- Submissions

Full Text
Research & Development in Material Science
Effect of Tempering Temperature on the Mechanical Properties of Quenched Hypoeutectoid Steel
Zakaria Boumerzoug*, Ridha Azzouz, and Abdelmadjid Attassi
Department of Mechanical Engineering, University of Biskra, Biskra, Algeria
*Corresponding author:Zakaria Boumerzoug, Mechanical Engineering Department, LMSM, University of Biskra, Algeria
Submission: March 16, 2026; Published: March 30, 2026

ISSN: 2576-8840 Volume 22 Issue 4
Abstract
The aim of this work is to study the influence of tempering temperature on the microstructure and hardness of a hypoeutectoid steel (0.38% C). This steel was previously water-quenched from its austenitizing temperature and then subjected to different tempering temperatures (from 100 to 700 C) for a fixed holding time. Analysis of the microstructure and Vickers hardness allowed us to observe the microstructural and mechanical changes occurring during the tempering treatment. As the temperature increases, the hardness decreases.
Keywords:Hypoeutectoid steel; Tempering temperature; Microstructure; Hardness
Introduction
Steel is present in many areas of our lives and in numerous industrial sectors due to its specific properties, such as good mechanical strength and ease of shaping, which make it the most widely used metallic material [1,2]. However, its mechanical properties can be improved either by adding alloying elements or by applying certain heat treatments. Among these heat treatments is quenching, which creates the martensite phase, a very hard phase [3,4]. However, the quenching process will cause the formation of internal stresses that can weaken the quenched steel part [5,6]. Therefore, it is necessary to apply a tempering treatment to eliminate these constraints [7,8]. It is noted that the tempering temperature is above the eutectoid line. There are three tempering treatment intervals: low-temperature tempering, medium-temperature tempering, and high-temperature tempering. It should be noted that each tempering temperature interval has an effect on the microstructure of the hardened steel and consequently on its mechanical properties. It is in this context that this study is devoted to the effect of tempering temperature on the microstructure and mechanical properties of a hypoeutectoid steel (0.38% C).
Experimental Procedure
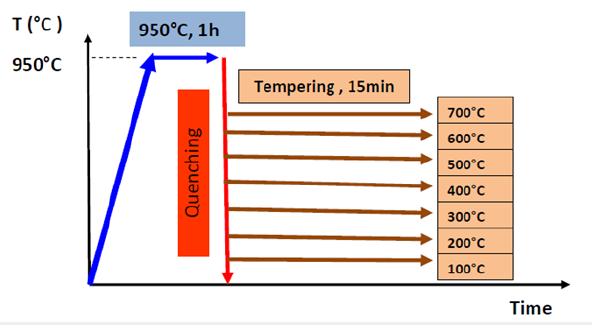
For this study, a hypoeutectoid steel (0.38% C) was chosen: The heat treatments applied to these steels are shown in Figure 1 and consist mainly of an austenitizing treatment at 950 C for 1h, followed by water quenching and then tempering for 15min at different temperatures i.e. from 100 to 700 °C. It is noted that the quenching process aims to form martensite, while the tempering aims to eliminate internal stresses.
Figure 1:Heat treatment cycles of the hypoeutectoid steel (0.38% C).
For these heat treatments, a programmable electric furnace with a maximum temperature Tmax = 1200 °C was used (Figure 2a). The study of microstructure requires well-polished surfaces, free of any scratches that could interfere with micrographic examination. To achieve this, the samples are mechanically polished on abrasive papers (from grit 120 to 2000), then with 0.5μm diamond paste. After each polishing step, the samples are cleaned with distilled water and dried. The surfaces are then chemically etched (Figure 2b) with 4% Nital (an alcoholic-nitric solution) composed of 4ml of nitric acid (HNO3), and 100ml of ethanol (C2H5OH).
Figure 2:(a): Electric furnace, and (b): Chemical etching of steel.

The first approach to understanding the various thermal and thermomechanical treatments the sample undergoes is its microstructure. For this reason, a Hund-type metallographic microscope equipped with a digital camera was used (Figure 3a), which allowed us to take micrographs of the structure of the studied samples at magnifications up to 800x.
Figure 3:(a): Metallographic microscope, and (b): Hardness tester.

The Vickers hardness (Hv) is the ratio between the applied load
and the projected area of the residual indentation:
Hv = (1.854F) / (9.81d²)
Where F is the applied load in Newtons (N) and the diagonal (d) of the indentation is in micrometers (μm). To measure the hardness of our steel, we used an Affri brand hardness tester with a digital display (Figure 3b). Vickers hardness measurements with a 200g load are used. Five to seven measurements were taken on each sample, and an average was calculated, discarding noncompliant values
Results and Discussion
Microscopic observations
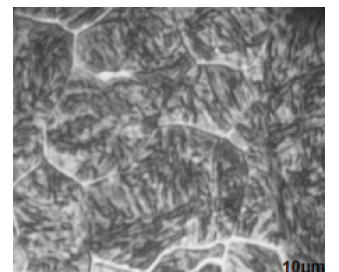
Quenching state of 0.38% C steel: In this part of the study, a homogenization treatment at 950 °C for 1 hour on the 0.38%C steel was performed, followed by very rapid cooling (water quenching at 25 °C). Observation under a metallographic microscope revealed the formation of martensite (Figure 4). It should be noted that the substructure of the martensitic crystals, as well as their morphology, can vary depending on the steel composition and the temperature of their formation. In our case, this martensite in the 0.38%C steel takes the form of laths. Furthermore, the grain boundaries are very apparent, with a white zone clearly visible in areas near these grain boundaries.
Figure 4:Microstructure of 0.38%C steel after homogenization for 1 hour at 950 °C followed by water quenching.
Tempering treatment of 0.38 % C steel: Tempering is a final heat treatment operation. Its purpose is to obtain the required mechanical properties; moreover, it completely or partially eliminates the internal stress caused by quenching. The reactions during tempering vary with time and especially with temperature. The microstructural evolution of 0.38% C steel during tempering at different temperatures of a previously quenched sample is shown in Figure 5.
Through these micrographs, we observe a change in the morphology of the initial structure (the martensite). We know that tempering at 250 °C reduces internal stress and transforms the quenching martensite into the tempering martensite. This is why we observe a slight change in the structure after this first tempering at 200 °C (Figure 5a). In the literature, this type of treatment (quenching + tempering at 2 0 °C) is called improvement. This structural change is very apparent if the tempering temperature is increased (Figure 5b-5g). At high temperatures, cementite forms, as it is known that from 450 to 500 °C, carbides transform into cementite following the reaction Carbide + ferrite → Fe3C, as in Figures 5g & 5f, where the microstructures are completely different from the initial microstructure. Figure 5g can only represent the orbit.
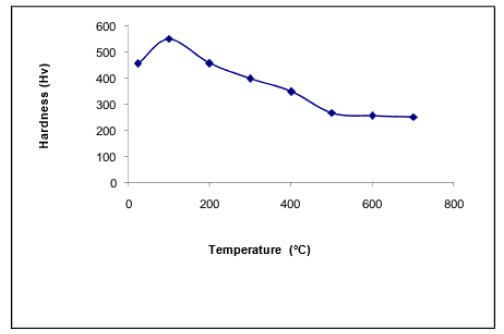
The variation in hardness of 0.38% C steel, previously quenched and then tempered at different temperatures, is illustrated in Figure 6. We note an increase in hardness followed by a decrease with increasing tempering temperature. The increase in hardness corresponds to the simultaneous formation of very fine ε carbide (the cause of hardness) and relatively soft, low- carbon martensite. The decrease in hardness corresponds to the transformation of the tempered martensite, and particularly its carbon depletion, because, according to the literature, the continuous carbon depletion of martensite is the main factor in the decrease in hardness above 150 °C. It has been found that the increase in tempering temperature in the range of 200-400 °C leads to insignificant decrease in strength and an increase in the impact of toughness [9]. The decrease in hardness can also be due to morphological change (coalescence of cementite) and structural change characterized by the transformation of tempered martensite into ferrite and formation of cementite, which will ultimately give a microstructure composed of ferrite and cementite (sorbite).
Figure 5:Microstructure of 0.38%C steel after homogenization for 1 hour at 950 °C and water quenching followed by tempering for 15 minutes at: (a) -100 °C (b) -200 °C (c) -300 °C (d) -400 °C (e) -500 °C (f) -600 °C and (g) -700 °C.

Figure 6:Vickers hardness variation curve of 0.38% C steel quenched in water from 950 °C and then tempered for 15min at different temperatures.
Conclusion
Based on study results of the effect of tempering temperature of 0.38 percent Carbon steel, it has been shown that the hardness of steel varies according to the tempering temperature. It increases and then decreases as the tempering temperature increases. Consequently, the user of this steel can choose the appropriate tempering temperature for a particular application.
References
- Wang Y, Feng C, Lin T, Zhu R, Zhang J, et al. (2025) A review of wear-resistant coatings for steel substrates: Applications and Metals 15(11): 1231.
- Oseme OS, Chinasa NC, Dickson Ofuyekpone O (2026) The role of carbon steel as a fundamental material in oil and gas industry applications: exploring the properties, performance, and protection strategies for enhanced durability in harsh environments. Nanotechnology and Nanomaterials.
- Khansaa DS, Bassam AA, Intesar NF (2018) Effect of quenching media on mechanical properties of medium carbon steel 1030. Journal of University of Babylon, Engineering Sciences 26(2): 412.
- Ejiko SO, Fatona AS, Ibikunle RA (2024) Investigation of quenching on microstructure and mechanical properties of mild steel. IOSR Journal of Engineering 14(4): 29-38.
- Chen Z, Peng Y, She X, Wang X, Liu H (2025) Quenching internal stress evolution and shape control in gigapascal ultra-high-strength martensitic steel. Metals 15(12): 1298.
- Samuel A, Prabhu KN (2022) Residual stress and distortion during quench hardening of steels: A Review. J of Materi Eng and Perform 31: 5161-5188.
- Carreno-Saavedra JI, Ros-Yanez T, García CI, Hernández-Durán EI, Iquilio RA, et al. (2023) Application of ultrafast heating and tempering to plate steel. Materials Science and Technology 39(16): 2417-2432.
- Sahoo G, Singh KK, Kumar V (2020) Quenched and tempered high strength steel: A review. Journal of Metals Materials and Minerals 30(4): 19-29.
- Zhang H, Mingshuai H, Zhiqiang M, Hui W, Guanqiao S, et al. (2022) Effects of quenching and tempering heat treatment processing on the microstructure and properties of high-strength hull steel. Metals 12(6): 914.
© 2026 © Zakaria Boumerzoug. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






