- Submissions

Full Text
Research & Development in Material Science
Study of Thermophysical Properties at High Temperatures for Some Nanomaterials
Ashwini Dixit* and Bindushekhar Sharma
Department of Physics, Dr. Bhimrao Ambedkar University, India
*Corresponding author:Ashwini Dixit, Department of Physics, I. B. S. Khandari Campus, Dr. Bhimrao Ambedkar University, Agra- 282002, India
Submission: June 05, 2024;Published: June 14, 2024

ISSN: 2576-8840 Volume 20 Issue 2
Abstract
A simple theoretical model is developed to study the effect of temperature on nanomaterials. It is found that the volume thermal experimental data for nanomaterials under study can be reproduced satisfactorily with the help of Liu model, which takes into account the temperature dependence of the Anderson- Grüneisen parameter. The Anderson model for investigating the temperature dependence of volume expansion of some nanomaterials viz. Zirconia, Ag, ZnO, NiO and Al compounds have been used by Kumar model to obtain results in an approximately fair agreement with the available experimental data. The model used by them is based on the assumption that the Anderson-Gru ̈neisen parameter does not depend on temperature.
Keywords:Nanomaterials; Volume thermal expansion; Anderson – Grüneisen parameter; Isobaric equation of state
Introduction
Nanomaterials are the central importance in the field of science and technology. The properties of nanomaterials are modified significantly at high temperatures [1,2]. Kumar et al. [3] have studied the temperature dependence of volume expansion of some nanomaterials viz. Zirconia, Ag, Zno, NiO and Al compounds using the Anderson formulation [4] which is based on the assumption that the Anderson- Grüneisen parameter Tδ is independent of temperature, i.e. Tδ does not change with the variation in temperature. It was found by Liu [5,6] that to take account of the temperature dependence of the Anderson- Grüneisen the Anderson formulation is necessary in order to improve the agreement between theoretical results and available experimental data.
Method of Analysis
Anderson and Zou [7] used the basic calculus to study the thermal properties of materials
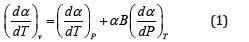
Where 𝛼 is the volume thermal expansion coefficient or simply the thermal Expansivity,

And B the isothermal bulk modulus given below

Eq. (1) has the status of an identity. The Anderson-Grneisen parameter δT is related to the temperature derivative of bulk modulus as follows [4]

Using another identity between thermodynamic quantities [8,9]
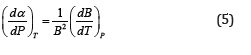
In Eq. (4), we get
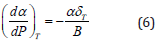
Using Eq. (5) in Eq. (1), we get
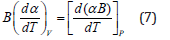
For most of the materials, the product remains constant in the high temperature region above the Debye temperature. Hence, Eq. (7) gives (dα/dT)V=0, and then Eq. (1) yields.
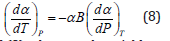
Equations (6) and (8) takes together yield

On integrating Eq. (9) with respect to temperature T between the limits T0 and a value of high temperature T, and taking a constant value of δT equal to δ0T , we get
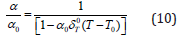
Taking Eq. (2) in Eq. (10), and then further integrating, we find
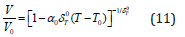
Where V0 is the volume V at T−T0, unified temperature. It is found [3] that the results for volume thermal expansion versus temperature calculated from Eq. (11) for the nanomaterials under study derivative significantly from the experimental data. This discrepancy appears due to the assumption that δT giving the following expression [5]
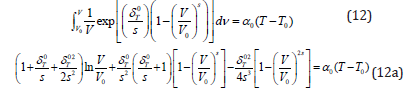
As in Eq. (12) the integral on left hand side, cannot be evaluated exactly with the help of an analytical method, and therefore [5,10] used an approximation procedure by truncating the expansion of the exponential function up to quadratic term in
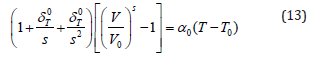
Where 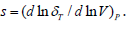 . “s” is the dimensionless
thermoelastic parameter whose [4] value is about s=+2, Eq.
(12a) is in fact an isobaric equation of state determining V/V0 as a
function of temperature along an isobar at P = 0 for different types
of materials.
. “s” is the dimensionless
thermoelastic parameter whose [4] value is about s=+2, Eq.
(12a) is in fact an isobaric equation of state determining V/V0 as a
function of temperature along an isobar at P = 0 for different types
of materials.
Discussion and conclusion
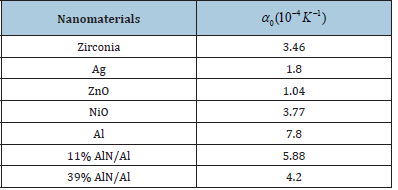
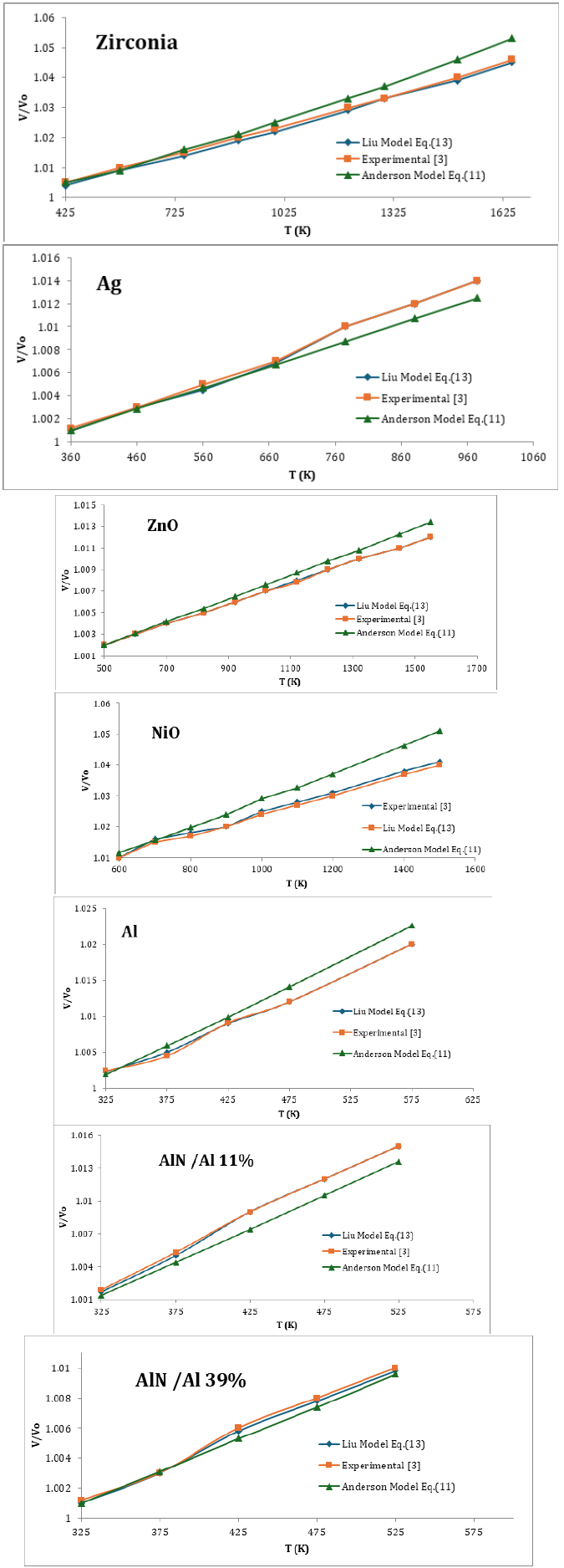
Value of δ0T , the Anderson- Grneisen at room temperature and atmospheric pressure remain almost between 4 and 6 for different types of materials [7]. For nanomaterials, accurate values of δ0T are not known, we have calculated V/V0 , taken three representative values of δ0T equal to 4, 5, and 6 for each material. Values of 0 α used as input in calculations are given in Table 1 of Kumar et al. [3],[11].
Table 1:Value of input parameters used in present work [3,11].
In order to demonstrate the validity of Eq. (13) and (11) are calculated as a function of temperature. Because one should check for consistencies using the same set only with Eq. (11) with experimental data [3], here we have used seven nanomaterials in present study, the value of α0 , δ0T are used as input parameters which are given in Table 1. Eq. (13) and (11) are used to compute temperature dependence of V/Vo at different temperature of nanomaterials. The result are compared with experimental data [3] we found Liu Model (13) is very consistent with experimental data [3] we can say that Liu Model can reproduced the experimental data in figure of merit The present work is simple and straightforward method to study the effect of temperature on nanomaterials due to the simplicity and applicability it may be the current interest to the researchers engaged in this field. To the best of our knowledge such simple methods are not yet available in the literature for nanomaterials (Tables 2-4; Figures 1-3).
Table 2:Temperature dependence of V/Vo of Eqs. (13) and (11) with the value δ0T=4
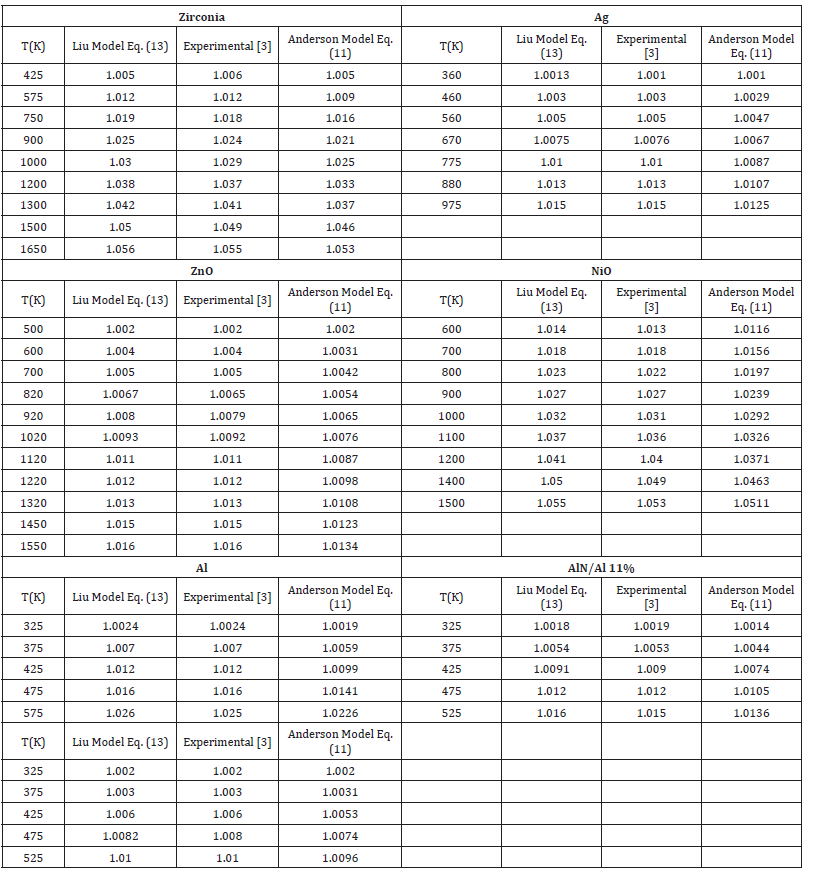
Table 3:Temperature dependence of V/Vo of Eqs. (13) and (11) with the value δ0T=5.
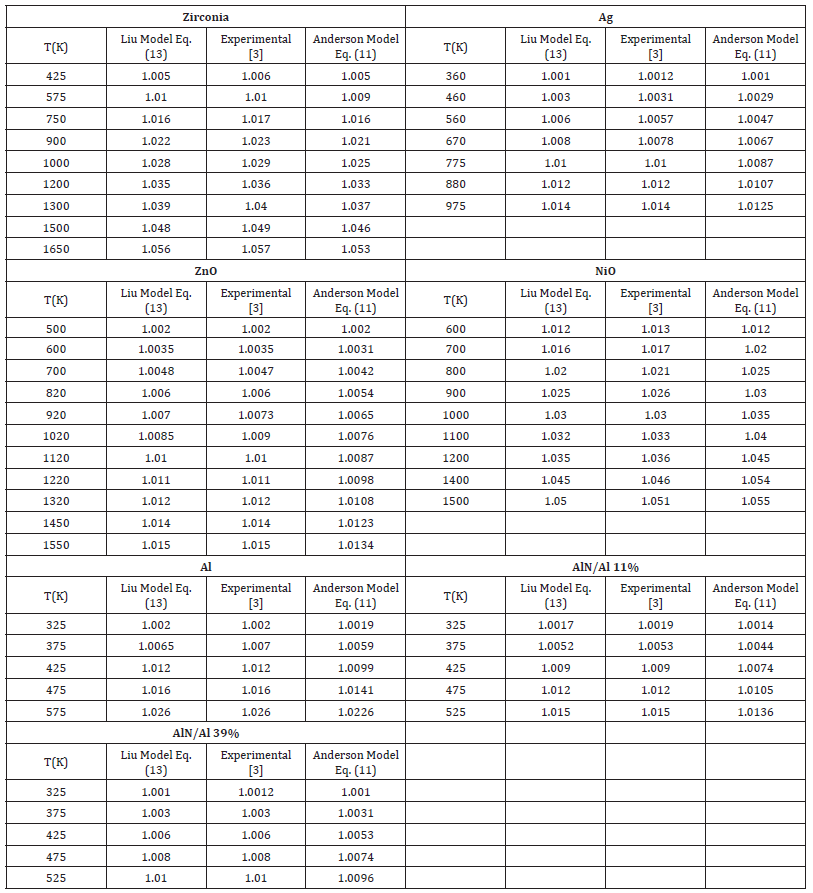
Table 4:Temperature dependence of V/Vo of Eqs. (13) and (11) with the value δ0T=6.
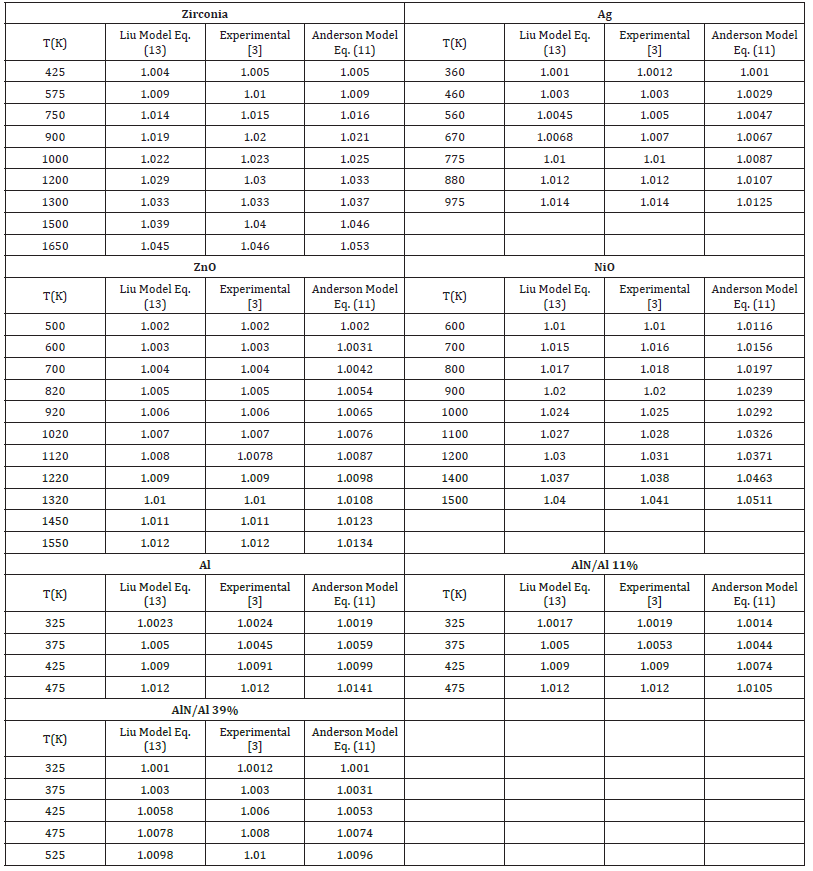
Figure 1:
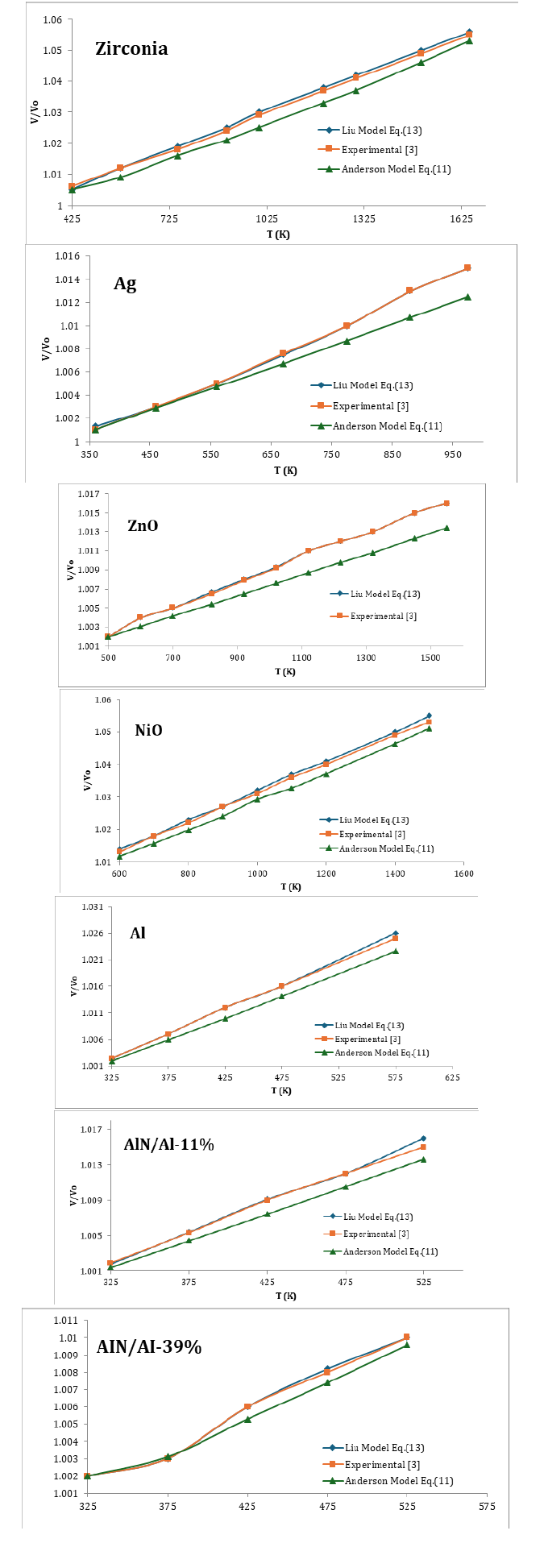
Figure 2:
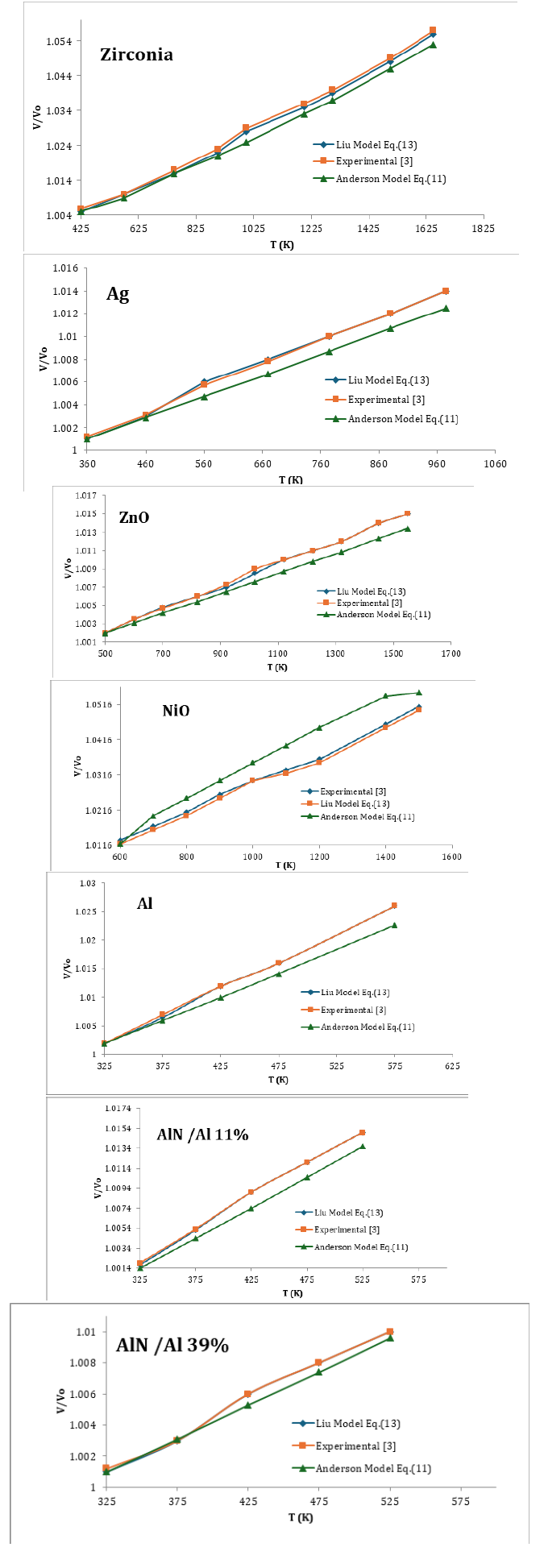
Figure 3:
References
- Bhagwat M, Ramaswami V (2004) Synthesis of nanocrystalline zirconia by amorphous citrate route: structural and thermal (HTXRD) studies. Mater Res Bull 39(11): 1627-1640.
- Hu Y, Tsai HL, Huang CL (2003) Phase transformation of precipitated TiO2 Mater Sci Eng A 344(1-2): 209-214.
- Kumar R, Bhatt A, Kumar M (2011) Theoretical formulation for the effect of temperature on nanomaterials. Modern Physics Letters B 25(27): 2149-2157.
- Anderson OL (1995) Equation of state for solid for geophysics and ceramic sciences. Oxford University Press, Oxford, England.
- Liu Q (2016) Temperature dependence of interatomic separation and bulk modulus for alkali halides. Int J Thermophys 37(65).
- Luo YX, Liu Q (2019) Response to the Remark on Temperature Dependence of Interatomic Separation and Bulk Modulus for Alkali Halides. Int J Thermophysics 40(34).
- Anderson OL, Zou K (1990) Thermodynamic functions and properties of MgO at high compression and high temperature. J Phys Chem Ref Data 19(69).
- Stacey FD, Davis PM (2004) High pressure equations of state with applications to the lower mantle and core. Phys Earth Planet Inter 142(3-4): 137-184.
- Stacey FD (2005) High pressure equations of state and planetary interiors. Rep Prog Phys 68(2): 341.
- Anand K, Singh MP, Sharma BS (2019) A Comment on the Paper Entitled “Temperature Dependence of Interatomic Separation and Bulk Modulus for Alkali Halides” Published in Int. J. Thermophys (2016) 37.65 DOI 10.1007/s10765-016-2069-6. Int J Thermophys 40(33).
- Singh M, Gupta BRK (2014) Study of thermoelastic properties of nanomaterials under high temperature. IJAET 6(6): 2514-2523.
© 2024 Ashwini Dixit. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






