- Submissions

Full Text
Polymer Science: Peer Review Journal
Electrochemical Formation of Iron- Based Metal-Polymer Nanocomposites in a Copolymer Matrix of 1-Vinyl-1,2,4- Triazole and N-Vinylpyridone
Sargsyan SH1*, Sargsyan AS2, Khizantsyan KM3, Sargsyan TS2, Aghajanyan IG1 and Margaryan KS2
1National Polytechnic University of Armenia, Armenia
2Yerevan State Medical University named after Mkhitar Heratsi, Armenia
3Yerevan branch of the Plekhanov Russian University of Economics, Armenia
*Corresponding author:S H Sargsyan, National Polytechnic University of Armenia, Yerevan, Armenia
Submission: February 18, 2026;Published: April 10, 2026

ISSN: 2770-6613 Volume6 Issue 2
Abstract
This work presents an electrochemical approach for producing iron-containing metal-polymer nanocomposites and surface coatings on electrodes made of pure iron, steel, and magnesium. The synthesis occurs during the simultaneous electrochemical copolymerization of 1-vinyl-1,2,4-triazole with N-vinylpyridone and the concurrent cathodic deposition of metallic species. Characterization by Electron Paramagnetic Resonance (EPR) and Transmission Electron Microscopy (TEM) revealed that the synthesized materials contain both zero-valent iron and iron oxides, predominantly exhibiting spherical morphologies. The resulting nanocomposites contain nanoparticles with diameters between 2 and 4nm and undergo multistage thermal degradation. The obtained hybrid materials are promising for use in catalysis, biomedicine, and advanced construction materials.
Keywords:Electrochemical copolymerization; Metal-polymer nanocomposite; Iron nanoparticles; 1-Vinyl-1,2,4-Triazole; N-Vinylpyridone
Introduction
Metal-polymer nanocomposites represent a class of hybrid materials in which nanoscale metallic particles are dispersed within a macromolecular polymer framework. The incorporation of metal nanoparticles often leads to strong interparticle coupling, which can obscure the behavior of isolated nanoparticles and give rise to new physicochemical properties that depend on the polymer environment [1,2]. Understanding the nature of these interactions is critical for the rational design of functional nanomaterials. Iron-based nanocomposites have attracted significant scientific attention due to their unique magnetic, electronic, and catalytic characteristics. Complexes of Fe³⁺ ions with gallium-containing polyvinylpyrrolidone derivatives, for instance, have been utilized as dual-function imaging agents for both photoacoustic and magnetic resonance diagnostics. These systems exhibit rapid biodegradation and excretion, demonstrating their potential suitability for medical applications [3].
Moreover, zero-valent iron nanoparticles have been reported to accelerate microbial denitrification in water purification systems [4,5]. In recent years, electrochemical methods have emerged as a versatile and controllable strategy for producing polymeric nanomaterials. Such approaches are increasingly employed to synthesize functional coatings, biocompatible composites, and nanosystems for biomedical and technological use [1-7]. Previous research has shown that iron nanoparticles can be incorporated into polymer matrices such as Poly(1-Vinyl-1,2,4-Triazole) (PVT) through the chemical reduction of Fe²⁺ salts with sodium borohydride [8], or into Polyvinylpyrrolidone (PVP) matrices via thermal decomposition of iron pentacarbonyl or iron acetylacetonate precursors [9-16]. These methods typically yield spherical nanoparticles of Fe2O3 or Fe3O4 within the 3-30nm range, displaying high colloidal stability and biocompatibility. Despite numerous studies on the chemical synthesis of iron nanocomposites in either PVT or PVP matrices, no systematic reports exist on their electrochemical formation within a copolymer framework of 1-Vinyl-1,2,4-Triazole (VT) and N-Vinylpyridone (VP). This copolymer system is of particular interest because of its hydrophilicity, non-toxicity, and presence of nitrogen-containing functional groups capable of coordinating with metal ions or nanoparticles. Such properties make VT-VP copolymers highly suitable for biomedical and catalytic applications [17-20]. The present study aims to demonstrate the electrochemical synthesis of iron-containing nanocomposites and thin-film coatings on metallic electrodes (Fe, Mg, and steel) through the concurrent processes of copolymerization of VT with VP and cathodic metal deposition. The resulting materials were characterized by using various spectroscopic and microscopic techniques to elucidate their composition, structure, and functional properties.
Experimental Section
Materials and methods
PElectrochemical polymerization was performed in a singlecompartment glass cell without a separating diaphragm. Elemental analysis was carried out using a FLASH EA 1112 Series analyzer (Germany). Ultraviolet spectra were obtained with a Perkin Elmer Lambda 35 UV/VIS Spectrophotometer (USA). Infrared (IR) spectra of the polymeric materials were recorded on Specord M-80 and Bruker Vertex 70 spectrometers (Germany) using KBr pellets prepared from finely ground samples. The metal content of the nanocomposites was determined by both elemental and atomic absorption spectroscopy using a Perkin Elmer Analyst 200 instrument (USA). Electron Paramagnetic Resonance (EPR) spectra were measured on a Bruker ELEXSYS E-580 (X-97PI) spectrometer (Germany) operating in continuous-wave mode. Transmission electron microscopy (TEM) was performed on a Leo 906E microscope (Germany) to assess nanoparticle morphology and size distribution. Thermal analysis was conducted on a MOM derivatograph (Hungary) under a heating rate of 5 °C min⁻¹. Electrical conductivity was determined using a Teraohmmeter E6- 13A (USSR). All reagents were of analytical grade and obtained from Sigma-Aldrich, used without further purification unless otherwise specified.
Monomer preparation
1-Vinyl-1,2,4-Triazole (VT) was synthesized and purified following the method described in [21]. N-Vinylpyridone (VP) (boiling point 91-92 °C at 1mm Hg) was prepared by a vinyl exchange reaction between pyridone and vinyl acetate, as previously reported in [22].
General procedure for electrochemical synthesis
Electrochemical copolymerization and metal deposition were conducted in a 50mL glass electrolytic cell equipped with two electrodes. The working electrode (1-2cm²) consisted of either pure iron, magnesium, or steel, while a platinum or glassy carbon plate (SU-12 or SU-20) served as the counter electrode.
The electrolytic solution contained:
a. 0.5-1molL⁻¹ of VT,
b. 0.5-1molL⁻¹ of VP,
c. 1.5-4mmolL⁻¹ of FeSO4·H2O,
d. 0.02-0.05 % of 4-Tert-Butylperoxy-4-Oxobutanoic
Acid (TBOPA) as the peroxide-type initiator, and in some
experiments, 0.05-0.07 % of chitosan was added to facilitate
iron reduction..
Electrolysis was performed at potentials from -0.1 to -1.2V vs. SCE or at constant current densities ranging from 1 to 20mAcm⁻², in either aqueous or aqueous-ethanol media. The chosen potential range corresponds to the reduction potentials of both the initiator and Fe²⁺ ions, promoting simultaneous copolymer formation and metal deposition. At low current densities (≤10mAcm⁻²), uniform nanocomposite coatings were deposited on the cathode surface. When j>10mAcm⁻², loose nanocomposite precipitates formed at the bottom of the electrochemical cell. After electrolysis, the cathode was removed, thoroughly rinsed with distilled water, and dried to a constant mass under ambient conditions. The obtained coatings exhibited a characteristic orange hue, indicative of iron incorporation. Mechanical, thermomechanical, and biocompatibility properties of similar polymeric coatings have been evaluated in our previous work. According to our findings, the electrode material had no significant effect on the composition, morphology, or particle distribution within the resulting nanocomposite.
Result and Discussion
During the electrochemical copolymerization of 1-Vinyl- 1,2,4-Triazole (VT) and N-Vinylpyridone (VP) in aqueous and aqueous–ethanolic media, iron-containing Nanocomposites (NCs) and Nanocomposite Coatings (NCCs) were formed. The process proceeded efficiently in the presence of a peroxide initiator (TBOPA), whose reduction potential is close to that of Fe²⁺ ions (-0.6 to -1.2V vs. SCE). By varying the monomer ratio, iron salt concentration, and applied current density, it was possible to control the metal content in the composites within the range of 0.4-6 wt%. The resulting materials exhibited an orange coloration and were fully soluble in water, indicating their homogeneous polymeric nature.
IR spectroscopic analysis
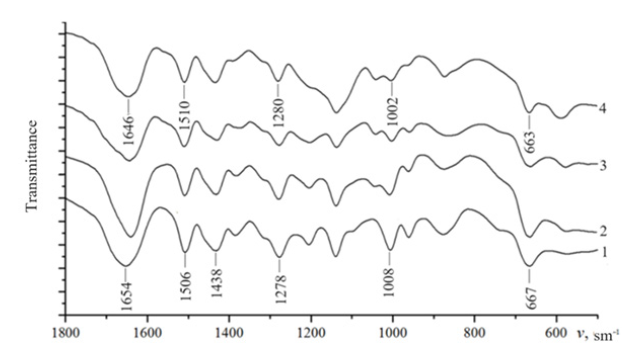
The infrared spectra of the VT-VP copolymers (Figure 1) showed characteristic vibrational bands associated with the triazole and pyridone rings: stretching and deformation modes at 1654, 1506, 1438, 1278, 1008, and 667cm⁻¹ corresponding to the C=N, C-N, N-N, C-H, and C=O groups. Upon coordination with iron species, slight shifts (by 2-12cm⁻¹) in these absorption bands were observed, suggesting the participation of nitrogen atoms in coordination with both Fe²⁺/Fe³⁺ ions and metallic Fe⁰ nanoparticles. Such spectral changes confirm the formation of chemical interactions between the polymer matrix and iron centers.
Figure 1:IR spectra of copolymers 1 - Fe nanocomposites%; 2 - 1,6%; 3 -3%; 4 - 5,8%.
Morphology and particle size distribution
Transmission Electron Microscopy (TEM) studies demonstrated that the synthesized nanocomposites consist of nearly spherical nanoparticles embedded in the polymer matrix (Figure 2). Alongside metallic Fe⁰ particles, iron oxides were also detected, primarily with a spherical morphology.
Figure 2:Electron micrograph (a) and histogram of the distribution of iron oxide nanoparticles by size (b) NC Fe - 1.6%.
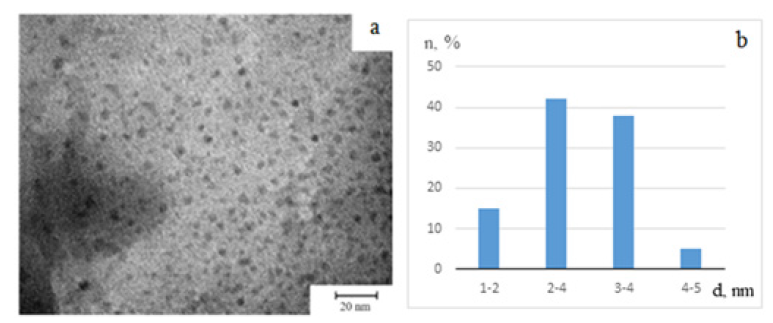
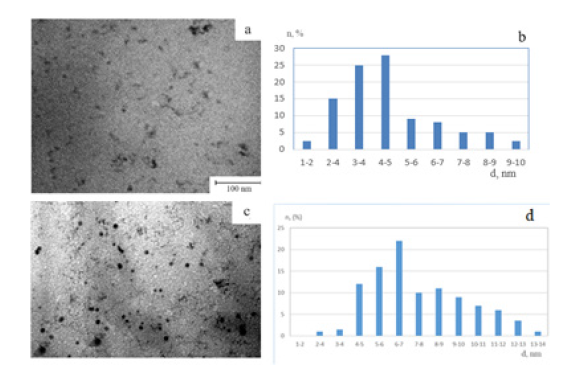
Quantitative analysis indicated that the ratio of iron oxides to metallic iron is approximately 65:35, reflecting the coexistence of both oxidized and zero-valent forms within the composite. Nanocomposites with lower iron content exhibited a more uniform nanoparticle distribution, with average sizes in the 1-6nm range. Increasing iron concentration led to a broader size distribution, reaching 14nm. This tendency arises because at higher metal loadings, a larger number of nucleation centers form simultaneously, which promotes particle aggregation and partial coagulation within the polymer network (Figure 3). At even higher iron concentrations, small clusters or aggregates composed of several nanoparticles were observed. These clusters, typically 50-80nm in diameter, remained stabilized by polymer chains, indicating strong coordination between the macromolecular framework and the metallic phase.
Figure 3:Electron micrographs (a, c) and histograms of the distribution of iron oxide nanoparticles by size (b, d), (a, c) - Fe - 3%, (c, d) - 5.8%.
EPR spectroscopic characterization
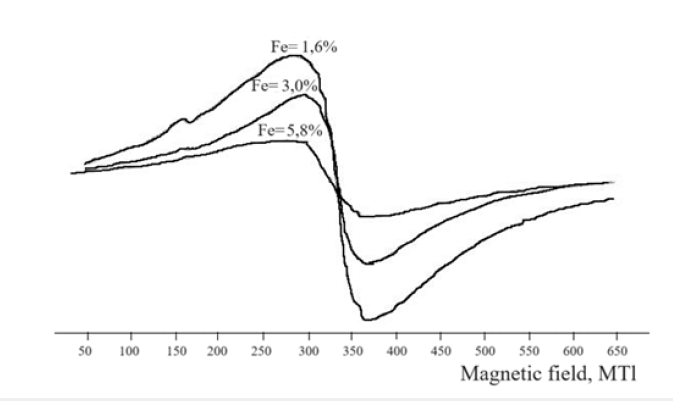
Electron paramagnetic resonance (EPR) spectra of the nanocomposites (Figure 4) displayed a broad asymmetric resonance line with an effective g-factor in the 2.04-2.10 range and a linewidth of 70-100mT, which is typical for high-spin iron centers. Additional signals with g-factors between 1.876 and 4.3 confirmed the coexistence of Fe³⁺ species in various environments [23,24]. The variation in g-values and line widths with increasing iron concentration reflects differences in nanoparticle size, morphology, and relaxation dynamics of the Fe-O bonds within the polymer matrix. In particular, a resonance signal at g=4.3 was attributed to γ-Fe₂O₃ nanoparticles stabilized in the polymer environment [25,26]. The nanocomposite containing approximately 1.6% Fe exhibited the broadest asymmetric line, corresponding to a near-equal proportion of Fe₂O₃ and Fe₃O₄ phases. In addition, the presence of a signal at g=1.9-2.1 provides direct evidence for zero-valent iron (Fe⁰), supporting the formation of core-shell nanostructures, where an iron core is surrounded by an oxide shell [27].
Figure 4:EPR spectrum of the NC (Fe) complex (%), 1 - 1,6; 2 - 3,0; 2 - 5,8.
Thermal stability
Thermogravimetric Analysis (TGA) results (Figure 5) demonstrated that the pristine VT-VP copolymer decomposes at higher temperatures (~325 °C) than the iron-containing nanocomposites (250-285 °C). Increasing the metal fraction resulted in a gradual reduction of decomposition temperature by 35-70 °C, suggesting that iron species catalyze polymer degradation processes.
Figure 5:TGA curves nanocomposite Fe - 3.0%.
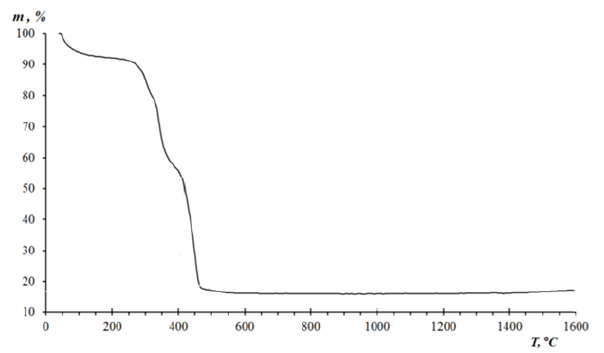
A small mass loss (~8%) occurred in the 60-155 °C range, corresponding to desorption of physically adsorbed water. Further decomposition between 240-385 °C was attributed to the elimination of side groups and functional fragments from polymer chains. The final oxidation of the hydrocarbon backbone occurred between 390-500 °C, accompanied by the evolution of CO₂ and other volatile products. These observations confirm a multistage thermal degradation pathway, with iron nanoparticles acting as active catalytic centers that promote oxidative and thermolytic processes.
Electrical conductivity
The electrical conductivity of the obtained nanocomposites was measured to be within 10⁻¹¹-10⁻¹⁰S·cm⁻¹, which is two to three orders of magnitude higher than that of the pristine copolymer (3.8×10⁻¹³S·cm⁻¹). The conductivity enhancement is attributed to tunneling electron transport between adjacent metallic nanoparticles dispersed in the polymer matrix [28]. This behavior demonstrates that even at low metal content, the nanocomposite forms quasi-continuous conductive pathways, making it potentially suitable for use in antistatic coatings and microelectronic applications.
Stability and morphological integrity
Preliminary tests revealed that both the composition and structure of the nanocomposites remained stable under ambient conditions for at least one month. No visible color changes or signs of oxidation were detected, confirming that the polymer matrix effectively prevents nanoparticle aggregation and surface oxidation.
Conclusion
This study confirms the feasibility of Synthesizing Iron- and Iron Oxide-Based Nanocomposites (NCs and NCCs) through electrochemical methods involving the copolymerization of 1-Vinyl- 1,2,4-Triazole (VT) with N-Vinylpyridone (VP). The process was carried out on electrodes composed of pure iron, magnesium, or steel, leading to the formation of polymer-stabilized nanostructured materials. EPR and TEM analyses demonstrated that the obtained nanocomposites contain both zero-valent iron (Fe⁰) and iron oxides (Fe₂O₃, Fe₃O₄). The data suggest that the particles possess a core–shell morphology, where the metallic core is surrounded by an oxide layer. The solubility of the materials in water is associated with intermolecular interactions between the VT-VP macromolecules and the dispersed metal nanoparticles, providing structural stability and homogeneity. Thermal and electrical studies showed that the introduction of iron significantly influences the physicochemical behavior of the copolymer matrix. While the presence of iron decreases thermal stability due to its catalytic role in degradation, it simultaneously enhances electrical conductivity by facilitating localized tunneling currents between neighboring nanoparticles. The developed electrochemical route offers a simple, controllable, and environmentally benign approach for producing iron-containing nanocomposites with tailored properties. These materials hold promise for applications in biomedicine, catalysis, and microelectronics, where combined magnetic and conductive characteristics are required.
References
- Gusev AI (2007) Nanomaterials, Nanostructures, Nanotechnologies. Fizmatlit, Moscow, Russia.
- Sergeev GB, Klabunde KJ (2013) Nanochemistry, (2nd edn), Elsevier.
- Chen L, Chen J, Qiu S, Wen L, Wu Y, et al. (2018) Biodegradable nanoagents with short biological half-life for SPECT/PAI/MRI multimodality imaging and PTT therapy of tumors. Small 14(4): 1702700.
- Jiang C, Liu Y, Chen Z, Megharaj M, Naidu R (2013) Impact of iron-based nanoparticles on microbial denitrification by paracoccus sp. YF1. Aquatic Toxicology 142(143): 329-335.
- Liu Y, Li S, Chen Z, Megharaj M, Naidu R (2014) Influence of zero-valent iron nanoparticles on nitrate removal by paracoccus sp. Chemosphere 108: 426-432.
- Lee N, Yoo D, Ling D, Cho MH, Hyeon T, et al. (2015) Iron oxide-based nanoparticles for multimodal imaging and magnetoresponsive therapy. Chemical Reviews 115(19): 10637-10689.
- Nikiforov VN, Filinova EY (2009) Biomedical applications of magnetic nanoparticles. In: Gubin SP (Ed.), Wiley-VCH.
- Prozorova GF, Korzhova SA, Emel’Yanov AI, Pozdnyakov AS, Khutsishvili SS, et al. (2013) Polymer nanocomposites with iron oxide nanoparticles. Russian Journal of Applied Chemistry 86: 1452-1455.
- Ha-Young L, Sang-Hoon L, Chenjie X, Xie J, Jin-Hyung L, et al. (2008) Synthesis and characterization of PVP-coated large-core iron oxide nanoparticles as MRI contrast agents. Nanotechnology 19(16): 165101.
- Young L, Nak Hyun L, Seo Jin A, Soon Hong Y, Byung Kook K, et al. (2006) Preparation and MRI effects of polyvinylpyrrolidone-coated iron oxide nanoparticles. J Biomed Mater Res B 79(1): 142-150.
- Huang H, Xie Q, Kang M, Zhang B, Zhang H, et al. (2009) Labeling transplanted islets with PVP-coated superparamagnetic iron oxide nanoparticles for in vivo MRI detection. Nanotechnology 20(36): 365101.
- Lu X, Niu M, Qiao R, Gao M (2008) Superdispersible PVP-Coated Fe₃O₄ nanocrystals prepared by a ‘one-pot’ reaction. J Phys Chem C 112(46): 14390-14394.
- Zhang B, Tu Z, Zhao F, Wang J (2013) Superparamagnetic iron oxide nanoparticles prepared via improved polyol method. Applied Surface Science 266: 375-379.
- Tu Z, Zhang B, Yang G, Wang M, Zhao F, et al. (2013) Synthesis of PEG/PVP coated superparamagnetic Fe₃O₄ nanoparticles for PH-sensitive drug release. Colloids and Surfaces A 436: 854-861.
- Jurkin T, Gotić M, Štefanić G, Pucić I (2016) Gamma-Irradiation synthesis of iron oxide nanoparticles in the presence of PEO, PVP or CTAB. Radiation Physics and Chemistry 124: 75-83.
- Xu Y, Zhang X, Hsing Y, Fang Y (2011) Ultrasonic-assisted synthesis of Fe nanoparticles in the presence of Poly(N-vinyl-2-pyrrolidone). Chinese J Chem 29(9): 1829-1836.
- Malekigorji M, Curtis ADM, Hoskins C (2014) The use of iron oxide nanoparticles for pancreatic cancer therapy. J Nanomed Res 1(1): 15-21.
- Prozorova GF, Korzhova SA, Pozdnyakov AS, Emelyanov AI, Ermakova TG, et al. (2015) Immunomodulatory properties of silver-containing nanocomposites based on polyvinyltriazole. Russ Chem Bull 64: 1437-1439.
- Kavas H, Durmus Z, Baykal A, Aslan A, Bozkurt A, et al. (2010) Synthesis and conductivity evaluation of PVTri-Fe₃O₄ J Non-Crystalline Solids 356(10): 484-489.
- Uzun L, Kara A, Osman B, Yilmaz E, Besirli N, et al. (2009) Removal of heavy metal ions by magnetic beads containing triazole chelating groups. J Appl Polym Sci 114(4): 2246-2253.
- Ermakova TG, Kuznetsova NP, Pozdnyakov AS, Larina LI, Korzhova SA, et al. (2016) 1-Vinyl-1,2,4-triazole in copolymerization with 1-Vinyl-4,5,6,7-tetrahydroindole: synthesis and properties of copolymers. Russ Chem Bull 65: 485-489.
- Lavlinskaya MS, Shatalov GV, Mokshina NY, Pakhomova OA, Kuznetsov VA (2016) Extraction of histidine and riboflavin by N-Vinylcaprolactam copolymers with 1-Vinyl- and 1-Methacryloyl-3,5-dimethylpyrazole. ChemChemTech 59(7): 55-60.
- Hevus I, Kohut A, Voronov A (2012) Micellar assemblies from amphiphilic polyurethanes for controlled synthesis of silver nanoparticles. J Nanopart Res 14: 820.
- Ulitin NV, Deberdeev RY, Deberdeev TR (2012) Kinetics of styrene radical polymerization under reversible chain transfer conditions. ChemChemTech 55: 9.
- Chakrabarti S, Mandal SK, Nath BK, Das D, Ganguli D, et al. (2003) Synthesis of γ-Fe₂O₃ nanoparticles coated on silica spheres: structural and magnetic properties. Eur Phys J B 34: 163-171.
- Vereshchagina TA, Anshits NN, Maksimov NG, Vereshchagin SN, Bayukov OA, et al. (2004) The nature and properties of iron-containing nanoparticles dispersed in aluminosilicate matrices. Glass Physics and Chemistry 30: 247-256.
- Amstad E, Textor M, Reimhult E (2011) Stabilization and functionalization of iron oxide nanoparticles for biomedical applications. Nanoscale 3(7): 2819-2843.
- Sargsyan SH, Sargsyan AS, Khizantsyan KM, Sargsyan TS, Aghajanyan IG, et al. (2024) Electrochemical synthesis of silver nanocomposites based on 1-vinyl-1,2,4-triazole and N-vinylcaprolactam. Polym Bull 10:
© 2026 Sargsyan SH. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






