- Submissions

Full Text
Trends in Textile Engineering & Fashion Technology
Preparation and Properties of PANI/ CFs Electrode Materials
Meijuan Cao*, Qiaole Song, Xiaopei Wu, Yun Xiang and Dongdong Qiu
School of Printing and packaging engineering, Beijing Institute of Graphic Communication, Beijing, China
*Corresponding author:Meijuan Cao, School of Printing and packaging engineering, Beijing Institute of Graphic Communication, No. 1, Xinghua Avenue (Section 2), Beijing, 102600, China
Submission: February 16, 2026; Published: March 02, 2026

ISSN 2578-0271 Volume11 Issue 4
Abstract
Polyaniline/cotton fibers (PANI/CFs) electrode materials were prepared by in situ chemical oxidative polymerization. The structure and properties of PANI were controlled from three perspectives, namely ammonium persulfate (APS), aniline (ANI) and dopant (H+) concentration, in order to optimize the PANI/ CFs electrode materials. When APS:ANI:H+=1:1:1, the maximum specific capacitance of the PANI/CFs electrode material was 301.02F g-1 (0.2mA cm-2). The capacity retention of the electrode material was 76.77% when the current density was increased to 1mA cm-2. This provides a simple, easy-to-use, and low-cost method for the preparation of flexible electrode materials.
Introduction
With the rapid development of economy and science and technology in the new era, people’s life is gradually becoming intelligent [1], subsequently increased is the interest in wearable electronic devices, which greatly stimulates the demand for new and flexible energy storage and conversion devices [2,3]. Flexible energy storage devices represented by flexible supercapacitors have high power density, fast charge and discharge speed, and long cycle life [4] Advantages, for people to further development of wearable electronic devices to lay the foundation. Flexible supercapacitors are generally composed of flexible electrodes, diaphragm and electrolyte. Flexible electrodes are the core and play a crucial role in the performance of supercapacitors [5] How to design and manufacture flexible electrode materials has become one of the research hotspots in the field of flexible supercapacitors.
As one kind of natural fiber, CFs are renewable, biodegradable, good biocompatible and high cost-effective [6] characteristic. It can satisfy people to the flexible electrode material with good mechanical performance, strong plasticity, low cost, safety and environmental protection [7] Performance pursuit. However, CFs belong to insulating materials, which need to be combined with electrochemical active materials to achieve capacitance performance, and then used to prepare electrochemical energy storage devices with high energy density and power density [8].
PANI is conjugated CPs with delocalized π electrons that can move freely within its unsaturated backbone, generating electrical access for the mobile charge carrier [9]. It is precisely because of this inherent electron / ionic property provided by its chain-like structure that PANI shows great electrode potential in the field of electrochemistry. Because conjugated polymers are prone to oxidation or reduction, “doping” can be used to change the redox state of PANI [10]. Thus modulating its electrical properties. Moreover, PANI also has rapidly reversible doping / dedoping kinetics with high potential for pseudocapenitor-type flexible supercapacitors.
This chapter proposes that PANI is polymerized on CFs by chemical oxidation to construct PANI / CFs electrode materials. The preparation process is shown in Figure 1, where CFs were first preprocessed and repolymerized PANI in situ on CNs. The electrode material combines the intrinsic flexibility of CFs and the intrinsic conductivity and pseudocapacitance advantages of PANI, and maximizes the efficiency of PANI / CFs electrode material by optimizing the polymerization conditions.
Figure 1:Schematic preparation of electrode materials for PANI / CFs.
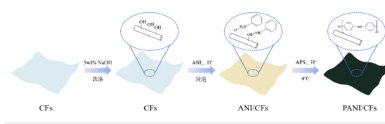
Material Preparation
Pretreatment of cotton fiber (CFs)
Arrange the NaOH solution with mass fraction of 5% and soak the CFs substrate for 10min to expand the fiber, remove impurities and improve the absorbibility of the fiber. It was then washed thoroughly with deionized water to keep the pH neutral and subsequently dried (35 ℃, 4h). Cut the pretreated cotton fabric into 5cm and weigh it for the next processing.
PANI/CFs Preparation of the electrode material
Preparation of PANI / CFs electrode materials with different concentrations of oxidant: Solution configuration: mix a certain mass of ammonium persulfate (APS) with 30ml HCl (0.075M) solution with a concentration gradient of 0.05mol/L, 0.1mol/L, 0.15mol/L, 0.2mol/L, and 0.25mol / L APS / HCl solution, and pre-cold. The quantified ANI monomer was mixed with 30ml HCl (0.075M) and sonicated for 20 min to an ANI / HCl solution of 0.15mol/L. The CFs were subsequently immersed in ANI / HCl for 30 min. PANI / CFs electrode material preparation: The precooled APS / HCl solution was slowly added to the ANI / HCl solution soaked with CFs in an ice bath, and the temperature was kept below 4 °C for the reaction time of 3h. After the reaction, the composite fabric was removed and washed with deionized water to remove oligomers or unfixed polymer adhering to it. The PANI / CFs electrode material was prepared in a 35 °C oven for 4h.
Preparation of PANI / CFs electrode materials with different concentrations of monomer: Different mole concentrations of ANI monomer solution configuration: a certain mass of ANI was mixed with 30ml of HCl (0.075M) solution and sonicated for 20 min to provide a concentration gradient of 0.05mol/L, 0.1mol/L, 0.15mol/L, 0.2mol/L, 0.25mol/L ANI / HCl solution. CFs were immersed in ANI / HCl solutions at different concentrations for 30 min. The quantified APS was mixed with 30ml of HCl (0.075M) in an APS ANI / HCl solution of 0.15mol/L and precooled.
Preparation of PANI / CFs electrode materials with different concentrations of dopants: Different molar concentrations of H+ Solution configuration: Quantitative ANI monomer was mixed with 30ml of different concentrations of HCl solution and sonicated for 20 min to configure ANI / HCl solution with 0.025mol/L, 0.05mol/L, 0.075mol/L, 0.1mol/L and 0.125mol/L. CFs were immersed in ANI / HCl solutions at different concentrations for 30 min. In addition, the quantitative APS was mixed with 30ml of HCl solution, equipped with a concentration gradient of 0.025mol/L, 0.05mol/L, 0.075mol/L, 0.1mol/L, 0.125mol/L of APS/HCl solution, and precooled.
Results and Discussion
Effect of oxidant concentration on the electrode material of PANI / CFs
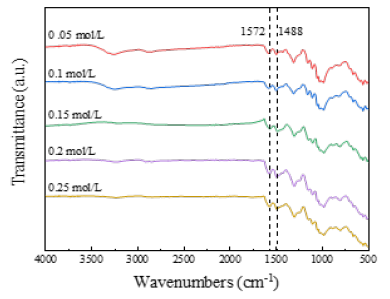
Physical structure and conductivity of PANI / CFs electrode materials: In the process of in situ polymerization of PANI, APS, as an oxidant, can oxidize ANI monomer, and trigger a polymerization reaction to promote the formation of intermolecular chemical bonds between ANI monomer, and finally the formation of PANI. Figure 2 is the FT-IR plot of PANI / CFs electrode materials prepared with different concentrations of oxidants. When the APS concentration was 0.05mol/L, 1572cm-1 And 1,488cm-1. The characteristic absorption vibration peak of quinone (oxidation) structure and benzene (reduction) structure appears respectively [140], show that PANI generation. With the increasing concentration of APS, more and more PANI polymerized, quinone structure (1572cm-1) And benzene structure (1488cm-1) The characteristic absorption vibration peak is enhanced accordingly.
Figure 2:FT-IR plots of PANI / CFs electrode materials prepared with different concentrations of oxidants.
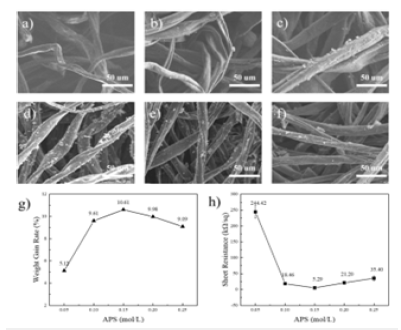
APS helps to control the rate and efficiency of polymerization reactions, thereby affecting the nature and structure of PANI [11]. Figure 3a-f shows the SEM plots of PANI / CFs prepared at 0mol/L, 0.05mol/L, 0.1mol/L, 0.15mol/L, 0.2mol/L, and 0.25mol/L, respectively. As shown in Figure 3a, when the APS concentration is 0mol/L, the CFss surface is smooth and no polymer is attached to it. With the addition of APS and increasing the increasing concentration, as shown in Figure 3b-f, there was gradually polymer to form attachment on the surface of CFs. When the APS concentration increased to 0.15mol/L and above, the increasing trend of PANI slowed down. Figure 3g shows the weight change of PANI / CFs electrode materials prepared by different concentrations of APS. According to the Figure, the load of PANI on CFs first increases and then decreases with the increase of APS concentration. When the APS increases to 0.15mol/L, the load of PANI on CFs is the maximum. This is because when the concentration of ANI in the reaction system is certain, excessive APS leads to excessive concentration of oxidant in the reaction system, while excessive oxidation can lead to breakage or rearrangement of the PANI molecular chain [12]. Which impeded the generation of PANI. Correspondingly, the square resistance of PANI / CFs electrode material also changes with the increase of APS concentration. As shown in Figure 3h, the square resistance value of PANI / CFs electrode material decreases and then increases with the increase of APS concentration. When the APS concentration is 0.15mol/L, the conductivity of PANI / CFs electrode material is relatively good.
Figure 3:a) 0mol/L, b) 0.05mol/L, c) 0.1mol/L, d) 0.15mol/L, e) 0.2mol/L, f) 0.25mol / L, g) weight gain and h) square resistance map of PANI / CFs electrode material.
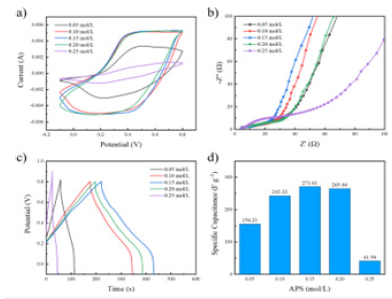
Electrochemical properties of the PANI / CFs electrode materials: Figure 4a is the CV curve of PANI / CFs electrode materials prepared with different concentrations of APS. As shown in Figure, when the scanning rate is 50mV / s, the area of CV curve increases with increasing APS concentration. The area of the CV curve reached its maximum when the APS concentration was 0.15mol/L. However, when the APS concentration continues to increase to 0.20mol/L and above, the area of the CV curve begins to decrease and the electrochemical performance decreases. This is because when the APS concentration is too low, there are not enough oxidants to participate in the reaction and the PANI yield is low. When the concentration of APS is too high, it will oxidize the polymer chain and break it into small molecules, resulting in the decrease of electrochemical properties. Figure 4b is the EIS curve of PANI / CFs electrode material. As shown in Figure, the curve shows a semicircular pattern in the high frequency area and a oblique line in the low frequency area. Where the high-frequency semicircle represents the charge transfer resistance, and the slope of the curve in the low-frequency region represents the ion diffusion resistance [13]. In the low frequency region, when the APS concentration is 0.05mol/L to 0.15mol/L, the semicircle becomes smaller and smaller, and when the APS concentration continues to increase to 0.25mol/L, the semicircle starts to increase instead, indicating that as the APS concentration increases, the interfacial charge transfer resistance decreases and then increases. In the high frequency region, the slope of the line shows the opposite trend to the semicircle in the low frequency region, and the slope first increases with the increase of the APS concentration. When the APS concentration increases to 0.15mol/L and above, the slope begins to decrease. When the APS concentration is 0.15mol/L, the semi-circle radius and the linear slope are larger, and the electrodes show more ideal capacitance performance. Figure 4c-d shows the GCD curves and the corresponding specific capacitance values of the PANI / CFs electrode materials prepared with different concentrations of APS, respectively. According to the Figure, the discharge time of PANI / CFs electrode material increases and then decreases with the concentration of oxidant APS. At the APS concentration of 0.15mol/L, 271.61Fg-1, The best electrochemical performance.
Figure 4:PANI / CNs electrode material a) CV curve (scanning rate 50mV / s), b) EIS curve, c) GCD curve (current density 0.8m Acm-2) And d) of the specific capacitance value.
Effect of monomer concentration on the electrode material of PANI / CFs
Morphological structure and electrical conductivity of PANI / CFs electrode materials: In the process of PANI synthesis by chemical oxidation method, ANI monomer participates in the reaction as an important substrate, and the molar ratio with APS greatly affects the yield and performance of PANI. Figure 5 shows the FT-IR plot of the PANI / CFs electrode material prepared with different concentrations of ANI, as shown at 1572cm at an ANI concentration of 0.05mol/L-1(Quinone-type structure), 1,488cm-1 The characteristic absorption vibration peak of PANI at (benzene structure) is not obvious. This is because when the APS concentration is certain and the ANI concentration is too low, the chain growth reaction of the PANI polymer chain is unfavorable. The characteristic absorption vibration peak of PANI was significantly enhanced with the increasing ANI concentration in the system. However, the ANI adsorbed by a certain volume of CFs substrate is limited. When the ANI concentration increases to 0.15mol/L and above, the characteristic absorption vibration peak of PANI does not change significantly.
Figure 5:FT-IR plots of PANI / CFs electrode materials prepared with different concentrations of the monomer.
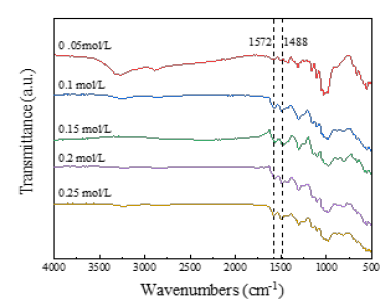
Figure 6:monomer concentration is a) 0mol/L, b) 0,) 0.05mol/L, c) 0.1mol/L, d) 0.15mol/L,) 0.2mol/L, e) 0.2mol/L, f) SEM map, g) weight gain and h) square resistance map of PANI / CFs electrode material at 0.25mol / L.
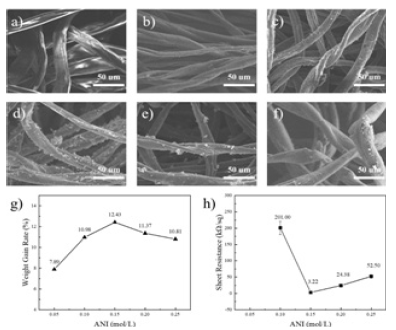
Figure 6a-f is the SEM diagram of the PANI / CFs electrode material prepared with different concentrations of ANI. As shown in Figure 6d, when the ANI concentration is 0.15mol/L, that is, the molar ratio between ANI and APS is 1:1, the loaded PANI on CFs is significantly better than the other concentrations. According to Figure 6g, the load of PANI on CFs first increases and then decreases with increasing ANI concentration. When the ANI concentration is 0.15mol/L, the weight gain rate of PANI / CFs electrode material can reach 12.43%. As shown in Figure 6h, the PANI / CFs electrodes did not show the conductivity when the ANI concentration was 0.05mol/L. When ANI concentration of 0.15mol/L, the square resistance drops to a minimum and later increases with increasing ANI concentration. According to the quantum chemistry theory, the longer the conjugated molecular chain is, the more likely it is to form charge carriers and facilitate the migration of charge carriers [14]. When the concentration of aniline monomer is large, the reaction system becomes sticky with the increase of reaction time and is prone to have side reactions, which is not conducive to the growth of the conjugated molecular chain of PANI, and the corresponding square resistance value is large and the conductivity is poor.
Electrochemical properties of the PANI / CFs electrode materials: Figure 7a shows the CV curves of the PANI / CFs electrode materials prepared at different ANI concentrations. As shown, at the ANI concentration of 0.05mol/L, the CV curve of the PANI / CFs electrode material shows no characteristic peak of the redox reaction. As the concentration of ANI increases to 0.15mol/L, the CV curve area of the electrode material is the largest, showing excellent electrochemical energy storage capacity. However, when the molar ratio of ANI to APS in the reaction system is greater than 1, the yield and mass of PANI decrease, reducing the charge transfer rate and electrochemical properties.
Figure 7:PANI / CNs electrode materials of different concentrations a) CV curve (scanning rate 50mV / s), b) EIS curve, c) GCD curve (current density 0.8mAccm-2) And d) of the specific capacitance value.
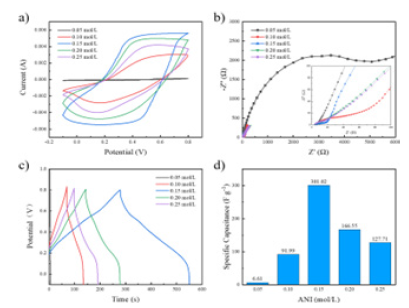
Figure 7b shows the EIS curves of the PANI / CFs electrode materials prepared at different ANI concentrations. As shown in the Figure, at an ANI concentration of 0.05mol/L, a maximum semicircle was observed in the high-frequency region with a large interfacial charge transfer resistance, which may be attributed to its poor conductivity. As the ANI concentration increases, the semicircle radius in the high frequency region begins to decrease until the ANI concentration reaches 0.15mol/L, and the latter semicircle radius begins to increase again. The linear slope in the low frequency area still increases and then decreases. When the ANI concentration is 0.15mol/L, the PANI / CFs electrode material has a longer vertical line, indicating that the capacitance performance of the electrode material is better, which is conducive to the rapid redox reaction [15], Improve the overall electrochemical properties. Figure 7c-d shows the GCD curves and the corresponding specific capacitance values of the PANI / CFs electrode materials prepared with different ANI concentrations. According to the Figure, at the concentration of ANI of 0.15mol/L, the PANI / CFs electrode material had the longest discharge time and the largest specific capacitance value of 301.02Fg-1. It shows the best electrochemical performance and is completely consistent with the results of CV and EIS curves.
Effect of dopant concentration on the material properties of PANI / CFs electrodes
Morphological structure and electrical conductivity of PANI / CFs electrode materials: Different concentrations of HCl solution were used as the dopant (H+), Additional positive charge can be introduced into the PANI polymer chain, changing its band structure and affecting its electronic structure and electron transport properties [16], and improve the electrical conductivity. Figures 8 show the different concentrations of H+FT-IR plots of doped prepared PANI / CFs, as shown, with H+. Increased concentration, 1572cm-1, 1488cm-1. The PANI characteristic absorption vibration peak at the place tends to increase, but when H+After concentrations greater than 0.1mol/L, the characteristic absorption vibration peak of PANI weakens, indicating that within a range, H+. For the chain growth response of PANI.
Figure 8:Different concentrations of dopant (H+) FT-IR plot of the electrode material prepared for PANI / CFs.
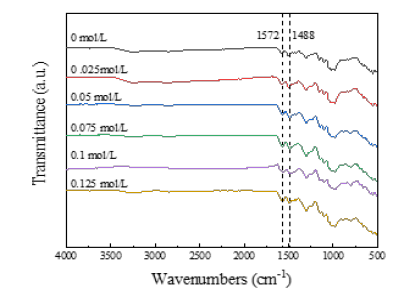
Figure 9a-f shows the different concentrations of H+SEM maps of the doped PANI / CFs prepared electrode material, as shown in Figure, with PANI attachment on the surface of CFs. The weight gain rate of Figure 9g PANI / CFs electrode material is shown along with H+Increase in concentration, PANI showed an increasing trend, when H+PANI loading was maximum on CFs at a concentration of 0.075mol/L. But when H+. When the concentration continued to increase, PANI did not increase, but tended to decrease. Figure 9h is the square resistance diagram of the PANI / CFs electrode material, as known from Figure, when H+PANI / CFs electrodes had the lowest square resistance at a concentration of 0.075mol/L. equal H+When the concentration is too low or too high, the square resistance value is relatively high. This is because when H+When the concentration is low, the reaction speed is slow, the chain growth reaction can be fully reacted, so PANI conductivity. But when H+ When the concentration is too large, it leads to an increase in the hydrochloric acid adduct [17], The conjugation degree of the molecular chain is weakened, so that the electrical conductivity is weakened.
Figure 9:The dopant (H+) Concentrations: a) 0mol/L, b) 0.025mol/L, c) 0.05mol/L, d) 0.075mol/L, 0.1mol/L, e) 0.1mol/L, f) SEM map of PANI / CFs electrode material prepared at 0.125mol/L, g) weight gain and h) square resistance map of PANI / CFs electrode material.
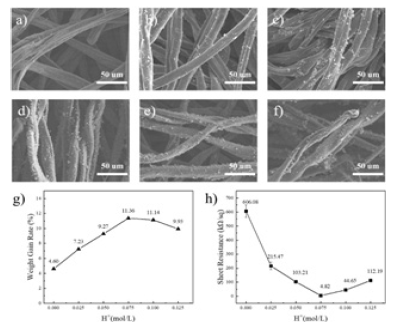
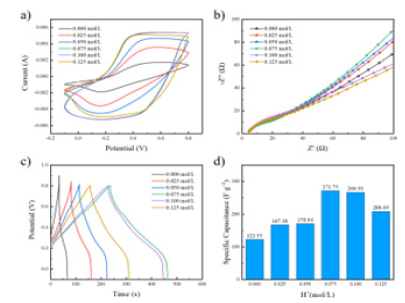
Figure 10a shows a different H+The CV curve of the PANI / CFs electrode material was prepared by concentration, as shown in Fig, and the CV curve with H+The concentration increase showed regular changes. The area of the CV curve of PANI / CFs electrode materials with H+The doping concentration increases, when H+At concentrations of 0.075mol/L and 0.1mol/L, the two groups had large and similar CV curves, with better electrochemical properties. equal H+The area of the CV curve decreases when the concentration increases to 0.125mol/L. Figures 10b are the different H+EIS curves of electrode materials of PANI / CFs. As shown in Fig, along with the H+As the concentration increases, the trend of the semicircle radius in the high frequency area in the curve is not obvious, and the slope of the curve in the low frequency region increases first and then decreases. equal H+The slope of the line is maximum at a concentration of 0.075mol/L. Combine the GCD curve and the corresponding specific capacitance value of PANI / CFs electrodes and comprehensively evaluate the capacitance performance, as shown in Figure 10c-d, with the same trend as the CV curve results, when H+At a concentration of 0.075mol/L, the PANI / CFs electrode material had the longest discharge time and the corresponding area ratio capacitance value, indicating its superior electrochemical properties.
Figure 10:Different dopants (H+) Concentration of PANI / CNs a) CV curve (scan rate 50mV / s), b) EIS curve, c) GCD curve (current density 0.8mA cm-2) And d) of the specific capacitance value.
Characterization analysis of PANI / CFs electrode materials and electrochemical performance analysis
Figure 11:The optical images and SEM maps of the CFs substrate and PANI / CFs electrode materials, respectively; c-f) are the EDS mapping images of the PANI / CFs electrode material.
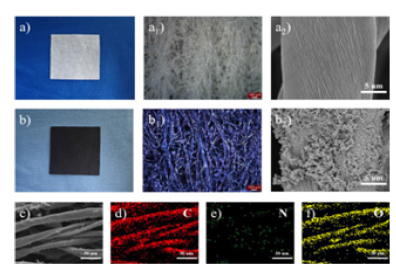
.Morphological analysis of the PANI / CFs electrode materials: Figure 11 shows the macroscopic and microscopic morphology of the CFs substrate and PANI / CFs electrode materials. See Figure 11a-a2 as shown, the substrate of CFs shows white features in appearance, with the white fiber network interwoven under optical microscope and the fiber surface polished under scanning electron microscope. After in situ polymerization of PANI, as shown in Figure 11b-b2, the appearance of the PANI / CFs electrode material shows black features, and the cotton fiber coated with PANI shows blue-purple under the optical microscope. For further observation, the PANI load on the CFs substrate can be visually seen under the scanning electron microscope. Figure 11c-f shows the EDS mapping image of the PANI / CFs electrode material. C and O elements mainly come from cotton fiber substrate, and N element, as PANI characteristic element, its distribution reflects that PANI is relatively evenly loaded on CFs, but the load is small.
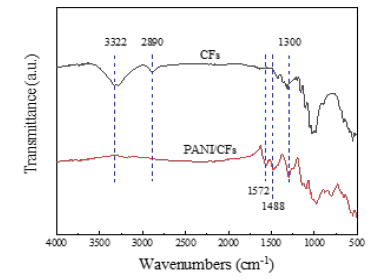
Structural analysis of the PANI / CFs electrode materials: FT-IR characterization of the CFs substrate and PANI / CFs electrode material structures by comparative analysis, as shown in Figure 12 at 1300cm in the infrared spectrum of the CFs substrate-1, 2890cm- 1 and 3322cm-1 dwell [18]. The C-H and-OH characteristic peaks of the molecular chains of cellulose in CFs appeared. However, in the IR spectrum of PANI / CFs, 2890cm-1 and 3322cm-1. The characteristic absorption peak of the cellulose molecular chain disappeared, 1572cm-1 and 1,488cm-1. The characteristic peak of polyaniline appears, and 1300cm-1. The characteristic peak at C-H was enhanced, indicating the successful generation of PANI on CFs.
Figure 12:FT-IR profiles of electrode materials for CFs and PANI / CFs.
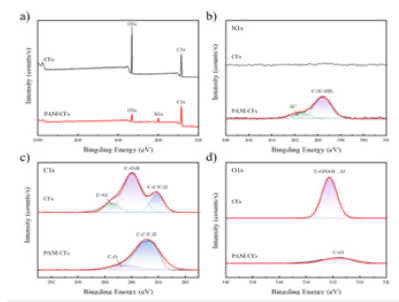
Surface composition analysis of PANI / CFs electrode materials: The surface composition analysis of the CFs substrate and PANI / CFs electrode materials was combined with X-ray photoelectron spectroscopy (XPS). Figure 13 are XPS spectra of CFs and PANI / CFs electrode materials and high-resolution fitted spectra of N, C, and O elements. As shown in Figure 13a, compared with the electrode material of CFs, the characteristic peaks of C1s and O1s were attenuated, especially the content of O element decreased significantly. Meanwhile, N1s characteristic peaks appear in parallel with PANI generation in the reaction system. In the N1s spectrum, as shown in Figure 13b, characteristic peaks at 399.54eV and 401.6eV appeared on the PANI / CNs electrode material, together with C-N / -NH-and-N in the PANI molecular chain, respectively+key [19] corresponding.
In the C1s spectrum, as shown in Figure 13c, C-O-H (286.0eV), C-C / C-H (284.1eV), C=O (287.5eV). in the molecular chain of CFs [20]. The characteristic peak is obvious. The C1s profiles of the PANI / CFs electrode material showed characteristic peaks at 284.8eV and 286.6eV, attributed to the binding energies of C-C / C-H and C-O bonds, respectively. Accordingly, in the O1s spectrum, as shown in Figure 9d, the CFs substrate has a characteristic peak at the binding energy of 532.33eV, corresponding to the C-O / O-H... O bond; the PANI / CFs electrode material has a characteristic peak with a binding energy of 531.6eV, corresponding to the C=O bond. This is because PANI chemically binds to the cellulose chain, and most of the hydroxyl group (-OH) on the primary cellulose chain is removed during the aniline polymerization process [21]. So that PANI is “connected” to the cellulose main chain, so the content of O element has decreased significantly. In addition, when PANI is combined with the cellulose molecular chain, C-O is broken, resulting in the rupture of a small part of cellulose, so the content of C element does not decrease after it “connecting” the cellulose molecular chain.
Figure 13:A) XPS full spectrum, b) C1s, c) N1s and d) O1s spectrum of CFs and PANI / CFs electrodes.
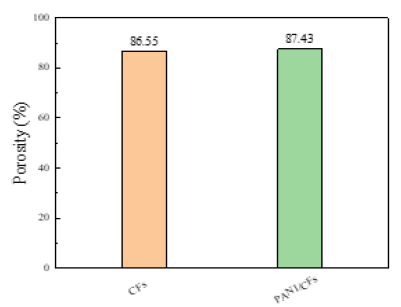
Porosity analysis of the PANI / CFs electrode material: The porosity can affect the electrochemical properties of the electrode material, and too low or too high porosity will cause mass transfer restriction or reduced energy density [8]. Figure 14 compares the porosity of CFs substrate and PANI / CFs electrode materials. As shown, the porosity of CFs substrate is 86.6% and PANI / CFs electrode material is 87.4%. The reasons for the increase in the porosity of PANI / CFs electrode materials: first, when the PANI is in situ polymerized on CFs, due to the shrinkage of fibers, the chemical binding of PANI and CFs leads to the rupture of a small number of cellulose molecules. After the pore increases, the contact between the electrode material and the electrolyte is increased to a certain extent, thus enhancing the electrochemical reaction
Figure 14:Porosity of the CFs substrate and PANI / CFs electrode materials.
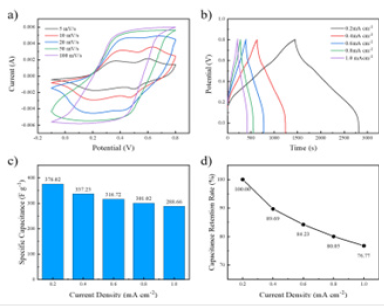
Electrochemical properties analysis of PANI / CFs electrode materials: To assess the multiplier ability of PANI / CFs electrode materials, PANI / CFs electrode materials were subjected to CV testing using different scanning rates. As shown in Figure 15a, as the scan rates 5,10,20,50 and 100mV / s increase. Figure 15b shows the GCD curve of PANI / CFs electrode materials at different current densities. As shown in Figure, with the gradual increase of current density, the charge and discharge time of the GCD curve gradually shortened and maintains symmetry, indicating its superior electrochemical reversibility. In order to more intuitively observe the multiplier performance of PANI / CFs electrode materials, as shown in Figure 15c-d, when the current density is 0.2mAccm-2. Then, the specific capacitance of the electrode material was calculated as 3 76 .02Fg-1. With the current density to 1mAccm-2, The capacitance retention rate of the electrode material was 76.77%.
Figure 15:PANI / CFs electrode material a) CV curve at different scanning rates, b) GCD curve at different current densities, c) specific capacitance and d) ratio performance.
Conclusion
In this chapter, PANI was prepared by in situ chemical oxidation polymerization and loaded on CFs to finally produce PANI / CFs electrode material. The PANI / CNs electrode material was optimized from three perspectives of oxidant, monomer and dopant. Through SEM, EDS and combining FT-IR and XPS tests, the test results showed that PANI was successfully grown on CFs, and also proved that APS oxidant, ANI monomer, H+dopants have important effects on the chemical state of PANI, the binding between PANI and CFs, and the conductivity and electrochemical properties of PANI / CFs electrode materials. The porosity of PANI / CFs electrode material was tested by pressing mercury and found that the porosity of this electrode material increased. When APS, ANI and H+When the molar ratio is 1:1:1, the electrochemical results show that the current density is 0.2 m Accm-2Below, the specific capacitance of the electrode material is 376.02Fg-1.
Author Contributions
We strongly encourage authors to include author contributions and recommend using CRediT for standardised contribution descriptions. Please refer to our general author guidelines for more information about authorship.
Conflicts of Interest
In accordance with our policy on Conflicts of interest please ensure that a conflict-of-interest statement is included in your manuscript here. Please note that this statement is required for all submitted manuscripts. If no conflicts exist, please state that “There are no conflicts to declare”.
Data Availability
A data availability statement (DAS) is required to be submitted alongside all articles. Please read our full guidance on data availability statements for more details and examples of suitable statements you can use.
Acknowledgement
This work was financially supported by the National Key R&D Program of China (2023YFE0113800) and BIGC Project (Ec202502).
References
- Zhang H, Zhu X, Tai Y, Zhou J, Li H, et al. (2023) Recent advances in nanofiber-based flexible transparent electrodes. International Journal of Extreme Manufacturing 5(3): 032005.
- Xu Y, Lin Z, Wei W, Hao Y, Liu S, et al. (2022) Recent progress of electrode materials for flexible perovskite solar cells. Nanomicro Lett 14(1): 117.
- Fan K, Li Z, Song Y, Xie W, Shao M, et al. (2020) Confinement synthesis based on layered double hydroxides: A new strategy to construct single‐atom‐containing integrated electrodes. Advanced Functional Materials 31(10): 10.
- Duan H, Liu Y, Zhao X (2023) Recent research progress of conductive polymer-based supercapacitor electrode materials. Textile Research Journal 93(15-16): 3884-3925.
- Wang X, Xu P, Zhang P, Ma S (2021) Preparation of electrode materials based on carbon cloth via hydrothermal method and their application in supercapacitors. Materials 14(23): 7148.
- Wang X, Yao C, Wang F, Li Z (2017) Cellulose‐based nanomaterials for energy applications. Small 13(42): 42.
- Dutta S, Kim J, Ide Y, Kim JH, Hossain SA, et al. (2017) 3D network of cellulose-based energy storage devices and related emerging applications. Materials Horizons 4(4): 522-545.
- Wang Z, Lee YH, Kim SW, Seo JY, Lee SY, et al. (2021) Energy‐storage materials: Why cellulose‐based electrochemical energy storage devices? Advanced Materials 33(28): 28.
- Van Tran V, Lee S, Lee D, Le TH (2022) Recent developments and implementations of conductive polymer-based flexible devices in sensing applications. Polymers 14(18): 3730.
- Etman AE-S, Ibrahim AM, Darwish FA-ZM, Qasim KF (2023) A 10 years-developmental study on conducting polymers composites for supercapacitors electrodes: A review for extensive data interpretation. Journal of Industrial and Engineering Chemistry 122: 27-45.
- Qiu B, Wang J, Li Z, Wang X, Li X (2020) Influence of acidity and oxidant concentration on the nanostructures and electrochemical performance of polyaniline during fast microwave-assisted chemical polymerization. Polymers 12(2): 310.
- Zhao Xiaoming, Sun Jiarui (2018) Progress of conducting PANI and its application in textile field. Journal of Textile Science and Engineering 35(01): 153-148.
- Vahid Mohammadi A, Moncada J, Chen H, Kayali E, Orangi J, et al. (2018) Thick and freestanding MXene/PANI pseudocapacitive electrodes with ultrahigh specific capacitance. Journal of Materials Chemistry A 6(44): 22123-22133.
- Xie Weibin, Wu Jianjian, Zhou Jiayi (2020) Preparation and properties of polyaniline / cotton composite fabric. Printing and Dyeing 46(03): 44-48.
- Chulkin P, Łapkowski M (2020) An insight into ionic conductivity of polyaniline thin films. Materials 13(12): 2877.
- Li B, Chen Y, Liu X, Li F, Wang J, et al. (2022) The infrared electrochromic properties based on acid doped-polyaniline films. Frontiers in Materials 9(1): 2.
- Zhou H, Hou L, Zhang W, Zhai HJ (2020) A dual-doped strategy to enhance the electrochemical performances of electropolymerized polyaniline electrodes for flexible energy storage. Materials Chemistry and Physics 240: 122259.
- Parvez MS, Rahman MM, Samykano M, Ali MY (2023) Electrochemical characterization and joule heating performance of polyaniline incorporated cotton fabric. Physics and Chemistry of the Earth 129: 103323.
- Deng J, Wang T, Guo J, Liu P (2017) Electrochemical capacity fading of polyaniline electrode in supercapacitor: An XPS analysis. Progress in Natural Science: Materials International 27(2): 257-260.
- Wang X, Li Q, Yang D, An X, Qian X (2022) Phytic acid doped polyaniline as a binding coating promoting growth of Prussian blue on cotton fibers for adsorption of copper ions. Coatings 12(2): 138.
- Chang Z, An X, Qian X (2020) High mass loading polyaniline layer anchored cellulose fibers: Enhanced interface junction for high conductivity and flame retardancy. Carbohydrate Polymers 230: 115660.
© 2026 Meijuan Cao. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






