- Submissions

Full Text
Significances of Bioengineering & Biosciences
Solar-and Wind-Powered Electrolysis Pathways for Green Hydrogen Production: A Comparative Assessment
Ameh Benson Agi1, Micheal Abimbola Oladosu2*, Moses Adondua Abah3, Clinton Arthur4, Salaudeen Mudashiru Opeyemi5, Timothy Kwame Asem6, Umeh Chisom7, Abimbola Mary Oluwajembola2, Olaide Ayokunmi Oladosu8 and Franklin Ogonna Ede9
1Department of Chemistry, College of Science, University of Siegen, Germany
2Department of Chemical Sciences, Faculty of Science, Anchor University, Nigeria
3Department of Biochemistry, Faculty of Pure and Applied Sciences, Federal University of Wukari, Nigeria
4Department of Chemistry, Faculty of Science, Eastern New Mexico University, USA
5Department of Mechanical Engineering, Faculty of Engineering and Technology, University of Ilorin, Nigeria
6Department of Chemistry and Biochemistry, Faculty of Science, University of Central Florida, USA.
7Department of Chemical Engineering, School of Engineering and Engineering Technology, Federal University of Technology Owerri, Nigeria
8Department of Computer Science, Faculty of Science and Technology, Babcock University, Nigeria
9Department of Computer Science, Faculty of Physical Sciences, University of Calabar, Nigeria
*Corresponding author:Micheal Abimbola Oladosu, Department of Chemical Sciences, Faculty of Science, Anchor University, Ayobo, Ipaja, Lagos, Nigeria
Submission: November 19, 2025; Published: March 02, 2026

ISSN 2637-8078Volume7 Issue 5
Abstract
Green hydrogen, generated through water electrolysis from renewable energy sources, is a crucial component in the worldwide shift towards a low-carbon future. This paper offers a thorough comparative evaluation of solar and wind-powered electrolysis systems, examining their technical efficacy, economic feasibility, and environmental consequences. Primary focal points encompass energy conversion efficiency, intermittency, hydrogen production yield, cost-effectiveness, and lifecycle emissions. The study emphasizes the benefits of Proton Exchange Membrane (PEM) electrolysers in accommodating fluctuating renewable energy sources and the distinct advantages of solar Photovoltaic (PV) and wind energy systems in various geographical and operational settings. The study examines system integration issues and hybrid configurations as methods to improve dependability and output continuity. The results offer practical guidance for politicians, investors, and engineers to facilitate the sustainable implementation of green hydrogen infrastructure according to particular geographical circumstances and energy characteristics.
Keywords: Green hydrogen; Electrolysis; Solar photovoltaic; Wind energy; PEM electrolyser, Renewable integration
Introduction
Green hydrogen, generated from renewable energy sources, has become a crucial facilitator of the worldwide shift towards a low-carbon economy. Among the diverse techniques for green hydrogen production, electrolysis driven by solar and wind energy emerges as the most viable approach owing to its sustainability, scalability, and alignment with decarbonization objectives [1-3]. Both technologies utilise electricity from renewable sources to electrolyse water into hydrogen and oxygen; however, their performance, cost-effectiveness, reliability, and environmental impact vary considerably between solar-powered and wind-powered electrolysis. This comparative analysis assesses the technological and economic feasibility of solar and wind energy for hydrogen generation, emphasising energy conversion efficiency, intermittency, cost per kilogram of hydrogen, geographical appropriateness, and lifecycle emissions. Comprehending these distinctions is essential for policymakers, investors, and engineers to choose the most suitable renewable resource for green hydrogen implementation across diverse geographies and applications.
Green hydrogen as a clean fuel
The escalating global energy demand, alongside rising concerns regarding climate change and fossil fuel depletion, has intensified the pursuit of sustainable and carbon-neutral energy carriers. Hydrogen has emerged as a critical solution due to its high energy density, versatility, and potential for zero-emission applications across multiple sectors [1-3]. When combusted or utilised in fuel cells, hydrogen produces only water vapour as a by-product, making it an ideal clean energy vector for decarbonising industries that are difficult to electrify directly. Currently, approximately 95% of global hydrogen production relies on fossil fuel-based processes, primarily Steam Methane Reforming (SMR), which generates substantial CO₂ emissions, approximately 9-12kg of CO₂ per kilogram of hydrogen produced [4]. This “grey hydrogen” contradicts global decarbonization efforts and climate targets. In contrast, green hydrogen, produced through water electrolysis powered entirely by renewable energy sources such as solar and wind, offers a truly sustainable pathway with near-zero carbon emissions throughout its lifecycle [1,3]. Green hydrogen presents transformative potential across diverse applications. In the transportation sector, hydrogen fuel cells can power heavy-duty vehicles, maritime vessels, and aviation, were battery electric alternatives face energy density limitations [1].
In industry, green hydrogen can replace fossil fuels and feedstocks in steel manufacturing, ammonia production, and chemical synthesis, enabling deep decarbonization of hard-to-abate sectors [1]. Additionally, hydrogen serves as an effective long-term energy storage medium, addressing the intermittency challenges of renewable electricity generation and facilitating grid stabilisation [3,5].
Despite its promise, the widespread adoption of green hydrogen faces several challenges. The technology remains more expensive than conventional hydrogen production methods, with costs heavily dependent on renewable electricity prices, electrolyser capital expenditure, and system efficiency [3,6]. Furthermore, the intermittent nature of solar and wind resources necessitates sophisticated integration strategies, including energy storage systems and hybrid configurations, to ensure continuous and economically viable hydrogen production [7]. Overcoming these technical and economic barriers is essential for establishing green hydrogen as a cornerstone of the global clean energy transition.
Overview of solar and wind-powered electrolysis
Electrolysis is a critical process in the creation of green hydrogen, a clean fuel that produces only water upon combustion. When fuelled by renewable energy sources such as solar and wind, electrolysis becomes a carbon-neutral technology that holds promise for decarbonising different sectors, including transportation, industry, and energy storage [1].
Principles of Electrolysis
Electrolysis is dividing Water (H₂O) into Hydrogen (H₂) and Oxygen (O₂) using an electric current. The process is aided by electrolysers, which may be classed into three basic types: Alkaline Electrolysers (AEL), Proton Exchange Membrane Electrolysers (PEM), and Solid Oxide Electrolysers (SOEC) [4]. Each method differs in operating circumstances, efficiency, and material needs, and all can be powered by renewable electricity.
Solar-powered electrolysis
Solar-powered electrolysis utilises electricity supplied from Photovoltaic (PV) panels or Concentrated Solar Power (CSP) systems to drive water splitting. PV systems are more often incorporated because of their modularity and lowering costs [8]. The efficiency of Solar-To-Hydrogen (STH) conversion is often restricted by the efficiency of both the PV cells and the electrolyser. Recent improvements in integrated photoelectrochemical systems have indicated potential for direct solar-to-hydrogen generation, but they are still mostly in the experimental phase [9].
Wind-powered electrolysis
Wind turbines generate power that can be used to operate electrolysers, especially in places with strong wind potential. Windpowered electrolysis systems can reach better capacity factors than solar-powered systems due to the more stable and often higher energy output of wind turbines, especially offshore. The intermittency of wind, however, can impair electrolyser functioning, which is why hybrid systems and energy storage technologies are commonly investigated [5].
Integration challenges and solutions
One of the biggest issues in linking intermittent renewable sources with electrolysis is the variable power supply, which can impair the operational efficiency and longevity of electrolysers [6]. Solutions include hybrid systems that combine solar and wind energy to smooth power input, energy storage (such as batteries or supercapacitors), and the use of advanced control systems to maximise electrolyser performance under varying loads [7]. Figure 1 shows the Basic diagram of electrolysis setups.
Figure 1:Basic diagram of electrolysis setups.
Technical framework
Electrolysis systems fuelled by solar and wind energy are engineered to transform renewable energy into hydrogen via the process of water splitting. The system typically consists of three fundamental components: the renewable energy source (solar photovoltaic or wind turbine), the power conditioning system, and the electrolyser unit.
Input of renewable energy
Solar-powered systems depend on Photovoltaic (PV) modules to produce Direct Current (DC) electricity. Conversely, wind turbines generally produce Alternating Current (AC) that requires conversion to Direct Current (DC) prior to utilization in electrolysis [5]. The variability and intermittency of these sources require the incorporation of energy storage devices or hybrid topologies to guarantee a reliable power supply [3].
Power conditioning system
A power conditioning unit, consisting of inverters, rectifiers, and controllers, is necessary for effective electrolyser functioning. These components govern voltage and current output to align with the input specifications of the electrolyser. Efficient power electronics enhance system stability and contribute to the reduction of hydrogen generation costs [10].
Electrolyser technology
Three primary categories of electrolysers utilized in hydrogen generation powered by renewable energy include alkaline electrolysers, Proton Exchange Membrane (PEM) electrolysers, and solid oxide electrolysers [11]. Alkaline electrolysers are more developed and economical, while PEM electrolysers provide greater current densities and better compatibility with variable renewable inputs [12]. PEM systems are especially appropriate for solar and wind applications because of their rapid response times and compact configuration [13].
System integration and control
The successful integration of the renewable source with the electrolyser necessitates sophisticated management algorithms to manage load fluctuations and enhance hydrogen production. Hybrid energy management solutions are progressively being formulated to address energy intermittency, hence improving reliability [5].
Solar PV vs. wind turbine integration
The incorporation of renewable energy sources into water electrolysis systems for hydrogen generation is contingent upon several parameters, including energy output characteristics, system compatibility, cost, and reliability. Solar Photovoltaic (PV) and wind turbine technologies have distinct benefits and obstacles in this setting.
Characteristics of energy output
Solar photovoltaic systems generate Direct Current (DC) electricity, which is immediately compatible with the specifications of Proton Exchange Membrane (PEM) electrolysers, hence minimizing the necessity for AC-DC conversion. Nonetheless, their energy production is sporadic and diurnal, contingent upon sunshine availability [3]. Conversely, wind turbines produce Alternating Current (AC), necessitating conversion to Direct Current (DC) for electrolysis. Wind energy may be more consistent and accessible during night-time; yet, it exhibits greater variability and is contingent upon location [5].
Efficacy and utilization
sun photovoltaic systems provide more predictability in energy generation, particularly in areas with elevated sun irradiation. This predictability streamlines system design and operation. Wind turbines generally provide superior capacity factors, particularly in coastal and elevated regions, indicating they can generate increased energy over time when optimally located [13].
Integration with electrolysers
PEM electrolysers are frequently used in solar and wind applications because of their rapid dynamic response and little start-up time, rendering them appropriate for variable energy input. Alkaline electrolysers are utilized, although they exhibit diminished responsiveness to fluctuating power inputs, particularly from wind systems [12].
Energy Input, Electrolyser Types
Characteristics of energy input
The characteristics of electrical energy produced by solar Photovoltaic (PV) systems and wind turbines directly affect the design and efficacy of water electrolysis devices. Solar photovoltaic systems produce Direct Current (DC) electricity, which is optimal for direct integration with Proton Exchange Membrane (PEM) electrolysers. The energy output is intermittent and diurnal, adhering to sun irradiance patterns [3]. Wind turbines provide Alternating Current (AC) power. This output must be transformed into direct current prior to being provided to electrolysers. Wind energy is frequently more unpredictable and location-specific, although it can function continuously, potentially enhancing total system utilisation [5]. Power conditioning systems, including inverters, rectifiers, and controllers, are essential for aligning fluctuating energy inputs with the specific demands of various electrolyser technologies [12].
Types of Electrolysers
Three principal categories of electrolysers are typically employed in conjunction with renewable energy systems:
Alkaline Electrolysers (AEL)
Utilise a liquid alkaline electrolyte (e.g., potassium hydroxide or sodium hydroxide). Established and economical technology with an extensive operational history. Optimally designed for consistent power inputs. Slow dynamic reaction renders them suboptimal for direct integration with variable renewable energy sources such as wind and solar without buffering measures [13].
Proton Exchange Membrane (PEM)
Electrolysers Utilise a robust polymer electrolyte. Compact, adaptable, and exceptionally sensitive, rendering them appropriate for sporadic inputs from solar photovoltaic and wind sources. Capable of functioning at elevated current densities and pressures. Presently more costly owing to the utilisation of rare materials (e.g., platinum) [12].
Solid Oxide Electrolysers (SOEC)
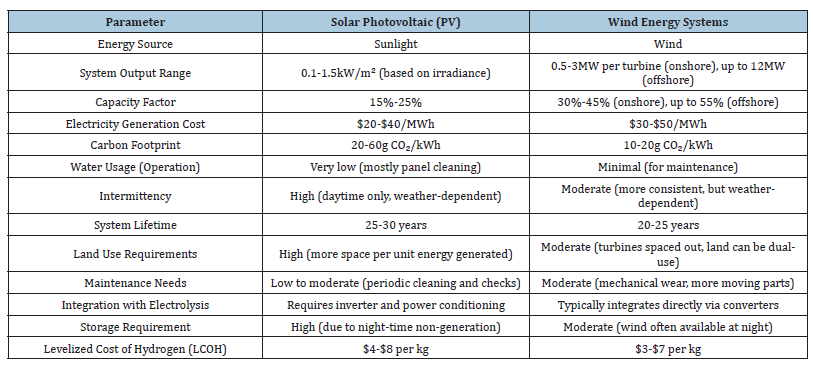
Function at elevated temperatures (600-850 °C). Provide exceptionally high efficiency using both electricity and thermal energy. Primarily in the research and pilot phase, not extensively implemented in existing solar or wind energy systems due to integration complexities and material difficulties [3]. The alignment between the energy characteristics of the renewable source and the electrolyser technology is essential [14]. PEM electrolysers are well-suited for direct integration with solar and wind energy due to their rapid load-following capability, whereas alkaline systems are more economical when energy input can be stabilised. Table 1: Comparative technical specifications. Table 1 gives the Comparative technical specifications.
Table 1:Comparative technical specifications [13-15].
Energy Efficiency and Yield
Energy efficiency and hydrogen yield are essential parameters for assessing the technical and economic feasibility of electrolysis systems utilising renewable energy. The parameters are affected by the electrolyser type, the renewable energy source, and the system setup.
Efficiency of electrolysis energy
The electrical efficiency of an electrolysis system is often described as the ratio of the energy content of the produced hydrogen, measured by its Lower Heating Value (LHV), to the electrical energy utilised in the water-splitting process. Alkaline Electrolysers (AEL) attain efficiencies of 60-70% based on Lower Heating Value (LHV) under steady-state circumstances [3]. Nonetheless, their efficiency markedly declines when subjected to variable energy inputs owing to their sluggish dynamic response [15]. Proton Exchange Membrane (PEM) Electrolysers have superior efficiency (65-75%), particularly under variable working circumstances. They can swiftly adapt to fluctuating solar and wind inputs, reducing energy losses and enhancing hydrogen production [16]. Solid Oxide Electrolysers (SOEC) can achieve efficiencies of 80- 90% by utilising both electrical and thermal energy; nevertheless, their adoption in renewable-powered systems remains limited due to the necessity for high-temperature operation [6].
Impact of renewable energy sources
Solar PV-powered electrolysis systems have reduced capacity factors (generally 15-25%), indicating they function at high power for only a limited duration. This impacts the overall hydrogen output unless supplemented by energy storage [9]. Wind-powered systems typically exhibit elevated capacity factors (25-45%), particularly in areas abundant in wind, resulting in increased annual hydrogen production. The erratic characteristics of wind may diminish overall efficiency if not adequately controlled [15].
Conversion efficiency, uptime, and intermittency
Conversion efficiency denotes the efficacy with which energy from a renewable source (solar or wind) is transformed into hydrogen via electrolysis. The efficiency of the electrolyser and the power conditioning system, which aligns renewable energy production with the electrolyser’s input specifications, both exert an effect. Solar photovoltaic systems often generate Direct Current (DC), which aligns well with Proton Exchange Membrane (PEM) electrolysers, exhibiting efficiency rates of 65-75% when functioning under optimal conditions [16]. Wind turbines generate Alternating Current (AC), necessitating conversion to Direct Current (DC). The conversion efficiency (from AC to DC) may incur extra losses; however, the overall system efficiency can remain elevated, contingent upon the location and design [14]. PEM electrolysers exhibit superior efficiency with variable inputs, whereas alkaline electrolysers have lower efficiency but are compatible with more stable energy sources, achieving efficiencies generally between 60- 70% [13].
Operational Continuity
Uptime refers to the duration a system is functional and generating hydrogen, usually expressed as a percentage of the entire time. It is determined by the accessibility of energy from renewable sources and the capability of the electrolyser system. Solar PV systems possess diurnal functionality, indicating they operate well solely during daylight hours and in the presence of adequate sunshine. Cloud cover and seasonal fluctuations may additionally diminish uptime. Wind turbines provide increased operational availability due to the consistent presence of wind both day and night. The variety of wind patterns, including calm times, can diminish uptime in specific places [14]. Hybrid systems that integrate solar and wind energy might enhance operational continuity, as one source may compensate for the lack of production from the other (e.g., solar energy is unavailable at night) [13]. Conversion efficiency, operational uptime, and intermittency are critical determinants influencing the success of solar and windpowered electrolysis systems. The intermittency of solar and wind energy can be alleviated by integrating both sources and utilising energy storage systems, thus enhancing operational continuity and overall system efficacy.
Impact on hydrogen yield
The efficiency of conversion directly affects the quantity of hydrogen generated per unit of input energy. Increased efficiency signifies that a greater proportion of electrical energy is transformed into chemical energy as hydrogen. PEM (Proton Exchange Membrane) electrolysers, exhibiting efficiencies of 65- 75%, optimise hydrogen production by reducing energy losses throughout the conversion process [15]. In contrast, alkaline electrolysers with efficiencies ranging from 60% to 70% generally yield less hydrogen for an equivalent energy input. In solar PVpowered electrolysis, improved conversion efficiency guarantees that a larger fraction of intermittent solar energy is effectively harnessed for hydrogen synthesis [13]. In wind-powered electrolysis, conversion efficiency may be compromised due to the necessity of AC to DC conversion, resulting in energy losses prior to reaching the electrolyser [14].
Influence of uptime on hydrogen production
Uptime, defined as the duration in which the system generates hydrogen, is directly correlated with the energy availability for the electrolysis process. Solar photovoltaic systems are constrained by the availability of sunlight and can generate hydrogen solely during daylight hours or when sunlight is adequate. This fundamentally diminishes both uptime and hydrogen yield. Energy storage or a hybrid system incorporating wind can enhance uptime and augment productivity by facilitating continuous hydrogen synthesis during periods of solar energy unavailability [9]. Wind turbine systems possess a greater uptime potential as wind energy is consistently accessible, both day and night. This results in increased hydrogen yields over time relative to solar-only systems, particularly in regions abundant in wind resources. In areas characterised by low or inconsistent wind, operational efficiency may diminish, hence impacting hydrogen production [14]. Hybrid solar-wind systems can enhance both operational availability and output by harmonising the variable generation from solar and wind sources. This facilitates a more consistent energy supply to the electrolyser, resulting in an increased overall hydrogen yield [13].
Influence of intermittency on hydrogen production
Intermittency denotes the variability in renewable energy production, which can result in fluctuations in the power provided to the electrolyser. Solar photovoltaic systems exhibit significant intermittency owing to variations in sunshine conditions, cloud cover, and diurnal cycles. This unpredictability results in fluctuations in energy input, which can diminish the efficiency of hydrogen production, particularly in alkaline electrolysers. In PEM electrolysers, the rapid response time can partially alleviate these variations; nonetheless, it still impacts the overall hydrogen yield until offset by energy storage [15]. The hydrogen yield of solar and wind-powered electrolysis systems is directly influenced by conversion efficiency, uptime, and intermittency. Enhanced conversion efficiency and increased uptime result in greater yields; however, intermittency may induce fluctuations and diminish yield until addressed by hybrid systems or storage solutions. Hybrid solar-wind systems are notably efficient in reducing intermittency, enhancing operational duration, and augmenting overall hydrogen production by delivering a more steady and continuous power source to electrolysers. Figure 2 illustrates the Efficiency curves over a year. The expense of hydrogen production through electrolysis is contingent upon various elements, including the capital expenditure of the electrolyser, the costs of renewable energy, and expenses related to system integration, such as energy storage or grid integration.
Figure 2:Efficiency curves over a year.
Capital and operational costs electrolysers
The capital expenditure for electrolysers, especially PEM electrolysers, presently exceeds that of alkaline electrolysers owing to the incorporation of precious metals (e.g., platinum). Recent estimates indicate that the capital cost for PEM electrolysers ranges from $500 to $800 per kW, but alkaline electrolysers are more economical, costing approximately $300 to $600 per kW [15,16]. Renewable Energy Source: The expenses associated with solar photovoltaic and wind energy have diminished considerably during the last ten years. The costs of solar photovoltaic systems have decreased to roughly $500-$1000 per installed kilowatt, whereas wind energy costs vary from $1000-$3000 per installed kilowatt, contingent upon location and size [14].
Total system cost
The overall expense of generating hydrogen by solar and windpowered electrolysis is contingent upon the system’s scale and energy source. Solar PV-powered systems may exhibit reduced start-up expenses; but they are more reliant on sunshine availability, thus necessitating storage solutions to guarantee uninterrupted hydrogen generation, thereby increasing prices. Wind energy systems, characterized by their reliability, typically exhibit greater uptime; however, they necessitate storage solutions during periods of low wind [13].
Hydrogen Production Expenses
The Levelized Cost of Hydrogen (LCOH) is a crucial metric, denoting the cost per kilogram of hydrogen generated over the system’s lifespan. According to current analyses: The Levelized Cost of Hydrogen (LCOH) from Solar PV and electrolysis is between $4 and $8 per kilogram [13]. The cost may be further diminished by enhanced electrolyser efficiency and reduced solar panel prices. The Levelized Cost of Hydrogen (LCOH) for wind-powered systems generally falls between $3 and $7 per kilogram, with opportunities for economies of scale in areas with substantial wind resources [14].
Ecological consequences of solar and wind-powered electrolysis
Solar photovoltaic systems and wind turbines are classified as low-carbon energy sources; however, their environmental implications necessitate evaluation over the entire lifecycle, from production to decommissioning.
Carbon footprint
Solar PV Systems: The fabrication of solar panels entails emissions, chiefly resulting from the energy-intensive processes involved in their production, such as silicon extraction and panel assembly. The carbon footprint of solar systems is typically low, generating approximately 20-60g CO2/kWh throughout their lifecycle, contingent upon the energy mix of the manufacturing site [9].
Wind turbines
Wind energy possesses a comparatively low lifespan carbon footprint, estimated at roughly 10-20g CO2/kWh. The principal emissions originate from the production of turbine components (steel, concrete, etc.), yet these are mitigated over the turbines’ 20- 25-year lifespan [15].
Waste management and recycling
The principal environmental issue associated with solar PV panels is their disposal upon reaching the end of their operational lives, which is generally 25 to 30 years. Panels have elements such as silicon, silver, and cadmium, which may present environmental hazards if inadequately recycled [6]. Recycling technologies are advancing, and considerable attempts are underway to create a circular economy for solar panel trash.
Wind Turbines: Wind turbine blades are built from composite materials, which might be challenging to recycle. Research on recycling technology is underway, and certain firms have been utilising recyclable materials [9]. The metallic constituents of the turbine (steel, copper) are typically more amenable to recycling. The expense associated with hydrogen production by renewablepowered electrolysis is diminishing, with wind energy often providing lower costs than solar energy due to its more consistent energy generation. However, both systems still face hurdles in terms of upfront capital expenses and the necessity for energy storage or grid connectivity to enable continuous hydrogen generation [2]. Environmental Impact: Solar and wind-powered systems are lowcarbon technologies that have comparatively less environmental effects relative to fossil fuel-based hydrogen production processes. Their primary environmental issues pertain to resource extraction, land utilization, and the recycling of equipment at the end of its lifecycle [2].
LCOH (Levelized Cost of Hydrogen)
The Levelized Cost of Hydrogen (LCOH) is a crucial economic
metric utilised to evaluate the cost-efficiency of hydrogen
generation systems, such as solar and wind-powered electrolysis.
It denotes the per-unit expense (usually per kilogram or per
megajoule) of hydrogen generated throughout the system’s
lifespan, encompassing all capital and operational expenditures,
including initial capital outlay, operational and maintenance
expenses, electricity charges, and energy storage costs (if relevant).
The Levelized Cost of Hydrogen (LCOH) is computed using a
formula analogous to the Levelized Cost of Energy (LCOE) and is
essential for evaluating various hydrogen production methods and
technologies.
Formula for Calculating Levelized Cost of Hydrogen (LCOH)
The formula for Levelized Cost of Hydrogen (LCOH) can be
articulated as:
𝐿𝐶𝑂𝐻 = Total Lifetime Expenditures of the Electrolysis System
Total Hydrogen Generated Throughout the System’s Lifespan
LCOH = Total Hydrogen Generated Throughout the System’s
Lifespan
Total Lifetime Expenses of the Electrolysis System
Location
Total Lifetime Costs encompass capital expenditures (cost of electrolysers, renewable energy systems, storage), operational and maintenance expenses, depreciation, and any financing charges.
Total Hydrogen Produced denotes the cumulative volume of hydrogen generated throughout the system’s lifespan, typically quantified in Kilograms (kg) or Megajoules (MJ) of hydrogen.
Determinants affecting LCOH
A multitude of factors influences the Levelized Cost of Hydrogen (LCOH) for solar and wind-powered electrolysis systems, including:
Initial investment
Capital expenditures encompass the expenses associated with electrolysers, renewable energy systems (such as solar photovoltaic or wind turbines), inverters, power conditioning apparatus, and perhaps energy storage devices. Elevated initial capital expenditures augment LCOH unless mitigated by enhancements in efficiency or economies of scale. PEM electrolysers generally incur higher costs than alkaline electrolysers; nevertheless, they can function at greater efficiency, potentially resulting in reduced LCOH in some scenarios.
Energy expenditures
The cost of electricity is a critical determinant of LCOH. The costs of renewable electricity from solar and wind have diminished over the past decade, enhancing their economic feasibility for electrolysis.
The costs of solar photovoltaic energy have decreased to $20- $40 per MWh, whereas wind energy ranges from $30-$50 per MWh in certain places [14]. The intermittency of both energy sources may necessitate energy storage or a hybrid system, thereby elevating costs.
System efficacy
The efficiency of the electrolyser is crucial in ascertaining the total hydrogen generated from a specific quantity of power. PEM electrolysers often function at elevated efficiency (65-75%), leading to increased hydrogen production per unit of power and a potential reduction in LCOH. The efficiency of a renewable energy system is contingent upon its capacity to deliver constant and highquality power, which fluctuates based on geographical location and meteorological circumstances.
Photovoltaic solar energy combined with electrolysis
The Levelized Cost of Hydrogen (LCOH) for solar photovoltaicpowered electrolysis is projected to be between $4 and $8 per kilogram of hydrogen [13]. In areas characterized by elevated solar insolation and reduced solar photovoltaic prices, the Levelized Cost of Hydrogen (LCOH) may reach as low as $3 to $4 per kilogram.
Wind energy with electrolysis
The Levelized Cost of Hydrogen (LCOH) for wind-powered electrolysis is often advantageous, ranging from $3 to $7 per kilogram of hydrogen, with areas exhibiting stable wind patterns attaining reduced prices [14].
Offshore wind farms, despite elevated construction expenditures, can yield substantial hydrogen generation at a reduced Levelized Cost of Hydrogen (LCOH) relative to terrestrial systems. Challenges in
Attaining Low LCOH intermittency and storage: The irregularity of solar and wind energy implies that electrolysers may not function continuously, potentially leading to diminished overall efficiency and elevated LCOH unless integrated with energy storage solutions such as batteries or hydrogen storage.
Economies of scale
Larger projects typically gain advantages from economies of scale, indicating that extensive hydrogen production systems can attain a lower Levelized Cost of Hydrogen (LCOH) due to diminished per-unit expenses for electrolysers and renewable energy systems. Technological advancements in electrolyser technology, like as more efficient PEM electrolysers, and enhancements in renewable energy generation, including reduced costs for solar panels and wind turbines, are anticipated to decrease the Levelized Cost of Hydrogen (LCOH) in the future. The Levelized Cost of Hydrogen (LCOH) is an essential metric for assessing the economic feasibility of hydrogen production from renewable sources. The cost of hydrogen produced by solar and wind-powered electrolysis systems can vary from $3 to $8 per kilogram, influenced by technology, location, and market conditions. Energy efficiency, system availability, and capital expenditures substantially affect these costs, with anticipated technological progress likely to reduce the LCOH in the forthcoming years, enhancing the competitiveness of green hydrogen against alternative energy sources.
Emission savings and water use
In evaluating hydrogen production methods, especially solar and wind-powered electrolysis, it is crucial to assess emission reductions and water consumption, two vital environmental factors that influence the sustainability of these technologies. Emission Savings in Solar and Wind-Powered Electrolysis a. Emission Reduction In comparison to hydrogen production derived from fossil fuels. Hydrogen generation using electrolysis, utilizing renewable energy sources such as solar and wind, yields no direct carbon emissions. This stands in stark contrast to traditional techniques such as Steam Methane Reforming (SMR), the predominant method for hydrogen synthesis, which generates substantial CO2 emissions. Steam Methane Reforming (SMR) is the predominant technique for the manufacture of grey hydrogen, accounting for over 95% of global hydrogen output. The procedure entails the extraction of hydrogen from natural gas, resulting in the emission of around 9-12kg of CO2 for each kilogram of hydrogen produced [4,12]
Solar and wind-powered electrolysis generates hydrogen with nearly negligible emissions, restricted to indirect emissions associated with the production, delivery, and installation of renewable energy infrastructure (i.e., embedded emissions). The lifecycle emissions linked to hydrogen generated using solar and wind-powered electrolysis often vary from 20 to 50g CO2 per kWh [10].
Potential for emission reduction
The emission reductions achieved with solar and wind-powered electrolysis are substantial. Global scaling of hydrogen production might lead to a decrease of millions of tons of CO2 per year, particularly in heavy industries such as steel manufacturing and transportation, which presently depend on hydrogen supplied from fossil fuels. Green Hydrogen: Hydrogen generated using renewable energy is termed green hydrogen, indicating the absence of direct CO2 emissions. The carbon footprint of green hydrogen may range from 0.1 to 0.3kg CO2 per kilogram of hydrogen, contingent upon the specific area and the energy composition of the electrolyser [14].
Water Utilisation in Solar and Wind-Powered Electrolysis
Electrolysis and water consumption
The electrolysis of water entails the dissociation of Water (H₂O) into hydrogen and oxygen, necessitating water as a feedstock. The quantity of water needed is contingent upon the electrolyser’s efficiency and the overall scale of hydrogen generation. The production of 1kg of hydrogen through electrolysis generally requires approximately 9 litres of water, contingent upon the type and efficiency of the electrolyser. This water is utilised in the electrolysis process and is not consumed; instead, it is dissociated into hydrogen and oxygen during electrolysis [13]. Comparative Analysis with Other Water-Intensive Processes Water Utilisation in SMR: Conventional hydrogen generation through steam methane reforming (grey hydrogen) necessitates considerable water, mainly for cooling and refining activities. This water usage is indirect, with a significant quantity needed for industrial cooling, particularly in large-scale hydrogen manufacturing facilities.
Water utilisation in renewable energy
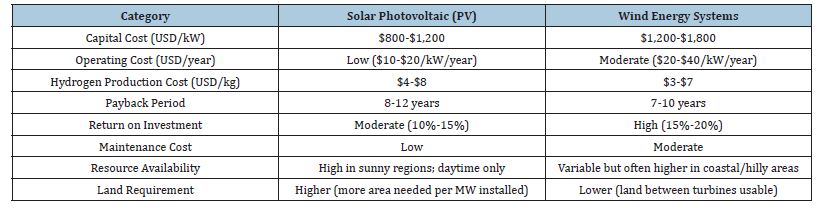
Solar photovoltaic and wind energy exhibit superior water efficiency relative to conventional power generation technologies such as coal and nuclear energy. For example: Solar PV systems often utilize minimal to no water, save for the negligible amount required for panel cleaning, which may often be diminished by the application of cleaning technology. Wind turbines exhibit minimal water use during operation, aside from infrequent maintenance and cleaning requirements. Consequently, when solar and wind energy are employed for electrolysis, the direct consumption of water is restricted to the electrolysis process and any water required for equipment cooling, if necessary. Renewable-powered electrolysis is comparatively more water-efficient than traditional hydrogen generation processes. Table 2 presents the Cost-benefit comparison.
Table 2:Cost-benefit comparison [13-16].
Future perspectives
The prospects for solar and wind-powered electrolysis in hydrogen generation are promising, with substantial breakthroughs anticipated. The global transition to green hydrogen is accelerating, with several trends, challenges, and opportunities influencing the advancement of this technology. Presented herein are several critical future outlooks for solar and wind-driven electrolysis.
Technological progressions
Enhancements in electrolyser technology: Innovative Electrolyser Configurations: The research aims to enhance the efficiency and cost-effectiveness of electrolysers. Advancements in alkaline and PEM (Proton Exchange Membrane) electrolysers, together with the progression of Solid Oxide Electrolysis Cells (SOECs), are anticipated to enhance efficiency, reduce prices, and prolong lifespan. PEM electrolysers are especially compatible with intermittent renewable energy sources such as solar and wind, due to their enhanced responsiveness to swift variations in power supply. Solid Oxide Electrolysers (SOECs), functioning at elevated temperatures, present the opportunity for enhanced efficiency in hydrogen production, especially when utilised with surplus heat from industrial operations [17].
Advanced catalysts
The innovation of non-precious metal catalysts for electrolysis has the potential to significantly lower the expense of electrolysers. Catalysts are essential for enhancing the efficiency of the electrolysis process, and advancements in this field might substantially reduce the cost per kilogram of hydrogen.
Energy storage solutions
Hybrid systems: To address the intermittency of solar and wind energy, forthcoming electrolysis systems may incorporate energy storage technologies, like batteries or hydrogen storage. These systems can provide a consistent supply of hydrogen even in the absence of power generation from renewable energy sources [18]. Extensive energy storage will be essential in stabilizing the electrical supply for electrolysis systems, enhancing the economic feasibility of hydrogen production. The Power-to-X (P2X) idea, which involves the conversion of renewable electricity into hydrogen or alternative synthetic fuels such as ammonia and synthetic methane, is expected to receive increased focus [1]. These technologies facilitate the efficient integration of intermittent renewable energy by turning surplus renewable energy into hydrogen or alternative storage mediums.
Expense minimization and financial feasibility
Economies of scale: The expansion of solar and wind-powered electrolysis plants will result in economies of scale in manufacturing, significantly decreasing the Levelized Cost of Hydrogen (LCOH). Mass production and extensive electrolyser initiatives will result in reduced capital and operational expenditures, hence decreasing the cost of hydrogen. Global Expansion: The rising global demand for green hydrogen will facilitate economies of scale, allowing manufacturers to create electrolysers at much reduced costs. Moreover, the declining expenses associated with renewable energy sources, such as solar photovoltaic and wind, will further reduce the cost of hydrogen production. Official Support and Market Demand Government Incentives: As nations establish carbon reduction objectives and net-zero pledges, there will be increased official backing for green hydrogen [1]. This may encompass subsidies, tax incentives, and research support for the expansion of hydrogen infrastructure. Carbon Pricing and Emission Regulations: Carbon pricing systems, like carbon taxes, may increase the cost of fossil fuel-based hydrogen generation, hence enhancing the demand for green hydrogen. This could enhance the commercial viability of solar and wind-powered electrolysis.
Integration with sectors and applications
Hydrogen as an eco-friendly fuel for heavy industry: Hydrogen generated using solar and wind-powered electrolysis has considerable promise for decarbonising heavy sectors that are challenging to electrify, including: Hydrogen can substitute coke in blast furnaces for steel manufacturing, markedly decreasing CO2 emissions. Green hydrogen can be utilised in the manufacturing of ammonia and several industrial operations within the chemicals and petrochemicals sector. The decarbonization of heavy industries with hydrogen are anticipated to be essential in achieving global climate objectives.
Transportation hydrogen fuel cells: The transportation sector’s pursuit of a diminished carbon footprint will likely position hydrogen fuel cells as crucial for long-haul trucking, maritime shipping, and aviation, where battery electric alternatives may be impractical due to limitations in energy density.
Hydrogen infrastructure: A comprehensive hydrogen fuelling infrastructure must be established to facilitate the expansion of the hydrogen economy. This encompasses the establishment of hydrogen refuelling stations and storage systems to facilitate both transportation and industrial uses.
Hybrid systems, policy incentives, storage options
Hybrid devices: The amalgamation of solar and wind energy
with energy storage devices (e.g., batteries or hydrogen storage)
can alleviate the intermittency of renewable energy, guaranteeing a
consistent supply of electricity for electrolysis [7]. Hybrid systems
enhance reliability, facilitating uninterrupted hydrogen production
even in times of diminished solar or wind energy.
Policy incentives: Governments are providing subsidies,
tax incentives, and research grants to facilitate the advancement
of green hydrogen. Carbon pricing and net-zero commitments
enhance the appeal of solar and wind-powered electrolysis by
diminishing the competitiveness of fossil-fuel-based hydrogen.
Storage alternatives: Energy storage is essential for
reconciling renewable energy generation with hydrogen production
requirements. Options comprise batteries (for short-term storage)
and hydrogen storage (for long-term energy storage). These
technologies facilitate a consistent hydrogen supply during periods
of variability in renewable energy production (Figure 3).
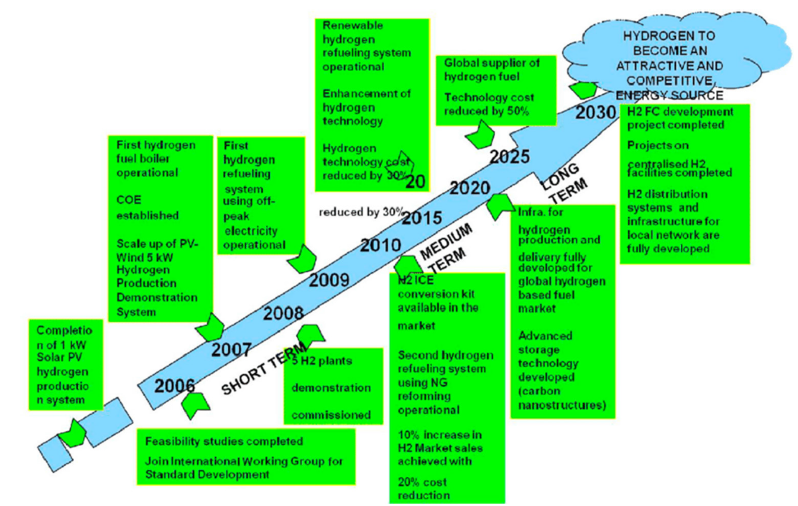
Figure 3:Roadmap for scalable green hydrogen infrastructure.
Acknowledgment
The authors acknowledge the contributions of researchers worldwide advancing AI applications in drug discovery and the open-source software community enabling these developments.
References
- Green hydrogen: A guide to policy making. IRENA.
- Abah MA, Paul-Okore RO, Oladosu MA, Agida OD (2025) Comparative study on green hydrogen production via solar vs wind electrolysis. Journal of e-Science Letters 6(4): 28.
- Ursua A, Gandia LM, Sanchis P (2012) Hydrogen production from water electrolysis: Current status and futures trends. Proceedings of the IEEE 100(2): 410-426.
- Buttler A, Spliethoff H (2018) Current status of water electrolysis for energy storage, grid balancing and sector coupling via power-to-gas and power-to-liquids: A review. Renewable and Sustainable Energy Reviews 82: 2440-2454.
- Milani D, Roshandel R, McNaughton R (2017) A comprehensive review of renewable-based hydrogen production and utilization strategies in fuel cell vehicles. Renewable and Sustainable Energy Reviews 73: 926-952.
- Schmidt O, Gambhir A, Staffell I, Hawkes A, Nelson J, et al. (2017) Future cost and performance of water electrolysis: An expert elicitation study. International Journal of Hydrogen Energy 42(52): 30470-30492.
- Zhang X, Zou D, Yang H, Wang X (2020) Hybrid solar and wind power system for hydrogen production in remote areas: An economic and reliability analysis. Renewable Energy 145: 184-196.
- NREL (2021) Solar-powered electrolysis for hydrogen production. National Renewable Energy Laboratory.
- Park JH, Kim KM, Lee SJ, Kim DW (2019) Progress and perspectives of photoelectrochemical water splitting for solar hydrogen generation. Advanced Functional Materials 29(16): 1807333.
- Turner JA (2004) Sustainable hydrogen production. Science 305(5686): 972-974.
- Kamavaram V, Mantha D, Reddy RG (2003) Electrorefining of aluminum alloy in ionic liquids at low temperatures. Journal of Mining and Metallurgy, Section B: Metallurgy 39(1-2): 43-58.
- Friedl R, Trimis D (2017) Assessment of hydrogen production technologies for a sustainable energy system. Energy Technology 5(1): 94-103.
- Gahleitner G (2013) Hydrogen from renewable electricity: An international review of power-to-gas pilot plants for stationary applications. International Journal of Hydrogen Energy 38(5): 2039-2061.
- Göransson L, Lundberg M, Johnsson F (2017) Cost-optimal design of a regional electricity system using hydrogen storage and wind power-a case study of Gotland. Energy 138: 1087-1098.
- Park S, Kim B, Lee J, Kang M (2019) Review of recent developments in photoelectrochemical water splitting using modified semiconductor photoelectrodes. Renewable and Sustainable Energy Reviews 82(1): 2580-2601.
- IEA (2022) Global hydrogen review 2022. International Energy Agency.
- Cappelletti G, Pasini A, Selli E (2022) Recent advances in solid oxide electrolysis cells for high-temperature hydrogen production. Renewable and Sustainable Energy Reviews 153: 111784.
- Ahmad MS, Ali M S, Abd Rahim N (2021) Hydrogen energy vision 2060: Hydrogen as energy Carrier in Malaysian primary energy mix-Developing P2G case. Energy Strategy Reviews 35: 100632.
© 2026 Micheal Abimbola Oladosu, This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






