- Submissions

Full Text
Researches in Arthritis & Bone Study
Extraosseous Membranous Ossification in Soft Tissue: A Case Series with Implications for Cardiac Calcification and the Oss Cordis
García-Palomeque JC1*, Cabezón Ruiz S2 and Treceño Fernández L1
1Anatomy Department and Human Embryology, Cádiz University, Spain
2Basic Sciences Department, School of Medicine, Cardiology Service, Virgen del Rocio University Hospital, Huelva University, Seville, Spain
*Corresponding author:García-Palomeque JC, Anatomy Department and Human Embryology, School of Medicine, Cádiz University, Spain
Submission: January 09, 2026;Published: February 24, 2026
Volume2 Issue3February 24, 2026
Abstract
Introduction: Endochondral and membranous ossification is the fundamental physiological mechanism
of bone growth; however, under certain conditions, it may be pathologically activated in extraosseous
tissues. This phenomenon has been closely associated with chronic inflammation and altered tissue
microenvironments and has been described in multiple anatomical locations, particularly in tissues
with synovial physiology. In the cardiovascular system, calcification and ossification of structures
such as cardiac valves, the fibrous skeleton and the development of the oss cordis represent clinical
manifestations of similar biological processes.
Objective: To describe the histopathological features of benign extraosseous cartilaginous lesions
associated with chronic inflammation in soft tissues and to explore their parallels with the mechanisms
involved in cardiac calcification and ossification, including oss cordis formation.
Methods: A retrospective analysis was conducted on seven cases of benign extraosseous cartilaginous
tumors with histological evidence of chronic inflammation. Specimens were processed using conventional
histological techniques and evaluated by an experienced pathologist, focusing on patterns of chondrogenic
differentiation and endochondral ossification.
Result: All cases demonstrated ectopic cartilaginous tissue with features consistent with organized
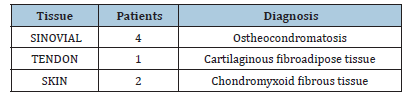
endochondral ossification. Lesions were in synovial tissue, tendon sheath, skin and showed
chronic inflammatory infiltrates in the surrounding stroma. Histopathological diagnoses included
osteochondromatosis, cartilaginous fibroadipose tissue and chondromyxoid fibrous tissue.
Conclusions: Chronic inflammation emerges as a central factor in the aberrant activation of chondrogenic
and osteogenic differentiation programs in soft tissues, leading to extraosseous endochondral ossification.
Histopathological and molecular parallels with cardiac calcification and ossification suggest a shared
etiopathogenic basis, with relevant implications for the development of therapeutic strategies targeting
early stages of the process.
Keywords:Endochondral ossification; Extraosseous ossification; Chronic inflammation; Synovial tissue; Cardiac calcification; Oss cordis
Introduction
Endochondral ossification is a highly regulated biological process that plays an essential role in the formation and growth of long bones during embryonic and postnatal development. This mechanism involves the sequential differentiation of mesenchymal cells into chondrocytes, subsequent chondrocyte hypertrophy, mineralization of the cartilaginous matrix and eventual replacement by mature lamellar bone. These events are tightly controlled by complex molecular signaling networks, including the BMP, TGF-β and Wnt/β-catenin pathways. Although endochondral ossification has traditionally been considered exclusive to skeletal tissue, accumulating evidence indicates that this program can be aberrantly activated in extraosseous tissues. In this context, many calcifications previously interpreted as passive dystrophic phenomena represent organized processes of ectopic chondrogenesis followed by endochondral ossification. This conceptual shift has enabled pathological calcification to be understood as a biologically active, dynamic and regulated process.
Tissues with synovial physiology-such as joints, tendon sheaths, and bursae-constitute an environment particularly susceptible to these phenomena. The combination of chronic mechanical stress, an extracellular matrix rich in proteoglycans and persistent proinflammatory cytokine signaling creates a microenvironment conducive to aberrant mesenchymal differentiation toward chondrogenic and osteogenic phenotypes. In this setting, chronic inflammation acts as a potent modulator of cellular plasticity. A similar paradigm shift has occurred in the cardiovascular system. Valvular calcification and ossification of the cardiac fibrous skeleton, historically regarded as passive degenerative processes, are now recognized as the result of active mechanisms that recapitulate stages of osteogenesis and endochondral ossification [1-6]. The formation of the oss cordis represents the most organized and advanced expression of this process. In the present study, we report a series of cases of extraosseous endochondral ossification in soft tissues, aiming to characterize their histopathological features and to discuss their implications in relation to cardiac calcification and ossification [7,8].
Materials and Methods
Study design and case selection
A retrospective study was conducted based on the analysis of seven cases of benign extraosseous cartilaginous lesions diagnosed histologically [9]. Cases were selected from archived material in the pathology department using strict inclusion criteria: Histological confirmation of a benign cartilaginous lesion, extraosseous location, origin in synovial or synovium-like tissues and evidence of chronic inflammation in the surrounding tissue.
Histopathological evaluation
All specimens were fixed in 10% buffered formalin and embedded in paraffin. Histological sections of 4μm thickness were prepared and stained with hematoxylin and eosin. Histological analysis focused on the identification of ectopic hyaline cartilage, the presence of endochondral ossification patterns-including chondrocyte hypertrophy, matrix calcification and lamellar bone formation-the type and intensity of the inflammatory infiltrate and the organization of the extracellular matrix [10-12]. Diagnostic evaluation was performed by a pathologist with expertise in soft tissue and cartilaginous pathology.
Result
In all seven cases, ectopic cartilaginous tissue with histological features consistent with organized endochondral ossification was identified. Lesions were in synovial tissue (four cases), tendon sheath (one case) and skin (two cases). Histopathological diagnoses included osteochondromatosis, cartilaginous fibroadipose tissue and chondromyxoid fibrous tissue (Figure 1). Chronic inflammatory infiltrates with a predominance of mononuclear cells were observed in the stroma adjacent to the cartilaginous tissue in all cases [13- 16]. Cartilaginous areas exhibited hypertrophic chondrocytes, focal matrix calcification and progressive transition toward lamellar bone trabeculae containing osteocytes within lacunae, confirming a clear pattern of extraosseous membranous ossification.
Figure 1:Histological features of the oss cordis.
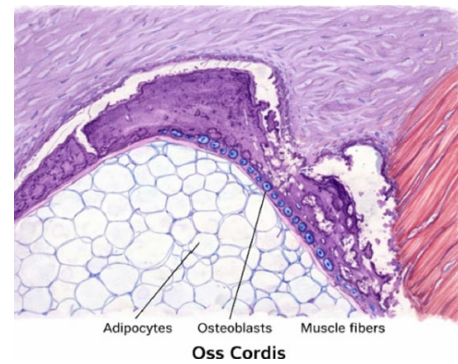
Histological Features of the Oss Cordis and Cardiac Tissue
From a histopathological perspective, the Oss cordis is characterized by the presence of mature bone tissue integrated within the cardiac fibrous skeleton [17,18]. This bone tissue displays an organized architecture, preceded by well-defined stages of hyaline cartilage formation with hypertrophic chondrocytes, extracellular matrix calcification and subsequent replacement by lamellar bone. Active vascularization and chronic inflammatory infiltrates are frequently observed in adjacent areas, indicating a dynamic and regulated process. Figure 1 illustrates a representative example of this histological pattern in a cardiac context [19]. These findings confirm that the Oss cordis does not represent an amorphous calcific deposit but rather a true phenomenon of ectopic endochondral ossification. The Sirtuin 1 (SIRT 1) gen encodes a NAD dependent deacetylase belonging to the sirtuin family, a group of proteins involved in epigenetic regulation, energy metabolism and cellular stress responses SIRT1 functions as a metabolic sensor that modulates gene expression through post-translational deacetylation of key transcription factors and regulatory proteins, including NF-kB, FOXO, transcriptions factors, p 53 and PGC-1 alfa [20-24]. Through these interactions, SIRT 1 plays a central role in the regulation of inflammation, oxidative stress, mitochondrial functions and cellular senescence SIRT 1 is widely expressed in cardiovascular tissues, where it contributes to the maintenance of cellular homeostasis and structural integrity.
In the context of valvular heart disease particularly calcific aortic valve disease, SIRT 1 exerts a predominantly protective role. Reduced expression of activity of SIRT 1, commonly observed during aging and in metabolic disorders, promotes osteogenic differentiation of valvular interstitial cells through upregulation of osteogenic signaling pathways involving transcription factor [25]. From a pharmacological perspective SIRT 1 activity can be modulated by both agonists and antagonists, most of which have been investigated in preclinical settings. Natural compounds such as resveratrol act as indirect activators of SIRT1, while synthetic small molecules including SRT1720 and SRT2104 exhibit greater specificity and potency [26-29]. These agents have demonstrated anti-inflammatory and anticalcific effects in experimental models. In constract, SIRT 1 inhibitor including sirtinol and selisistat supress sirt 1 activitty and have the oppositive effect (Table 1).
Table 1:Tissue patients diagnosis synovial.
Discussion
The findings of this case series support the hypothesis that chronic inflammation in soft tissues with synovial or synoviumlike physiology can activate aberrant mesenchymal differentiation programs, leading to ectopic cartilage formation and subsequent endochondral ossification. This process represents an active, highly organized phenomenon regulated by molecular signals shared with physiological osteogenesis. In the cardiovascular field, numerous studies have demonstrated that valvular calcification and ossification of the cardiac fibrous skeleton follow a similar pathogenic sequence, characterized by inflammation, recruitment of progenitor cells, osteochondrogenic differentiation and organized mineralization [30]. The expression of key regulatory factors such as BMPs, osteopontin and Runx2 in calcified valves further reinforces the analogy between these processes. The oss cordis represents the structural extreme of this pathological spectrum. From a translational perspective, distinguishing between passive dystrophic calcification and active endochondral ossification is essential. Given that chronic inflammation constitutes a common denominator, therapeutic strategies aimed at modulating inflammatory responses or interfering with osteochondrogenic signaling pathways may have potential applications in both articular disorders and degenerative cardiovascular diseases.
Conclusion
Extraosseous endochondral ossification in soft tissues associated with synovial physiology is an active process driven by chronic inflammation and aberrant mesenchymal differentiation. Histopathological and molecular parallels with cardiac calcification and ossification, including the Oss cordis, suggest shared etiopathogenic mechanisms. This model provides a robust foundation for exploring therapeutic interventions targeting early stages of the process.
References
- Aikawa E, Whittaker P, Farber M, Padera RF, Aikawa M, et al. (2006) Human semilunar cardiac valve remodeling by activated cells from fetus to adult: Implications for postnatal adaptation, pathology and tissue engineering. Circulation 113(10): 1344-1352.
- Rajamannan NM, Evans FJ, Aikawa E, Grande-Allen KJ, Demer LL, et al. (2011) Calcific aortic valve disease: Not simply a degenerative process. N Engl J Med 364: 146-156.
- Otto CM, Prendergast B (2014) Aortic-valve stenosis- from patients at risk to severe valve obstruction. N Engl J Med 371(8): 744-756.
- Mohler ER, Gannon F, Reynolds C, Keane MG, Kaplan FS, et al. (2001) Bone formation and inflammation in cardiac valves. Circulation 103(11): 1522-1528.
- Towler DA (2013) Molecular and cellular aspects of calcific aortic valve disease. Circ Res 113(2): 198-208.
- Sage AP, Tintut Y, Demer LL (2010) Regulatory mechanisms in vascular calcification. Nat Rev Cardiol 7(9): 528-536.
- Mathieu P, Boulanger MC (2014) Basic mechanisms of calcific aortic valve disease. Can J Cardiol 30(9): 982-993.
- Boström K, Watson KE, Horn S, Herman IM, Demer LL, et al. (1993) Bone morphogenetic protein expression in human atherosclerotic lesions. J Clin Invest 91(4): 1800-1809.
- Hinton RB, Yutzey KE (2011) Heart valve structure and function in development and disease. Annu Rev Physiol 73: 29-46.
- Chen JH, Yip CY, Sone ED, Simmons CA (2009) Identification and characterization of aortic valve mesenchymal progenitor cells with osteogenic calcification potential. Am J Pathol 174(3): 1109-1119.
- New SE, Aikawa E (2011) Molecular imaging insights into early inflammatory stages of arterial and aortic valve calcification. Circ Res 108(11): 1381-1391.
- Mohler ER (2004) Mechanisms of aortic valve calcification. Am J Cardiol 94(11): 1396-1402.
- Dweck MR, Boon NA, Newby DE (2012) Calcific aortic stenosis: A disease of the valve and the myocardium. J Am Coll Cardiol 60(19): 1854-1863.
- Rajamannan NM (2011) Calcific aortic valve disease: Cellular origins of valve calcification. Arterioscler Thromb Vasc Biol 31(12): 2777-2782.
- Bobryshev YV, Lord RS, Warren BA (1999) Calcified deposit formation in human aortic valves. Cardiovasc Res 41: 200-210.
- Aikawa E, Libby P (2004) Atherosclerotic calcification: Bone formation in the artery wall. Curr Opin Lipidol 15: 555-561.
- Hortells L, Sosa C, Guillén N, Lucea S, Millán Á, et al. (2018) Identifying early pathogenic events during vascular calcification. J Mol Cell Cardiol 121: 34-44.
- Yutzey KE, Demer LL, Body SC, Huggins GS, Towler DA, et al. (2014) Calcific aortic valve disease: A consensus summary from the alliance of investigators on calcific aortic valve disease. Circ Res 114: 1104-1115.
- Durham AL, Speer MY, Scatena M, Giachelli CM, Shanahan CM (2018) Role of smooth muscle cells in vascular calcification. Nat Rev Cardiol 15: 387-398.
- Anderson HC (1995) Molecular biology of matrix vesicles. Clin Orthop Relat Res 314: 266-280.
- Boskey AL (2004) Biomineralization: Conflicts, challenges and opportunities. J Cell Biochem 92: 1161-1170.
- Kirsch T (2015) Regulation of chondrocyte differentiation. Bone 75: 55-61.
- Steiner I, Krbal L, Rozsypal J (2007) Ossification of the cardiac fibrous skeleton in humans. Virchows Arch 451: 709-717.
- Simionescu A, Simionescu DT, Deac R (2009) Biology of calcified aortic valve disease. Cardiovasc Pathol 18: 1-7.
- Miller JD, Weiss RM, Heistad DD (2011) Calcific aortic valve stenosis: Methods, models and mechanisms. Circ Res 108(11): 1392-1412.
- Kaden JJ, Bickelhaupt S, Grobholz R, Haase KK, Sarikoc A, et al. (2004) Expression of bone sialoprotein and bone morphogenetic protein-2 in calcific aortic stenosis. J Heart Valve Dis 13(4): 560-566.
- Peacock JD, Levay AK, Gillaspie DB, Tao G, Lincoln J (2010) Reduced Sox9 function promotes heart valve calcification phenotypes invivo. Circ Res 106(4): 712-719.
- Artiach G, Carracedo M, Clària J, Bäck M (2020) Omega-3 polyunsaturated fatty acids in cardiovascular disease: Insights from resolution of inflammation. Kardiol Pol 78: 93-95.
- Towler DA, Shao JS, Cheng SL, Pingsterhaus JM, Loewy AP (2006) Osteogenic regulation of vascular calcification. Ann N Y Acad Sci 1068: 327-333.
- Demer LL, Tintut Y (2011) The roles of lipid oxidation products and rank signaling in atherosclerotic calcification. Circ Res 108: 1482-1493.
© 2026 García-Palomeque JC. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






