- Submissions

Full Text
Trends in Telemedicine & E-health
The Future of Organoids Engineering: Biomaterials, Mechanobiology, Personalized Therapeutics
Zarif Bin Akhtar1* and Ahmed Tajbiul Rawol2
1Department of Computing, Institute of Electrical and Electronics Engineers, USA
2Department of Computer Science, American International University-Bangladesh, Bangladesh
*Corresponding author:Zarif Bin Akhtar, Department of Computing, Institute of Electrical and Electronics Engineers, USA
Submission: December 11, 2025;Published: March 23, 2026

ISSN: 2689-2707 Volume 6 Issue 4
Abstract
Organoid engineering has emerged as a powerful platform for disease modeling, drug discovery and personalized medicine by enabling three-dimensional in-vitro systems that closely recapitulate human tissue architecture and function. This investigation critically examines recent advances in organoid bioengineering with a focused emphasis on biomaterials, mechanobiological regulation and micro engineered platforms that enhance organoid maturation, functionality and reproducibility. Particular attention is given to the role of mechanical cues, ultrasound-based stimulation and acoustofluidic technologies in directing cellular organization and mechan transduction within organoid systems. The integration of organoids with organ-on-a-chip technologies and embedded biosensors is discussed as a strategy for real-time functional monitoring and improved experimental control, addressing key limitations related to variability and scalability. Additionally, current challenges in large-scale organoid manufacturing and clinical translation are analyzed, alongside emerging solutions aimed at standardization and personalized therapeutic applications. By consolidating advances across biomaterials science, mechanobiology, and microfluidics, this review highlights how engineered organoid platforms are reshaping precision medicine and provides a structured perspective on future directions in organoidbased biomedical research..
Keywords:Artificial intelligence; Biomedical engineering; Deep learning; Machine learning; Mechanobiological engineering; Organoids engineering
Introduction
Organoid engineering has rapidly evolved into a cornerstone of modern biomedical research, offering three-dimensional (3D) in-vitro tissue models that more accurately recapitulate human organ architecture and function than conventional two-dimensional cultures. These systems have become indispensable for disease modeling, drug discovery and the development of personalized therapeutic strategies, bridging a critical gap between simplified cell cultures and complex in-vivo models [1-3]. As organoid research continues to expand, there is a growing need for engineering-driven approaches that enhance reproducibility, functional maturity and translational relevance. Recent progress in this field has been driven by the convergence of biomaterials science, mechanobiology and microengineering technologies. Biomaterial-based Extra-Cellular Matrix (ECM) systems provide structural and biochemical cues that guide organoid self-organization, while mechanobiological regulation introduces controlled physical forces that influence cellular differentiation, tissue patterning and maturation [4-6]. Techniques such as microfluidic confinement, acoustofluidic and ultrasound-mediated stimulation have enabled precise manipulation of mechanical signaling pathways, offering new opportunities to regulate organoid development beyond biochemical control alone. Despite these advances, several limitations continue to hinder the widespread application of organoids in translational and clinical settings. Organoid cultures often exhibit substantial variability in size, architecture and functional output, limiting reproducibility and scalability [7-9]. In addition, the absence of vascularization, restricted nutrient transport and limited access to real-time functional readouts constrain long-term culture and physiological relevance. Addressing these challenges requires integrated engineering solutions that combine controlled microenvironments with continuous monitoring and standardized fabrication strategies.
Organoids-on-a-chip platforms have emerged as a promising response to these challenges by integrating organoid cultures with micro engineered systems capable of regulating flow, mechanical stimulation and biochemical gradients [10-12]. The incorporation of embedded biosensors further enables real-time, label-free monitoring of organoid behavior, improving experimental precision and facilitating dynamic assessment of drug responses and disease phenotypes. Such platforms offer a pathway toward scalable, reproducible, and patient-specific organoid models suitable for precision medicine applications. This exploration presents a focused evaluation of engineering strategies that are shaping the future of organoid technologies, with particular emphasis on biomaterials design, mechanobiological modulation, and organoid-on-a-chip integration. By synthesizing recent advances and identifying current limitations, this work aims to provide a structured framework for understanding how engineered organoid systems can be optimized for disease modeling, drug development and personalized therapeutics.
Methods and Experimental Analysis
This investigative exploration adopts a step-by-step structured methodological framework to analyze and synthesize recent engineering strategies in organoid research, with an emphasis on biomaterials, mechanobiological regulation and micro engineered platforms for personalized medicine. Rather than reporting new experimental data with publicly available datasets, this investigation mainly outlines the conceptual and analytical approaches used to evaluate current methodologies, identify technological limitations, and assess translational potential across organoid systems. With the emergence of Generative Artificial Intelligence (GAI) acceleration, the domains of biomedical engineering can progressively advance in the upcoming years..
Cell sources and culture platforms
The analysis considers organoid models derived from wellestablished pluripotent stem cells, adult stem cells and patientderived cells, as reported in the literature. Particular emphasis is placed on culture systems designed to replicate tissue-specific microenvironments through defined media formulations and Extra-Cellular Matrix (ECM) mimetics. Biomaterial-based scaffolds, including natural and synthetic hydrogels, are evaluated for their role in supporting organoid structural integrity, cellular differentiation, and functional maturation.
Mechanobiological regulation and characterization
Mechanobiological modulation is examined as a key regulatory factor influencing organoid development. The reviewed methodologies include the application of controlled mechanical forces using microfluidic devices, stretchable substrates and bioreactor-based systems to simulate physiologically relevant conditions. Advanced imaging and biomechanical characterization techniques described in prior studies are analyzed to assess how mechanical cues affect tissue organization, mechan transduction pathways and maturation dynamics within organoids.
Ultrasound stimulation and acoustofluidic approaches
Ultrasound-based stimulation and acoustofluidic technologies are reviewed as emerging tools for noninvasive control of organoid microenvironments. The analysis focuses on approaches that leverage ultrasound to activate mechanosensitive ion channels and to manipulate cellular positioning within three-dimensional cultures. Acoustofluidic systems enabling precise cell patterning, microenvironmental control and integration with sensing elements are evaluated for their potential to enhance reproducibility and scalability.
Multi-organoid systems and scalable manufacturing
Strategies for engineering interconnected multi-organoid systems are examined with respect to vascularization, interorganoid communication and functional coupling. The framework reviews scalable production methods, including automated bioreactor platforms and high throughput micro engineered systems, that aim to improve consistency across organoid batches. Emphasis is placed on approaches that balance throughput with the preservation of biological fidelity.
Validation criteria and ethical considerations
Validation across reviewed studies is assessed using reported structural, functional and molecular benchmarks that define organoid fidelity. Criteria such as reproducibility, responsiveness to mechanical and biochemical stimuli and relevance to human physiology are considered central to translational applicability. Ethical considerations, including the responsible use of stem cellderived materials and compliance with regulatory standards, are discussed in the context of advancing organoid-based technologies toward clinical and personalized medicine applications.
Background Research and Available Knowledge
Mechanobiology as a foundation for organoid engineering
Mechanobiology examines how physical forces and mechanical properties regulate cellular behavior, tissue organization and disease progression. In native tissues, cells continuously experience mechanical cues such as tension, compression, shear stress and matrix stiffness, all of which influence differentiation, migration and functional maturation. Unlike purely biochemical models, mechanobiological frameworks emphasize that alterations in cellular mechanics and Extra-Cellular Matrix (ECM) architecture play central roles in development, regeneration and pathological states including fibrosis, cardiovascular disease and cancer. Within organoid systems, mechan transduction-the process by which cells convert mechanical stimuli into biochemical signals-has emerged as a critical determinant of tissue self-organization [1-11]. Mechanosensitive pathways involving focal adhesion complexes, cytoskeletal remodeling and nuclear deformation regulate gene expression and spatial patterning. These processes are particularly relevant for replicating morphogenetic events observed during embryonic development, where mechanical forces act alongside biochemical gradients to guide tissue formation.
Mechanical cues, ultrasound and acoustofluidic technologies
Recent advances have expanded the toolkit for delivering controlled mechanical stimuli to organoids. Ultrasound-based approaches enable noninvasive modulation of mechanosensitive ion channels and intracellular signaling pathways, offering precise spatiotemporal control without direct physical contact [12-22]. Similarly, acoustofluidic systems leverage acoustic waves to manipulate cells, control microenvironmental organization and enhance reproducibility in three-dimensional cultures. These technologies provide distinct advantages over static culture systems by enabling dynamic regulation of mechanical forces that more closely resemble in-vivo conditions. The ability to fine-tune mechanical stimulation has shown promise in improving organoid maturation, tissue architecture and functional stability, while also supporting scalable and automated culture workflows.
Organ-on-a-chip and micro engineered platforms
Organ-on-a-chip technologies integrate microfluidics, biomaterials and living cells to recreate physiologically relevant microenvironments. When combined with organoid cultures, these platforms offer enhanced control overflow, nutrient delivery, mechanical stress and biochemical gradients [23-33]. Such integration addresses key limitations of conventional organoid systems, including variability, limited lifespan and restricted functional readouts. Micro engineered organoid-on-a-chip platforms further enable the incorporation of biosensors for realtime monitoring of metabolic activity, barrier function and tissue responses to external stimuli. These systems are particularly valuable for drug screening and disease modeling, as they provide continuous, quantitative data while maintaining biological relevance.
Biomaterials and extracellular matrix engineering
Biomaterials play a central role in organoid engineering by providing structural support and instructive cues that regulate cell fate and tissue organization. Advances in synthetic and hybrid ECM materials have enabled more precise control over stiffness, viscoelasticity and biochemical composition, reducing batchto- batch variability associated with naturally derived matrices. Stimuli-responsive and “active” biomaterials further extend these capabilities by dynamically altering mechanical properties in response to external triggers such as light, magnetic fields, or acoustic stimulation. These materials facilitate the study of temporal mechan transduction and mechanical memory, offering new insights into how cells adapt to changing physical environments during development and disease progression.
Challenges and knowledge gaps
Despite significant progress, several challenges remain in translating mechanobiological insights into robust organoid platforms. Reproducing native tissue complexity requires precise coordination of mechanical, biochemical and cellular interactions, which remain difficult to standardize across laboratories. Variability in organoid size, structure and functional output continues to limit reproducibility, particularly in high-throughput and personalized medicine applications. Furthermore, the longterm effects of dynamic mechanical stimulation and the integration of multiorgan interactions are not yet fully understood. Addressing these gaps will require interdisciplinary efforts combining biomaterials engineering, microfabrication, computational modeling and systems biology to develop standardized, scalable and physiologically relevant organoid systems.
Engineering Advanced Organoid Systems
Biomaterials, tissue engineering and stem cell integration
Advances in organoid bioengineering have been driven by the integration of tissue engineering principles, stem cell technologies and engineered microenvironments designed to more closely replicate native tissue structure and function. Modern organoid systems extend beyond self-organizing cell aggregates, incorporating biomaterials, mechanical regulation and scalable fabrication strategies to enhance reproducibility, maturation and translational relevance.
Bioengineering strategies for advanced organoid development
Recent progress in three-Dimensional (3D) culture technologies has enabled the generation of organoids with increased structural and functional complexity. Engineering approaches now focus on controlling both biochemical and physical determinants of organoid development, including Extra-Cellular Matrix (ECM) composition, stiffness and spatial organization. Synthetic and hybrid biomaterials have emerged as effective alternatives to biologically derived matrices, offering tunable mechanical properties and improved batch-to-batch consistency. Bio fabrication techniques such as micropatterning, 3D bioprinting and microfluidic confinement enable precise control over cell-cell and cell-matrix interactions. These methods support organ-specific patterning and facilitate the formation of organized tissue architectures, improving the fidelity of organoid models for disease modeling and drug screening. Dynamic culture systems further enhance these outcomes by introducing controlled mechanical and fluidic stimuli that better mimic physiological conditions.
Tissue engineering contributions to organoid maturation
Tissue engineering principles play a critical role in overcoming key limitations of conventional organoid cultures, particularly related to nutrient diffusion, structural stability and long-term viability. Biomaterial scaffolds derived from biodegradable polymers, ceramics and composite systems provide mechanical support while allowing gradual remodeling during tissue maturation. Advances in scaffold porosity, surface functionalization and degradation kinetics have improved cell survival and tissue integration. Stem cell–based organoid systems benefit significantly from engineered microenvironments that regulate lineage specification and functional maturation. Mechanical cues such as matrix stiffness and cyclic strain have been shown to influence stem cell fate decisions, reinforcing the importance of mechanobiological regulation in tissue development. The incorporation of angiogenic cues and vascular-mimetic structures further enhances nutrient transport and functional longevity, addressing one of the major bottlenecks in scaling organoid systems.
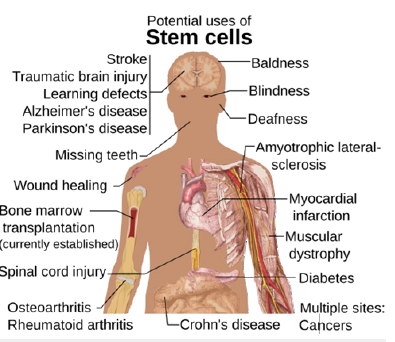
Stem cells as a core component of engineered organoids
Stem cells remain central to organoid technology due to their self-renewal capacity and differentiation potential. Adult stem cells, pluripotent stem cells and patient-derived Induced Pluripotent Stem Cells (iPSCs) enable the generation of tissue-specific organoids for personalized medicine applications. Engineered culture systems improve the expansion and differentiation of these cells by providing defined mechanical and biochemical environments that reduce variability. Stem cell-derived organoids have demonstrated utility in modeling genetic diseases, cancer progression and tissue regeneration. In regenerative medicine, organoid-based strategies offer a renewable source of functional tissue for transplantation and therapeutic testing. However, translating these systems into clinical applications requires stringent control over differentiation efficiency, genomic stability and functional consistency-challenges that are increasingly addressed through engineered culture platforms and standardized protocols.
Clinical translation, challenges and ethical considerations
Despite rapid advancements, several challenges remain in translating engineered organoid systems into clinical and industrial settings. Variability in organoid formation, incomplete cellular composition and limited maturation continue to constrain reproducibility. Additionally, ethical concerns surrounding stem cell sourcing and the proliferation of unregulated stem cell therapies underscore the need for rigorous oversight and regulatory alignment. Engineering-driven solutions, including automated culture systems, integrated biosensing and standardized biomaterial platforms-are critical for addressing these limitations. By combining stem cell biology with tissue engineering and bioengineering technologies, advanced organoid systems are moving toward greater scalability, reliability and clinical relevance. To provide further information concerning the matters Figures 1-3 provides illustrative responses to the prospects.
Figure 1:Insights towards advanced organoids.
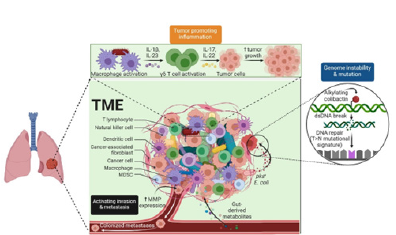
Figure 2:Advances towards tissue engineering.
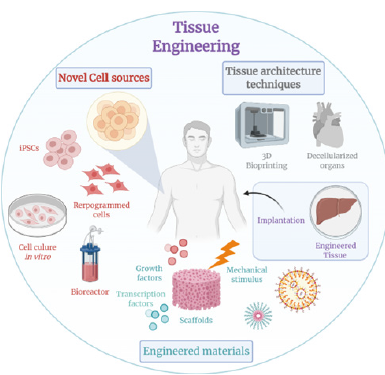
Figure 3:Potential uses of stem cells.
Mechanobiology of Tumor Progression
ECM dynamics, external forces, active biomaterial models
Mechanical cues within the tumor microenvironment play a central role in regulating cancer initiation, progression and metastasis. Beyond genetic and biochemical alterations, tumor cells respond dynamically to physical factors such as Extra-Cellular Matrix (ECM) stiffness, topography, interstitial fluid pressure and externally applied forces. These mechanobiological signals influence cellular proliferation, migration and survival, making them critical considerations in the design of physiologically relevant organoid-based cancer models.
ECM mechanics and tumor cell behavior
The biomechanical properties of the ECM are key regulators of tumor growth and tissue invasion. Increased matrix stiffness, often resulting from collagen cross-linking and ECM remodeling, promotes malignant transformation and enhances metastatic potential. Tumor cells exhibit differential sensitivity to substrate rigidity, with some populations displaying rigidity-dependent proliferation while others maintain growth across a broad mechanical range. These responses are mediated through focal adhesion complexes, cytoskeletal contractility and mechanosensitive signaling pathways that transmit external forces to the nucleus. Topographical features of the ECM further regulate cancer cell behavior by directing cell polarity, migration patterns and invasion pathways. Aligned collagen fibers, for example, facilitate directional tumor cell migration, while micro and nanoscale surface features modulate adhesion and proliferation. Understanding how ECM architecture integrates with mechanical stiffness is essential for reproducing tumor-specific microenvironments in engineered organoid systems.
External mechanical forces and mechan transduction
In addition to matrix-derived cues, externally applied mechanical forces such as compression, stretching, and shear stress significantly influence tumor progression. Elevated interstitial fluid pressure in solid tumors generates compressive forces that can activate proliferative signaling pathways and alter tissue organization. Mechanical stretching has been shown to modulate mechan transduction regulators, including YAP/TAZ, enabling cancer cells to bypass growth-inhibitory signals and re-enter the cell cycle. These mechan transudative processes highlight the importance of dynamic mechanical regulation in cancer biology. Importantly, cancer cells can develop mechanical memory, whereby prior exposure to specific mechanical environments alters future responses to physical stimuli. Capturing these temporal and adaptive behaviors requires experimental platforms capable of controlled and repeatable mechanical modulation.
Active biomaterials for modeling tumor mechanobiology
Active biomaterials offer powerful tools for recreating dynamic tumor microenvironments by enabling precise control over mechanical properties and external force application. Stimuliresponsive hydrogels and mechanically tunable matrices allow researchers to modulate stiffness, viscoelasticity and stress profiles in real time. These systems provide a means to study how temporal changes in mechanical cues influence tumor cell behavior, differentiation and therapeutic response. Mechanosensitive pathways involving integrins, cytoskeletal networks and ion channels such as Piezo proteins are central to these interactions. Active biomaterials facilitate the investigation of these pathways by decoupling matrix stiffness from force generation, allowing systematic analysis of mechan transduction mechanisms. When integrated with organoid cultures, such platforms enable more physiologically relevant cancer models that reflect the dynamic nature of tumor progression.
Implications for organoid-based cancer models
Figure 4:Insights towards tumor growth & proliferation process.
Figure 5:The process of ECM mechan sensing & external forces.
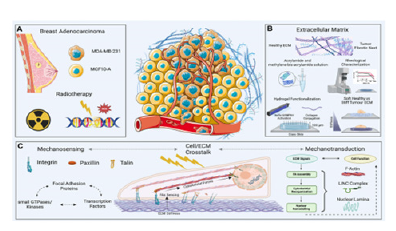
The integration of mechanobiology and active biomaterial systems into tumor organoid platforms enhances their relevance for disease modeling and drug testing. By incorporating ECM remodeling, external mechanical forces and dynamic mechanical modulation, engineered organoids can better replicate in-vivo tumor behavior. These advances improve the predictive power of organoid-based cancer models and support the development of mechanomedicine strategies that target biomechanical vulnerabilities in tumors. Despite these advances, challenges remain in standardizing mechanical parameters across experimental platforms and translating mechanobiological insights into clinically actionable therapies. Continued interdisciplinary efforts combining biomaterials engineering, microfabrication and computational modeling are essential for refining tumor organoid systems and advancing their application in precision oncology. To better understand the matters of perspectives Figures 4-6 sheds insight into the relationship between the retrospectives.
Figure 6:The process of ECM mechan sensing & external forces.
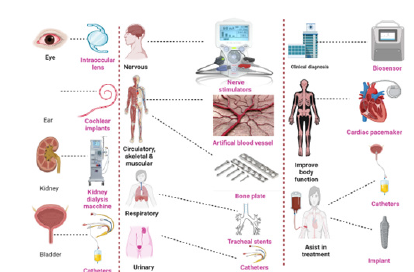
Organoid-on-a-Chip Systems: Current Limitations and Future Directions
Organoid-on-a-chip technologies represent a convergence of organoid biology and microengineering, offering enhanced control over the physical and biochemical microenvironment of three-dimensional tissue models. While these systems have significantly advanced vitro modeling capabilities, several technical and biological challenges continue to limit their reproducibility, scalability and translational impact.
Biological and structural limitations of current organoid systems
Despite their physiological relevance, many organoid models lack key features of native tissues, including vascularization, immune components, stromal interactions and long-term functional stability. The absence of perusable vasculature restricts nutrient and oxygen transport, leading to necrotic cores and limiting organoid size and lifespan. In addition, organoids frequently remain developmentally immature, often resembling fetal rather than adult tissue phenotypes. Structural heterogeneity is another persistent challenge. Variability in organoid size, cellular composition and spatial organization arises from stochastic selforganization processes and inconsistent culture conditions. This heterogeneity complicates quantitative analysis and undermines reproducibility, particularly in high-throughput drug screening and personalized medicine applications.
Engineering constraints and platform-level challenges
From an engineering perspective, integrating organoids into microfluidic platforms introduces additional complexities. The design of microchannels must balance physiological relevance with manufacturability and robustness, while commonly used materials such as Poly Di-Methyl Siloxane (PDMS) present challenges related to small-molecule absorption and long-term stability. Standardizing culture media and flow conditions for multi-organoid or multiorganoid systems remains difficult due to competing metabolic and environmental requirements across different types of tissue. Moreover, functional assessment of organoid-on-a-chip systems is often limited by reliance on optical imaging, which provides restricted insight into dynamic physiological processes. The integration of biosensors for real-time monitoring of metabolic activity, barrier integrity and biochemical signaling is still in early stages and requires further optimization for reliability and scalability.
Strategies to reduce variability and enhance reproducibility
Micro engineered platforms offer promising solutions to many of these limitations by enabling precise control over organoid formation, culture conditions and mechanical stimulation. Automated fluid handling, digital microfluidics and high-density microfabricated arrays have demonstrated improved consistency in organoid size and morphology. Size-selective trapping and controlled perfusion further enhances uniformity and supports long-term culture. Advances in defined biomaterials and synthetic extracellular matrices also contribute to reduced variability by minimizing batch-to-batch differences inherent in biologically derived matrices. When combined with standardized differentiation protocols, these approaches support more reproducible and scalable organoid production.
Toward multiorgan integration and clinical translation
Future directions in organoid-on-a-chip research emphasize the integration of multiple organ systems to model interorgan communication and systemic drug responses. Multiorgan platforms connected through controlled microfluidic circulation enable physiologically relevant crosstalk while maintaining compartmentalization. However, achieving functional synchronization across tissues and establishing unified culture conditions remain significant challenges. Clinical translation will require not only technical refinement but also regulatory alignment, cost-effective manufacturing and robust validation standards.
The integration of computational modeling, artificial intelligence and machine learning offers promising avenues for optimizing system design, automating data analysis and improving predictive accuracy. By addressing current biological and engineering limitations through interdisciplinary innovation, organoid-on-achip systems are positioned to play a transformative role in disease modeling, drug development and precision medicine. Continued efforts toward standardization, scalability and functional validation will be essential for realizing their full translational potential.
Result and Finding
This exploration presents a synthesis of key insights derived from the integrated analysis of recent advances in organoid engineering, organoid-on-a-chip technologies and mechanobiological regulation. Rather than reporting original experimental outcomes, the findings summarize consistent trends, technological impacts and translational implications identified across the evaluated information. To provide further information concerning the matters of perspectives Figures 7-12 along with Table 1 gives an overview summary of the findings concerning the research investigative explorations.
Figure 7:Current organoid systems and their associated approaches.
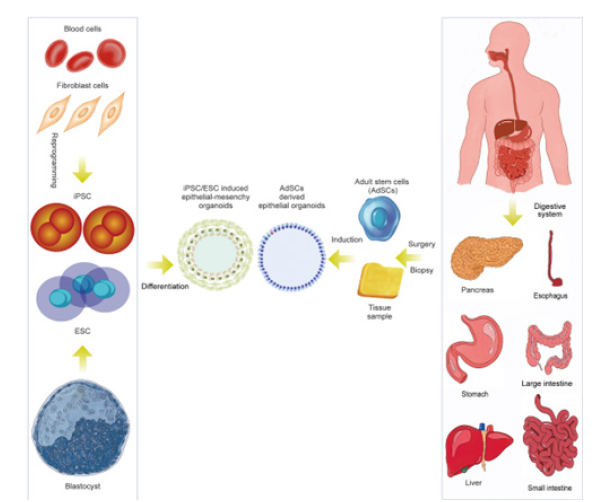
Figure 8:A summarization of the findings involved concerning the research explorations [1].
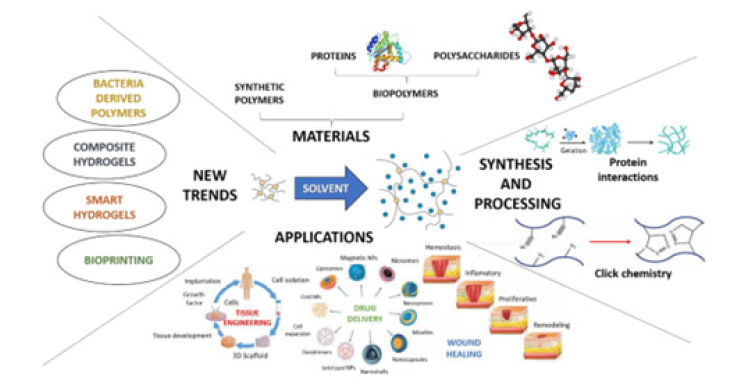
Figure 9:A summarization of the findings involved concerning the research explorations [2].
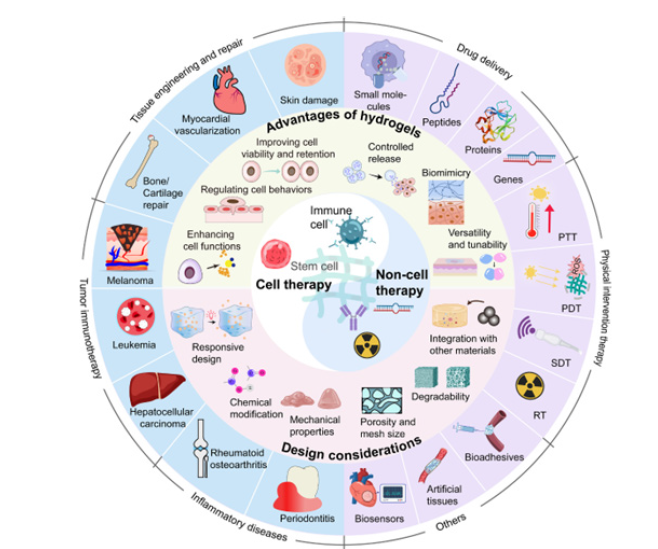
Figure 10:A summarization of the findings involved concerning the research explorations [3].
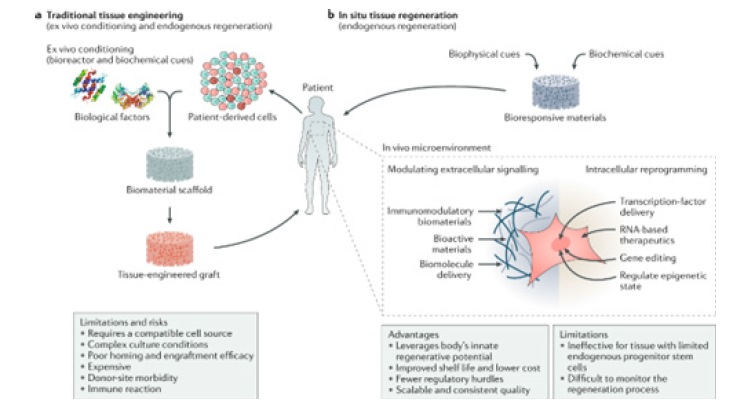
Figure 11:A summarization of the findings involved concerning the research explorations [4].
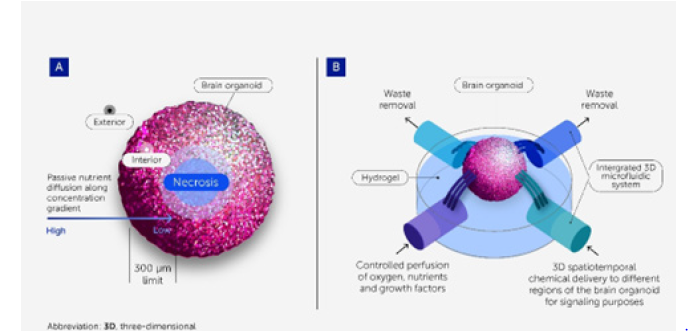
Figure 12:A summarization of the findings involved concerning the research explorations [5].
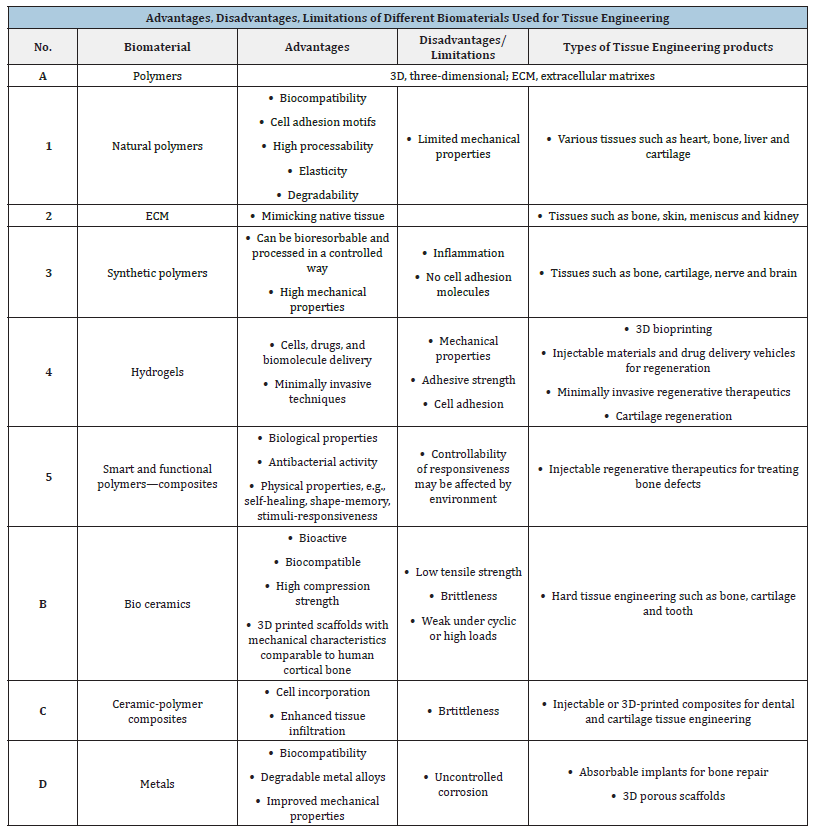
Table 1:The different types of biomaterials used towards tissue engineering.
Enhanced physiological relevance through engineered microenvironments
A consistent finding across organoid studies is that engineered microenvironments significantly improve structural organization and functional maturity. The incorporation of defined biomaterials controlled mechanical cues, and dynamic perfusion enhances tissue polarization, cellular differentiation and long-term viability. Organoid-on-a-chip platforms, in particular, demonstrate improved physiological relevance by regulating flow, nutrient delivery and mechanical stimulation, thereby addressing limitations observed in static culture systems.
Improved reproducibility and experimental control
Micro engineered culture systems contribute to reduced variability in organoid size, morphology and functional output. Techniques such as automated fluid handling, size-controlled trapping and standardized biomaterial matrices promote greater consistency across experimental batches. These advances support more reliable comparative analyses in drug screening and disease modeling, a critical requirement for translational and industrial applications.
Integration of biosensing and real-time functional monitoring
The integration of biosensors within organoid-on-a-chip platforms enables continuous, label-free monitoring of tissue function. Electrochemical and optical sensing approaches provide real-time insights into metabolic activity, barrier integrity and cellular responses to pharmacological stimuli. These capabilities improve data richness while reducing reliance on endpoint assays, supporting dynamic assessment of treatment effects and disease progression.
Advances in cancer modeling and mechanobiological insights
Mechanobiology-informed organoid systems offer enhanced modeling of tumor progression by incorporating extracellular matrix remodeling, mechanical stress and dynamic force application. Such systems capture key aspects of tumor behavior, including altered mechan transduction, invasive phenotypes and adaptive responses to mechanical cues. These findings highlight the importance of mechanical regulation in cancer organoid models and suggest new opportunities for mechanomedicine-based therapeutic strategies.
Scalability and personalized medicine applications
Scalable organoid production remains a central focus of current research. Automated bioreactors and high-throughput microfluidic platforms enable the expansion of patient-derived organoids while maintaining biological fidelity. These systems support personalized medicine by facilitating individualized drug response profiling and disease modeling. However, achieving widespread clinical adoption will require further standardization, validation and regulatory alignment.
Discussions and Future Directions
The convergence of organoid biology with bioengineering, mechanobiology, and microfabrication has significantly advanced the development of physiologically relevant in-vitro models. While organoid technologies have demonstrated clear advantages over traditional culture systems, their translational impact depends on overcoming persistent challenges related to reproducibility, scalability and functional maturation. A central theme emerging from this review is the importance of engineered microenvironments in guiding organoid development. Biomaterials with well-defined mechanical and biochemical properties, combined with dynamic mechanical regulation, enable more consistent tissue organization and enhance functional fidelity. Mechanobiological control, in particular, provides a critical layer of regulation that complements biochemical signaling and allows more accurate modeling of developmental processes and disease states. Despite these advances, clinical translation remains constrained by technical and logistical barriers. Long-term viability, vascular integration and multiorgan coordination continue to limit the physiological relevance of current systems. In addition, variability across platforms and laboratories highlights the need for standardized protocols, defined matrices and reproducible fabrication strategies. Addressing these challenges will require coordinated efforts across materials science, bioengineering and stem cell biology.
Looking forward, the integration of organoid-on-a-chip platforms with computational modeling, artificial intelligence and machine learning represents a promising direction for accelerating discovery and improving predictive power. Data-driven optimization of culture conditions, automated image and signal analysis, and in silico modeling of mechanobiological processes can enhance experimental efficiency while reducing costs. These approaches are particularly relevant for personalized medicine, where patient-specific organoids can be used to guide therapeutic decision-making. Future research should also prioritize regulatory alignment and ethical oversight to support clinical adoption. Establishing clear validation benchmarks, safety standards and quality control metrics will be essential for translating organoidbased systems into reliable tools for drug development and regenerative therapies. As interdisciplinary collaboration continues to strengthen, engineered organoid platforms are poised to play an increasingly important role in precision medicine, disease modeling and the reduction of animal-based testing.
Conclusion
Organoid engineering has progressed from self-organizing three-dimensional cultures to highly engineered systems that integrate biomaterials, mechanobiology and microfabrication to better replicate human tissue structure and function. This review synthesizes recent advances demonstrating how controlled microenvironments, dynamic mechanical regulation and organoidon- a-chip technologies improve reproducibility, functional maturity and translational relevance. A key insight emerging from this analysis is that mechanical cues are as critical as biochemical signals in directing organoid development and disease modeling. Engineered biomaterials and active substrates enable precise regulation of extracellular matrix properties and external forces, while micro engineered platforms facilitate real-time monitoring and standardized culture conditions. Together, these approaches address long-standing limitations related to variability, scalability and physiological fidelity.
Despite substantial progress, challenges remain in achieving long-term stability, multiorgan integration, and clinical translation. Overcoming these barriers will require standardized fabrication protocols, defined biomaterial systems and rigorous validation frameworks. The integration of computational modeling, artificial intelligence and high throughput microengineering is expected to further enhance predictive accuracy and accelerate personalized medicine applications. The continued convergence of engineering principles with organoid biology is reshaping in-vitro modeling and precision therapeutics. As these technologies mature, engineered organoid platforms are poised to become essential tools in drug development, disease modeling and regenerative medicine, offering robust alternatives to traditional experimental models.
Supplementary Information
The various original data sources some of which are not all publicly available, because they contain various types of private information. The available platform provided data sources that support the research exploration findings and information of the research investigations are referenced where appropriate.
Acknowledgments
The author would like to acknowledge and thank the GOOGLE deep mind research with its associated pre-prints access platforms. This research exploration was investigated under the platform provided by GOOGLE Deep Mind which is under the support of GOOGLE Research and the GOOGLE Research Publications within the GOOGLE Gemini platform. Using their provided platform of datasets and database associated files with digital software layouts consisting of free web access to a large collection of recorded models that are found within research access and its related open-source software distributions which is the implementation for the proposed research exploration that was undergone and set in motion. There are many data sources, some of which are resourced and retrieved from a wide variety of GOOGLE service domains as well. All the data sources which have been included and retrieved for this research are identified, mentioned and referenced where appropriate. ChatGPT was used in order to check for any grammatical errors concerning the whole manuscript.
Declarations
Funding
No Funding was provided for the conduction concerning this research.
Conflict of interest/competing interests
There is no Conflict of Interest or any type of Competing Interests for this research.
Ethics approval
The author declares no competing interests in this research.
Consent to participate
The author has read, approved the manuscript and have agreed to its publication.
Consent for publication
The author has read, approved the manuscript and have agreed to its publication.
Availability of data and materials
The various original data sources some of which are not all publicly available, because they contain various types of private information. The available platform provided data sources that support the exploration findings and information of the research investigations are referenced where appropriate.
Code availability
Mentioned in detail within the Acknowledgements section.
Author’s contributions
Described in detail within the Acknowledgements section.
References
- Zarif BA (2025) Artificial intelligence within medical diagnostics: A multi-disease perspective. Artificial Intelligence in Health 2(3): 44-62.
- Zhong Z, Yang S, Chen F, Du ZH, Liu N, et al. (2025) Molecular engineering strategies of cationic oligo (p-phenylene ethynylene)s for enhancing sensitive discrimination of multiple hazardous explosives. Chemical Engineering Journal 510: 161558.
- Akhtar ZB (2024) The design approach of an artificial intelligent (AI) medical system based on electronical health records (EHR) and priority segmentations. J Eng 2024(4): e12381.
- Zhan J, Wang S, Li X, Zhang J (2025) Molecular engineering of functional DNA molecules toward point-of-care diagnostic devices. Chemical Communications 61(22): 4316-4338.
- Rani A, Zafar F, Hussain R, Zafar WUI, Khanum A, et al. (2024) Molecular engineering of benzothiadiazole core based non-fullerene acceptors to tune the optoelectronic properties of perovskite solar cells. Computational and Theoretical Chemistry 1237: 114637.
- Lin N, Tsuji M, Bruzzese I, Chen A, Vrionides M, et al. (2024) Molecular engineering of emissive molecular qubits based on spin-correlated radical pairs. Journal of the American Chemical Society 147(13): 11062-11071.
- Cheng Y, Wang T, Zhu H, Hu X, Mi J, et al. (2025) Molecular engineering of amino acid crystals with enhanced piezoelectric performance for biodegradable sensors. Angewandte Chemie 64(15): e202500334.
- Liu Y, Song Y, Zhu ZH, Ji C, Li J, et al. (2025) Twisted‐planar molecular engineering with sonication‐induced J‐aggregation to design near‐infrared J‐aggregates for enhanced phototherapy. Angew Chem Int Ed Engl 137(7): e202419428.
- Sun W, Xue B, Fan Q, Tao R, Wang C, et al. (2020) Molecular engineering of metal coordination interactions for strong, tough, and fast-recovery hydrogels. Science Advances 6(16): eaaz9531.
- Lv G, Zhu J (2025) Intrinsic thermal conductivity of molecular engineered polymer. Advanced Functional Materials 36(6): 2420708.
- Wan Q, Zhang R, Zhuang Z, Li Y, Huang Y, et al. (2020) Molecular engineering to boost AIE‐active free radical photo generators and enable high‐performance photodynamic therapy under hypoxia. Advanced Functional Materials 30(39): 2002057.
- Yang R, Ai D, Fan S, Zhang W, Yang X, et al. (2025) Aramid dielectric co-polymer: From molecular engineering to roll-to-roll scalability for high-temperature capacitive energy storage. Energy & Environmental Science 18(8): 3718-3729.
- Huang Y, Liu T, Huang Q, Wang Y (2024) From organ-on-a-chip to human-on-a-chip: A review of research progress and latest applications. ACS Sensors 9(7): 3466-3488.
- Sun S (2024) Research of organ-on-a-chip and its application. E3S Web of Conferences 553: 05012.
- Sadeghzade S, Hooshiar MH, Akbari H, Tajer MHM, Sahneh KK, et al. (2024) Recent advances in organ-on-a-chip models: How precision engineering integrates cutting edge technologies in fabrication and characterization. Applied Materials Today 38: 102231.
- Zhao Y, Landau S, Okhovatian S, Liu C, Lu RXZ, et al. (2024) Integrating organoids and organ-on-a-chip devices. Nature Reviews Bioengineering 2(7): 588-608.
- He C, Lu F, Liu Y, Lei Y, Wang X, et al. (2024) Emergent trends in organ-on-a-chip applications for investigating metastasis within tumor microenvironment: A comprehensive bibliometric analysis. Heliyon 10(1): e23504.
- Song SH, Jeong S (2024) State-of-the-art in high throughput organ-on-chip for biotechnology and pharmaceuticals. Clin Exp Reprod Med 52(2):114-124.
- Sean G, Banes AJ, Gangaraju R (2024) Organoids and tissue/organ chips. Stem Cell Research & Therapy 15(1): 241.
- Li X, Zhu H, Gu B, Yao C, Gu Y, et al. (2024) Advancing intelligent organ‐on‐a‐chip systems with comprehensive in situ bioanalysis. Advanced Materials 36(18): 2305268.
- Nasiri R, Zhu Y, de BNR (2024) Microfluidics and organ-on-a-chip for disease modelling and drug screening. Biosensors 14(2): 86.
- Gunasekaran BM, Srinivasan S, Ezhilan M, Rajagopal V, Nesakumar N (2024) Advancements in organ‐on‐a‐chip systems: Materials, characterization, and applications. Chemistry Select 9(40): e202403611.
- Huang X, Lou Y, Duan Y, Liu H, Tian J, et al. (2024) Biomaterial scaffolds in maxillofacial bone tissue engineering: A review of recent advances. Bioactive Materials 33: 129-156.
- Akhtar ZB, Stany VR (2020) The design approach of an artificial human brain in digitized formulation based on machine learning and neural mapping. International Conference for Emerging Technology pp. 1-7.
- Bagherpour R, Bagherpour G, Mohammadi P (2025) Application of artificial intelligence in tissue engineering. Tissue Engineering Part B: Reviews 31(1): 31-43.
- Bauso LV, La FV, Longo C, Calabrese G (2024) Bone tissue engineering and nanotechnology: A promising combination for bone regeneration. Biology 13(4): 237.
- Angolkar M, Paramshetti S, Gahtani RM, Al SM, Hani U, et al. (2024) Pioneering a paradigm shift in tissue engineering and regeneration with polysaccharides and proteins-based scaffolds: A comprehensive review. International Journal of Biological Macromolecules 265: 130643.
- Akhtar ZB, Rozario VS (2025) AI perspectives within computational neuroscience: EEG integrations and the human brain. Artificial Intelligence and Applications 3(2): 145-160.
- Chen A, Wang W, Mao Z, He Y, Chen S, et al. (2024) Multimaterial 3D and 4D bioprinting of heterogenous constructs for tissue engineering. Advanced Materials 36(34): e2307686.
- Gou Y, Huang Y, Luo W, Li Y, Zhao P, et al. (2024) Adipose-derived mesenchymal stem cells (MSCs) are a superior cell source for bone tissue engineering. Bioactive Materials 34: 51-63.
- Xu C, Liu Z, Chen X, Gao Y, Wang W, et al. (2024) Bone tissue engineering scaffold materials: Fundamentals, advances, and challenges. Chinese Chemical Letters 35(2): 109197.
- Jafari A, Vahid NS, Kaviani M, Saleh W, Wong N, et al. (2024) Formulation and evaluation of PVA/gelatin/carrageenan inks for 3d printing and development of tissue‐engineered heart valves. Advanced Functional Materials 34(7): 2305188.
- Shan BH, Wu FG (2024) Hydrogel‐based growth factor delivery platforms: Strategies and recent advances. Advanced Materials 36(5): 2210707.
© 2026 Zarif Bin Akhtar. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






