- Submissions

Full Text
Trends in Textile Engineering & Fashion Technology
Optimization of Conditions and Methodological Validation for Determining the Effective Content of Disperse Dyes by Spectrophotometry
Yue Liu1,2* and Lingling HU1,2
1College of Textile Science and Engineering, Shaoxing University, Shaoxing, Zhejiang, 312000, China
2Key Laboratory of Clean Dyeing and Finishing Technology of Zhejiang Province, Shaoxing, Zhejiang, 312000, China
*Corresponding author:Yue Liu, College of Textile Science and Engineering, Shaoxing University, Shaoxing, Zhejiang, 312000, China Key Laboratory of Clean Dyeing and Finishing Technology of Zhejiang Province, Shaoxing, Zhejiang, 312000, China
Submission: January 19, 2026;Published: January 29, 2026

ISSN 2578-0271 Volume11 Issue 3
Abstract
Disperse dyes are non-water soluble specialized dyes used for the dyeing of polyethylene terephthalate fibers. Typically, the quantification of their effective content is achieved through a color comparison method subsequent to the dyeing of polyester fiber textiles. However, this conventional method is notably complex, labor-intensive, and economically inefficient. This article explores the feasibility of using spectrophotometry to determine the effective content of dispersed dyes, using a mixture of dispersed blue 291G filter cake, different proportions of dispersant MF, sodium lignosulfonate dispersant, and corresponding commercial dispersed dyes as examples. The results demonstrate that optimizing the DMF/ water solvent ratio, calibrating the dye’s spectral absorption curve, and executing linear regression on “dye absorbance-dye concentration” yields a maximum deviation of +0.8314% and a minimum of +0.016% in the effective dye content measured by spectrophotometry, as compared to the color comparison method. This underscores the substantial consistency between the two methodologies. The spectrophotometry approach not only offers high precision but also significantly reduces testing duration. For large-scale assessment of effective dye content in disperse dyes, spectrophotometry provides significant advantages including rapid analysis, elevated accuracy, and enhanced environmental sustainability.
Keywords: UV visible absorption spectrophotometry; Disperse dyes; Effective content; Rapid determination; Lambert-Beer law
Introduction
Polyethylene terephthalate (PET) fibers [1,2] have tightly arranged macromolecular chains, high crystallinity, and strong hydrophobicity. The disperse dyes used for dyeing these fibers typically contain few strong water-soluble groups and have relatively low molecular weight. Consequently, these dyes are insoluble or only slightly soluble in water, but they can dissolve in organic solvents such as acetone and N,N-dimethylformamide. After the synthesis of the raw dye, disperse dyes require the addition of a certain proportion of dispersants and other surfactants, followed by grinding [3], crystal transformation [4,5], and standardization to produce commercial dyes with excellent dyeing properties. The effective content of disperse dyes is an essential quality control metric for assessing their color-rendering ability. According to the Chinese national standard GB/T2394 “Determination of color and strength of disperse dyes”[6], this effective content is determined by dyeing samples using the high-temperature and high-pressure method, followed by visual comparison [7] or instrumental K/S value [8] analysis. Assessment is permissible only if the sample’s coloration “closely”, “slightly”, or “moderately resembles” the standard sample; conversely, if the coloration “deviates markedly” or “significantly”, evaluating the strength is not feasible. This process is complex and timeconsuming. In this context, this paper examines a simple and rapid method for determining the effective content of disperse dyes, using Disperse Blue 291G dye, dispersant MF, and sodium lignosulfonate as experimental subjects. This method aims to facilitate the quality control of disperse dyes while aligning with the ecological development concepts of energy conservation and emission reduction in textile printing and dyeing.
Spectrophotometric analysis operates on the principles of the Lambert-Beer law, which establishes a direct correlation between the concentration of a dye and its absorbance of monochromatic light [9,10]. When a beam of monochromatic light traverses a homogenous solution, a portion of the light is absorbed, while the remainder is transmitted through the solution. Let I0 denote the intensity of the incident light, and I represent the intensity of the light transmitted through the solution. The absorbance (A) is defined by the relationship A=lgT=lgI/I0, which, following mathematical transformation, yields Equation (1):
A = −1gT =ε cl (1)
Wherein: T denotes transmittance, c represents the concentration of the substance (mol/L), ε is the molar absorption coefficient (L/(mol·cm)), and l signifies the path length (cm). The formula delineates that, under specified conditions, the absorbance (A) of the dye exhibits a linear proportionality to both the concentration of the dye or other absorbing substances within the solution and the path length of the light through the solution.
Experimental
Experimental reagents
Disperse Blue 291G filter cake and commercial dye, sourced from Hangzhou FuLaiente Fine Chemical Co., Ltd., are classified as industrial grade, with their structural formula depicted in Figure 1. Dispersant MF, sodium lignosulfonate, sodium dithionite, N,Ndimethylformamide, glacial acetic acid, and sodium hydroxide, all procured from Aladdin Reagent (Shanghai) Co., Ltd., are of analytical grade.
Figure 1:Structural formula of disperse blue 291G.
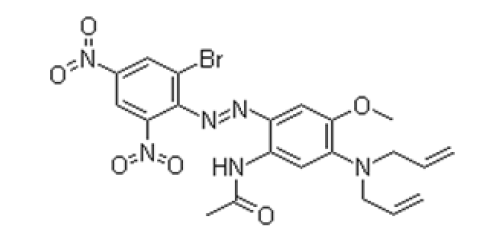
Commercial disperse dyes are primarily formulated from a blend of disperse dyes and environmentally sustainable dispersants [11-13], exhibiting high dispersion efficacy in precisely calibrated proportions. To ensure robust comparability in the experiments, the employed dye comprised a mixture of disperse dye filter cake and two types of dispersants routinely utilized in the manufacture of commercial dyes. This mixture was formulated into a slurry and subsequently ground. The grinding parameters were established at a rotational speed of 200r/min and a grinding duration of 7 hours, with a mass ratio of grinding media to mixed dye of 10:1. The grinding media was composed of a combination of 3mm zirconia beads and 1mm zirconia beads, maintained at a 1:1 mass ratio.
The four experimental disperse dyes are named Dye1, Dye 2, Dye 3, and Dye 4. The compositions are delineated as follows: Dye 1 comprises Disperse Blue 291G (filter cake) and dispersant MF, formulated in a 1:1 mass ratio. Dye 2 is composed of Disperse Blue 291G (filter cake) and dispersant MF, mixed in a 1:2 mass ratio. Dye 3 includes Disperse Blue 291G (filter cake) and dispersant sodium lignosulfonate, blended in a 1:1 mass ratio. Dye 4 consists of Disperse Blue 291G (filter cake) and dispersant sodium lignosulfonate, configured in a 1:2 mass ratio.
Experimental testing instruments
Floor-standing planetary ball mill, model PM400, manufactured by Retsch, Germany. Oscillating dyeing machine, model L-12, produced by Xiamen Ruibi Precision Machinery Co., Ltd. Oil bath high-temperature dyeing machine, model H-24CF, from Xiamen Ruibei Precision Machinery Co., Ltd. Forced convection universal oven, model UF260Plus, engineered by MEMMERT, Germany. Malvern laser particle size analyzer, model Master Sizer 3000, by Malvern Instruments Ltd., UK. UV-Vis spectrophotometer, model UV2600, produced by SHIMADZU, Japan. Color measurement and matching system, model 600, developed by Data color, USA. Ultrasonic cleaner, model SCQ-9201E, by Shanghai Shengyan Ultrasonic Instrument Co., Ltd. Precision electronic balance, model FA204S, manufactured by METTLER TOLEDO, Switzerland.
Preferred DMF/water mixed solvent
To ensure complete dissolution of the dye and dispersant within the disperse dye, thereby forming a true solution compliant with the Lambert-Beer law prerequisites [14,15], an optimal DMF/water ratio is imperative. The experimental design establishes a gradient of DMF/water concentration ratios at 100/0, 80/20, 60/40, 50/50, 40/60, and 20/80, excluding a 0/100 ratio due to the low solubility of disperse dyes. Variations in the maximum absorption wavelength (λmax) will be observed when the dye is not completely dissolved, as opposed to its fully dissolved state; these variations will guide the determination of the suitable DMF/water ratio.
Testing of absorption spectra curves for dyes and dispersants
To precisely delineate the relationship between dye concentration and its absorbance, both the dye and dispersant concentrations are established within a low range. The concentration gradient for Disperse Blue 291G filter cake has been methodically set at 0.004, 0.006, 0.008, 0.010, 0.012, 0.014, 0.016, 0.018, and 0.020g/L. The concentration series for dispersant MF is designed to be 0.010, 0.015, 0.020, 0.025, 0.030, 0.040, 0.050, and 0.060g/L. The concentration gradient for dispersant sodium lignosulfonate is strategically arranged from 0.010 to 0.080g/L in increments reflecting precise control. A specific concentration from these series is selected to analyze the absorption spectrum curve, thereby determining the corresponding maximum absorption wavelength (λmax).
Testing of dye strength using high-temperature highpressure dyeing method
In this comparative analysis, the effective content of disperse dyes is quantified utilizing an instrumental colorimetric method, in accordance with the established intensity testing standards previously outlined.
(1) Dyeing Concentration: For the dyeing process, each fabric
sample is meticulously weighed to 1g (±0.005g). The dye
concentration gradient is methodically established at intervals
of 0.3%, 0.5%, 0.7%, 0.9%, 1.0%, 1.1%, 1.3%, 1.5%, 2.0%, 2.5%,
3.0%, 3.5%, and 4.0% (o.w.f).
(2) Dyeing Process and Procedure: Employing a disperse dye
concentration of x% (o.w.f.) and a liquor ratio of 1:50, the pH
is adjusted to 4-5 using acetic acid. The dyeing commences
at room temperature, with the temperature incrementally
increased to 90 ℃ at a rate of 3 ℃/min, followed by a rise to
130 ℃ at 1 ℃/min, sustained for 30 minutes, and subsequently
cooled to 80 ℃ at 2 ℃/min, culminating with a water wash.
(3) Reduction Cleaning Procedure: The process involves
treating the dyed samples with sodium dithionite and sodium
hydroxide, each at 2g/L concentration, with a liquor ratio of
1:50, at 85 ℃ for 10 minutes, followed sequentially by rinsing
in cold water, washing in hot water, and drying of dyed samples.
(4) Calculation of Effective Content: The effective content of
disperse dyes is determined by measuring the K/S values of
the dyed samples. Employing Dye 1 as the reference standard,
the effective content of each dye is calculated. The relative
effectiveness, denoted as ST and expressed as a percentage, is
derived using Equation (2):
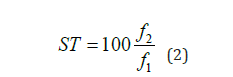
where f2 and f1 signify the K/S values of the test dye and the standard dye, respectively.
Results and Discussion
Particle size distribution of experimental disperse dyes
The formulation of commercial disperse dyes necessitates small particle sizes and a narrowly defined particle size distribution. When particle sizes are excessively large, this results in a reduced dye uptake rate and may lead to complications such as dye aggregation and sedimentation during the dyeing process, culminating in the appearance of color spots on the fabric. Conversely, overly small particles tend to aggregate into larger formations throughout the dyeing process. Consequently, the commercial viability of disperse dyes is contingent upon the incorporation of high-dispersion, environmentally benign dispersants [11-13] and the application of suitable grinding techniques [15]. Analysis of the absorption spectrum curves of the experimental commercial disperse dyes reveals that they are composed of a single dye component synergistically blended with a dispersant in precise proportions. To reduce the difference between the mixed dye in the filter cake and the commercial dye, it is necessary to grind the abovementioned dyes to obtain the appropriate dye particle size. In this study, the dyes were formulated by integrating commonly utilized dispersants-MF and sodium lignosulfonate-with dye filter cake in designated proportions, subsequently subjected to grinding. The particle size distributions of the resultant experimental dyes postgrinding are depicted in Figure 2.
Figure 2:Particle size distribution diagram of experimental disperse dyes after grinding.
(a) Dye 1; (b) Dye 2; (c) Dye 3; (d) Dye 4.
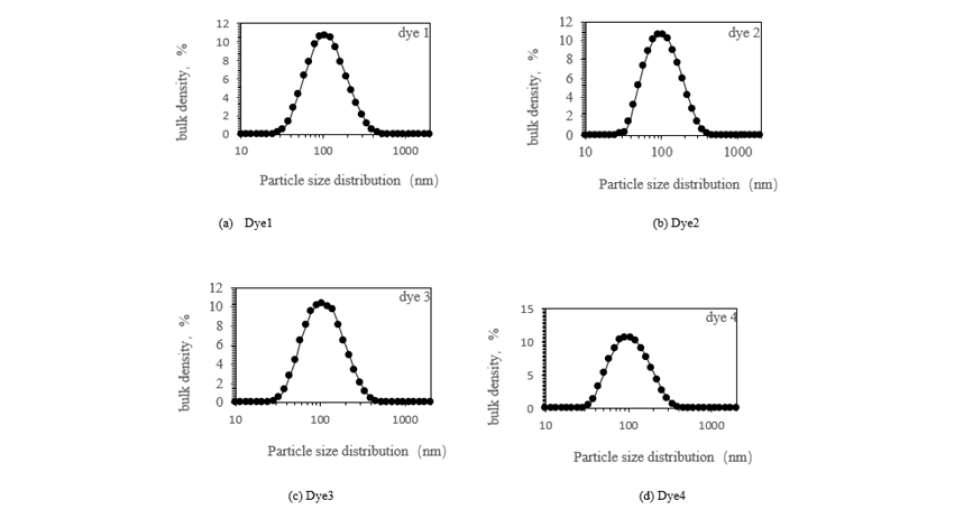
As depicted in Figure 2, the particle size distribution of the four experimental disperse dyes is basically similar, predominantly centers around 100nm, with all particle sizes remaining below 1000nm. This size range is smaller than that typically observed in commercial dyes, suggesting enhanced dyeing application performance for the experimental dyes.
Selection of DMF/water mixed solvent ratio
The maximum absorption wavelengths for dyes 1, 2, 3, 4, and the commercial dye across various DMF/water solvent systems were systematically analyzed, with the findings presented in Table 1.
Table 1:Maximum absorption wavelength of disperse blue 291G experimental dye in different DMF/water mixed solvents.
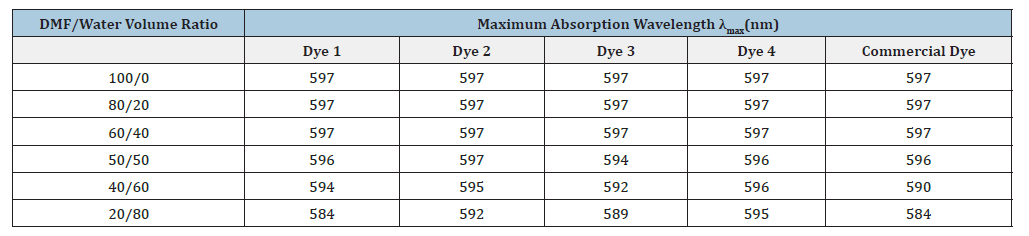
Analysis of the data presented in Table 1 reveals that the maximum absorption wavelengths of the disperse dyes remain consistent across the 100/0, 80/20, and 60/40 DMF/water solvent mixtures. This observation suggests that variations within these specific solvent mixtures do not markedly impact the dye’s absorption characteristics within a practical range. Given the practical considerations for the use of organic solvents, the optimal experimental solvent mixture has been established as DMF/ water=60/40.
Absorption spectra curves of dyes and dispersants
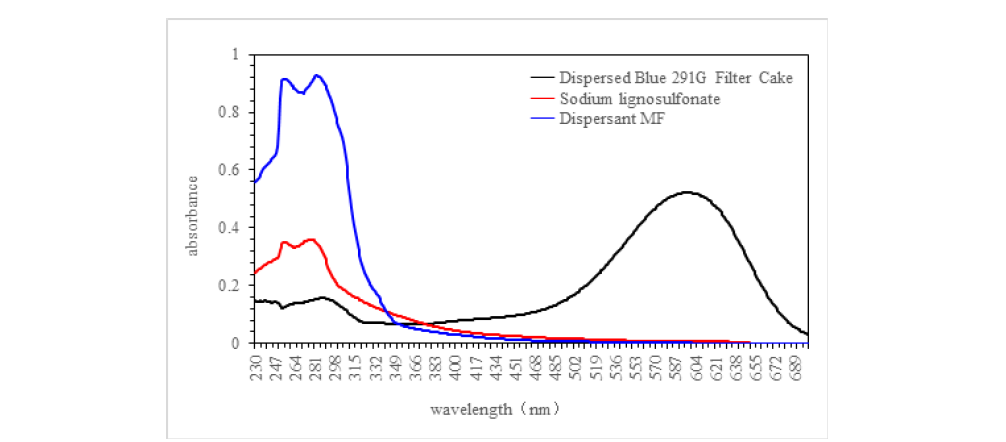
To determine the maximum absorption wavelengths for Disperse Blue 291G filter cake, dispersant MF, and dispersant sodium lignosulfonate, the absorption spectra of these solutions were analyzed in a DMF/water =60/40 mixed solvent. The findings are presented in Figure 3. As depicted in Figure 3, the maximum absorption wavelengths identified for dispersant MF, sodium lignosulfonate, and Disperse Blue 291G are 283nm, 277nm, and 597nm, respectively.
Figure 3:Absorption spectrum curve of dispersant MF, sodium lignosulfonate and disperse blue 291G filter cake.
Linear fitting of “absorbance-concentration” and calculation of molar absorptivity for each substance
Utilizing the established maximum absorption wavelengths for Disperse Blue 291G filter cake, dispersant MF, and dispersant sodium lignosulfonate, specifically λ1=597nm, λ2=283nm, and λ3=277nm, the absorbance of Disperse Blue 291G filter cake at varying concentrations was systematically evaluated. The experimental findings are detailed in Table 2.
Table 2:Absorbance corresponding to the concentration of disperse blue 291G filter cake at different wavelengths.
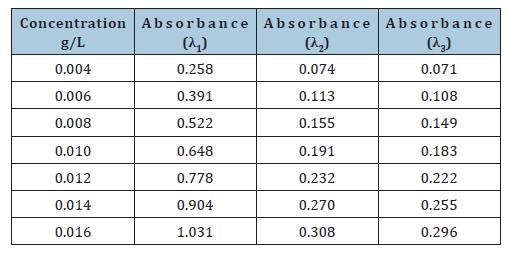
For Disperse Blue 291G filter cake at its maximum absorption wavelength (λ1), a linear regression was applied to model the relationship between absorbance (y) and concentration (x), resulting in the equation y=64.682x. This model demonstrated an exceptionally high linear correlation coefficient, R²=0.9999. According to Lambert-Beer’s law and the established linear fitting, the molar absorption coefficient (εfilter) at 597nm for Disperse Blue 291G filter cake is calculated as 64.682.
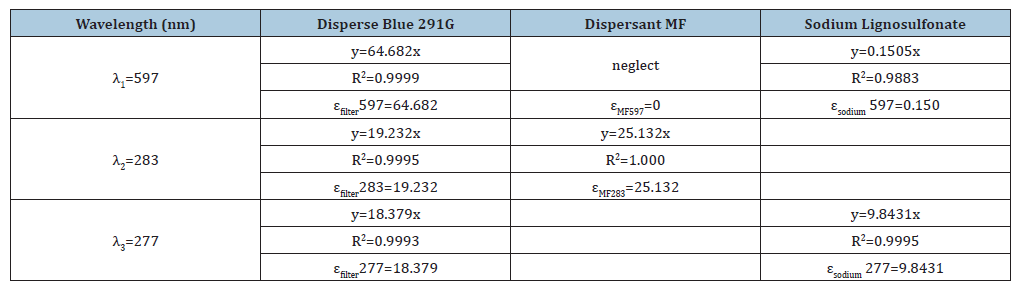
Drawing upon the relevant experimental data and the previously described calculation model, the linear “absorbanceconcentration” relationship for Disperse Blue 291G filter cake at the maximum absorption wavelengths (λ2 in dispersant MF and λ3 in sodium lignosulfonate) has been established. Furthermore, the molar absorption coefficients (εfilter 283, εfilter 277) for the dye filter cake at these specified maximum absorption wavelengths have been accurately quantified.
Following the methodology of the aforementioned experiment and utilizing the same calculation model, the linear “absorbanceconcentration” relationship for various concentrations of dispersant MF at wavelengths λ1 and λ2 has been derived. Additionally, the molar absorption coefficients for dispersant MF at these specific maximum absorption wavelengths, εMF597 and εMF283, have been precisely determined. The data reveal that the absorbance of dispersant MF at wavelength λ1 is negligible, effectively zero, thus its impact on the solution’s absorbance at λ1 can be disregarded, with εMF597 determined to be 0. Similarly, employing the same experimental framework, the “absorbanceconcentration” linear relationship for sodium lignosulfonate across a range of concentrations at wavelengths λ1 and λ3, along with the corresponding molar absorption coefficients, εsodium 597 and εsodium 277, have been established. The detailed relationships and corresponding data are presented in Table 3.
Table 3:Linear fitting relationship of “absorbance-concentration” and molar absorptivity of disperse blue 291G, dispersant MF, and sodium lignosulfonate at different wavelengths.
Calculation of effective content of disperse dyes by spectrophotometry
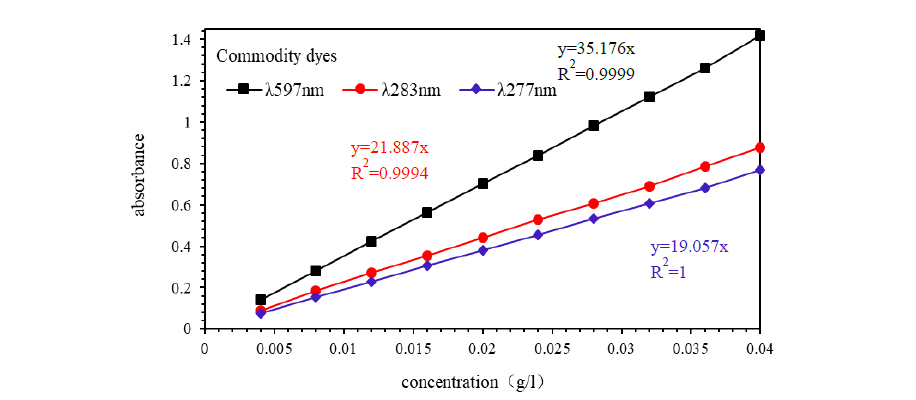
Utilizing Dye 1 as the standard reference and designating Dyes 2, 3, and 4, along with commercial dyes, as test subjects, the absorbance measurements for Dye 1 at 597nm and 283nm, Dye 2 at 597nm and 283nm, Dye 3 at 597nm and 277nm, Dye 4 at 597nm and 277nm, and commercial dyes at 597nm, 283nm, and 277nm were conducted at various concentrations. These measurements are graphically represented in Figures 4&5.
Figure 4:Linear relationship between absorbance and concentration of experimental dyes.
(a) Dye 1; (b) Dye 2; (c) Dye 3; (d) Dye 4.
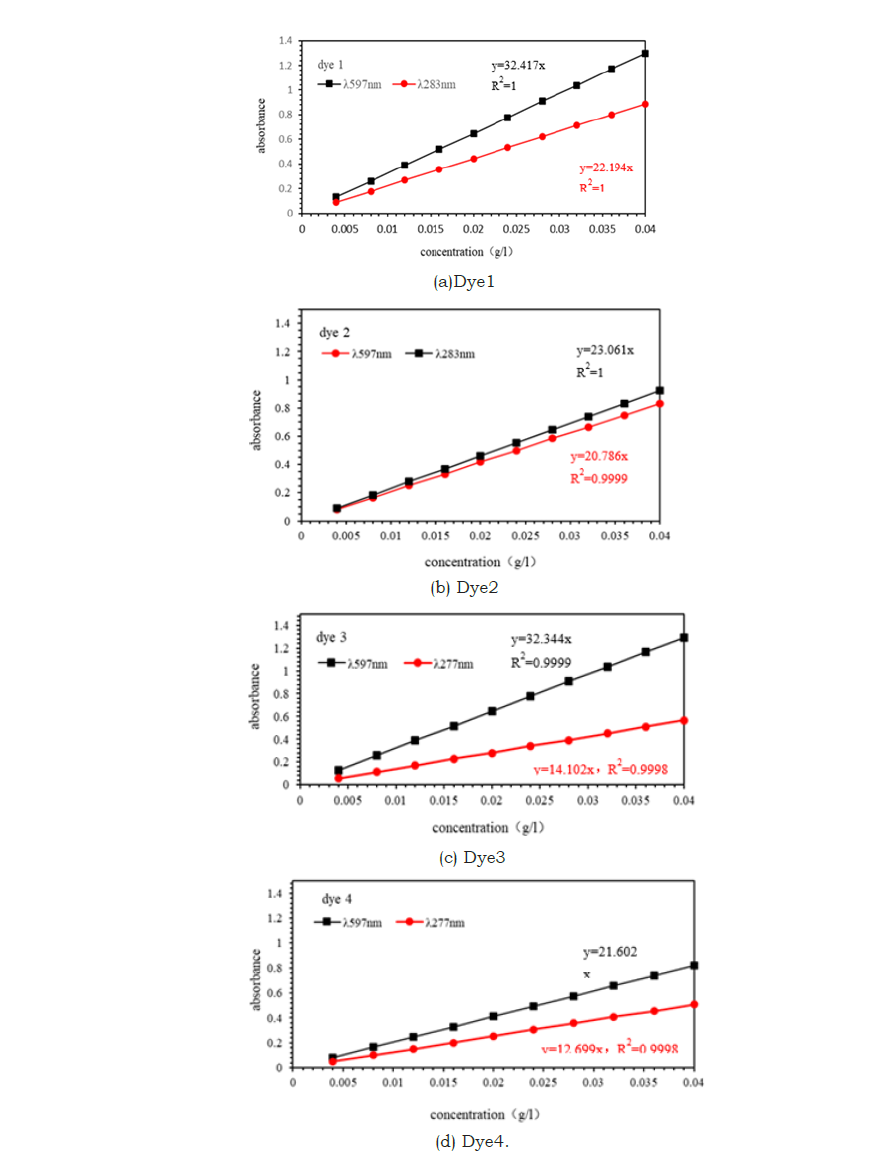
Figure 5:Linear relationship between absorbance and concentration of commodity dyes.
The thickness of the cuvette used in the experiments was consistently maintained at 1cm. Building on Equation (1), Equations (3), (4), and (5) were derived to further explore the relationships:
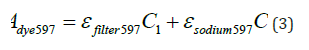
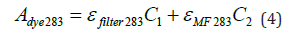
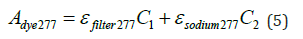
Where, C1 represents the concentration of the disperse dye (g/L), and C2 denotes the concentration of the dispersant (g/L).
Utilizing the established “dye concentration-absorbance” fitting relationship, the absorbance for any given dye concentration can be accurately determined. The dye concentration values are maintained at 0.004, 0.008, 0.012, 0.016, 0.020, 0.024, 0.028, 0.032, 0.036, and 0.040g/L for consistency in the analysis. By concurrently solving Equations (3) and (4), along with Equations (3) and (5), the concentrations C1 and C2 for each dye sample can be accurately calculated. The ratio of the test sample to the effective component at the same concentration quantifies the effective content of the disperse dye as determined by the spectrophotometric method, as delineated in Equation (6):
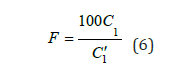
In the formula, F denotes the effective content of the disperse dye as determined by the spectrophotometric method, while C1 and C1′ represent the concentrations of the test dye and the standard dye, respectively.
Consequently, the spectrophotometric analysis revealed that the effective content of dye 2 is 64.00, dye 3 is 99.75, dye 4 is 63.06, and the commercial dye registers at 109.5.
Calculation of disperse dye strength by K/S value method
In order to more accurately determine the effective content of experimental dispersed dyes, experiments were conducted on the dyeing enhancement capability of commercial dispersed dyes, and the results are shown in Figure 6.
Figure 6:Enhancement capability of commercial dyes.
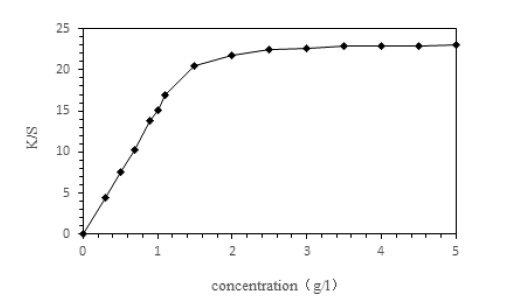
As depicted in Figure 6, the K/S value of the commercial dye on polyester fabric demonstrates a linear relationship with the dye concentration, exhibiting a critical turning point at approximately 1.5% (o.w.f). To maintain consistency with the concentrations employed in the spectrophotometric experiments, dye concentrations for subsequent dyeing trials were established at 0.3, 0.5, 0.7, 0.9, and 1.1% (o.w.f). The K/S values for samples dyed with Dyes 1, 2, 3, 4, and the commercial dye are presented in Table 4.
Table 4:The K/S value of dyed samples with different experiments.
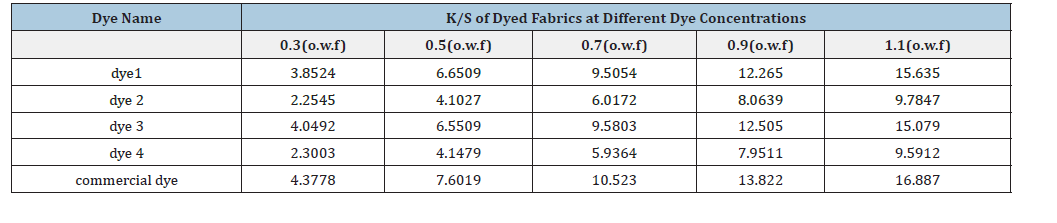
Upon fitting and analyzing the K/S values of the five dyes, the following results were derived: dye 2 yielded an effective content of 63.39, dye 3 recorded 99.42, dye 4 measured 62.54, and the commercial dye achieved an effective content of 110.4.
Analysis of effective content of disperse dyes obtained by two methods
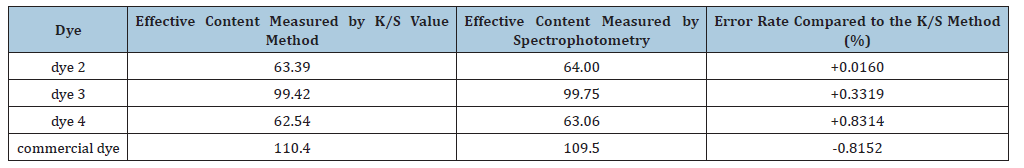
The variances in the effective content of the dyes, as calculated by the two distinct methods, are detailed in Table 5. According to the data, when compared to the standard GB/T 2394 “Determination of Color Strength and Tone of Disperse Dyes” using the K/S value method, the spectrophotometric method exhibits a maximum error in the effective content of the experimental disperse dyes of +0.8314%, and a minimum error of +0.016%.
Table 5:Differences in effective dye content measured by two methods.
Conclusion
This research employed the spectrophotometric method to assess the effective content of disperse dyes, specifically focusing on disperse blue 291G, dispersant MF, and dispersant sodium lignosulfonate. The findings are as follows:
(1) When benchmarked against the standard GB/T 2394
“Determination of Color Strength and Tone of Disperse Dyes”
employing the K/S value method, the spectrophotometric
approach exhibited a maximum deviation of +0.8314% and a
minimum deviation of +0.016% in determining the effective
content of the tested disperse dyes.
(2) Optimization of the DMF/water mixed solvent, calibration
of dye spectral absorption curves, and linear regression of
the “absorbance-dye concentration” relationship facilitated
the spectrophotometric method’s robust alignment with
the standard K/S value method in evaluating the effective
content of disperse dyes. This substantiates the feasibility of
the spectrophotometric method for determining the effective
content of disperse dyes. Owing to its rapid throughput and high
consistency, the spectrophotometric method is particularly
advantageous for large-scale analyses and additionally offers
significant benefits in reducing environmental impact and
energy consumption.
References
- (2023) Analysis of the current situation and prospects of the fiber market in 2023.
- Jingsi Q, Yue L (2021) Research progress on the superfine-dispersion and its particle-size effects of disperse dyes. Journal of Textile Research 42(8): 194-201.
- Naifeng Z, Weisong F, Zhiyong T, Huaxiang C, Lingling Z, et al. (2016) Precipitation of disperse dye black slurry based on diameter analysis. Journal of Textile Research 37(4): 80-85.
- Liang J, Zhong Y, Mao Z, Xu H, Zhang L, et al. (2018) Effect of crystal form on dyeing behavior of disperse dyes. Journal of Textile Research 39(7): 69-73.
- Wang R, Li M, Tian A, Wang CX, Fu SH (2021) Relationship between disperse yellow 6GSL crystal form and thermal stability of its dispersions. Journal of Textile Research 42(5): 96-102.
- GB/T2394-2013, disperse dyes Disperse dyes, determination of shade and intensity.
- GB/T2374-2007, General conditions for dyeing conditions.
- GB/T6688-2008, disperse dyes Dyes-Determination of relative intensity and color difference-Instrumental method.
- Zhang J, Zhang Y, Hu X (2017) Method of mixed dye liquor concentration detection based on spectral optimization and multi-level support vector machine. Joural of Textile Research 38(7): 90-94.
- Golob V, Lidija T (1999) VIS absorption spectrophotometry of disperse dyes. Dyes & Pigments 40(2): 211-217.
- Gharanjig H, Gharanjig K, Sina T (2013) Stability of dye dispersions in the presence of some eco-friendly dispersing agents. Journal of Surfactants and Detergents 16(6): 849-856.
- El-Wahab HA, Nasser AM, Elbary HMA, Abd Elrahman M, Hassanein M (2020) Effect of the modified dispersing agent and milling time on the properties and particle size distribution of inkjet ink formulation for textile printing. Pigment & Resin Technology 50(4): 356-366.
- Gharanjig K, Dadras FS, Sadeghi-Kiakhani M, Tafaghodi S (2013) Stability of dye dispersions in the presence of various surface active agents and additives. Journal of Dispersion Science and Technology 34(3): 381-388.
- Zevatskii YE, Lysova SS, Skripnikova TA, Vorona SV, Myznikov LV (2024) Bouguer-Lambert-Beer law of absorption: Spectrophotometry in electrolyte solutions. Russian Journal of Physical Chemistry A 98: 242-247.
- Song Jiangting; Jin Fujiang;Zhou Lichun et al. (2021) A study of soft sensor model of single-component dye concentration based on optical quantum tunneling penetration. Chinese Journal of Scientific Instrument 41(8): 238-248.
- Yang Junhao, Qian Wei (2014) Progress in ultrafine, nano level grinding technique of pigments and dyes. Shanghai Dyestuffs 42(5): 69-75.
© 2026 Yue Liu. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






