- Submissions

Full Text
Surgical Medicine Open Access Journal
Simultaneous Multisystemic Tuberculosis and Metastatic Ductal Carcinoma: An Uncommon Case Report
Amina El Alami¹*, Oumaima Fakir³, Othmane Echarfaoui², Hanaa Lazhar³, Aziz Slaoui³, Aziz Baidada³ and Jaouad Kouach²
¹Gynaecology-Obstetrics and Endocrinology Maternity Souissi, Mohammed V University, Morocco
²Department of Gynecology-Obstetrics, Mohammed V University, Morocco
³Gynaecology-Obstetrics and Endoscopy Maternity Souissi, Mohammed V University, Morocco
*Corresponding author:Amina El Alami, Gynaecology-Obstetrics and Endocrinology Maternity Souissi, Mohammed V University, Rabat, Morocco
Submission: January 15, 2026;Published: January 30, 2026

ISSN 2578-0379 Volume6 Issue2
Abstract
Background: Morocco is considered an endemic country to tuberculosis and breast cancer as they
both are considered major health issues. Their coexistence is scarce and makes diagnosis exacting since
symptoms, clinical and imaging investigations are non-specific of neither of these two pathologies.
Case report: We hereby present one of the rare cases exhibiting the coexistence of a metastatic breast
carcinoma in a patient with multisystemic tuberculosis.
Conclusion: Although clinical and imaging is not specific, histopathology bestows a definitive diagnosis.
An astute clinical judgement is required to ensure that the diagnosis is made as early as possible so that
treatment can be initiated promptly.
Keywords:Tuberculosis-Ductal; Carcinoma-Anatomopatholoy
Abbreviations:TST: Mantoux Tuberculin Skin Test; TB: Tuberculosis; BT: Breast Tuberculosis; CT: Computed Tomography; SBR: Bloom-Richardson System; ER: Estrogen Receptor; PR: Progesterone Receptor; MCM: Multidisciplinary Consultation Meeting; WHO: World Health Organization
Introduction
Bayle first described the association of TB and carcinoma almost 200 ago [1]. The coexistence of tuberculosis and breast cancer, as rare as it is, has been extremely haunting for clinicians, radiologists, surgeons, anatomopathologists and oncologists. The absence of pathognomonic symptoms alongside clinical and imaging findings are straining the path towards an accurate diagnosis. Histological and bacteriological means are very beneficent. The coexistence of these two pathologies in one organism makes the course of treatment challenging. Through this article, we disclose a case of multisystemic tuberculosis coexisting with an invasive metastatic ductal carcinoma.
Case Report
We hereby present the case of a 54 years old woman referred by the pulmonology unit to the gynecology department for a consult after fortuitously discovering a high malignancy risk lesion in her mammogram. The patient had been menopausal for the last 6 years after one vaginal delivery 20 years ago. She breastfed for 6 months. The patient never complained of any breast lump and had no nipple discharge. She never had a proper screening of cervical nor breast cancer. She was following a 6 months antibiotic course for pulmonary Tuberculosis (TB) that was confirmed based on a positive Mantoux Tuberculin Skin Test (TST) followed by a sputum analysis. She was admitted 3 days before at the TB unit subsequently to a drastic worsening of her general state of health. She complained of a 15 kilograms weight loss in the last 4 months, fatigue, chronic lumbar pain, profuse sweating and intermittent fever despite responsibly taking her antibiotics (isoniazid and rifampicin).
Upon her complaints, a sputum smear test was performed which came back positive and a whole Body-Scan was conducted. Thoracic Computed Tomography (CT) showed a calcified micronodule in the outer segment of the middle lobe, bilateral axillary nodes ranging from 0.5 to 9mm and a right mammary macrocalcification. Abdominal and pelvic CT revealed an abnormal hypodense lesion located on the right anteroinferior liver segment (V) with focal biliary tract enlargement associated with bilateral simple kidney cysts (Bosniak 1). Her pelvic CT showed an endometrial thickening of 17mm. Bone Window CT exhibited multiple peripheral and axial skeletal osteoblastic and osteolytic lesions particularly seated on the vertebral body of the 5th lumbar vertebrae disrupting the cortex. In order to further explore her mammary findings, a sonomammography was performed manifesting bilateral heterogenous hypoechogenic lesions with irregular borders associated to calcifications in the right mammary gland. Mammography on cranio-caudal and oblique projection showed an ill-circumcised lesion with mild density and fibrous spicules of the medial upper quadrant in left breast and an oval mass with wild density with fibrous spicules and macrocalcification in the upper outer quadrant in the right breast surrounded by edema (Figure 1).
Figure 1:Mammography on cranio-caudal and oblique projection showing: a) Blue arrow: ill-circumcised lesion with mild density and fibrous spicules of the medial upper quadrant in left breast and an oval mass with wild density with fibrous spicules b) White arrow: macrocalcification in the upper outer quadrant in the right breast surrounded by edema.
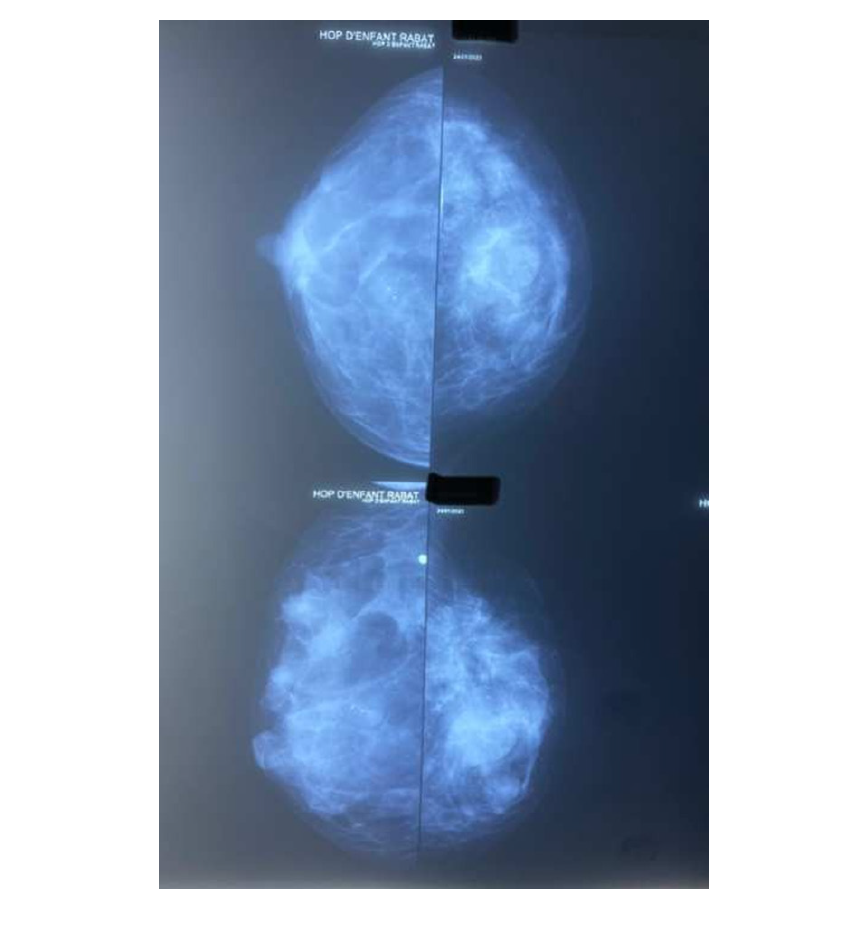
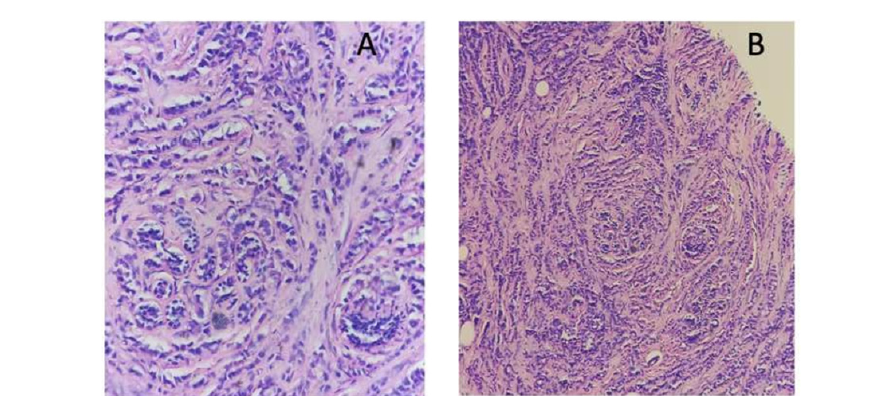
Based on the results above, the patient was referred to our gynecological outpatient’s unit for probable breast tuberculosis. General and physical examination reported a cachectic patient. Her temperature was 37.7° Celsius and her vitals were within normal range. Breast examination revealed bilateral multiple palpable masses with blurry limits. There was no ulceration or retraction of the adjacent skin. There was no nipple retraction. Many bilateral lymph nodes were felt upon axillary examination. A trucut biopsy on right mammary gland confirmed an invasive ductal carcinoma. according to the Nottingham modification of the Bloom- Richardson System (SBR), the tumor was SBR grade II (Figure 2). Immunohistochemistry expression of the Estrogen Receptor (ER) and Progesterone Receptor (PR) was positive. Herceptest staining was negative. An ultrasound-guided biopsy of the liver and hysteroscopy-guided biopsy of the endometrium were performed concluding to a metastasizing ductal carcinoma. Multidisciplinary Consultation Meeting (MCM) decided that the best course of action would be palliative chemotherapy and antituberculous drugs with a very poor prognosis.
Figure 2:Representative micrographs of the breast tumor (hematoxylin-eosin): a) Tumor cells are arranged in single files, cords and single cells, concentrically around normal ducts (targetoid appearance) b) Tumor cells are discohesive, monomorphic and lacking marked atypia.
Discussion
Tuberculosis and breast cancer are major health issues. The World Health Organization (WHO) states breast cancer as the world’s most prevalent cancer with 2.3 million women diagnosed in 2020 and TB as the ninth leading cause of death worldwide with over 25% of TB deaths occurring in the African region. Morocco is considered an endemic country with a total of 29327 cases of TB diagnosed in 2021 declared by the Ministry of Health and Social Protection and 11747 cases of female breast cancer of all ages in 2020 [2,3]. In previously conducted literature research, there are many reported cases about TB being misdiagnosed [4]. Paradoxically, in our case, a metastatic invasive breast cancer went unnoticed because of a pre-existing systemic TB.
Breast Tuberculosis (BT) is an uncommon localization of Mycobacterium tuberculosis bacillus infection [5]. BT is commonly a secondary location subsequent to an underlining infection via a direct hematogenous spread [6,7]. On a molecular level, malignancy is developed in an environment prone to chronic inflammatory conditions increasing the risk for genetic errors such as the relationship between gastroesophageal reflux and oesophageal cancer [8]. However, there is no evidence that tuberculosis is carcinogenic [9,10]. The relationship between TB and breast malignancy is set to be very complex and can be categorized in 3 different types of association: Either cancer is developed on the background of a previous tuberculous infection, or their coexistence is incidental, or tuberculosis is complicating a pre-existing cancer secondary to the immunosuppressive effects of therapy [11]. In our case, a pre-existent pulmonary tuberculosis was complicated 4 months after initial diagnosis by a metastatic invasive ductal carcinoma.
Nick-named a “great masquerader” by Gon and al, the nonspecific clinical and imaging characteristics of TB alongside clinical and imaging similarities between TB and malignancy are the primary cause of misled diagnosis [12,13]. Typical symptoms of TB such as general fatigue fever, night sweats and weight loss are rarely present. As a matter of fact, symptoms vary among patients [14,15]. As it is, our patient had many constitutional typical TB symptoms: Weight loss accounted for 15 kilograms, fever as intermittent as it is, profuse sweating and a notable declining of her general health. Therefore, the aforementioned symptoms led to believe first and foremost to a non-response to first-line anti-tuberculosis treatment. This hypothesis was emphasized by the positive sputum smear test. Physical examination in BT usually finds an isolated breast mass -without an associated sinus- usually firm, ill-defined, irregular with probable fixation to the skin mimicking a breast cancer [16]. Moreover, the presence of associated axillary adenopathy complicates the diagnosis. Besides, the absence of nipple and areola complex involvement is rare in BT [6,15]. Ergo, regarding our patient, one of the criteria that led to suspect a BT was the presence of bilateral axillary adenopathies after a period of 4 months and the integrity of the skin and areola.
Imaging features of BT are non-specific. BT ultrasonography usually shows heterogenous hypoechogenic lesions with irregular borders as it was the case in our case study [16,17]. Mammography findings are as blurry and confusing as ultrasonography. It usually displays many dense solitary lesions with fibrous spicules surrounded by edema and nipple retraction which is similar to mammography findings in breast carcinoma [16,17]. Regarding our case, mammography showed similar characteristics. Nevertheless, the presence of calcifications was the main reason why our hypothesis shifted towards a breast cancer rather than a breast tuberculosis. Aside from mammo-sonography, Computed Tomography (CT) and Magnetic Resonance Imaging (MRI) do not improve diagnostic accuracy [14]. Given the abstruseness and equivocacy of clinical and imaging findings, histopathologic and bacteriological proof are sustainable means to confirm a diagnosis. The detection of acid-fast bacilli is pathognomonic but unfortunately may be missing sometimes [15]. The low number of mycobacteria in clinical specimen alongside the non-specificity of conventional methods in many studies are the main factor inflicting such high numbers of misdiagnosed cases [18].
The key element for a differential diagnosis is biopsy of the lesion justifying the tru-cut biopsy in our patient [19]. One of the most crucial key-points is the importance of axillary lymph nodes metastasis in breast carcinoma staging. Since TB causes adenopathies, it mimics breast cancer and may lead to an overstaging, ergo unfitting therapy course [20]. Therapy wise, there are two situations regarding the co-existence of systemic TB and breast cancer. In one hand, if breast cancer is clinically operable, radical mastectomy should be performed, followed by post-operative antituberculous chemotherapy for 18 months. In the other hand if cancer is incurable, palliative measures combined with antituberculous drugs are indicated [21]. The latter was the course of treatment chosen by our department after consulting with the oncology department.
Conclusion
It’s safe to say that the co-existence of tuberculosis and breast cancer in the same patient or even in the same breast is very rare and raises difficulties in diagnostic and therapeutic management [22]. Nevertheless, all clinicians should be aware of the challenges regarding a proper and prompt diagnosis given the clinical and radiological similarities between breast cancer and breast tuberculosis. Histological and bacteriological tools are the cornerstones to confirm a diagnosis. Multidisciplinary management is a requirement in order to offer optimal therapy and follow-up. This work has been reported in line with the SCARE 2020 criteria [23].
Declarations
Conflicts of interest
N/A.
Sources of funding
N/A.
Ethical approval
Ethics approval has been obtained to proceed with the current study.
Consent
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in- Chief of this journal.
Guarantor of submission
The corresponding author is the guarantor of submission.
Acknowledgement
None.
Availability of data and materials
Supporting material is available if further analysis is needed.
Provenance and peer review
Not commissioned, externally peer-reviewed.
References
- Patil S, Gajanan H (2014) Cavitary lung disease: Not always due to tuberculosis! primary lung cancer with smear positive pulmonary tuberculosis-a case report. American Journal of Medical Case Reports 2(8): 164-166.
- (2021) The global cancer observatory, international agency for research on cancer. World Health Organization.
- (2021) Breast cancer.
- Tian-Xing H, Fang G, Huang Y, Chun-Mei H, Chen W, et al. (2020) Misdiagnosis of a multi-organ involvement hematogenous disseminated tuberculosis as metastasis: A case report and literature review. Infect Dis Poverty 9: 66.
- Oucharqui S, Adil H, Benaissa E, Bssaibis F, En-Nafaa I, et al. (2021) Breast tuberculosis: A forgotten diagnosis. ID Case 26: e01341.
- Shinde SR, Chandawarkar RY, Deshmukh SP (1995) Tuberculosis of the breast masquerading as carcinoma: A study of 100 patients. World J Surg 19(3): 379-381.
- Göksoy E, Düren M, Durgun V, Uygun N (1995) Tuberculosis of the breast. Eur J Surg 161(7): 471-473.
- Schottenfeld D, Beebe-Dimmer J (2006) Chronic inflammation: A common and important factor in the pathogeniss of neoplasia. Ca Cancer J Clin 56(2): 69-83.
- Salemis NS, Razou A (2010) Coexistence of breast cancer metastases and tuberculosis in axillary lymph nodes-A rare association and review of the literature. Southeast Asian J Trop Med Public Health 41(3): 608-613.
- Bouhout T, Serji B, Egyir EU, Amri BE, Bouhout I, et al. (2017) Breast tuberculosis: about a case. Pan Afr Med J 28: 183.
- Rizzo G, Colli F, Marco PD, Brocca AL, Militello G, et al. (2020) An unusual presentation of breast tuberculosis: A case report. Clin Case Rep 9(1): 210-212.
- Gon S, Bhattacharyya A, Majumdar B, Kundu S (2013) Tubercular mastitis: A great masquerader. Turk Patoloji Derg 29(1): 61-63.
- Gupta S, Singh VJ, Bhatia G, Dhuria K (2014) Primary tuberculosis of the breast manifested as abscess: A rare case report. Acta Med Indones 46(1): 51-53.
- Akçay MN, Sağlam L, Polat P, Erdoğan F, Albayrak Y, et al. (2007) Mammary tuberculosis-importance of recognition and differentiation from that of a breast malignancy: Report of three cases and review of the literature. World J Surg Oncol 5: 67.
- Khanna R, Prasanna GV, Gupta P, Kumar M, Khanna S, et al. (2002) Mammary tuberculosis: Report on 52 cases. Postgrad Med J 78(921): 422-424.
- Harris SH, Khan MA, Khan R, Haque F, Syed A, et al. (2006) Mammary tuberculosis: Analysis of thirty-eight patients. ANZ J Surg 76(4): 234-237.
- Lee CH, Sharif SZ, (2016) Primary Breast Tuberculosis (TB) in a patient with known invasive breast carcinoma: A case report. Med J Malaysia 71.
- Kayali S, Alhamid A, Kayali A, Danial AK, Alsaman MZB, et al. (2020) Primary tuberculous mastitis: The first report from Syria. Int J Surg Case Rep 68: 48-51.
- Miller RE, Salomon PF, West JP (1971) The coexistence of carcinoma and tuberculosis of the breast and axillary lymph nodes. Am J Surg 121(3): 338-340.
- Pujani M, Khan S, Hassan MJ, Jetley S, Raina PK, et al. (2015) Coexistence of metastatic breast carcinoma and primary tuberculosis in axillary lymph nodes: A report of a rare case. Breast Dis 35(3): 195-198.
- Pamateck S, Sidibe BY, Diakité I, Saye Z, Darar IA, et al. (2022) Association of metastasis and axillary lymph node tuberculosis in breast cancer: Clinical case and review of the literature. Research 11: 101-108.
- Chottanapund S, Wongtawatchai P (2004) Tuberculosis axillary lymphadenitis coexistence in patient with invasive ductal carcinoma of the breast: A case report. The Thai Journal of Surrgery 25: 121-124.
- Agha RA, Franchi T, Sohrabi C, Mathew G, Kerwan A, et al. (2020) The scare group the SCARE 2020 guideline: Updating consensus Surgical Case Report (SCARE) guidelines. Int J Surg 84: 226-230.
© 2026 Amina El Alami. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






