- Submissions

Full Text
Research & Development in Material Science
Study of the Ternary System Tb-Sb-Se Along the Section Sb2Se3-TbSe
Zakir Ismailov*, Zivar Hasanova, Rakhila Mirzaeva and Rahman Fatullzade
Department of General and Inorganic Chemistry, Baku State University, Azerbaijan
*Corresponding author:Zakir Ismailov, Department of General and Inorganic Chemistry, Baku State University, Azerbaijan
Submission: March 26, 2026;Published: April 16, 2026

ISSN: 2576-8840 Volume 22 Issue 5
Abstract
Chemical interactions in the Sb2Se3-TbSe system were studied using physicochemical analysis methods (differential thermal analysis, X-ray diffraction, microstructural analysis, microhardness measurements, and density determination). Based on the analysis results, a phase diagram of the Sb2Se3-TbSe system was constructed. It was established that the Sb2Se3-TbSe cross section is a quasi-binary cross section of the Tb-Bi-Se ternary system and is eutectic. In the Sb2Se -based system, a solid solution region containing 8mol.% TbSe forms at room temperature. The solid solution area, determined by microstructural analysis, is 17mol.% at a eutectic temperature of 823K and 5mol.% TbSe at room temperature. The phase diagrams show that a new ternary compound with incongruent melting properties, TbSb4Se7, is formed in the Sb2Se3-TbSe section as a result of a peritectic reaction at 863K with a component ratio of 1:2. X-ray diffraction analysis revealed that TbSb4Se7 crystallizes in an orthorhombic system with lattice parameters a=16.762; b=23.860; c=4.137Å. The solubility of TbSe in Sb2Se3 3.0mol% at 300K. Eutectic point coordinates: 10-15mol.% TbSe, temperature: ~823K.
Keywords:System; Analysis; Crystallization; Phase; Diagram; Temperature
Introduction
Modern scientific and technological progress, including the exploration of outer space, is inextricably linked with the development of semiconductor technology [1-4]. The rapid development of semiconductor technology has been the main impetus for the search for complex semiconductor materials [5-8]. However, the growing demand for materials in semiconductor technology is not yet fully met due to the lack of materials possessing different combinations of optical, magnetic, and electrical properties. These requirements for materials open up new challenges for chemical engineers, including the synthesis of new substances with desired properties [9-11].
Chalcogenides of rare earth elements, in particular antimony and bismuth, have valuable optical and electrophysical properties for use in thermoelectric and optical devices [12-15]. Therefore, the study of the nature of the chemical interaction of the Sb2Se3-TbSe system is relevant.
Experimental Part
The system was synthesized in a sealed quartz glass ampoule with air removed to 0.133Pa. The furnace was heated very slowly. Particularly after 200 °C, the furnace temperature increased by 100 °C above the melting point of the substance and maintained at this temperature for 1 hour. Cooling of the alloy was accomplished by turning off the furnace. After synthesis, the alloys were heat treated for 500 hours at the solidus temperature in a quartz ampoule under vacuum conditions.
For the synthesis of samples, terbium metal ingots TbM-1 purity 99.9%, stibium grade GOST 1089-82 and selenium GOST ГОСТ 3778-98. were used. The synthesis mode was selected based on the physicochemical properties of the elementary components, binary compounds. The alloys were obtained by direct fusion of the components in evacuated quartz ampoules at 1500K, followed by slow cooling with the furnace turned off.
The study was conducted using differential thermal analysis, high-temperature differential thermal analysis, X-ray diffraction (XRD), microstructural analysis (MSA), and microhardness measurement. An etchant of the composition 0.1∙K2Cr2O7∙H2SO4+ H2O (1:1) was used to study the microstructure of the alloys. Thermal analysis was performed on an NTP-73 pyrometer and a Thermoscope-2. Accuracy ±5.0 (Pt-Pt/Rh thermocouples were used).
High-temperature thermal analysis (VTTA) was performed using a VTA-987 pyrometer (using W-W/Re thermocouples). X-ray phase analysis of the alloys was performed using a D2 Phaser and Bruker D8 diffractometer (CuK α-radiation). Microstructural analysis of alloys (MSA) was performed on a MIM-7 microscope.
Results and Discussion
The Tb-Sb-Se (Terbium-Antimony-Selenium) system is a ternary semiconductor system being studied for the creation of new functional materials. The focus is on the study of phase equilibria, particularly along the Sb2Se3-TbSe cross-section, where a ternary compound and a eutectic mixture are formed. These equilibria are studied using differential thermal analysis and X-ray diffraction. Selenium was added in excess to maintain the stoichiometric composition. Thermograms of the alloys show that the observed heating effects are endothermic and reversible. The results of DTA, microhardness and density measurements of the alloys of the Sb2Se3-TbSe system are presented in Table 1.
Table 1:Results of DTA, microhardness and density of Sb2Se3 -TbSe system alloys.
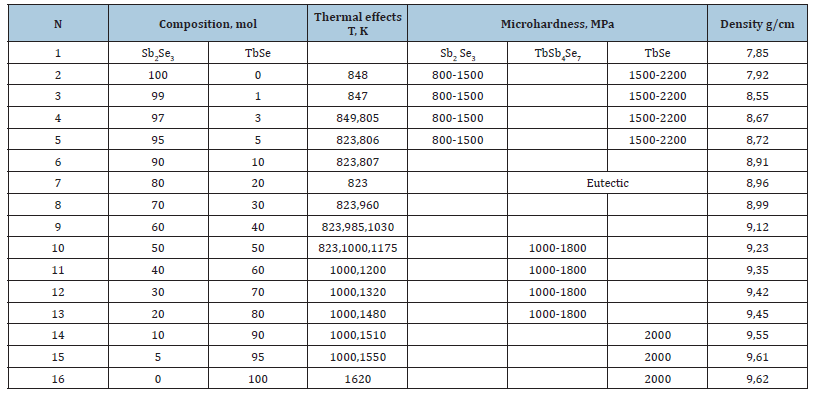
The x-ray density of the compound is 9.76g/cm3, and the pycnometric density is 9.45g/cm3. A solubility range of 3mol% TbSe based on Sb2Se3 was found in the system. A solubility range of 3mol% TbSe based on Sb2Se3 was found in the system. To determine the solubility limit, samples of 1mol% TbSe based on Sb2Se3 were synthesized and placed in ice water after heat treatment for 250 hours. As a result of microstructural analysis, it was mainly determined that the solubility was 5mol% TbSe based on Sb2Se3 at 823K, and when the temperature decreased, the solubility decreased to 3mol% TbSe.
Based on the powder method, the structure type and lattice constants of the triple compound were determined, and it was determined that the compound crystallizes in rhombic syngonia. Lattice parameters are a=16.762; b=23.860; c=4.137Å.
As can be seen from Figure 1, the system is quasi-binary and
eutectic. Based on Sb₂Se₃ a solubility field of 3mol.% TbSe is
formed in the system at 300K. In the system between Sb₂Se₃ and
TbSe, the eutectic crystallizes at a temperature of 823K with a
content of 20mol.% TbSe. To determine the boundary of the solid
solution based on Sb₂Se₃. 4 samples with a content of 0.5mol.%
were synthesized, which were subjected to heat treatment at
temperatures of 400, 500, 600 and 700K for 300 hours. Each
sample is subjected to microscopic quality analysis. According to
the results of microscopic analysis, it was established that at the
eutectic temperature (823K), the solubility is 5mol.% in terms of
Sb₂Se₃.
Eutectic point coordinates:
Composition: 10-15mol.% TbSe
Temperature: ~823K
Figure 1:Phase diagram of the Sb2Se3-TbSe system.

Conclusion
1 . The Sb2Se3 -T b S e s ys t e m wa s s t u d i e d by c o m p l ex
methods of physicochemical analysis in the entire
solidity interval, and the phase diagram of the system was
constructed.
2. An incongruent melting compound containing TbSb4S7 is
formed in the system in a 1:2 ratio of components. It was
determined that the compound crystallizes in rhombic
syngonia by setting the crystal lattice parameters
a=16.762; b=23.860; c=4.137 Å.
3. It was determined that Sb2Se3 based on the eutectic
temperature, the solubility was 5mol%, and as the
temperature decreased, the solubility decreased to 3mol%
TbSe. The electrophysical properties of some samples based
on Sb2Se3 were measured at room temperature and it was
determined that they are “p” type semiconductors and have
thermoelectric properties.
References
- Goltsman BM, Kidinov VA, Smirnov IA (1972) Semiconductor thermoelectric materials based on Bi2Te3. 320.
- Yarembash EI, Eliseev AA (1977) Chalcogenides of rare earth elements. Nauka, Moscow, Russia, p. 275.
- Sadygov FM, Jafarova EK, Babanly MB (2002) Character of phase formation in Sb2Se3(Bi2Se3)-TmSe systems. Inorg Chemistry 47(10): 1713.
- Sadygov FM, Ilyasly TM, Nasibova LE, Aliev II (2013) Physico-chemical study of the Sb2Se3-Nd2Se3 Inorg Chemistry 58(9): 1253-1256.
- Sadygov FM, Mamedova SG, Babanly MB, Ilyasly TM (2001) Phase equilibria in the Ce-Sb-Se system along the Sb2Se3-Ce2Se3 Inorg Chemistry 46(8): 1382-1383.
- Jeffrey Snyder G, Eric S Toberer (2008) Complex thermoelectric materials. Nature Materials 7: 105-114.
- Sadygov FM, Ilyasly TM, Ganbarova GT, Zlomanov VP, Aliev II (2017) Physical and chemical study of the Sb2Se3-Nd2Se3 Inorg Mater 53(7): 681-685.
- Shurova MA, Andreev OV, Kharitontsev VB (2014) Phase equilibria in the Bi2Se3-Sm2Se3 Bulletin of the Tyumen State University. Socio-economic and legal research 5: 113-121.
- Kost ME, Shilov AA, Mikheeva VI (1983) and other chemistry of rare elements, compounds of rare earth elements hydrides, borides, carbides, phosphides, chalcogenides M: science. P. 272.
- Zhang H, Liu CX, Qi XL, Dai X, Fang Z, et al. (2009) Topological insulators in Bi2Se3, Bi2Te3 and Sb2Te3 with a single Dirac cone on the surface. Nature Physics 5(6): 438-442.
- Nikiforov VN, Morozkin AV, Irkhin VYu (2013) Thermoelectric properties of rare-earth alloys. Physics of Metals and Materials Science 114(8): 711-720.
- Chih-hao L, Hsin-jay W, Sinn-Wen C (2013) Liquidus projection of the ternary Bi-SbTe. Thermoelectric Material System.
- Babanly MB, Makhumudova MA, Aliev ZS, Imamaliyeva SZ (2011) Thermodynamic properties of phrases in the Yb-Bi-Se system. Inorg Mater 47(4): 352-355.
- Andreev OV, Kharitsontsev VB, Elyshev AV (2013) Compositions of phrases in the interaction of rare-earth metals with selenium. Russ J Inorg Chem 58: 910-915.
- Zakir Ismailov, Zivar Hasanova, Rakhila Mirzaeva (2025) Character of chemical interaction in Bi2Se3-Tb2Se3 Research & Development in Material science 21(4): 2568-2571.
© 2026 © Zakir Ismailov. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






