- Submissions

Full Text
Techniques in Neurosurgery & Neurology
Unilateral Microscopic Approach for Bilateral Decompression and Removal of Massive Lumbar Disc Herniations Associated with Incomplete Cauda Equina Syndrome: Description and Evaluation of a Novel Technique
Mario Carvi Nievas1,2,3* and Eleftherios Archavlis1,2,4
1 Department of Neurosurgery, Interdisciplinary Spine Center, Elisabethen Hospital, Germany
2 European University Cyprus, School of Medicine, Germany
3 JW Goethe University, Germany
4 IU University of Applied Sciences, Germany
*Corresponding author:Mario Carvi Nievas, Department of Neurosurgery, Interdisciplinary Spine Center, Elisabethen Hospital, Frankfurt, Germany
Submission: February 17, 2026;Published: March 03, 2026

ISSN 2637-7748
Volume6 Issue2
Abstract
Background: To describe and evaluate a unilateral microscopic surgical approach for bilateral
decompression and removal of massive lumbar disc herniations causing incomplete cauda equina
syndrome, with the dual objective of rapid neurological recovery and minimization of postoperative
segmental instability.
Methods: A retrospective review was conducted of all patients treated over the past eight years by the
senior authors in whom this technique was employed to reverse presenting symptomatology and reduce
the risk of postoperative instability.
Result: Fourteen patients presenting with massive lumbar disc herniation and incomplete cauda equina
syndrome underwent surgery using the described approach. In all cases, bilateral neural decompression
was achieved without technical difficulty, resulting in marked enlargement of the spinal canal and
no radiographic evidence of postoperative instability. Neurological signs and symptoms improved
immediately following surgery. At a minimum follow-up of one year in nine of the fourteen patients, no
complaints suggestive of recurrent disc herniation or segmental instability were observed.
Conclusion: The unilateral microscopic approach employed by us for bilateral decompression and
removal of massive lumbar disc herniations associated with incomplete cauda equina syndrome
represents a safe, effective and stability-preserving alternative to more invasive decompression or fusion
techniques currently in use.
Keywords: Unilateral microscopic approach; Bilateral lumbar discectomy; Massive lumbar disc herniation; Cauda equina syndrome
Introduction
Patients presenting with chronic low back pain and lower limb numbness who suddenly experience symptom exacerbation accompanied by urinary retention or other signs of cauda equina compression require urgent Magnetic Resonance Imaging (MRI) [1,2]. When imaging reveals significant lumbar canal stenosis secondary to a massive disc herniation at the lower lumbar levels, these patients are at considerable risk of progressing to complete cauda equina injury, which may result in irreversible neurological damage [3-11]. Recent studies have demonstrated that the lumbar spinal canal diameter in such cases is often narrower than that observed in patients with asymptomatic disc herniations [12]. Furthermore, many of these massive herniations are associated with osteophytic growths that further compromise canal diameter. Therefore, it appears logical to take advantage of disc removal to simultaneously enlarge the stenotic canal. Importantly, patients with the aforementioned clinical presentation should not be confused with individuals who display similar radiological findings but lack clinical features of cauda equina compression. The latter should, as a rule, undergo initial conservative management before surgical intervention is considered [13-16]. However, given the medical and legal implications associated with cauda equina syndrome, urgent surgical decompression is indicated whenever even partial or incomplete cauda equina syndrome is present in conjunction with MRI evidence of massive dural sac compression at the affected lumbar level. A variety of surgical strategies have been reported in the literature, including posterior decompression with or without discectomy [8,10,11] decompression combined with Posterior Lumbar Interbody Fusion (PLIF), [17] Transforaminal Lumbar Interbody Fusion (TLIF) [18,19] and more recently, endoscopic excision with or without fusion [7,8]. To the best of our knowledge and understanding, a microscopic technique that allows the removal of a herniated lumbar disc bilaterally through a unilateral approach has not yet been described in the literature. Considering the favourable outcomes achieved with the unilateral microscopic approach described herein, the authors present their experience and provide a detailed account of the technique.
Methods
A retrospective analysis was performed of all cases treated over the last eight years in which the described unilateral microscopic approach was utilized. The objective was to alleviate presenting neurological deficits while minimizing the risk of postoperative segmental instability. Due to the urgency of surgical decompression, the severity of neurological deficits and the retrospective nature of the analysis-conducted with full anonymization of patient dataformal informed consent for study inclusion was not obtained. Medication type and dosage at admission were recorded and served as additional parameters in evaluating treatment efficacy. Pain intensity at admission and during treatment was routinely documented using the Visual Analog Scale (VAS), ranging from 0 (no pain) to 10 (worst imaginable pain) [20]. Functional disability was assessed using the Oswestry Disability Index (ODI) [21] which consists of 10 items addressing back pain intensity and its impact on daily activities. Each item includes six response options scored from 0 (no disability) to 5 (maximum disability). ODI was recorded at admission, at discharge and during follow-up controls. A minimum difference of 15 points between preoperative and postoperative scores was interpreted as a clinically significant improvement. All patients underwent urgent MRI at admission due to symptoms suggestive of cauda equina involvement. Digitally archived studies were used to categorize the degree of lumbar disc herniation using the AFFM-YOLOv8 system [22]. Given the notable overweight status of most patients at admission, Body Mass Index (BMI) and habitual physical activity levels were also recorded.
Patient population
All patients were admitted on an emergency basis. They reported progressive exacerbation of chronic low back pain and sciatica over several months prior to admission, which had become intolerable and immobilizing. Regardless of the presence or absence of sensorimotor deficits in the lower extremities, all patients reported urinary or fecal dysfunction (retention or incontinence), as well as other manifestations of cauda equina syndrome, including perianal or saddle anesthesia and in some cases, erectile dysfunction.
Surgical technique
Disc removal was performed using a modification of the “overthe- top” technique to achieve bilateral decompression through a unilateral microscopic approach in patients with lumbar spinal canal stenosis [23-26]. Under general anesthesia, a minimal midline skin incision was made over the affected level. Sharp dissection of the fascia from the midline was followed by blunt mobilization of the paravertebral muscles laterally, allowing placement of a unilateral muscle retractor. After exposure of the interlaminar window, the surgical level was confirmed using intraoperative fluoroscopy. A limited sublaminar undercutting of the superior and inferior lamina was initiated on the side of dominant radicular pain. The ligamentum flavum was perforated bluntly and removed, along with any granulation tissue contributing to canal narrowing, until a small window was created permitting visualization of the dural sac. Subarticular undercutting of the ipsilateral facet was performed to expose the corresponding nerve root at the disc level, completing ipsilateral decompression. After medial retraction of neural elements, the posterior longitudinal ligament was incised ipsilaterally and herniated disc material was removed as medially as possible using angled disc forceps. Following ipsilateral decompression, the operating table was tilted approximately 25 degrees contralaterally and the microscope repositioned. The base of the spinous process was thinned using a high-speed drill. Selective microscopic bony decompression of the contralateral posterior arch was performed by drilling the inner surfaces of the lamina and facet joint with a 2-mm burr, creating an approximately 3-mm working corridor around the contralateral posterior aspect of the dural sac. With gentle depression of the dural sac, the contralateral nerve root trajectory could be visualized. Using a fine, angled nerve hook, the surgeon explored residual disc prominence and compression. When significant contralateral compression was confirmed, particularly in medially herniated discs with osteophytic overgrowth, the contralateral posterior longitudinal ligament was incised and residual disc material was removed using inverted angled forceps and a 2mm Kerrison punch for osteophytes. At completion, the dural sac and nerve roots were bilaterally decompressed, demonstrating visible expansion and pulsation. Final epidural hemostasis was achieved with bipolar coagulation. A subfascial, non-suction drain was placed and the wound was closed in layers, with intracutaneous absorbable skin suturing.
Postoperative management
Patients were mobilized 12 hours after surgery without restrictions in range of motion. Prior to discharge, standard lumbar spine radiographs were obtained to exclude postoperative segmental instability. In obese patients, a hypocaloric diet tailored to individual weight reduction was initiated. During wound healing, isometric exercises targeting paraspinal and abdominal musculature were introduced to promote spinal stabilization.
Results
Nine of the fourteen patients completed a minimum followup of one year. The mean age at admission was 48 years; ten patients were female and four males. All patients underwent progressively intensified physiotherapy in the weeks following surgery until restoration of normal physiological mobility was achieved. Notably, most patients shared similar clinical characteristics, including obesity (mean body mass index [BMI] 33), low levels of physical activity, working-age status (23-65 years) and a prior history of chronic low back pain and sciatica in the months preceding admission, frequently accompanied by medication overuse. Symptoms of neurogenic claudication were often masked by baseline physical inactivity. Sensorimotor deficits of the lower extremities were present in only half of the patients and were typically of recent onset. Magnetic Resonance Imaging (MRI) demonstrated remarkable similarity across cases. In the
Illustrative Cases
Case 1
majority (10 patients), degenerative pathology was confined to a single lumbar level, most commonly between L4 and L5, with less frequent extension to the lumbosacral level. All patients exhibited chronic, massive subligamentous disc migration at the affected level. In twelve cases, this was accompanied by osteophytic deformation of adjacent vertebral bodies, resulting in absolute spinal canal stenosis with compression and stretching of cauda equina fibers. According to the AFFM-YOLOv8 MRI-based grading system, two patients presented with type 2B disc extrusion (broad-based, circumferential extension partially exceeding the facet line), while twelve patients exhibited type 3A extrusion, in which disc material occupied nearly the entire canal and extended beyond facet margins. Preoperatively, nine patients presented with bladder retention and incontinence consistent with Cauda Equina Syndrome-Retention (CES-R), whereas five demonstrated incomplete bladder dysfunction consistent with Cauda Equina Syndrome-Incomplete (CES-I). All of them improved after surgery until they achieved complete remission. Functional and pain outcomes were highly favorable. The mean preoperative Oswestry Disability Index (ODI) was 84.08, decreasing to 37 at first postrehabilitation follow-up. Mean preoperative Visual Analog Scale (VAS) score for back pain was 8.4, improving to 2.78 at follow-up. Mean preoperative VAS for leg pain was 8.8, improving to 1.67. No complications were identified that would necessitate additional studies or any form of surgical revision.
Figure 1:The preoperative T2W magnetic resonance imaging mid sagittal (a) and axial (b) views showing the massive disc herniation. Intraoperative photomicrographs: c: showing ipsilateral disc removal to the surgical approach with a medially angled disc forceps and the aspirator assisting in the retraction of the dural sac; d: after tilting the table about 25 degrees exposure of the contralateral residual disc herniation using a fine, angled nerve hook; c: extraction of the contralateral disc material
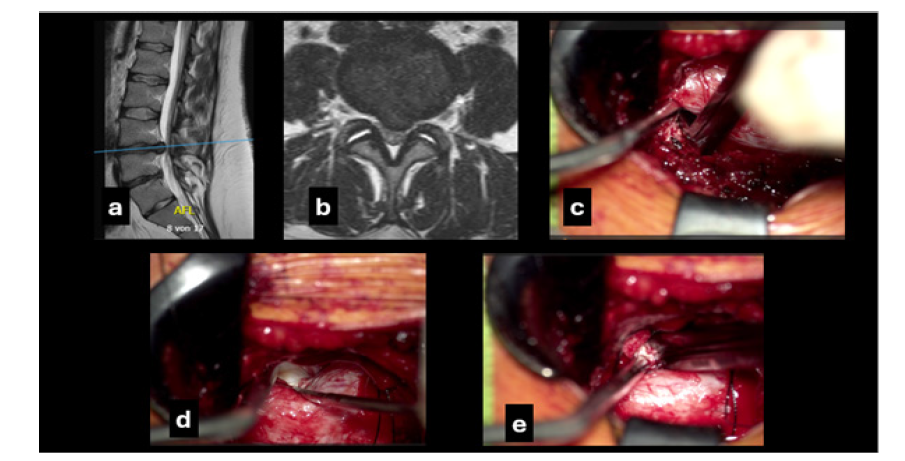
A 34-year-old obese female (BMI 33.7) presented emergently with severe immobilizing low back pain (VAS 9) and left anterior thigh and lower leg pain (VAS 7), present for several weeks, with occasional milder contralateral symptoms. ODI was 82. She received ibuprofen 800mg three times daily and tapentadol 50 mg twice daily without relief. Neurological examination revealed left L4 motor deficit (knee extension 3/5), left thigh hypoesthesia and urinary incontinence. MRI demonstrated a massive L4-L5 disc herniation classified as type 2B (AFFM-YOLOv8), with lumbarization of S1. Postoperatively, complete remission of neurological deficits was observed, along with progressive weight reduction. Postrehabilitation ODI was 48. VAS improved to 3 for back pain and 2 for leg pain (Figure 1).
Case 2
A 23-year-old obese female (BMI 35.8) was admitted emergently with severe low back pain (VAS 8) and left S1 sensorimotor deficit (VAS 9). She reported three months of progressive back pain and lower limb numbness, followed by one week of stress urinary incontinence. ODI was 87. Her medical history included depressive episodes with cyclothymia and posttraumatic stress disorder under treatment. Chronic opioid use and medication overuse were documented. Neurological examination revealed left L5-S1 hypoesthesia, reduced perineal sensation and left foot plantar flexion weakness (3/5). MRI showed progression of a previously identified L4-L5 disc herniation with subtotal canal obstruction, classified as type 3A (AFFM-YOLOv8). Surgery was performed urgently. The procedure was technically successful and complication-free. Although medication overuse required gradual regulation, neurological deficits almost completely resolved postoperatively. ODI after rehabilitation was 39. VAS improved to 4 for back pain and 3 for leg pain, accompanied by progressive weight reduction (Figure 2).
Figure 2:The preoperative T2W magnetic resonance imaging mid sagittal (a) and axial (b) views showing the massive disc herniation at the level L4-L5; (c) intraoperative lateral X-ray showing the unilateral muscle retractor and a Kocher clamp applied to the L4 lamina to confirm the level of access. Intraoperative photomicrographs: (d) shows how the sequestered disc tissue can be progressively and ipsilaterally removed in one large block with a disc forceps and without additional retractions; (e) after tilting the table about 25 degrees and positioning two cotton pads above and below the contralateral disc space and only aided in retraction by the tip of the suction device, the residual protrusion of the posterior longitudinal ligament is being incised with a scalpel to extract the herniated disc.
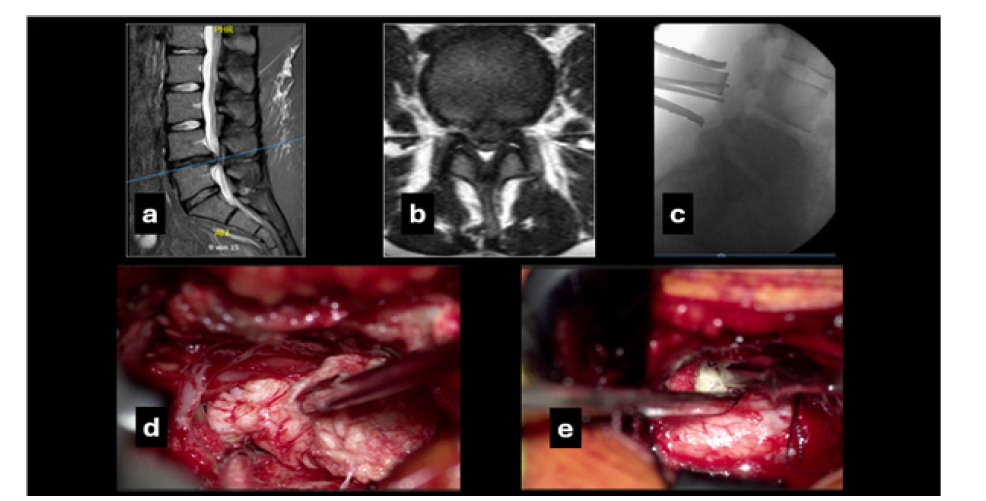
Case 3
A 65-year-old obese male (BMI 31.3) presented emergently with severe immobilizing low back pain (VAS 8) and bilateral sciatica, more pronounced on the right (VAS 9). He reported months of right lumbosacral radiculopathy and recent bowel and bladder dysfunction. Neurological examination revealed right foot dorsiflexion weakness (2/5), right L5-S1 hypoesthesia and decreased perineal sensation. MRI demonstrated a massive L4-L5 disc herniation with subtotal canal obstruction, classified as type 3A (AFFM-YOLOv8). Postoperatively, bowel and bladder function improved and neurological deficits regressed significantly. At follow-up, complete neurological recovery and progressive weight reduction were documented. Post-rehabilitation ODI was 41. VAS improved to 3 for back pain and 2 for leg pain (Figure 3).
Figure 3:The preoperative T2W magnetic resonance imaging mid sagittal (a) and axial (b) views showing the massive disc herniation and the osteophyte growth that accompanies disc extrusion at the level L4-L5; (c) coronal T2-weighted MRI of the lumbosacral spine. The bright vertical column in the midline represents Cerebro Spinal Fluid (CSF) within the thecal sac. The CSF column appears constricted at the L4-L5 level, with a focal indentation consistent with severe central canal stenosis produced by the large disc herniation combined with the degenerative changes. Intraoperative photomicrographs: (d) it shows how the herniated disc is being removed ipsilaterally with a disc forceps and the help of the aspirator tip, which is slightly retracting the dural sac; (e) after tilting the table about 25 degrees and having partially removed the material from the contralateral disc, the osteophytes are removed with a Kerrison punch, while the nerve and dural structures are held slightly apart with the tip of the aspirator.
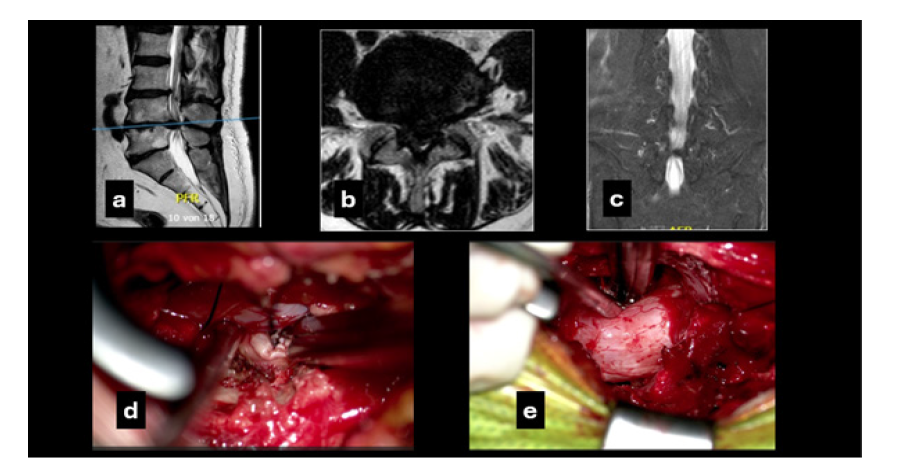
Discussion
In contrast to typical degenerative lumbar spinal stenosis, patients with massive anteromedial disc herniation and secondary canal stenosis often develop acute neurological deterioration superimposed on chronic symptoms. They frequently present emergently with severe lumbago, bilateral radiculopathy and not uncommonly incomplete cauda equina syndrome. The presence of bladder and bowel dysfunction in the context of lumbar disc herniation generally mandates urgent surgical decompression, ideally within 48 hours, to optimize neurological recovery [27]. Although delayed improvement has been reported [28,29] expectant management carries a significant risk of irreversible neurological damage [3-8,30-32]. The patient population analyzed in this study presented substantial comorbid challenges, including obesity, diabetes, hypertension and frequent polypharmacy. Obesity increases the risk of wound complications, recurrent herniation and postoperative instability. Nevertheless, the progression to cauda equina syndrome leaves no reasonable alternative to surgical intervention. The choice of surgical techniques remains critical. Isolated laminectomy without instrumentation may fail to address the specific pathology observed in these patients [33]. Similarly, simple posterior decompression with or without discectomy may be insufficient in obese individuals, given the risk of recurrent herniation and instability.
On the other hand, prophylactic fusion in the absence of true instability may accelerate adjacent segment degeneration and initiate a cascade of further fusion procedures. Therefore, interbody fusion techniques such as posterior lumbar interbody fusion [17] or transforaminal lumbar interbody fusion [18,19] should be reserved for cases with demonstrable instability. Bilateral partial laminectomy with bilateral discectomy [34] has become widely adopted as a less invasive approach for the treatment of midline disc herniations and is likely the technique most used by surgeons in these patients. In recent years, however, increasing interest has emerged in decompression via Unilateral Biportal Endoscopy (UBE). This technique has been reported as effective for the treatment of lumbar spinal stenosis combined with MSU-1 lumbar disc herniation and has also been applied in patients presenting with cauda equina syndrome [35-38]. Without diminishing its merits, Yuan et al. [39] analyzed UBE and identified several disadvantages when comparing it with single channel foraminal endoscopic techniques. Among the reported drawbacks were greater surgical trauma due to the creation of two incisions, muscle detachment from the vertebral lamina, establishment of two operative corridors and partial laminar resection. These factors may contribute to increased bleeding, a higher risk of iatrogenic injury and postoperative anemia. A direct comparison between UBE and the microscopic technique employed in our series remains challenging. Although UBE provides enhanced visualization and a smaller access corridor for disc removal, the underlying technical principles differ substantially, as do the clinical and surgical characteristics of the patient populations reported to date.
Our microscopic approach enables direct in situ visualization and creates an expanded working space using an over-the-top drilling technique, thereby allowing less traumatic manipulation of compressed neural structures. In contrast, UBE relies on the creation of a viewing portal for the endoscopic camera and a separate working portal for surgical instruments, typically utilizing continuous saline irrigation under pressures of 30-40mmHg to maintain an operative field. Beyond these conceptual differences in decompression strategy, the surgical challenges also vary. The removal of extruded and sequestered discs within the spinal canal, as described by Cheetri et al. [37] or most migrated discs, as reported by Park et al. [38], differs substantially from the management of massively contained disc herniations constrained by the posterior longitudinal ligament and frequently associated with osteophytic overgrowth, as observed in most of our patients. Patient characteristics further complicate direct comparisons. Obesity was prevalent in our cohort, with a mean body mass index of 33, whereas only two of the 32 patients in Park’s series reached comparable values [38]. A noteworthy distinction concerns complication rates. In our series, no complications requiring further investigation or surgical revision were observed. In contrast, Park et al. [38] reported incidental durotomy (6.3%), facet joint injury (6.3%) and asymptomatic radiographic hematoma (21.9%). These findings may suggest that UBE, given its relatively narrow working corridor, requires extensive decompression and tissue manipulation to avoid surgery-related injuries. Additionally, the etiology of persistent lower-limb dysesthesia reported in some UBE-treated patients remains insufficiently clarified. Unlike microscopic techniques, UBE commonly employs radiofrequency cauterization for intraoperative hemostasis, which may represent a potential contributing factor. Nonetheless, ongoing refinements of UBE-including variations enabling bilateral discectomy [40,41]-underscore the evolving role and versatility of endoscopic approaches. Despite their advantages, however, these techniques involve a considerable learning curve, increased equipment costs and potential limitations in emergency settings [42]. Furthermore, during early adoption, endoscopic approaches may carry a higher risk of dural injury or cerebrospinal fluid leakage and can be technically demanding in cases with severe inflammatory adhesions or calcified disc material [43,44].
In contrast, the modified microscopic unilateral over-the-top decompression technique presented in this study allows bilateral removal of massively extruded lumbar discs, as well as resection of anterior osteophytes contributing to canal narrowing. This bilateral canal enlargement preserves most posterior stabilizing structures. In cases of circumferential compression with prominent anteromedial disc extrusion, this strategy may be preferable to extensive bilateral laminectomy. Preservation of extra canalicular osteophytic bridges located anteriorly and laterally, along with the posterior facet-laminar complex, contributes to postoperative segmental stability. Considering that emergency discectomy has been associated with higher recurrence and early readmission rates [45], thorough bilateral removal of degenerative disc material (as performed in our series), may contribute to reducing recurrence. The rapid clinical improvement observed in our patients was consistently reflected in ODI and VAS scores at one-year follow-up. Structured rehabilitation and progressive postoperative weight reduction likely contributed to the absence of subsequent fusion procedures, recurrent herniations or segmental instability. We believe that reducing spinal mechanical stress through weight loss and occupational modification further alleviated pain and may have delayed or partially reversed degenerative changes at adjacent levels. Ultimately, optimal outcomes depend not only on timely and technically appropriate surgical intervention but also on comprehensive postoperative management aimed at mitigating modifiable risk factors and preventing recurrence.
Conclusion
The unilateral microscopic approach employed by us for bilateral decompression and removal of massive lumbar disc herniations associated with incomplete cauda equina syndrome represents a safe, effective and stability-preserving alternative to more invasive decompression or fusion techniques currently in use.
References
- Hussain MM, Razak AA, Hassan SS, Choudhari KA, Spink GM (2018) Time to implement a national referral pathway for suspected cauda equina syndrome: Review and outcome of 250 referrals. Br J Neurosurg 32(3): 264-268.
- Motiei-Langroudi R, Sadeghian H, Ekanem UO, Safdar A, Grossbach AJ, et al. (2023) Predicting the need for surgery in patients with lumbar disc herniation: A new internally validated scoring system. Asian Spine J 17(6): 1059-1065.
- Kumar V, Baburaj V, Rajnish RK, Dhatt SS (2022) Outcomes of cauda equina syndrome due to lumbar disc herniation after surgical management and the factors affecting it: A systematic review and meta-analysis of 22 studies with 852 cases. Eur Spine J 31(2): 353-363.
- Louison R, Barber JB (1968) Massive herniation of lumbar discs with compression of the cauda equina--a surgical emergency; Report of two cases. J Natl Med Assoc 60(3): 188-190.
- Hogan WB, Kuris EO, Durand WM, Eltorai AE, Daniels AH (2019) Timing of surgical decompression for cauda equina syndrome. World Neurosurg 132: e732-e738.
- Lam J, De Souza R, Laycock J, Perera D, Burford C, et al. (2020) Patient-reported bladder, bowel and sexual function after cauda equina syndrome secondary to a herniated lumbar intervertebral disc. Spinal Cord Inj Rehabil 26(4): 290-303.
- Yunoki M, Umakoshi M, Tatano M, Imoto R (2021) Cauda equina syndrome due to lumbar disc herniation and ossification of the yellow ligament. Surg Neurol Int 12: 202.
- Ahn UM, Ahn NU, Buchowski MS, Garrett ES, Sieber AN, et al. (2000) Cauda equina syndrome secondary to lumbar disc herniation: A meta-analysis of surgical outcomes. Spine 25: 1515-1522.
- Nakamura K, Arizono T, Inokuchi A, Hamada T, Imamura R (2021) Massive lumbar disc herniation causing cauda equina syndrome that presents as bladder and bowel dysfunction in the absence of lower extremity weakness. Cureus 13(9): e17952.
- Jeon CH, Chung NS, Son KH, Lee HS (2013) Massive lumbar disc herniation with complete dural sac stenosis. Indian J Orthop 47(3): 244-249.
- Dhole K, Sakhare K, Sakharkar N, Agrawal P, Chaudhary S, et al. (2025) Late presentation of cauda equina syndrome with isolated bladder bowel incontinence in massive lower lumbar disc herniation: A series of three cases. J Orthop Case Rep 15(2): 95-100.
- Korse NS, Kruit MC, Peul WC, Vleggeert-Lankamp CLA (2017) Lumbar spinal canal MRI diameter is smaller in herniated disc cauda equina syndrome patients. PLoS One 12(10): e0186148.
- Hong SJ, Kim DY, Kim H, Kim S, Shin KM, et al. (2016) Resorption of massive lumbar disc herniation on MRI treated with epidural steroid injection: A retrospective study of 28 cases. Pain Physician 19(6): 381-388.
- U EC, Shetty A, Craig PR, Chitgopkar SD (2018) An observation of massive lumbar disc prolapse. J Spine Surg 4(3): 583-587.
- Fan Z, Xu N, Qi J, Su H, Wang H (2023) Regression of a large prolapsed lumbar disc herniation achieved by conservative treatment: A case report and literature review. Heliyon 9(9): e20041.
- Naidoo D (2021) Spontaneous and rapid resolution of a massive lumbar disc herniation. Surg Neurol Int 12: 352
- Satoh I, Yonenobu K, Hosono N, Ohwada T, Fuji T, et al. (2006) Indication of posterior lumbar interbody fusion for lumbar disc herniation. J Spinal Disord Tech 19(2): 104-108.
- Zhao CQ, Ding W, Zhang K, Zhao J (2016) Transforaminal lumbar interbody fusion using one diagonal fusion cage with unilateral pedicle screw fixation for treatment of massive lumbar disc herniation. Indian J Orthop 50(5): 473-478.
- Gupta A, Chhabra HS, Nagarjuna D, Arora M (2021) Comparison of functional outcomes between lumbar interbody fusion surgery and discectomy in massive lumbar disc herniation: A retrospective analysis. Global Spine J 11(5): 690-696.
- Scott J, Huskisson EC (1976) Graphic representation of pain. Pain 2(2): 175-184
- Fairbank JC, Pynsent PB (2000) The Oswestry Disability Index. Spine 25(22): 2940-2952.
- Wang Y, Yang Z, Cai S, Wu W, Wu W (2025) MRI grading of lumbar disc herniation based on AFFM-YOLOv8 system. Sci Rep 15(1): 32880.
- Mayer HM, Korger A (2009) Microsurgical decompression of degenerative lumbar spinal stenosis. Eur Spine J 18(12): 1989-1990.
- Sasai K, Umeda M, Maruyama T, Wakabayashi E, Iida H (2008) Microsurgical bilateral decompression via a unilateral approach for lumbar spinal canal stenosis including degenerative spondylolisthesis. J Neurosurg Spine 9(6): 554-559
- Raffo CS, Lauerman WC (2006) Predicting morbidity and mortality of lumbar spine arthrodesis in patients in their ninth decade. Spine 31(1): 99-103.
- Schöller K, Steingrüber T, Stein M, Vogt N, Müller T, et al. (2016) Microsurgical unilateral laminotomy for decompression of lumbar spinal stenosis: Long-term results and predictive factors. Acta Neurochir 158(6): 1103-1113.
- Kalidindi KK, Sath S, Vishwakarma G, Chhabra HS (2020) Magnetic resonance imaging findings in intervertebral disc herniation: Comparison of canal compromise and canal size in patients with and without cauda equina syndrome. Surg Neurol Int 11: 171.
- Lai XW, Li W, Wang JX, Zhang HJ, Peng HM, et al. (2017) Delayed decompression for cauda equina syndrome secondary to lumbar disc herniation: Long-term follow-up results. Nan Fang Yi Ke Da Xue Xue Bao 37(9): 1143-1148.
- Sath S (2020) Does surgical decompression alleviate neglected cauda equina syndromes attributed to lumbar disc herniation and/or degenerative canal stenosis? Surg Neurol Int 11: 278.
- Chau AM, Xu LL, Pelzer NR, Gragnaniello C (2014) Timing of surgical intervention in cauda equina syndrome: A systematic critical review. World Neurosurg 81(3-4): 640-650.
- Dhatt S, Tahasildar N, Tripathy SK, Bahadur R, Dhillon M, et al. (2011) Outcome of spinal decompression in cauda equina syndrome presenting late in developing countries: Case series of 50 cases. Eur Spine J 20: 2235-2239.
- Aly TA, Aboramadan MO (2014) Efficacy of delayed decompression of lumbar disk herniation causing cauda equina syndrome. Orthopedics 37(2): e153-e156.
- Lawal YZ, Maitama MI, Ejagwulu FS, Lawal A (2019) Open laminectomy for lumbar spinal canal stenosis due to prolapsed lumbar intervertebral discs. West Afr J Med 36(1): 83-87.
- Eule JM, Breeze R, Kindt GW (1999) Bilateral partial laminectomy: A treatment for lumbar spinal stenosis and midline disc herniation. Surg Neurol 52(4): 329-337.
- Zhao F, Qiu X, Yuan J, Liu R, Wei X, et al. (2025) Early effectiveness of posterior 180-degree decompression via unilateral biportal endoscopy in treatment of lumbar spinal stenosis combined with MSU-1 lumbar disc herniation. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi 39(6): 735-740.
- Hung WP, Kavishwar RA, Kim HS, Jie Chin BZ (2023) Technical feasibility of combined uniportal unilateral laminotomy with bilateral decompression and interlaminar lumbar discectomy surgery for cauda equina syndrome due to lower lumbar disc herniation. N Am Spine Soc J 16: 100290.
- Chhetri P, Thapa BB, Shakya AR, Rai B (2025) Cauda equina syndrome due to lumbar disc herniation managed with unilateral biportal endoscopic discectomy: A case report and review of literature. Int J Surg Case Rep 137: 112-115.
- Park SM, Lee HJ, Park HJ, You KH, Jung JH, et al. (2025) Biportal endoscopic lumbar discectomy surgery in patients with cauda equina syndrome caused by lumbar herniated intervertebral disc: A retrospective multi-center cohort study. J Orthop Surg Res 20(1): 172.
- Yuan C, Wen B, Lin H (2022) Clinical analysis of minimally invasive percutaneous treatment of severe lumbar disc herniation with ube two-channel endoscopy and foraminal single-channel endoscopy technique. Oxid Med and Cell Longev.
- Meng SW, Peng C, Zhou CL, Tao H, Wang C, et al. (2021) Massively prolapsed intervertebral disc herniation with interlaminar endoscopic spine system Delta endoscope: A case series. World J Clin Cases 9(1): 61-70.
- Feng Z, Zhao Z, Cui W, Meng X, Hai Y (2024) Unilateral biportal endoscopic discectomy versus microdiscectomy for lumbar disc herniation: A systematic review and meta-analysis. Eur Spine J 33(6): 2139-2153.
- Alostaz M, Derman P, Lipson P, Du J, Gardocki R, et al. (2025) Attitudes regarding barriers to entry and the learning curve associated with endoscopic decompression-only surgery: An international survey. Spine J 25(5): 983-995.
- Yadav YR, Parihar V, Kher Y, Bhatele PR (2016) Endoscopic interlaminar management of lumbar disease. Asian J Neurosurg 11(1): 1-7.
- Zhou C, Zhang G, Panchal RR, Ren X, Xiang H, et al. (2018) Unique complications of percutaneous endoscopic lumbar discectomy and percutaneous endoscopic interlaminar discectomy. Pain Physician 21(2): E105-E112.
- Reito A, Kyrölä K, Pekkanen L, Paloneva J (2020) 30-Day recurrence, readmission rate and clinical outcome after emergency lumbar discectomy. Spine 45(18): 1253-1259.
© 2026 Mario Carvi Nievas. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






