- Submissions

Full Text
Significances of Bioengineering & Biosciences
Artificial Intelligence in Multimodal Retinal Imaging: A Systematic Review of Diagnostic Applications in Ocular and Systemic Diseases
Ameh Benson Agi1, Micheal Abimbola Oladosu2*, Moses Adondua Abah3, Dominic Agida Ochuele3, Abimbola Mary Oluwajembola2, Olaide Ayokunmi Oladosu4, Bukola Oluwaseyi Olufosoye5 and Olamide Yosola Falana6
1Department of Chemistry, College of Science, University of Siegen, Germany
2Department of Chemical Sciences, Faculty of Science, Anchor University, Nigeria
3Department of Biochemistry, Faculty of Pure and Applied Sciences, Federal University of Wukari, Nigeria
4Department of Computer Science, Faculty of Science and Technology, Babcock University, Nigeria
5Department of Medical Microbiology, Faculty of Medical Laboratory Sciences, Ambrose Alli University, Nigeria
6Department of Public Health, Teesside University, UK
*Corresponding author:Micheal Abimbola Oladosu, Department of Chemical Sciences, Faculty of Science, Anchor University, Ayobo, Ipaja, Lagos, Nigeria
Submission: November 19, 2025; Published: March 11, 2026

ISSN 2637-8078Volume7 Issue 5
Abstract
Progress in Artificial Intelligence (AI) has transformed medical imaging, with retinal imaging becoming a crucial diagnostic tool for ocular and systemic illnesses. This systematic review examines the incorporation of AI in multimodal retinal imaging, which includes fundus photography, Optical Coherence Tomography (OCT), OCT angiography, and fluorescein angiography, along with its diagnostic applications for conditions such as diabetic retinopathy, age-related macular degeneration, glaucoma, and systemic diseases like cardiovascular and neurodegenerative disorders. A thorough examination of contemporary AI models reveals elevated sensitivity and specificity, frequently matching or surpassing that of experienced ophthalmologists. The paper emphasises AI’s ability to automate illness detection, improve early diagnosis, and facilitate scalable screening in resource-constrained environments. Nevertheless, obstacles persist, such as restricted dataset heterogeneity, absence of multicentre validation, and complications with clinical integration and algorithm transparency. Future investigations should prioritise the creation of different datasets, the standardisation of validation methodologies, and the development of interpretable AI systems that integrate effortlessly into clinical processes. AI-enhanced multimodal retinal imaging offers substantial potential for revolutionising precision diagnoses in ophthalmology and systemic medicine..
Keywords:Artificial intelligence; Multimodal retinal imaging; Diabetic retinopathy; Systemic diseases; Deep learning
Introduction
Artificial Intelligence (AI) has rapidly transformed the field of medical imaging, offering new opportunities for enhanced diagnosis, prognosis, and disease monitoring. In ophthalmology, multimodal retinal imaging, which integrates data from diverse imaging modalities such as Optical Coherence Tomography (OCT), fundus photography, fluorescein angiography, and OCT angiography, provides comprehensive insights into retinal health. Leveraging AI, particularly machine learning and deep learning algorithms, to analyse these multimodal datasets has significantly improved the detection and classification of both ocular diseases (e.g., diabetic retinopathy, age-related macular degeneration, and glaucoma) and systemic conditions (e.g., hypertension, diabetes, and neurodegenerative disorders) with retinal manifestations [1,2]. This systematic review explores the diagnostic applications of AI in multimodal retinal imaging, highlighting how these technologies enhance diagnostic accuracy, enable early disease detection, and support clinical decisionmaking in both ocular and systemic healthcare contexts [3,4].
Retinal imaging and its diagnostic potential
Retinal imaging has become a crucial diagnostic instrument in ophthalmology and systemic medicine because of the retina’s distinctive accessibility and its representation of vascular and neurological health. Conventional methods, including fundus photography, Fluorescein Angiography (FA), and Optical Coherence Tomography (OCT) have historically been employed to identify and observe retinal diseases. Fundus photography offers a comprehensive picture of the retinal surface, essential for identifying diabetic retinopathy, age-related macular degeneration, and hypertensive retinopathy [5]. Fluorescein angiography provides high-resolution images of the retinal blood vessels, facilitating the detection of ischemia, neovascularization, and leakage [6]. Among these technologies, OCT is distinguished by its non-invasive, cross-sectional imaging capabilities, facilitating detailed observation of retinal layers and structural alterations at a micron scale [7]. It is particularly essential for the diagnosis and monitoring of macular oedema, glaucoma, and retinal detachment. Recently, OCT Angiography (OCTA) has enabled clinicians to evaluate microvascular circulation without dye injection, hence enhancing early diagnosis and illness management [8].
Retinal imaging extends beyond ocular disorders. Research indicates that retinal biomarkers, specifically vessel diameter, tortuosity, and microaneurysms, may signify systemic illnesses such as diabetes, hypertension, cardiovascular disease, and cognitive decline, including Alzheimer’s disease [9-11]. The capacity to identify systemic disorders using retinal imaging is gaining acknowledgement, as the retina serves as a “window” into the body’s microcirculation. The incorporation of Artificial Intelligence (AI) is enhancing the efficacy of retinal imaging. AI algorithms can autonomously identify disease characteristics with elevated sensitivity and specificity, providing scalable and economical solutions for screening and diagnosis [1,12]. Consequently, the diagnostic capability of retinal imaging is being enhanced, reinforcing its increasing significance in personalised medicine and public health.
AI in Ophthalmology
Artificial Intelligence (AI) is transforming ophthalmology by augmenting diagnostic precision, optimising screening efficacy, and facilitating individualised patient treatment. Utilizing Machine Learning (ML) and Deep Learning (DL) methodologies, AI systems can efficiently analyse extensive ophthalmic imaging data, including fundus photographs, Optical Coherence Tomography (OCT), and fluorescein angiography, exceeding the speed and accuracy of conventional manual interpretation [12]. A significant use of AI in ophthalmology is the early diagnosis of Diabetic Retinopathy (DR). AI algorithms trained on retinal fundus pictures can detect diabetic retinopathy with sensitivity and specificity akin to that of human specialists, providing scalable screening options, particularly in resource-constrained environments [1]. AI techniques have been created for glaucoma identification by examining optic nerve head morphology and retinal nerve fibre layer thickness in OCT scans [13]. In Age-Related Macular Degeneration (AMD), artificial intelligence models facilitate classification and progression forecasting by examining retinal anatomy and patterns of fluid buildup [14]. Additionally, AI facilitates retinal vascular analysis to deduce systemic illnesses, including cardiovascular risk, anaemia, and neurological disorders such as Alzheimer’s disease [9,10]. Artificial intelligence is progressively incorporated into clinical decision support systems, allowing ophthalmologists to triage patients more effectively and prioritise urgent cases. In addition to diagnoses, AI enables automated treatment planning, such as scheduling anti-VEGF medication for macular degeneration, by forecasting disease activity based on sequential imaging data [14]. With ongoing regulatory approvals and real-world validation, AI is anticipated to play a pivotal role in precision ophthalmology, enhancing accessibility and diminishing preventable blindness worldwide [3].
Objective of the Review
The main aim of this systematic review is to rigorously
assess and integrate current literature about the use of Artificial
Intelligence (AI) in multimodal retinal imaging for diagnosing
ocular and systemic illnesses. The review specifically intends to:
i. Classify and delineate the many AI algorithms employed
in retinal image processing across distinct imaging modalities,
such as fundus photography, OCT, OCT Angiography (OCTA),
and Fluorescein Angiography (FA).
ii. Evaluate the diagnostic efficacy (e.g., sensitivity,
specificity, accuracy) of AI-driven systems in identifying retinal
disorders, including diabetic retinopathy, glaucoma, and agerelated
macular degeneration.
iii. Investigate the function of retinal imaging
artificial intelligence in the identification of systemic
diseases, encompassing cardiovascular, metabolic, and
neurodegenerative disorders.
iv. Assess the clinical efficacy, obstacles, and constraints of
incorporating AI into standard ophthalmic practice.
v. Outline prospective avenues for research and development
in AI-enhanced retinal diagnostics, emphasising multimodal
integration and personalized therapy.
Methodology
Research design
This work was conducted as a systematic review in accordance with the PRISMA 2020 standards to discover, assess, and synthesize current literature about the utilization of Artificial Intelligence (AI) in multimodal retinal imaging for diagnostic applications in ocular and systemic illnesses.
Search strategy
A thorough literature search was performed across the
electronic databases: PubMed, Scopus, Web of Science, and IEEE
Xplore, encompassing papers from January 2010 to May 2025. The
search phrases comprised various combinations of the specified
keywords and Boolean operators:
a. “Artificial Intelligence” OR “AI” OR “Machine Learning” OR
“Deep Learning”
b. “Retinal Imaging” OR “Fundus Photography” OR “Optical
Coherence Tomography” OR “OCT” OR “OCT Angiography” OR
“Fluorescein Angiography”
c. “Diagnosis” OR “Detection”
d. “Ocular Disease” OR “Systemic Disease”
Reference lists of selected papers were meticulously examined to locate further pertinent studies.
Inclusion criteria
Studies were incorporated if they:
i. Utilised AI algorithms to examine retinal pictures for
diagnostic applications.
ii. Employed a minimum of one retinal imaging technique
(e.g., fundus photography, OCT, OCTA, or fluorescein
angiography).
iii. Concentrated on the identification or categorisation of
ocular or systemic ailments.
iv. Documented quantitative performance indicators (e.g.,
accuracy, sensitivity, specificity, AUC).
v. Published in peer-reviewed journals and composed in
English.
Exclusion criteria
Studies were omitted if they:
i. Were editorials, reviews, letters, or conference abstracts
lacking full papers?
ii. Concentrated on animal models or non-human datasets.
iii. Did not report diagnostic performance measures.
iv. Were either duplicated or did not possess full-text
accessibility.
Data extraction and administration
Two independent evaluators assessed all titles and abstracts according to the established inclusion and exclusion criteria. Subsequently, full-text publications were examined for final inclusion. Data were retrieved into a standardised format encompassing: authors and year of publication; imaging modality utilised; applied AI methodology/architecture; target ailments; characteristics of the dataset (dimensions, origin); performance indicators (e.g., sensitivity, specificity, area under the curve); and verification techniques. Discrepancies were rectified by dialogue or arbitration by a third reviewer.
Evaluation of quality
The methodological quality of the included studies was evaluated using the QUADAS-2 technique (Quality Assessment of Diagnostic Accuracy Studies). Each study was assessed across four domains: Patient selection, index test, reference standard, and flow and timing.
Data integration
A narrative synthesis was conducted because of the variability in AI models, imaging techniques, and outcome metrics. Research was categorised by imaging technique and illness classification. Summary tables were generated to compare diagnostic performance across studies where adequate uniformity was present.
Types of Retinal Imaging Modalities
Progress in retinal imaging has facilitated the collection of high-resolution, multimodal data essential for diagnosing and monitoring ocular and systemic illnesses. The predominant imaging modalities utilised in artificial intelligence-assisted diagnostics are summarised below and in Table 1.
Table 1:Imaging modalities, clinical use, and key references.
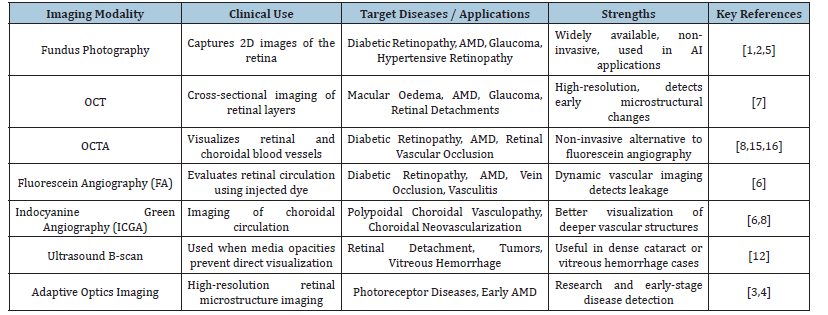
Fundus imaging
Fundus photography produces two-dimensional colour photographs of the retina, offering an extensive view of the optic disc, macula, blood vessels, and retinal periphery. It is extensively utilized for the screening and diagnosis of conditions such as Diabetic Retinopathy (DR), Age-Related Macular Degeneration (AMD), and glaucoma. Artificial intelligence algorithms, especially Convolutional Neural Networks (CNNs), have demonstrated exceptional efficacy in identifying pathological characteristics such as microaneurysms, haemorrhages, and neovascularization in fundus pictures [1,2].
Optical Coherence Tomography (OCT)
OCT is a non-invasive imaging modality that delivers crosssectional images of the retina with micrometer precision [7]. It facilitates comprehensive viewing of the retinal layers, permitting early identification of structural alterations linked to disorders such as AMD, macular oedema, and glaucoma. Deep learning models have been created to categorise OCT images and segment pathological characteristics, including fluid accumulation and retinal layer thinning, with great precision [6,15].
Retinal Angiography
Retinal angiography encompasses several techniques: Fluorescein Angiography (FA) involves the administration of fluorescein dye to obtain dynamic pictures of retinal blood circulation, mostly utilised for identifying leakage, ischemia, and neovascularization [6]. Indocyanine Green Angiography (ICGA) is employed to elucidate choroidal circulation, particularly advantageous in the diagnosis of choroidal neovascular membranes. Optical Coherence Tomography Angiography (OCTA) is a dye-free technique that uses motion contrast to visualise blood flow in the retina and choroid, enabling the detection of microvascular abnormalities [8,15]. AI-based interpretation of angiographic images has been applied for vessel segmentation, detection of nonperfusion areas, and classification of disease severity in diabetic retinopathy, retinal vein occlusion, and hypertensive retinopathy [16]. Table 1 highlights the Imaging Modalities and Clinical Use.
AI Models Used in Diagnosis
The incorporation of Artificial Intelligence (AI) into multimodal retinal imaging has markedly improved the diagnosis of ocular and systemic disorders in terms of speed, precision, and scalability. Diverse AI models have been created and refined for distinct imaging modalities and disease-specific applications.
Convolutional Neural Networks (CNNs)
Convolutional Neural Networks (CNNs) are the predominant deep learning architecture employed in retinal image analysis. They are especially proficient in autonomously extracting hierarchical characteristics from fundus pictures and OCT images. CNNs have attained expert-level proficiency in identifying diabetic retinopathy, age-related macular degeneration, and glaucoma from fundus images [1,2]. CNNs are also employed for the segmentation of retinal layers and the identification of biomarkers, including drusen, fluid pockets, and retinal thinning in OCT scans [6,16].
Transformers
Transformers, first designed for natural language processing, have lately been repurposed for medical imaging owing to their capacity to simulate long-range dependencies in data. Vision Transformers (ViTs) have been utilised for fundus and OCT images, demonstrating performance that is comparable to or exceeds that of CNNs, especially in tasks necessitating spatial context and multimodal data integration [17,18]. Transformers offer enhanced interpretability and generalizability, particularly in multicentre datasets characterised by varying image quality.
Hybrid models
Hybrid models integrate Convolutional Neural Networks (CNNs) with other architectures such as Recurrent Neural Networks (RNNs), Transformers, or attention processes to improve diagnostic precision. The integration of CNNs and RNNs facilitates the analysis of time-series OCT data for predicting temporal illness progression [18]. Transformer-CNN hybrids are utilized to integrate funds and OCT properties for comprehensive analysis [19]. Fusion techniques in hybrid models can amalgamate multimodal imaging data, clinical metadata, and genomic information to generate more comprehensive diagnostic predictions [20-22].
Disease-specific models
AI systems are frequently trained and refined for certain diseases with extensive annotated datasets. For Diabetic Retinopathy (DR), Google’s research team and other groups have built robust CNN models that classify DR into multiple severity levels [1,6]. For Age-Related Macular Degeneration (AMD), OCT-based artificial intelligence models can identify AMD and distinguish between its dry and wet variants [14,23]. For glaucoma, AI models utilizing optic nerve head and retinal nerve fibre layer data can assess intraocular pressure and the risk of visual field loss [13,20]. Systemic disease AI models can identify cardiovascular risk factors, anaemia, and chronic kidney disease through fundus images [10,11]. Figure 1 presents the AI pipeline for Retina image analysis, while Table 2 gives the Summary of AI Models and Target Diseases.
Figure 1:AI Pipeline for retinal image analysis. Deep learning-based automated retinal image analysis system showing classified retinal fundus images according to the International Clinical Diabetic Retinopathy Severity Scale, including the algorithm’s training, validation, and testing workflow. DR=Diabetic Retinopathy; NPDR=Non-Proliferative Diabetic Retinopathy; PDR=Proliferative Diabetic Retinopathy [19].
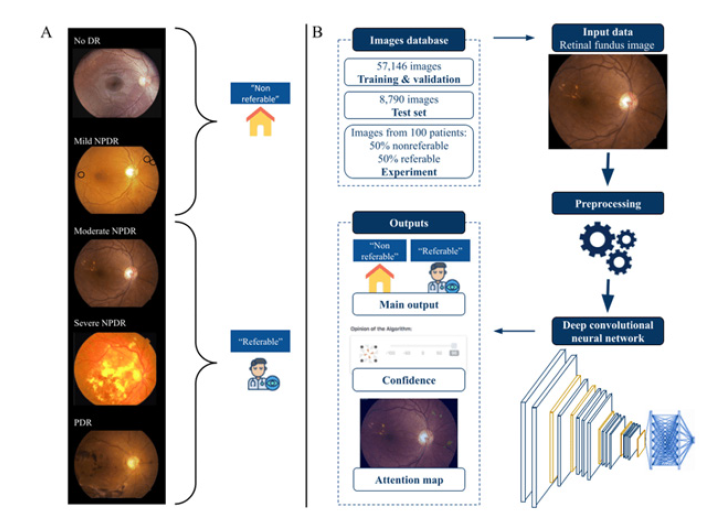
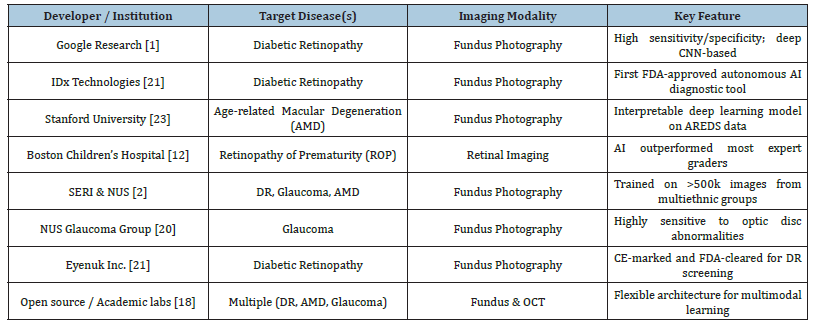
Table 2:Summary of AI models and target diseases.
Multi Disease Detection Applications
Multidisease detection applications refer to software tools or systems, frequently utilizing Artificial Intelligence (AI), Machine Learning (ML), and/or data analytics, that can concurrently detect or forecast many diseases from medical data. These applications are increasingly pertinent in contemporary healthcare owing to the escalating demand for efficient, precise, and scalable diagnostic solutions.
Diabetic retinopathy
Diabetic Retinopathy (DR) is a primary contributor to preventable blindness among diabetic individuals, advancing through microvascular injury to the retina. AI-driven retinal image analysis can identify diabetic retinopathy in conjunction with other visual conditions such as glaucoma, as well as cardiovascular risk factors inferred from retinal vasculature [1,2]. IDx-DR is an FDAsanctioned artificial intelligence system for independent diabetic retinopathy screening [21]. Google’s system identifies more than 50 ocular disorders, including diabetic retinopathy [1,6].
Glaucoma
Glaucoma is a persistent optic neuropathy marked by elevated intraocular pressure and loss of vision fields, frequently advancing without symptoms. Detection generally encompasses OCT imaging, visual field evaluation, and optic nerve head examination, all of which are compatible with AI-driven multimodal systems [13,20]. Certain AI platforms evaluate glaucoma alongside diabetic retinopathy and AMD, and some further associate ocular biomarkers with neurodegenerative disorders [2,12].
Cardiovascular and neurodegenerative markers
Cardiovascular markers, including atrial fibrillation, myocardial infarction, and heart failure, are major contributors to global mortality. AI models can evaluate these conditions, while retinal imaging AI systems also assess cardiovascular risks from fundus images [10,11]. For neurodegenerative conditions such as Alzheimer’s and Parkinson’s disease, AI evaluates brain imaging (MRI, PET), speech patterns, gait, and digital cognitive assessments to detect neurodegeneration, which can be integrated with cardiovascular and ocular data for comprehensive health monitoring [9,11]. Multi disease detection applications are revolutionising clinical diagnostics by enabling simultaneous, AI-assisted screening for several illnesses. These technologies enhance diagnostic efficiency and accuracy while facilitating early intervention, especially in disadvantaged or resource-constrained areas. The integration of imaging, signal processing, and intelligent algorithms signifies a potential future for personalized and comprehensive healthcare [3,4,22].
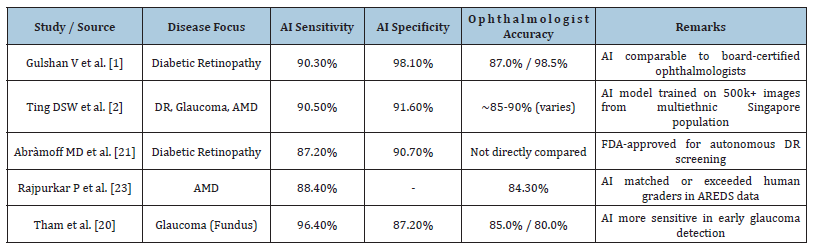
Comparison with Human Graders
AI-driven multidisease detection systems have demonstrated diagnostic efficacy comparable to, and in certain instances exceeding, that of qualified human evaluators. For example, Google’s deep learning system for detecting diabetic retinopathy attained sensitivity and specificity akin to those of board-certified ophthalmologists [1]. Likewise, FDA-sanctioned systems such as IDx-DR have equalled or surpassed the precision of human clinicians in practical applications [21]. These AI solutions provide benefits in efficiency, uniformity, and scalability, particularly in resource-constrained environments where human evaluators are restricted. In contrast to human experts, AI is unaffected by fatigue or inconsistencies among observers. Nonetheless, human evaluators demonstrate superiority in interpretability, contextual clinical reasoning, and managing uncommon or borderline cases, domains in which AI continues to encounter difficulties [22]. A hybrid paradigm that combines the efficiency of AI with human expertise is widely regarded as the ideal approach for clinical implementation [3,12]. Table 3 highlights the Diagnostic Accuracy of AI Models, while Table 4 gives the Comparison between AI vs. Ophthalmologist Accuracy Rates.
Table 3:Diagnostic accuracy of AI models (sensitivity, specificity).
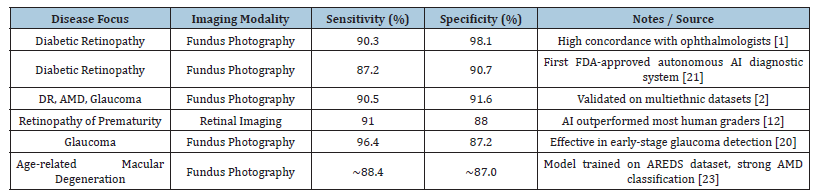
Table 4:AI vs. ophthalmologist accuracy rates.
Research Gaps
Notwithstanding substantial progress, some essential deficiencies persist in the advancement and implementation of multi disease detection applications:
Dataset diversity
Many current AI models are developed using datasets that exhibit inadequate variety in race, age, disease severity, and imaging modalities. This constrains generalizability across diverse demographics and healthcare environments. Many retinal image datasets are derived from individual countries or hospital systems, heightening the likelihood of algorithmic bias and diminishing efficacy among underrepresented populations [2,22].
Multicenter validation
A deficiency exists in extensive, prospective multicenter validation studies. Numerous algorithms are evaluated in isolated, controlled settings and not thoroughly examined across diverse geographic regions, institutions, and device types (Table 5). In the absence of multicenter validation, evaluating the actual performance, robustness, and reproducibility of these systems is challenging [12,22].
Table 5:Future research recommendations.
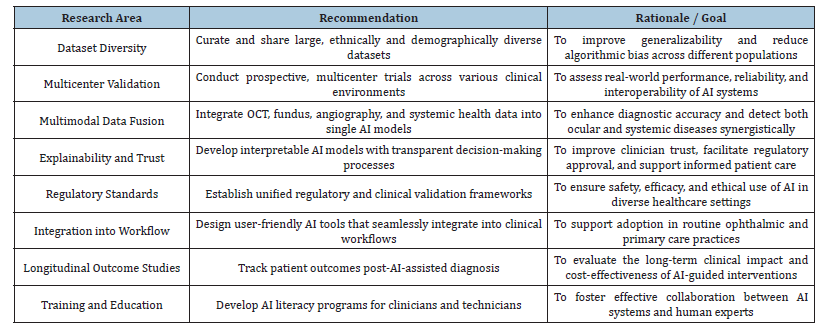
Explainability and clinical integration
Many AI models function as “black boxes,” making it difficult for clinicians to understand and trust their outputs. Developing interpretable and explainable AI architectures is critical for regulatory approval and safe clinical deployment [3,22]. Table 4 presents the future research and recommendations.
Conclusion
Artificial Intelligence (AI) has become a transformative instrument in multimodal retinal imaging, markedly improving the diagnostic framework for ocular and systemic illnesses. By integrating deep learning algorithms with advanced imaging techniques such as fundus photography, OCT, and angiography, AI systems have exhibited expert-level precision in identifying conditions such as diabetic retinopathy, glaucoma, age-related macular degeneration, and systemic diseases, including cardiovascular and neurodegenerative disorders. This systematic analysis emphasizes that AI provides significant benefits regarding speed, scalability, and consistency, rendering it especially beneficial in screening programs and resource-constrained environments. Furthermore, multimodal imaging techniques enhance diagnostic accuracy by acquiring complementary structural and vascular data from the retina. Notwithstanding these advancements, the review also highlights significant limitations, including insufficient dataset diversity, restricted multicenter validation, and issues related to explainability and clinician trust. These deficiencies must be rectified to guarantee enhanced generalizability, equity, and secure clinical integration. In summary, AI-enhanced multimodal retinal imaging possesses significant potential to transform early disease identification and individualised healthcare. Future research must prioritise enhancing data variety, executing extensive real-world validations, and promoting transparent AI systems that can be seamlessly incorporated into healthcare processes. A collaborative framework integrating AI efficiency with clinical experience is crucial for transforming this technology into sustainable and equitable healthcare solutions.
Acknowledgement
The authors acknowledge the contributions of researchers worldwide advancing AI applications in drug discovery and the open-source software community enabling these developments.
References
- Gulshan V, Peng L, Coram M, Stumpe MC, Wu D, et al. (2016) Development and validation of a deep learning algorithm for detection of diabetic retinopathy in retinal fundus photographs. JAMA 316(22): 2402-2410.
- Ting DSW, Cheung CY, Lim G, Tan GSW, Quang ND, et al. (2017) Development and validation of a deep learning system for diabetic retinopathy and related eye diseases using retinal images from multiethnic populations with diabetes. JAMA 318(22): 2211-2223.
- Oganov AC, Seddon I (2023) Artificial intelligence in retinal image analysis: Development, advances, and challenges. Survey of Ophthalmology 68(5): 905-919.
- Chen KY, Chan HC, Chan CM. (2025) Can artificial intelligence with multimodal imaging outperform traditional methods in predicting age-related macular degeneration progression? A systematic review and exploratory meta-analysis. BMC Medical Informatics and Decision Making 25: 321.
- Abràmoff MD, Niemeijer M, Suttorp-Schulten MSA, Viergever MA, Russell SR, et al. (2008) Evaluation of a system for automatic detection of diabetic retinopathy from color fundus photographs in a large population of patients with diabetes. Diabetes Care 31(2): 193-198.
- De Fauw J, Ledsam JR, Romera-Paredes B, Nikolov S, Tomasev N, et al. (2018) Clinically applicable deep learning for diagnosis and referral in retinal disease. Nature Medicine 24(9): 1342-1350.
- Huang D, Swanson EA, Lin CP, Schuman JS, Stinson WG, et al. (1991) Optical coherence tomography. Science 254(5035): 1178-1181.
- De Carlo TE, Romano A, Waheed NK, Duker JS (2015) A review of Optical Coherence Tomography Angiography (OCTA). International Journal of Retina and Vitreous 1(1): 5.
- Cheung CY, Xu D, Cheng CY, Sabanayagam C, Tham YC, et al. (2021) Retinal microvascular changes and risk of dementia in a multi-ethnic Asian population: The Singapore epidemiology of eye diseases study. JAMA Network Open 4(5): e218758.
- Poplin R, Varadarajan AV, Blumer K, Liu Y, McConnell MV, et al. (2018) Prediction of cardiovascular risk factors from retinal fundus photographs via deep learning. Nature Biomedical Engineering 2(3): 158-164.
- Wang J, Wang YX, Zeng D, Zhu Z, Li D, et al. (2025) Artificial intelligence-enhanced retinal imaging as a biomarker for systemic diseases. Theranostics 15(8): 3223-3233.
- Li JO, Liu H, Ting DSW, Jeon S, Chan RVP, et al. (2021) Digital technology, tele-medicine and artificial intelligence in ophthalmology: A global perspective. Progress in Retinal and Eye Research 82: 100900.
- Asaoka R, Murata H, Iwase A, Araie M, Fujino Y. (2019) Detecting preperimetric glaucoma with standard automated perimetry using deep learning classifier trained with retinal nerve fiber layer thickness data. Ophthalmology 126(1): 117-127.
- Schmidt-Erfurth U, Bogunović H, Sadeghipour A, Schlegl T, Langs G, et al. (2018) Machine learning to analyze the prognostic value of current imaging biomarkers in neovascular age-related macular degeneration. Ophthalmology Retina 2(1): 24-30.
- Spaide RF, Klancnik JM, Cooney MJ (2015) Retinal vascular layers imaged by optical coherence tomography angiography. JAMA Ophthalmology 133(1): 45-50.
- Yin X, Chen Y, Zhang Q, Wang RK, Li D (2020) Vessel segmentation in optical coherence tomography angiography images using a novel convolutional neural network architecture. IEEE Transactions on Medical Imaging 39(3): 731-743.
- Dosovitskiy A, Beyer L, Kolesnikov A, Weissenborn D, Zhai X, et al. (2021) An image is worth 16x16 words: Transformers for image recognition at scale. International Conference on Learning Representations (ICLR).
- Sükei E, Rumetshofer E, Schmidinger N (2024) Multi-modal representation learning in retinal imaging using self-supervised learning for enhanced clinical predictions. Scientific Reports 14: 26802.
- Noriega A, Meizner D, Camacho D, Enciso J, Quiroz-Mercado H, et al. (2021) Screening diabetic retinopathy using an automated retinal image analysis system in independent and assistive use cases in Mexico: Randomized controlled trial. JMIR Formative Research 5(8): e25290.
- Tham YC, Cheng CY, Wong TY (2020) Deep learning in detecting glaucomatous optic neuropathy on fundus photographs. Ophthalmology 127(8): 1091-1093.
- Abràmoff MD, Lavin PT, Birch M, Shah N, Folk JC (2018) Pivotal trial of an autonomous AI-based diagnostic system for detection of diabetic retinopathy in primary care offices. NPJ Digital Medicine 1(1): 39.
- Daich Varela M, Sen S, De Guimaraes TAC, Kabiri N, Pontikos N, et al. (2023) Artificial intelligence in retinal disease: Clinical application, challenges, and future directions. Graefe's Archive for Clinical and Experimental Ophthalmology 261(11): 3283-3297.
- Rajpurkar P, Irvin J, Ball RL, Zhu K, Yang B, et al. (2022) Deep learning for ophthalmology: Deep see net on age-related eye disease study (AREDS). NPJ Digital Medicine 5(1): 32.
© 2026 Micheal Abimbola Oladosu, This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






