- Submissions

Full Text
Research & Development in Material Science
Martensitic Transformation in a Single-Crystal Cu-Al-Ni Shape-Memory Alloy
Zakaria Boumerzoug* and Ibtissem Debbache
Department of Mechanical Engineering, LMSM, Uiversity of Biskra, Algeria
*Corresponding author:Zakaria Boumerzoug, Mechanical Engineering Department, LMSM, University of Biskra, Algeria
Submission: April 02, 2026;Published: April 27, 2026

ISSN: 2576-8840 Volume 22 Issue 5
Abstract
Shape Memory Alloys (SMA) are functional materials known for their shape memory behavior. They are considered smart materials because they regain their shape through simple heat or mechanical treatments. This entire behavior is linked to reversible martesitic transformation. The progress of this martensitic transformation depends on several parameters, such as the effect of thermal or mechanical treatments. It is in this context that this research was carried out. This work aims to study a martensitic transformation in a single-crystal Cu-16.3wt. Al-2.6wt. Ni shape-memory alloy after different heat and thermo-mechanical treatments. The experimental techniques used were optical microscopy, energy-dispersive X-ray spectroscopy, and Vickers microhardness tests. It has been found that the martensitic transformation and its reverse reaction is achieved by simple heating and cooling, and it is characterized by a double memory effect. Aging treatment has an effect on the hardness and the type of martensite.
Introduction
Shape Memory Alloys (SMAs) are functional materials recognized for their shape memory properties, which result from a phase transition between two solid phases: austenite and martensite [1]. These shape memory alloys are also called smart materials (SMMs) [2-5], because they can regain their original shape through reversible martensitic phase transformations triggered by a thermal or mechanical effect [6]. Shape Memory Alloys (SMAs) offer distinct advantages and appeal to engineers due to their broad applicability to contemporary engineering challenges. SMAs are becoming increasingly essential for improving desired properties by replacing traditional engineering materials. This innovative material has been the subject of much research in multiple fields, including robotics, microsystems, biomedical engineering, structural applications, and transportation sectors such as automotive and aerospace [7]. They were discovered by the Swedish metallurgist, Arne Olander, in 1932 [3,8-11]. He observed the rubbery behavior of the gold-cadmium alloy [8,11].
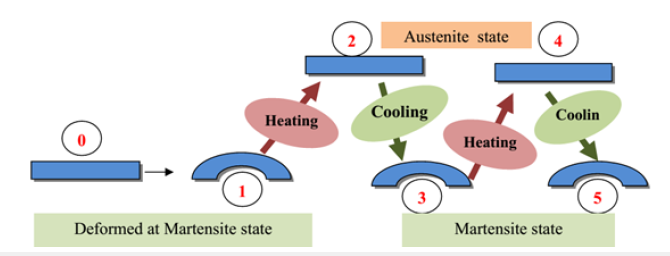
Shape memory alloys undergo a solid-solid phase transition as the temperature increases, moving from a martensitic state at low temperature to an austenitic state. Similarly, under the effect of heat, alloys deformed in the martensitic phase can regain their initial shape through their transformation into the austenitic phase [12]. The austenite-martensite transformation is characterized by the temperatures Ms and Mf, which correspond to the temperature range where martensite forms and develops from austenite. The reverse transformation, martensite-austenite, is characterized by the temperatures As and Af, which correspond respectively to the beginning and end of the transition from martensite to austenite [13]. Shape memory can occur through various mechanisms, notably Two-Way Shape Memory Effect (TWSME), which is highly sought after in artificial semiconductor materials. This mechanism is characterized by a spontaneous change in shape during heating and cooling, without external intervention. It requires a prior “learning” or “training” process to create directed internal stresses. Figure 1 illustrates this mechanism: if, for example, a shape-memory alloy bar (step 0) is deformed into a martensitic state (step 1) and then heated to the austenite formation zone (step 2), it will return to its original shape. If this bar is then cooled back to the martensite formation zone (step 3), it will return to its deformed shape. Subsequently, a simple heating and cooling cycle is sufficient to restore its previous shape as mentioned at step 4 and 5.
Figure 1:Illustration of Two-Way Shape Memory Effect (TWSME).
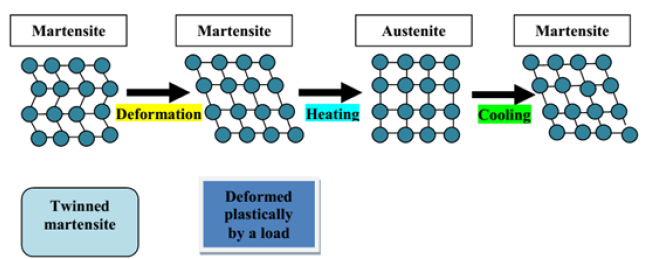
This mechanism can be represented at the scale of the crystal structure of the alloy as shown in Figure 2. The transformation of the crystal lattice is shown at each stage. It is important to note that the initial martensite (undeformed plastically) is called twinned martensite. Under the effect of deformation, it becomes untwinned martensite. Heating will cause the formation of the austenite phase, which has a different crystallographic structure. Then, cooling again forms the martensite phase.
Figure 2:Representation crystallographic of Two-Way Shape Memory Effect (TWSME),
Nickel-titanium (Nitinol) and Copper-based (Cu-Al-Ni, Cu-Zn- Al) alloys are the most common shape memory alloys (SMAs) [14]. Over the past decade, copper-based Shape Memory Alloys (SMAs) have emerged as promising materials for various applications, including sensors, actuators, and high-damping materials. Cu-Al-Ni AMFs have attracted particular interest due to their better thermal stability compared to other copper-based AMFs. Cu-Al-Ni AMF is an excellent candidate, exhibiting a remarkable shape memory effect and super elastic behavior in a narrow temperature range of 373 to 473K. The addition of nickel in the Cu-Al binary system lowers the high transformation temperature and stabilizes the austenitic phase [15]. Shape memory alloys (CuAlNi) possess a hightemperature austenitic phase with a Body-Centered Cubic (BCC) structure. Upon cooling, this phase transforms into a more ductile martensitic phase, generally monoclinic or orthorhombic [16].
The ranges of temperature at which austenite and martensite are stable depend primarily on the chemical composition and the thermomechanical history of the alloy [1]. It is in this context that this research was carried out. The objective of this research work is to study a martensitic behavior in single-crystal Cu-16.3wt. Al- 2.6wt. Ni after different heat treatments. Furthermore, structural evolution was also observed during the application of external stress.
Experimental Procedure
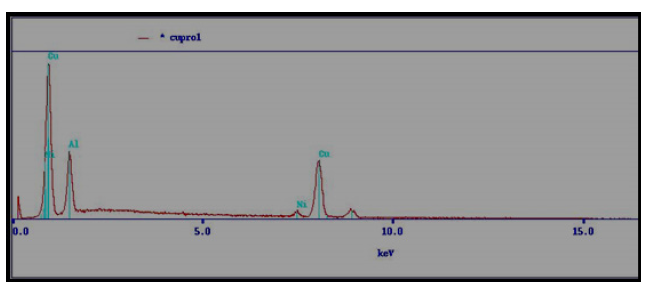
This work aims to study a martensitic transformation phenomenon in a single-crystal Cu-16.3wt. Al-2.6wt. Ni shapememory alloy. The elements present in the alloy studied were confirmed by energy-dispersive X-ray spectroscopy (EDS) of a Joel JSM-5900 LV scanning electron microscope (Figure 3).
Figure 3:Spectrum obtained by elemental EDS microanalysis of the Cu-81.1 wt. Al-16.3 wt. Ni alloy.
In order to monitor the evolution of the structure during thermal and thermomechanical cycles, i.e. in situ, the setup shown in Figure 4 was chosen. A device tool of heating ad device for a plastic deformation were added to the optical microscope.
Figure 4:Setup for monitoring the microstructural evolution of the sample during different heat treatments.

The evolution of the microstructure of samples subjected to different heat treatments was monitored using a Hund-type metallographic microscope. Observations focused primarily on the shape of the martensite platelets. Hardness measurements were carried out on a Vickers AFFRI type microhardness tester. Three hardness measurements were taken for each sample.
Results and Discussion
Effect of a thermal cycle
A sample of the Cu-81.1wt. Al-16.3wt. Ni alloy underwent a heating cycle from 15 °C to 107 °C and vice versa. Figure 5 illustrates the microstructure of the martensite obtained after cooling process and the austenite phase obtained after heating beyond 107 °C. The martensite has a lamellar shape. The microstructure of the martensitic phase corresponds to the typical martensitic βʹ microstructure of a Cu-Al-Ni alloy observed in the study by Mazzer et al. [17]. Generally, for Cu-based SMAs, before the martensitic transformation, the austenitic β phase undergoes an intermediate transition during cooling [18]. This alloy is characterized by bidirectional shape memory, as it exhibits a shape memory effect both during heating and cooling and without the application of external force (intrinsic bidirectional effect).
Figure 5:Microstructural evolution of the Cu-81.1wt% Al-16.3wt% Ni alloy, which underwent a heating cycle from 15 °C to 107 °C followed by a cooling to 17 °C.

Effect of plastic deformation
In this part of this investigation, the effect of an applied force on the formation of martensite has been studied.
Case 1: Effect of plastic deformation at a temperature of 18 °C
In this part of the study, successive plastic deformations were applied to a sample at a temperature of 18 °C (Figure 6). It was observed that the number of martensite platelets increased with the intensity of the plastic deformation (indicated by blue arrows). Furthermore, other martensite platelets appeared, exhibiting different orientations, as indicated by the arrow. It was established that the application of plastic deformation to Shape Memory Alloys (SMAs) in the martesitic phase can induce the appearance of martensite platelets with a preferred crystallographic orientation.
Figure 6:The microstructural evolution of the Cu-81.1wt% Al-16.3wt% Ni alloy, which underwent successive plastic deformations by rolling at a temperature of 18 °C.

Case 2: Effect of plastic deformation during heating process
The sample subjected to stress is gradually heated from 18 °C to 37 °C. Figure 7 illustrates the microstructural evolution of this alloy. The progressive disappearance of the previously formed martensite platelets begins at 29 °C and ends at 37 °C. It can be concluded that this process reflects the reversibility of this phenomenon.
Figure 7:The microstructural evolution of the Cu- 81.1wt% Al-16.3wt% Ni alloy, progressively heated from temperature from 18 °C to 37 °C.

Structural evolution during aging at 200 °C
Before the aging treatment, the alloy was homogenized at 800 °C and then quenched in ice water. Figure 8 shows the structural state after aging at 200 °C for 2 hours. We observe a twin-type structure. The homogenization temperature must not be too high to avoid any influence on grain size, especially for polycrystals. However, a temperature that is too low does not allow for good homogenization. Therefore, we chose a temperature of 800 °C for this alloy; this seemed necessary to completely eliminate precipitates and defects from the parent phase. Quenching the sample is necessary to obtain a metastable β phase and to prevent martensite stabilization. Indeed, a sample quenched from 800 °C to room temperature no longer exhibits martensitic transformation. Presumably, the absence of this transformation is due to a high concentration of vacancies. This quenching is therefore necessary to eliminate these vacancies by diffusion and to give the β phase a well-defined L21 or DO3 order. It can also be noted that the transformation temperature Ms is lower the higher the disorder level [19].
Figure 8:Microstructure of a Cu-81.1wt% Al-16.3wt% Ni alloy homogenized at 800 °C for 5min followed by quenching in ice water and then aged at 200 °C for 2 hours.

Tempering after quenching restores order and eliminates quenching vacancies that are supersaturated, thus preventing martensite stabilization problems [20]. Figure 8 presents microstructure of a Cu-81.1wt% Al-16.3wt% Ni alloy homogenized at 800 °C for 5min followed by quenching in ice water and then aged at 200 °C for 2 hours. The microstructure in (b) is the 1000x magnification of an area of the microstructure (a). This microstructure is known as twined martensite and has been mentioned in some shape memory alloys [21-24]. Twin martensite in Shape Memory Alloys (SMA) refers to a low-temperature crystal structure where martensite variants form alternating mirror patterns (“twins”) to minimize internal stress [25].
The evolution of hardness during this aging treatment is shown in Figure 9. We note that the hardness decreases and then gradually increases. It has been found that ageing a water-quenched (from 800 °C) Cu-Al-based shape memory alloy at 200 °C primarily induces the decomposition of the metastable martensite (or retained β1 parent phase) into stable, equilibrium phases. This process involves the precipitation of ɑ (fcc, low Al) and У2 (Cu9Al4, high Al) phases [26].
Figure 9:Evolution of microhardness as a function of heat treatment of a Cu-81.1wt. Al-16.3wt. Ni sample homogenized at 800 °C for 5 min followed by quenching in ice water and then aged at 200 °C for 2 hours.

Conclusion
This work aims to study a martensitic transformation in a
single-crystal Cu-16.3wt. Al-2.6wt. Ni shape-memory alloy after
different heat and thermo-mechanical treatments. The most
important results are as follows:
a) The martensitic transformation and its reverse reaction
are achieved by simple heating and cooling, that is to say that it is
characterized by a double memory effect.
b) Twined martensite was observed.
c) The sample subjected to deformation stress promotes
the formation of other martensite variants, which disappear upon
heating of the same sample.
References
- Nespoli A, Ninarello D, Fanciulli C (2023) A review on shape memory alloys with martensitic transition at cryogenic temperatures. Metals 13(7): 1311.
- Ayinde W, Camur H, Savas M (2021) A brief review of characteristics and applications of shape memory alloys in engineering and related fields. International Journal of Mechanical Engineering and Technology (IJMET) 12(9): 34-43.
- Jani JM, Leary M, Subic A, Gibson MA (2014) A review of shape memory alloy research, applications and opportunities. Materials & Design 56: 1078-1113.
- Shukla U, Garg K (2023) Journey of smart material from composite to Shape Memory Alloy (SMA), characterization and their applications – A review. Smart Materials in Medicine 4: 227-242.
- Hubert A, Calchand N, Gorrec YL, Gauthier J (2012) Magnetic shape memory alloys as smart materials for micro-positioning devices. in: Advanced Electromagnetics Symposium, TELECOM PARISTECH, Paris, France. Pp: 1-10.
- Ahmed A, Pranto S I, Al Faruq MH, Kasfia MTT, Rashid AB (2026) A comprehensive overview of shape memory alloys: Recent advances in fabrications, properties, and applications for next-generation smart systems. Next Materials 10: 101441.
- Rodinò S, Maletta C (2024) Design considerations and applications of shape memory alloy-based actuation in morphing structures: a systematic review. Prog Eng Sci 1(4).
- Concilio A, Lecce L (2015) Historical background and future perspectives. Shape Memory Alloy Engineering: For Aerospace. Structural and Biomedical Applications, pp. 3-52.
- Balasubramanian M, Srimath R, Vignesh L, Rajesh S (2021) Application of shape memory alloys in engineering - A review. J Phys Conf Ser.
- Mehta K, Gupta K (2019) Fabrication and processing of shape memory alloys. Springer Briefs in Applied Sciences and Technology: Manufacturing and Surface Engineering. Springer International Publishing.
- Christian JW (2002) The theory of transformations in metals and alloys. pp. 1102-1113.
- Mevada H, Liu B, Gao L, Hwang Y, Takeuchi I, et al. (2024) Elastocaloric cooling: A pathway towards future cooling technology. Int J Refrig 162: 86-98.
- Otsuka K, Ren X (2005) Physical metallurgy of Ti-Ni-based shape memory alloys. Prog Mater Sci 50(5): 511-678.
- Chen F, Tian Z, Liu P, Han W, Yi X, et al. (2026) Research advances and practical applications of Cu-Al-Mn shape memory alloys. Copper - History, Properties and Applications.
- Maalolan B, Santosh S (2024) A review on CuAlNi shape memory alloys: Production methods, applications and current trends. Materials Today: Proceedings.
- Dasgupta R (2014) A look into Cu-based shape memory alloys: Present scenario and future prospects. Journal of Materials Research 29(16): 1681-1698.
- Mazzer EM, Gargarella P, Cava RD, Bolfarini C, Galano M, et al. (2017) Effect of dislocations and residual stresses on the martensitic transformation of Cu-Al-Ni-Mn shape memory alloy powders. Journal of Alloys and Compounds 723: 841-849.
- Recarte V, Pérez-Sáez RB, Bocanegra EH, Nó ML, San-Juan J (2002) Influence of Al and Ni concentration on the martensitic transformation in Cu-Al-Ni shape-memory alloys. Metall Mater Trans A 33: 2581-2591.
- Belkahla S (1985) Thermal degradation of the shape memory effect of a ternary alloy: CuZnAl. Master's Institute of Materials Physics of Annaba, Algeria.
- Contardo L (1988) Study of stability education treatments and origin of the two-way shape memory effect in a Cu-Zn-Al alloy. Doctoral thesis, INSA (Lyon), p. 118.
- Bronstein E, Faran E, Shilo D (2019) Analysis of austenite-martensite phase boundary and twinned microstructure in shape memory alloys: The role of twinning disconnections. Acta Materialia 164: 520-529.
- Shamsolhodaei A, Zarei-Hanzaki A, Abedi H R, Safde A, Peterlechner M (2022) Twin-based martensite stabilizing and improving the shape memory response of near equiatomic NiTi alloy through multi-axial forging. Journal of Materials Research and Technology 16: 39-46.
- Rogovoy AA (2024) Microstructural model of twinning and detwinning processes of the martensitic phase in shape memory alloys. Journal of Samara State Technical University, Ser. Physical and Mathematical Sciences 28(3): 416-444.
- Enkovaara J, Ayuela A, Zayak AT, Entel P, Nordström L, et al. (2004) Magnetically driven shape memory alloys. Materials Science and Engineering: A 378(1-2): 52-60.
- Nnamchi P, Younes A, González S (2019) A review on shape memory metallic alloys and their critical stress for twinning. Intermetallics 105: 61-78.
- Al-Haidary JT, Mustafa AM, Hamza AA (2017) Effect of aging on corrosion behavior of martensite phase in Cu-Al-Be shape memory alloy. Journal of Material Sciences & Engineering 6(6): 1-7.
© 2026 © Zakaria Boumerzoug. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






