- Submissions

Full Text
Trends in Telemedicine & E-health
Device-Associated Fungal Bloodstream Infections and Central Nervous System Invasion: Mechanisms, Immunopathogenesis and Stewardship Strategies
Gargee Anand and Rijhul Lahariya*
All India Institute of Medical Sciences Patna, India
*Corresponding author:Dr. Rijhul Lahariya, All India Institute of Medical Sciences, India
Submission: March 20, 2026;Published: April 06, 2026

ISSN: 2689-2707 Volume 6 Issue 4
Abstract
Healthcare-Associated Infections (HAIs), particularly device-associated infections, remain a major cause of morbidity and mortality in hospitalized patients. Among these, Central Line-Associated Blood-Stream Infections (CLABSIs) are well recognized; however, the contribution of fungal pathogens has gained increasing clinical importance. Candida species, including Candida albicans, Candida parapsilosis and the emerging multidrug-resistant Candida auris, are primary etiological agents capable of colonizing indwelling medical devices and forming resilient biofilms. These biofilms facilitate persistent bloodstream infections and enable hematogenous dissemination. In rare but severe cases, fungal pathogens can invade the Central Nervous System (CNS), resulting in meningoencephalitis. Mechanistically, CNS invasion occurs through multiple pathways, including direct endothelial invasion, paracellular traversal and the “Trojan horse” mechanism via infected phagocytes. Neurotropic fungi such as Cryptococcus neoformans demonstrate a high propensity for blood-brain barrier penetration, whereas device-associated fungi may access the CNS through colonized neurosurgical hardware such as ventricular drains and shunts. Host immune responses, including innate defenses and adaptive T-cell-mediated immunity, play a critical role in controlling infection but may also contribute to neuroinflammation. Clinical outcomes are often poor, with high mortality rates, particularly in immune-compromised individuals. Complications such as hydrocephalus and cerebral abscesses further worsen prognosis. Antifungal stewardship is therefore essential, emphasizing early diagnosis, targeted therapy and therapeutic drug monitoring. A comprehensive understanding of fungal pathogenesis, host-pathogen interactions and optimized stewardship strategies is crucial to improving outcomes in these life-threatening infections..
Keywords:Device-associated infections; Fungal bloodstream infections; Candida species; Meningoencephalitis; Antifungal stewardship
Introduction
Healthcare Associated Infections (HAIs) are defined as infections acquired during medical care that were neither present nor incubating at the time of admission, and they are linked to increased morbidity, mortality, prolonged hospitalization and higher healthcare costs [1]. Device-associated infections significantly contribute to ICU morbidity, with hundreds of cases reported in large cohorts of patients with invasive devices [2,3]. Central Line-Associated Blood-Stream Infections (CLABSIs) are among the most significant HAIs globally and are strongly associated with adverse outcomes in hospitalized patients [4,5]. While bacterial pathogens remain the predominant cause of device-related bloodstream infections, fungal pathogens, particularly Candida spp., have gained increasing clinical importance, accounting for approximately 20% of bloodstream infections in ICU patients and strongly associated with invasive devices such as central venous catheters [6,7]. Fungal pathogens, particularly Candida spp., are recognized causes of BSIs associated with intravascular devices, with rising incidence in hospital settings [8-10]. Fungal BSIs frequently involve Candida species, which can colonize indwelling devices such as central venous catheters, ventricular assist devices and Cerebro-Spinal Fluid (CSF) shunts [11]. These infections can disseminate hematogenously, occasionally crossing the Blood Brain Barrier (BBB) to cause severe meningoencephalitis [12]. Neurotropic fungi such as Cryptococcus neoformans classically invade via the bloodstream, whereas device associated fungi such as Candida spp. may use colonized CSF shunts or drains as conduits for CNS entry [6,13]. The clinical consequences are profound: Delayed diagnosis, therapeutic complexity and high morbidity and mortality, particularly in immunocompromised hosts [1]. Fungal neuroinfections, although relatively rare, are life-threatening, with cryptococcal meningitis alone causing ~220,000 cases and 180,000 deaths annually worldwide, highlighting the need for rapid diagnosis and targeted antifungal therapy [14-16].
Recent increases in device utilization have expanded opportunities for fungal biofilm-mediated infections, with Candida spp. responsible for ~5.4-16% of catheter-related bloodstream infections, where biofilm formation on devices facilitates immune evasion, persistent fungemia and dissemination to deep organs including the CNS [17-19]. The ability of fungi to adhere to foreign surfaces, form resilient biofilms and resist antifungal agents compounds the risk of invasive disease progression and complicates management [1]. This narrative review aims to synthesize current knowledge on mechanisms of fungal dissemination in device associated settings, the profile of organisms implicated, host immune responses, clinical prognosis and the critical role of antimicrobial stewardship in optimizing outcomes.
Mechanism of BSI Causing Meningoencephalitis
Device colonization and biofilm formation
The initiation of a device associated fungal infection typically involves colonization of abiotic surfaces by fungal cells [20]. Species such as Candida albicans, Candida parapsilosis and the emerging multidrug resistant Candida auris exhibit strong adhesive properties and the capacity to form structured biofilms on device surfaces [20,21]. Biofilms protect fungi from host immune defenses and enhance tolerance to antifungal agents, creating a persistent reservoir for bloodstream seeding [22]. Species such as Candida albicans, Candida parapsilosis and the emerging multidrug-resistant Candida auris exhibit substantial biofilm-forming capacity, with reported rates of approximately 30-50% in C. albicans, 66-82% in C. parapsilosis, and up to 60-90% in C. auris isolates, of which nearly 25-30% are strong biofilm producers [23,24]. In addition, non albicans Candida species collectively demonstrate biofilm formation in up to 80-90% of clinical isolates, reflecting their increasing role in device-associated infections [21]. Other opportunistic fungi, including Aspergillus spp., also form structurally complex biofilms in a majority of isolates, further contributing to device colonization [25]. These biofilms provide a protective extracellular matrix that limits antifungal penetration and shields fungal cells from host immune responses, leading to significantly increased antifungal tolerance and persistent infection. Consequently, indwelling medical devices act as reservoirs for sustained fungal shedding into the bloodstream, promoting candidemia and other invasive fungal infections. Once fungi gain access to the bloodstream, they may disseminate hematogenously. Although rare, this systemic spread can lead to seeding of the CNS, especially in immunocompromised individuals or patients with breaches in meninges or neurosurgical hardware [20,26].
Hematogenous dissemination and blood-brain barrier penetration
Once established in the bloodstream, fungal pathogens such as Candida and Cryptococcus species can disseminate systemically [27]. Mechanisms of dissemination include direct invasion of endothelial barriers, survival within phagocytes (the “Trojan horse” model), and paracellular traversal of endothelial tight junctions [12]. Cryptococcal species are particularly adept at crossing the blood brain barrier, often within macrophages, leading to meningitis or meningoencephalitis [12]. Postbiotics, particularly short-chain fatty acids derived from gut microbiota, modulate the gut-brain axis by regulating systemic immune responses and neuroinflammatory pathways, which may influence host susceptibility and inflammatory response to hematogenous fungal dissemination in device-associated BSIs [28,29].
Device associated CNS infections
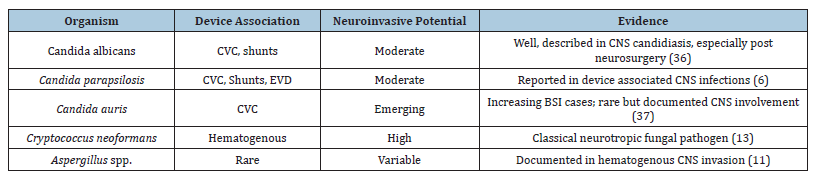
Indwelling CNS devices such as External Ventricular Drains (EVDs), Ventriculo-Peritoneal (VP) shunts and Ommaya reservoirs provide direct access to the CNS and can be focal points for fungal colonization and infection [6]. Infections may begin at the device interface and then propagate along catheter tracks into CSF spaces, leading to meningeal inflammation and brain parenchymal involvement [6]. Case reports highlight the rare but clinically significant role of fungi in device-associated CNS infections, with Candida parapsilosis identified as a cause of VP shunt and EVD infections, including recent reports emphasizing its occurrence in neurosurgical patients despite its rarity, while emerging data on Candida auris document multidrug-resistant meningitis secondary to infected shunt systems, with only a limited number of cases reported globally, underscoring its evolving neuroinvasive potential [30-33]. Table 1 depicts various fungal BSI causing organisms leading to CNS infections.
Table 1:Key pathogens in device‑associated fungal BSIs with CNS involvement.
Action of the Immune System in Fungal Device BSI and CNS Infection
Innate immune defenses
The innate immune system represents the first line of defense against fungal invasion [34]. Neutrophils, macrophages, and dendritic cells recognize fungal pathogen associated molecular patterns via pattern recognition receptors such as Toll like receptors and C type lectin receptors [35]. Neutrophils can phagocytose yeast cells and release reactive oxygen species and neutrophil extracellular traps to limit fungal spread [27]. Trained immunity, a form of innate immune memory mediated through epigenetic and metabolic reprogramming of monocytes and macrophages, enhances antifungal host responses following prior exposure to fungal components such as β-glucan [36-38]. However, in device associated infections, biofilms can shield fungal cells from phagocytosis, enabling persistent bloodstream infection [22].
Adaptive immune responses
CD4+ T cells and Th1 responses are critical for controlling systemic fungal infections. Interferon γ and other pro inflammatory cytokines enhance macrophage fungicidal activity [39,40]. Clinical and translational studies demonstrate that restoration of CD4⁺ T cell responses reduce fungal burden, but paradoxically, approximately 10-30% of patients with cryptococcal meningitis develop Immune Reconstitution Inflammatory Syndrome (IRIS) following immune recovery, reflecting excessive Th1-driven inflammation [41]. Meta-analytic data further indicate that cryptococcal IRIS carries a mortality of ~20.8%, compared to ~4.5% across all IRIS forms, underscoring its severity [42]. Mechanistically, Th1 polarization with interferon-γ production promotes macrophage fungicidal activity but also drives recruitment of inflammatory myeloid cells and cytokine-mediated tissue injury within the central nervous system [43]. In CNS fungal disease, immune responses can be a double-edged sword: While necessary for pathogen clearance, they can contribute to inflammation of the meninges and cerebral tissue, exacerbating clinical severity [44].
Immunocompromised hosts
Patients with neutropenia, HIV/AIDS, immunosuppressive therapies, or critical illness have impaired innate and adaptive responses [45,46]. Cohort data indicate that in HIV-associated cryptococcal disease, median CD4 counts are often <50 cells/mm³, reflecting profound immunosuppression, while mortality remains high at ~30% despite treatment [47]. This immunosuppression facilitates fungal BSI establishment, persistence, and dissemination to deep organs including the CNS [48]. Mechanistically, fungal pathogens exploit impaired immune surveillance to cross the blood–brain barrier via transcellular migration, paracellular entry, or “Trojan horse” transport within infected phagocytes [49]. Consequently, these vulnerabilities enable the development of meningoencephalitis, particularly in patients with severe immune deficits or disrupted host barriers [50].
Prognosis
Clinical outcomes
Fungal BSIs carry high mortality, particularly when associated with invasive devices and underlying critical illness. Candida BSI alone has been associated with mortality rates ranging from 30- 60%, a figure that increases dramatically with CNS involvement or delayed treatment [9]. Device associated fungal infection of Ventricular Assist Devices (VADs) also carries significant mortality and morbidity, and the presence of Candida species can lead to endocarditis and reduced long term survival in VAD recipients [51]. Hence early detection and prompt initiation of appropriate therapy in bloodstream infections are critical to limit hematogenous dissemination and reduce the risk of metastatic complications, including central nervous system involvement [52,53].
Complications
Complications of fungal meningoencephalitis are common and clinically significant, with studies reporting hydrocephalus in approximately 9-63% of patients with cryptococcal meningitis, reflecting one of the most frequent and life-threatening sequelae requiring cerebrospinal fluid diversion [54-57]. In cohort analyses of CNS cryptococcosis, cerebral abscesses (cryptococcomas) occur in ~12% of cases, while hydrocephalus and raised intracranial pressure remain dominant contributors to morbidity [58]. Longterm outcomes are also substantial, with systematic review data showing that neurological impairment persists in 19-70% of survivors, highlighting the burden of chronic deficits [59]. These conditions often require prolonged antifungal therapy and sometimes surgical intervention such as device removal or debridement [56,60].
Importance of antimicrobial stewardship
Antimicrobial Stewardship Programs (ASP) have traditionally focused on bacterial pathogens; however, the rise of invasive fungal infections underscores the need for robust antifungal stewardship [61]. Appropriate antifungal stewardship involves optimizing diagnosis, prescribing targeted antifungal agents, minimizing inappropriate use, and monitoring therapeutic drug levels to avoid toxicity [62].
Benefits of stewardship
i. Improved diagnosis: Utilization of biomarkers (e.g., βD
glucan), molecular diagnostics, and rapid species identification
to confirm fungal infection and species susceptibility [63].
ii. Rational use of antifungals: Tailoring therapy based on
susceptibility patterns to avoid unnecessary broad-spectrum
antifungals that drive resistance [62].
iii. Therapeutic drug monitoring: Ensuring appropriate
serum drug levels for agents like amphotericin B or triazoles to
maximize efficacy while minimizing toxicity [64].
iv. Outcome improvement: Stewardship interventions have
been shown to increase appropriate antifungal use and reduce
inappropriate prescriptions, a key component in invasive infection
management [65]. Importantly, timely and targeted therapy
may limit hematogenous dissemination and reduce secondary
complications, including central nervous system involvement.
Emerging evidence suggests that preservation of blood-brain
barrier integrity and Neuro-Vascular Unit (NVU) function is a
critical determinant of CNS susceptibility, further underscoring the
importance of early, optimized intervention.
Conclusion
Device-associated fungal bloodstream infections represent a complex interplay between pathogen virulence, device-related biofilm biology, and host immune status. This review highlights that Candida species, particularly C. albicans, C. parapsilosis and the emerging C. auris, along with other opportunistic fungi, possess strong biofilm-forming capacity that facilitates persistence on indwelling devices and sustained bloodstream seeding. Hematogenous dissemination, aided by mechanisms such as endothelial invasion and “Trojan horse” transport, enables these pathogens to breach the blood-brain barrier and establish meningoencephalitis, particularly in immunocompromised hosts. While innate and adaptive immune responses are essential for fungal clearance, dysregulated inflammation, such as IRIS, can exacerbate CNS injury, underscoring the dual role of host immunity. Clinically, these infections are associated with high mortality, frequent complications including hydrocephalus and long-term neurological deficits, and often require prolonged antifungal therapy alongside surgical interventions. Importantly, this review underscores the growing clinical significance of fungal pathogens in device-associated infections and the need for integrated antifungal stewardship strategies to enable early diagnosis, targeted therapy and improved outcomes. As device use and high-risk patient populations continue to expand, addressing these infections will require coordinated advances in diagnostics, therapeutics and infection control practices.
Statements and Declarations
Competing interests
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Funding
No funding was received.
Authors contribution
Rijhul Lahariya and Gargee Anand contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Gargee Anand and Rijhul Lahariya. The first draft of the manuscript was written by Rijhul Lahariya and Gargee Anand. Rijhul Lahariya and Gargee Anand both contributed equally to the work*. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Acknowledgement
None
References
- Anand G, Lahariya R (2025) Healthcare-associated infections. GMS Hyg Infect Control 20: 1-9.
- Anand G, Lahariya R, Sarfraz A, Thakuria B, Kokkayil P, et al. (2026) Antimicrobial resistance profiles and mortality rates in intensive care unit patients having central line associated blood stream infection: A temporal analysis. GMS Hyg Infect Control 21: 1-8.
- Menon G, Subramanian A, Baby P, Daniel N, Radhika R, et al. (2020) Incidence of device associated-healthcare associated infections from a neurosurgical intensive care unit of a tertiary care center: A retrospective analysis. Anesth Essays Res 14(3): 454-460.
- Anand G, Lahariya R, Priyadarshi K, Sarfraz A (2025) From access to reserve: Antimicrobial resistance among etiological agents of central line-associated bloodstream infections in the view of WHO’s AWaRe antimicrobial spectrum. GMS Hyg Infect Control 20: 1-9.
- Lahariya R, Anand G, Sarfraz A, Tiewsoh JBA, Kumar A (2025) CLABpredICU---AI-driven risk prediction for CLABSI in intensive care units based on clinical and biochemical parameters. Am J Infect Control 53(8): 875-880.
- Bhalla GS, Malik M, Sarao MS, Bandyopadhyay K, Singh P, et al. (2018) Device-associated central nervous system infection caused by candida parapsilosis. Cureus 10(8): e3140.
- Pérez MA, Hoffmann WJ, Pérez JC, Adelman MW (2025) Invasive candidiasis in critically Ill Patients: Fundamental concepts and future directions. Chest p. S0012-3692(25)05954-9.
- Deobrat CM, Nayanjyoti K, Lokesh G, Lipika M, Prabhat S (2025) A Comprehensive review of candidemia and invasive candidiasis in adults: Focus on the emerging multidrug-resistant fungus candida auris. Diseases 13(4): 93.
- Deng J, Liu Y, Shu L, Zhou D, Xie Y, et al. (2025) The prevalence, patterns, and antifungal drug resistance of bloodstream infection-causing fungi in Sichuan Province, China (2019-2023): A retrospective observational study using national monitoring data. Front Microbiol 16: 1616013.
- Anand G, Lahariya R, Priyadarshi K, Sarfraz A (2025) Emerging threat of WHO priority pathogens in ICU-associated CLABSI and CAUTI: An integrated analysis of resistance patterns, epidemiological trends, and stewardship strategies. Acta Clin Belg 80(5): 135-145.
- Góralska K, Blaszkowska J, Dzikowiec M (2018) Neuroinfections caused by fungi. Infection 46(4): 443-459.
- Anand G, Lahariya R (2025) Bloodstream infection-induced neuroinflammation: From systemic infection to brain invasion. Curr Microbiol 83(1): 47.
- Lino R, Guimarães AR, Sousa E, Azevedo M, Santos L (2024) Emerging fungal infections of the central nervous system in the past decade: A literature review. Infect Dis Rep 16(5): 952-976.
- Masouris I, Klein M, Schichor C, Stemmler J, Koedel U, et al. (2025) Clinical, diagnostic and therapeutic challenges in CNS fungal infections: A single-center retrospective data analysis. J Neurol 272(3): 214.
- Anand G, Lahariya R, Sarfraz A, Tiewsoh JBA (2025) Early screening of central line-associated bloodstream infections: A novel comparative analysis of AISI, SII, and SIRI as predictive biomarkers. Am J Infect Control 53(12): 1320-1324.
- Dao A, Kim HY, Garnham K, Kidd S, Sati H, et al. (2024) Cryptococcosis-a systematic review to inform the world health organization fungal priority pathogens list. Med Mycol 62(6): myae043.
- Kardas N, Wyroba N, Sordyl R, Antkowiak L, Mandera M (2025) Fungal infections of cerebrospinal fluid shunt in pediatric patients: A systematic literature review with a case report. World Neurosurg 204: 124567.
- Bloch K, Hasbun R (2021) Central nervous system infections associated with neurologic devices. Curr Opin Infect Dis 34(3): 238-244.
- Devrim İ, Celebi MY, Karakaya N, Sahinkaya S, Acar SO, et al. (2023) The evaluation of Candida-related central line-related bloodstream infections in pediatric cancer patients: A pre- and post-intervention study. J Infect Prev 24(5): 219-222.
- Nett JE, Andes DR (2020) Contributions of the biofilm matrix to candida pathogenesis. J Fungi 6(1): 21.
- Romera D, Aguilera CJJ, Gadea I, Viñuela SL, García RJ, et al. (2019) Candida auris: A comparison between planktonic and biofilm susceptibility to antifungal drugs. J Med Microbiol 68(9): 1353-1358.
- Johnson CJ, Cabezas OJ, Kernien JF, Wang SX, Beebe DJ, et al. (2016) The extracellular matrix of candida albicans biofilms impairs formation of neutrophil extracellular traps. PLoS Pathog 12(9): e1005884.
- Khari A, Biswas B, Gangwar G, Thakur A, Pria R (2023) Candida auris biofilm: A review on model to mechanism conservation. Expert Rev Anti Infect Ther 21(3): 295-308.
- Arévalo JBV, Admella J, Blanco CN, Torrents E (2023) Culture media influences candida parapsilosis growth, susceptibility, and virulence. Front Cell Infect Microbiol 13: 1323619.
- Morelli KA, Kerkaert JD, Cramer RA (2021) Aspergillus fumigatus biofilms: Toward understanding how growth as a multicellular network increases antifungal resistance and disease progression. PLoS Pathog 17(8): e1009794.
- Chatzimoschou A, Giampani A, Meis JF, Roilides E (2021) Activities of nine antifungal agents against candida auris biofilms. Mycoses 64(4): 381-384.
- Strickland AB, Shi M (2021) Mechanisms of fungal dissemination. Cell Mol Life Sci CMLS 78(7): 3219-3238.
- Dalile B, Van OL, Vervliet B, Verbeke K (2019) The role of short-chain fatty acids in microbiota-gut-brain communication. Nat Rev Gastroenterol Hepatol 16(8): 461-478.
- Lahariya R, Anand G, Kumari B, Priyadarshi K (2026) Postbiotics and the gut-brain axis: A mechanistic review on modulating neuroinflammation and cognitive aging. J Neuroimmunol 413: 578870.
- Fadel H, Moon SJ, Klinger NV, Chamiraju P, Eltahawy HA, et al. (2018) Candida parapsilosis Infection of ventriculoperitoneal shunt in adult: Case report and literature review. World Neurosurg 119: 290-293.
- Bagheri F, Cervellione KL, Maruf M, Marino W, Santucci T (2010) Candida parapsilosis meningitis associated with shunt infection in an adult male. Clin Neurol Neurosurg 112(3): 248-251.
- Sridharan S, Govindaswamy A, Rajendran A, Roopesh KVR (2024) Challenges in management of Candida auris meningitis secondary to infected ventriculoperitoneal shunt - A case report. Indian J Med Microbiol 48: 100555.
- Ikizoglu E, Arslan M, Guzel I, Kizmazoglu C, Oguz VA, et al. (2025) Candida parapsilosis: A rare culprit of shunt infection in an adult. Case Rep Surg 2025: 6687581.
- O’Brien D, Stevens NT, Lim CH, O’Brien DF, Smyth E, et al. (2011) Candida infection of the central nervous system following neurosurgery: A 12-year review. Acta Neurochir (Wien) 153(6): 1347-1350.
- Jelkin GL, Heineman AL, Beckett HT, Goddard CM, Ngwenya LB (2026) Intracranial candida auris infection: A case report and scoping review. World Neurosurg 206: 124775.
- Netea MG, Joosten LAB, Latz E, Mills KHG, Natoli G, et al. (2016) Trained immunity: A program of innate immune memory in health and disease. Science 352(6284): aaf1098.
- Lahariya R, Anand G, Kumari B (2025) Trained Immunity in sepsis: Exploring the molecular link to long-term cardiometabolic disorders. Immunol Res 73(1): 139.
- Dos SJC, Barroso FAM, Teodoro SMV, Cirovic B, de BLCJ, et al. (2019) β-Glucan-Induced trained immunity protects against leishmania braziliensis infection: A crucial role for IL-32. Cell Rep 28(10): 2659-2672.e6.
- Mukaremera L, Nielsen K (2017) Adaptive immunity to cryptococcus neoformans infections. J Fungi 3(4): 64.
- Lionakis MS, Drummond RA, Hohl TM (2023) Immune responses to human fungal pathogens and therapeutic prospects. Nat Rev Immunol 23(7): 433-452.
- Hain S, Fu MS, Wigg L, George L, Lecky D, et al. (2025) Brain-infiltrating CD4 T cells drive inflammatory microglia proliferation during cryptococcal meningitis in mice. Nat Commun 16(1): 8995.
- Khaw YM, Aggarwal N, Barclay WE, Kang E, Inoue M, et al. (2020) Th1-Dependent cryptococcus-associated immune reconstitution inflammatory syndrome model with brain damage. Front Immunol 11: 529219.
- Mohamed SH, Nyazika TK, Ssebambulidde K, Lionakis MS, Meya DB, et al. (2022) Fungal CNS infections in Africa: The neuroimmunology of cryptococcal meningitis. Front Immunol 13: 804674.
- Neal LM, Xing E, Xu J, Kolbe JL, Osterholzer JJ, et al. (2017) CD4+ T cells orchestrate lethal immune pathology despite fungal clearance during cryptococcus neoformans meningoencephalitis. mBio 8(6): e01415-e01417.
- Anandani G, Bhise M, Agarwal A (2025) Invasive fungal infections and the management in immunocompromised conditions. J Fam Med Prim Care 14(7): 2643-2652.
- Badiee P, Hashemizadeh Z (2014) Opportunistic invasive fungal infections: Diagnosis & clinical management. Indian J Med Res. 139(2): 195-204.
- Person AK, Crabtree RB, Kim A, Veloso V, Maruri F, et al. (2023) Cryptococcal meningitis and clinical outcomes in persons with human immunodeficiency virus: A global view. Clin Infect Dis 76(12): 2116-2125.
- Reyes EY, Shinohara ML (2022) Host immune responses in the central nervous system during fungal infections. Immunol Rev 311(1): 50-74.
- Liu TB, Perlin DS, Xue C (2012) Molecular mechanisms of cryptococcal meningitis. Virulence 3(2): 173-181.
- Chen Y, Shi ZW, Strickland AB, Shi M (2022) Cryptococcus neoformans infection in the central nervous system: The battle between host and pathogen. J Fungi (Basel) 8(10): 1069.
- Anderson R, Pouch S, Busch L, Hayes T, Sennhauser S, et al. (2025) Candidemia in left ventricular assist device recipients: Incidence, risk factors, and outcomes. Open Forum Infect Dis 12(5): ofaf251.
- Pappas PG, Kauffman CA, Andes DR, Clancy CJ, Marr KA, et al. (2016) Clinical practice guideline for the management of candidiasis: 2016 update by the infectious disease’s society of America. Clin Infect Dis 62(4): e1-e50.
- Lahariya R, Anand G (2025) Early detection of central line-associated bloodstream infection in intensive care unit patients using the systemic inflammatory response index (SIRI). GMS Hyg Infect Control 20: Doc77
- Sivasubramanian G, Kadakia S, Kim JM, Pervaiz S, Yan Y, et al. (2023) Challenges in the long-term management of patients with coccidioidal meningitis: A retrospective analysis of treatment and outcomes. Open Forum Infect Dis 10(6): ofad243.
- Yampolsky C, Corti M, Negroni R (2010) Fungal cerebral abscess in a diabetic patient successfully treated with surgery followed by prolonged antifungal therapy. Rev Iberoam Micol 27(1): 6-9.
- Young RF, Gade G, Grinnell V (1985) Surgical treatment for fungal infections in the central nervous system. J Neurosurg 63(3): 371-381.
- Liliang PC, Liang CL, Chang WN, Chen HJ, Su TM, et al. (2003) Shunt surgery for hydrocephalus complicating cryptococcal meningitis in human immunodeficiency virus-negative patients. Clin Infect Dis 37(5): 673-678.
- Beardsley J, Sorrell TC, Chen SCA (2019) Central nervous system cryptococcal infections in non-HIV infected patients. J Fungi 5(3): 71.
- Pasquier E, Kunda J, De BP, Loyse A, Temfack E, et al. (2018) Long-term mortality and disability in cryptococcal meningitis: A systematic literature review. Clin Infect Dis 66(7): 1122-1132.
- Chen M, Chen C, Yang Q, Zhan R (2020) Candida meningitis in neurosurgical patients: A single-institute study of nine cases over 7 years. Epidemiol Infect 148: e148.
- Anand G, Lahariya R, Priyadarshi K (2026) The rise of WHO-priority pathogens in central line associated bloodstream infection: Challenging the AWaRe paradigm in critical care. World J Microbiol Biotechnol 42(2): 55.
- Gupta AK, Mann A, Polla RS, Wang T (2024) Navigating fungal infections and antifungal stewardship: Drug resistance, susceptibility testing, therapeutic drug monitoring and future directions. Ital J Dermatol Venereol 159(2): 105-117.
- Hare D, Coates C, Kelly M, Cottrell E, Connolly E, et al. (2020) Antifungal stewardship in critical care: Implementing a diagnostics-driven care pathway in the management of invasive candidiasis. Infect Prev Pract 2(2): 100047.
- Ashbee HR, Barnes RA, Johnson EM, Richardson MD, Gorton R, et al. (2014) Therapeutic drug monitoring (TDM) of antifungal agents: Guidelines from the British society for medical mycology. J Antimicrob Chemother 69(5): 1162-1176.
- Sachdev J, Gourav S, Xess I, Soneja M, Punjadath S, et al. (2025) Impact of an institutional antifungal stewardship program on antifungal usage and outcomes in patients with invasive fungal infections. Med Mycol 63(2): myaf003.
© 2026 Rijhul Lahariya. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






