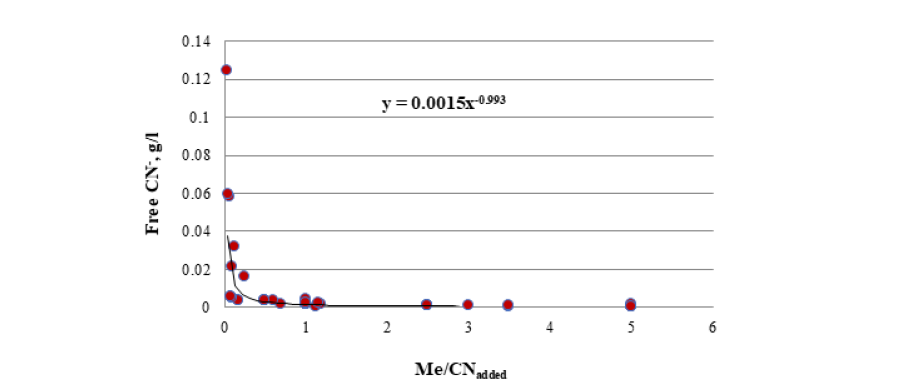
The results of free cyanide quantification by the volumetric technique in both unitary and binary solutions are presented in a previous communication [11]. By including in a single figure all the Me/CNadded ratios for all solutions, unitary and binary, the result is as shown in Figure 3. The above figure shows that all the relationships fit reasonably with the same function, which allows establishing the concentration of free cyanide for any concentration of cyanicidal metal and the cyanide added to the aqueous system. By knowing the concentration of cyanicidal metals in the system, it is possible to determine the quantity of cyanide that must be added so that the free cyanide is less than or close to 0.001g/l, which will help to avoid excess added cyanide, which can promote the passivation of the cyanidation reactions, as well as the contamination of soils and groundwater. Due to the shape of the curve in the relationship shown in Figure 3, it is established that a limitation of this technique is the adequate determination of free cyanide if the CN− is less than 0.001g/l and in any case, the quantification must be carried out in aliquots with a certain dilution factor less than 1.0.
Figure 3:Free CN− quantification results for all Me/CNadded ratios tested in this work.
Conclusion
After applying the standardized method of dissociable cyanides in weak acids to determine free or residual cyanides in water, the following conclusion is derived: All the Me/CNadded ratios included in this research work, reasonably adjusted to the same function that allows establishing the g/l of free cyanide in the residual solution once the cyanide reacts with the cyanicidal metals contained in the reaction system. This conclusion leads to the assertion that it is possible to avoid the excessive addition of cyanide, which inhibits the dissolution of cyanide species, passivating the reactions between cyanide and cyanicide metals contained, for example, in a mineral, as well as the high contamination of groundwater and soil. A limitation of this technique is proposed when the concentration of free cyanide is less than 0.001g/l; it is recommended that the quantification should be carried out in aliquots with a certain dilution factor less than 1.0.
Acknowledgment
Authors deeply thanks the UMSNH for the support received for carrying out this research work.
References
- Chumpitaz PH, Rodriguez CE, Andia PG (2022) Significant reduction in cyanide consumption in gold leaching allowed considerable savings in operating costs at the La Arena Mine Processing Plant. Technical Report 2022, Panamerican Silver-La Arena Mine, Canada.
- Standard NMX-AA-058-SCFI-2001. Official Gazette of the Federation.
- ASTM Designation: D2036-09 (2022) Standard test methods for cyanides in water. US Department of defense. iTeh Standards.
- Cobos H, Fabián O, Reyes T, Luis J, García F, et al. (2007) Electrochemical behavior of copper in cyanide solutions. Science and Technology 13(36): 239-244.
- Kurowa K, Masaki Y, Koga Y, Deming TJ (2013) Self-Assembly of discrete metal complexes in aqueous solution via block copolypeptide amphiphiles. International Journal of Molecular Sciences 14(1): 2022-2035.
- Rawashdeh-Omary MA, Omary MA, Patterson HH (2000) Oligomerization of Au(CN)2- and Ag(CN)2-Ions in solution via ground-state aurophilic and argentophilic. J Am Chem Soc 122(42): 10371-10380.
- Monica CV, Avila Y, Mojica R, Avila M, Reguera E (2023) A typical metal‐center coordination in two cyanide‐based coordination polymers. Chemistry Select 8(32).
- Gabler N, Hartley J, Frisch G (2017) Voltammetric and spectroscopic study of ferrocene and hexacyanoferrate and the suitability of their redox couples as internal standards in ionic liquids. Physical Chemistry Chemical Physics 19(42): 28841-28852.
- Ching Bin K, Te Ling L, Jian Lian C (2018) Capacitively coupled plasma discharge of ionic liquid solutions to synthesize carbon dots as fluorescent sensors. Nanomaterials 8(6):
- Barani K, Torkashvand J, Rouhbakhsh H (2023) Characterization of gold and copper complexes in cyanide-clycine systems in cyanide-glycine system by UV.V is spectroscopy. Journal of Chemical Technology and Metallurgy 58(5): 906-912.
- Esmeralda Y García A, Ramiro Escudero G, Rosa E Pérez S, Martín Reyes P, Roberto Guerra G (2024) Determination of the appropriate metal/cyanide ratio for the reaction of gold and cyanicidal metals in synthetic solutions quantification of free cyanide by colorimetry. Advances in Mining & Mineral Engineering 1(3): 1-10.
© 2025 Ramiro Escudero G. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






