- Submissions

Full Text
Progress in Petrochemical Science
Theoretical Investigation on Cs2CO3-Mediated [4+2] Annulation of Ynone and α, α-Dicyanoalkene Leading to Highly Functionalized M‑Terphenyl
Nan Lu*
College of Chemistry and Material Science, Shandong Agricultural University, China
*Corresponding author:Nan Lu, College of Chemistry and Material Science, Shandong Agricultural University, Taian 271018, P. R. China
Submission: November 11, 2025;Published: November 21, 2025

ISSN 2637-8035Volume7 Issue 4
Abstract
The first theoretical investigation on Cs2CO3-mediated [4+2] annulation of α, α-dicyanoalkene and ynone was provided by our DFT calculation. Via Cs2CO3-mediated deprotonation, the α, α-dicyanoalkene is initially converted into vinylogous carbanion from methyl group. Then 4-membered ring carbanion stabilized by two electron-withdrawing cyano groups is generated through nucleophilic attack of methylene on ynone followed by an intramolecular nucleophilic addition. Next the ring opening occurs from strained 4-membered ring. Subsequently the 6-membered ring closure is accomplished via a second intramolecular nucleophilic attack of methylene carbanion to cyano group determined to be rate-limiting for the whole process. Finally, the desired product m-terphenyl is obtained through aromatization of the central ring and afterwards protonation together with recovered Cs2CO3.
Keywords:[4+2] annulation; Intramolecular rearrangement; M-terphenyl; Aromatization; Ring opening
Introduction
As a class of aromatic hydrocarbons, terphenyls are characterized by three benzene rings with the terminal one ortho-, meta- or para-substituents relative to the central one. Although p-terphenyl derivatives are natural predominant and recently identified, m-terphenyl derivatives are scarce and has attracted much attention in synthetic chemistry [1]. In addition to exceptional optical and photophysical characteristics [2], a variety of applications have been reported such as laser dyes, OLEDs, textile dye carriers and agents for heat storage, transfer [3]. Furthermore, m-terphenyl moiety displays biological activities in natural compounds [4]. Sawayama reported syntheses of naturally occurring terphenyls [5]. Patrick discovered antiprotozoal activity of dicationic m-terphenyl [6]. Rajakumar found anti-inflammatory activity and toxicity of some novel bis-oxy cyclophane diamides [7]. Samshuddin gave biological evaluation of some functionalized terphenyl derivatives [8]. There is also pharmacological evaluation of m-terphenyl amines as cyclooxygenase inhibitors [9]. New monodentate ruthenium (II) complexes containing terphenyl arenes have cytotoxicity, cellular uptake and DNA interactions stabilizing low-coordinate main-group and transition metals [10]. In synthesis of conjugated polymers, m-terphenyls are employed as smart materials owing to unique optical properties [11]. In the field of synthetic methods, there are reaction of β-cyclo-hexanedione and its ethyl enol ether with phenylmagnesium bromide and two-aryne sequence [12,13].
Adrio discovered aryl cross-coupling reaction using aryl halides to generate central aromatic ring [14]. Shu developed base-mediated synthesis of highly functionalized 2-amino-nicotinonitriles from α-keto vinyl azides and α, α-dicyanoalkenes [15]. Cao reported synthesis of dibenzofurans via benzannulation of 2-nitrobenzofurans and alkylidene malononitriles [16]. Recently, Gopi realized one-pot regioselective synthesis of meta-terphenyls via [3+3] annulation of nitro-allylic acetates with alkylidene-malononitriles [17]. Then Davas published divergent approach to highly substituted arenes via [3+3] annulation of vinyl sulfoxonium ylides with ynones [18]. Similar progresses are Tf2O-induced selective 1, 3-transposition/cyclization of ynones and sequential condensation/bi-annulation reactions of β-(2-aminophenyl)-α, β-ynones with 1, 3-dicarbonyls [19,20]. Clarke realized atom and step economic synthesis of functionalized piperidines using fivecomponent condensation [21]. Wang gave cerium ammonium nitrate-catalyzed multicomponent reaction for efficient synthesis of functionalized tetrahydropyridines [22].
The mechanism of this annulation was researched in synthesis of bridged ring system initiated by ring expansion of indene-1,3- dione and AgNO3-catalysed intramolecular cyclization to access cyclopentanones and spiro-cyclopentanones [23,24]. Another breakthrough was Cs2CO3-catalyzed [4+2] annulation of ynone and α, α-dicyanoalkene [25]. Although desired m-Terphenyl was synthesized, how intramolecular rearrangement followed by ring cyclization was achieved for the whole process? What’s the function of Cs2CO3 in base-mediated deprotonation converting α, α-dicyanoalkene into vinylogous carbanion? How 4-membered ring carbanion and 6-membered intermediate were obtained via ring opening in consecutive mode? How Cs2CO3 is recovered along with final aromatization of central ring?
Computational Details
The geometry optimizations were performed at the B3LYP/ BSI level with the Gaussian 09 package [26,27]. The mixed basis set of LanL2DZ for Cs and 6-31G(d) for other non-metal atoms [28- 32] was denoted as BSI. Different singlet and multiplet states were clarified with B3LYP and ROB3LYP approaches including Becke’s three-parameter hybrid functional combined with Lee-Yang-Parr correction for correlation [33,34]. The nature of each structure was verified by performing harmonic vibrational frequency calculations. Intrinsic Reaction Coordinate (IRC) calculations were examined to confirm the right connections among key transition-states and corresponding reactants and products. Harmonic frequency calculations were carried out at the B3LYP/BSI level to gain Zero- Point Vibrational Energy (ZPVE) and thermodynamic corrections at 383K and 1atm for each structure in 1, 4-dioxane. The solvationcorrected free energies were obtained at the B3LYP/6-311++G (d, p) (LanL2DZ for Cs) level by using Integral Equation Formalism Polarizable Continuum Model (IEFPCM) in Truhlar’s “density” solvation model [35-37] on the B3LYP/BSI-optimized geometries. As an efficient method of obtaining bond and lone pair of a molecule from modern ab initio wave functions, NBO procedure was performed with Natural Bond Orbital (NBO3.1) to characterize electronic properties and bonding orbital interactions [38,39]. The wave function analysis was provided using Multiwfn_3.7_dev package [40] including research on Frontier Molecular Orbital (FMO).
Results and Discussion
The mechanism was explored for Cs2CO3-mediated [4+2] annulation of α, α-dicyanoalkene 1 and ynone 2 leading to m terphenyl 3 (Scheme 1). As is illustrated by black arrow of Scheme 2, first, via Cs2CO3-mediated deprotonation, α, α-dicyanoalkene 1 is converted into vinylogous carbanion A from methyl group of 1. Then, the nucleophilic attack of A on ynone 2 leads to intermediate B followed by an intramolecular nucleophilic addition forming 4-membered ring carbanion intermediate C stabilized by two electron-withdrawing cyano groups. Next, the ring opening of C occurs from strained 4-membered ring giving intermediate D, which turns into E through another intramolecular nucleophilic attack of methyl carbanion to cyano group. Finally, the desired product m-terphenyl 3 is achieved through aromatization of the central ring from E and protonation afterwards along with recovery of Cs2CO3. Figure 1 listed schematic structures of optimized TSs in Scheme 2. Table 1 gave activation energy for all steps.
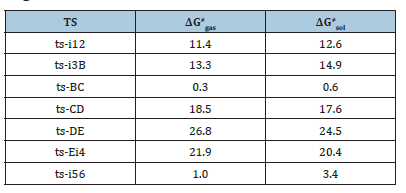
Table 1:The activation energy (in kcal mol-1) of all reactions in gas and solvent.
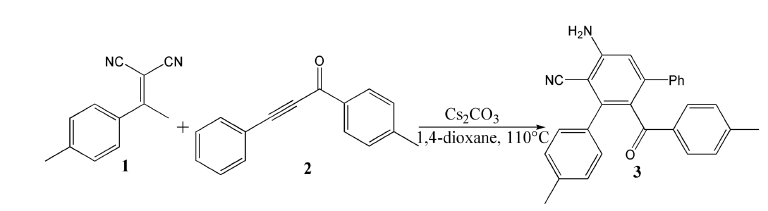
Scheme 1:Cs2CO3-mediated [4+2] annulation of α, α-dicyanoalkene 1 and ynone 2 leading to functionalized m-terphenyl 3.
Scheme 2:Proposed reaction mechanism of Cs2CO3-mediated [4+2] annulation of 1 and 2 to access 3. TS is named according to the two intermediates it connects.
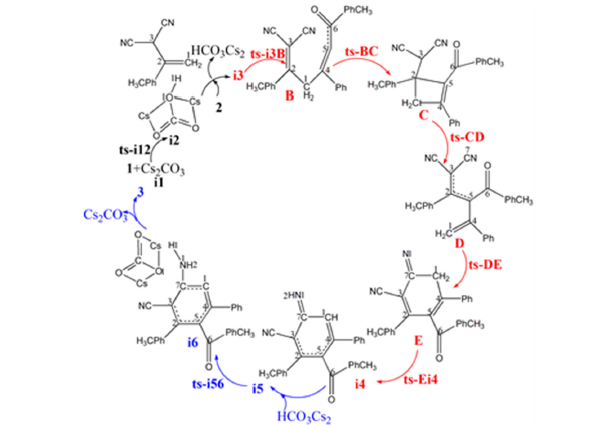
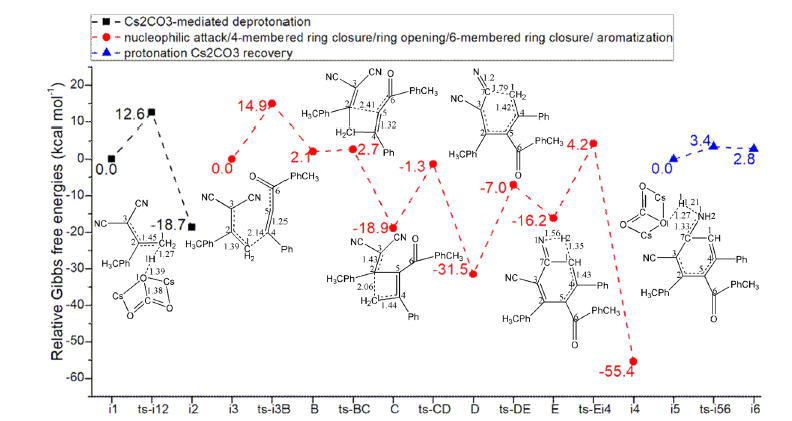
Figure 1:Relative Gibbs free energy profile in solvent phase starting from complex i1, i3, i5 (Bond lengths of optimized TSs in Å).
Cs2CO3-mediated deprotonation
In step 1, α, α-dicyanoalkene 1 and Cs2CO3 forms intermediate i1, from which the deprotonation occurs via ts-i12 with the activation energy of 12.6kcal mol-1 exothermic by -18.7kcal mol-1 producing vinylogous carbanion A (black dash line of Figure 1). The transition vector corresponds to proton H1 on methyl group captured by carbonyl of Cs2CO3 denoted as C1···H1···O1 (1.27, 1.39 Å) (Figure S1a). Along with the shortening of C1-C2 from single bond to double (1.45 Å), methyl is converted into methylene in A, which is reactive and ready to initiate next step.
Nucleophilic attack/4-membered ring closure/ring opening/6-membered ring closure
Then, the intermediate i3 binding A and ynone 2 is taken as new starting point of next five steps (red dash line of Figure 1). The nucleophilic attack of A to 2 leads to intermediate B via ts-i3B with activation energy of 14.9kcal mol-1 endothermic by 2.1kcal mol-1 in step 2. The transition vector not only includes C1 bonding with terminal alkyne C4 but resultant elongation of C1-C2 from double to single and C4-C5 from triple to double (2.14, 1.39, 1.25 Å). Thus, complex B is obtained with typical C1-C4 single bond. Subsequently, an intramolecular nucleophilic addition takes place to accomplish 4-membered ring closure via ts-BC in step 3 with low activation energy of 0.6kcal mol-1 exothermic by -18.9kcal mol-1 delivering carbanion intermediate C. From detailed atomic motion of transition vector, the negative alkyne C5 is approaching C2 together with assisted elongation of C2-C3 and C6-C5 from double to single (2.41 Å) (Figure S1b). Once the 4-membered ring is closed involving C5-C2 single bond, negative charge is transferred at C3 stabilized by two electron-withdrawing cyano groups. Next, from strained 4-membered ring, the ring opening of C is readily via ts- CD in step 4 with activation energy of 17.6kcal mol-1 exothermic by -31.5kcal mol-1 affording intermediate D. The transition vector suggests facile cleavage of C1···C2 single bond driven by enhanced C3-C2 and C1-C4 from single to double (2.06, 1.43, 1.44 Å) (Figure S1c).
Clearly, the much more stable of D compared with C is expected as a result of ring tension release. The negative charge is located at methylene C1 in D. Another intramolecular nucleophilic attack through methylene carbanion to cyano group happens via ts-DE with increased activation energy of 24.5kcal mol-1 exothermic by -16.2kcal mol-1. D turns into E realizing 6-membered ring closure in step 5. The transition vector contains linkage of C1···C7 as well as stretching of C1···C4 from double to single and C7···N1 from triple to double (1.79, 1.42, 1.2 Å) (Figure S1d). The 6-membered ring is yielded with typical C1-C7 single bond in E.
Aromatization/protonation- Cs2CO3 recovery
Then in step 6, the central ring of E undergoes aromatization via ts-Ei4 with reduced activation energy of 20.4kcal mol-1 exothermic by -55.4kcal mol-1 yielding intermediate i4. The transition vector suggests apparent proton donation by methylene C1 to imine N1 through transfer mode C1···H2···N1 and concerted shortening of C1-C4 from single bond to double (1.35, 1.56, 1.43 Å) (Figure S1e). The preliminarily protonation is realized via this step effectively pull the entire process with huge released energy. On the basis of previous step, the product m-terphenyl 3 is finally achieved through afterwards proton transfer in step 7 (blue dash line of Figure 1). The transition vector reveals proton H1 provided by O1 of HCO3Cs2 to N1 via O1···H1···N1 along with of slight elongation of N1-C7 (1.27, 1.21, 1.33 Å) (Figure S1f). The last step happens via ts-i56 with small barrier of 3.4kcal mol-1 endothermic by 2.8kcal mol-1 giving i6, from which the catalyst Cs2CO3 is simultaneously recovered. Comparatively, the 6-membered ring closure of step 5 is determined to be rate-limiting for Cs2CO3-mediated [4+2] annulation producing m terphenyl.
Conclusion
In summary, the first theoretical investigation was provided by our DFT calculation on Cs2CO3-mediated [4+2] annulation of α, α-dicyanoalkene and ynone. The α, α-dicyanoalkene is initially converted into vinylogous carbanion from methyl group via Cs2CO3-mediated deprotonation. Then, the nucleophilic attack of methylene on ynone followed by an intramolecular nucleophilic addition leads to 4-membered ring carbanion stabilized by two electron-withdrawing cyano groups. Next, the ring opening occurs from strained 4-membered ring. Subsequently, the second intramolecular nucleophilic attack of methylene carbanion to cyano group accomplishes 6-membered ring closure determined to be rate-limiting for the whole process. Finally, the desired product m-terphenyl is obtained through aromatization of the central ring and afterwards protonation assisted by Cs2CO3 recovery.
Electronic Supplementary Material
Supplementary data available: [Computation information and cartesian coordinates of stationary points; Calculated relative energies for the ZPE-corrected Gibbs free energies (ΔGgas) and Gibbs free energies (ΔGsol) for all species in solution phase at 383K].
Author Contributions
Conceptualization, Nan Lu; Methodology, Nan Lu; Software, Nan Lu; Validation, Nan Lu; Formal Analysis, Nan Lu; Investigation, Nan Lu; Resources, Nan Lu; Data Curation, Nan Lu; Writing-Original Draft Preparation, Nan Lu; Writing-Review & Editing, Nan Lu; Visualization, Nan Lu; Supervision, Nan Lu; Project Administration, Nan Lu. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by Key Laboratory of Agricultural Film Application of Ministry of Agriculture and Rural Affairs, P.R. China.
Conflict of Interest
The authors declare no conflict of interest.
References
- Liu JK (2006) Natural terphenyls: Developments since 1877. Chem Rev 106(6): 2209-2223.
- Xiao X, Luo J, Gan Z, Jiang W, Tang Q (2020) Metal-free and solvent-free synthesis of m-terphenyls through tandem cyclocondensation of aryl methyl ketones with triethyl orthoformate. RSC Adv 10(20): 12113-12118.
- Sasabe H, Seino Y, Kimura M, Kido J (2012) A m-terphenyl-modified sulfone derivative as a host material for high-efficiency blue and green phosphorescent OLEDs. Chem Mater 24(8): 1404-1406.
- Kikuchi H, Matsuo Y, Katou Y, Kubohara Y, Oshima Y (2012) Isolation, synthesis and biological activity of biphenyl and m-terphenyl-type compounds from Dictyostelium cellular slime molds. Tetrahedron 68(43): 8884-8889.
- Sawayama Y, Tsujimoto T, Sugino K, Nishikawa T, Isobe M, et al. (2006) Syntheses of naturally occurring terphenyls and related compounds. Biosci Biotechnol Biochem 70(12): 2998-3003.
- Patrick DA, Ismail MA, Arafa RK, Wenzler T, Zhu X, et al. (2013) Synthesis and antiprotozoal activity of dicationic m-terphenyl and 1, 3-dipyridylbenzene derivatives. J Med Chem 56(13): 5473-5494.
- Rajakumar P, Padmanabhan R, Rajesh N (2012) Synthesis, study on anti-arthritic, anti-inflammatory activity and toxicity of some novel bis-oxy cyclophane diamides. Bioorg Med Chem Lett 22(11): 3770-3775.
- Samshuddin S, Narayana B, Sarojini BK, Shetty DN, Suchetha KN (2012) Synthesis, characterization and biological evaluation of some new functionalized terphenyl derivatives. Int J Med Chem 2012: 530392.
- Bauer JD, Foster MS, Hugdahl JD, Burns KL, May SW, et al. (2007) Synthesis and pharmacological evaluation of m-terphenyl amines as cyclooxygenase inhibitors. Med Chem Res 16: 119-129.
- Bugarcic T, Novakova O, Zerzánková L, Vrána O, Kašpárková J, et al. (2008) Cytotoxicity, cellular uptake and DNA interactions of new monodentate ruthenium (II) complexes containing terphenyl arenes. J Med Chem 51(17): 5310-5319.
- Wu CA, Chou HH, Shih CH, Wu FI, Cheng CH, et al. (2012) Synthesis and physical properties of meta-terphenyl oxadiazole derivatives and their application as electron transporting materials for blue phosphorescent and fluorescent devices. J Mater Chem 22(34): 17792-17799.
- Woods GF, Tucker IW (1948) The reaction of β-cyclohexanedione (dihydroresorcinol) and its ethyl enol ether with phenylmagnesium bromide. J Am Chem Soc 70(6): 2174-2177.
- Du CF, Hart H, Ng KD (1986) A one-pot synthesis of m-terphenyls, via a two-aryne sequence. J Org Chem 51(16): 3162-3165.
- Adrio LA, Míguez JA, Hii KK (2009) Synthesis of terphenyls. Org Prep Proced Int 41(5): 331-358.
- Shu K, Shao J, Li H, Chen B, Tang P, et al. (2016) Base-mediated synthesis of highly functionalized 2-aminonicotinonitriles from α-keto vinyl azides and α, α-dicyanoalkenes. RSC Adv 6(54): 49123-49126.
- Cao D, Chen D, Chen G, Mo H, Xia Z, et al. (2020) Synthesis of dibenzofurans derivatives via benzannulation of 2-nitrobenzofurans and alkylidene malononitriles. Asian J Org Chem 9(10): 1610-1616.
- Gopi E, Namboothiri IN (2014) One-pot regioselective synthesis of meta-terphenyls via [3+3] annulation of nitroallylic acetates with alkylidenemalononitriles. J Org Chem 79(16): 7468-7476.
- Davas DS, Gopalakrishnan DK, Bar K, Kumar S, Karmakar T, et al. (2023) Divergent approach to highly substituted arenes via [3+3] annulation of vinyl sulfoxonium ylides with ynones. Org Lett 25(50): 8992-8996.
- Lan HL, Liu W, Liu W, Peng J, Ying B, et al. (2024) Tf2O-induced selective 1, 3-transposition/cyclization of ynones in DMF. Organic Chemistry Frontiers 12(4): 1167-1176.
- Marsicano V, Arcadi A, Chiarini M, Giancarlo F, Antonella G, et al. (2021) Sequential condensation/biannulation reactions of β-(2-aminophenyl)-α, β-ynones with 1, 3-dicarbonyls. Organic & Biomolecular Chemistry 23(19): 5177-5190.
- Clarke PA, Zaytzev AV, Whitwood AC (2007) Pot, Atom and Step Economic (PASE) synthesis of highly functionalized piperidines: A five-component condensation. Tetrahedron Letters 48(30): 5209-5212.
- Wang HJ, Mo LP, Zhang ZH (2011) Cerium ammonium nitrate-catalyzed multicomponent reaction for efficient synthesis of functionalized tetrahydropyridines. ACS Combinatorial Science 13(2): 181-185.
- Mu Y, Yao Q, Yin L, Fu S, Wang M, et al. (2021) Atom-economic synthesis of highly functionalized bridged ring systems initiated by ring expansion of indene-1, 3-dione. J Org Chem 86(9): 6755-6764.
- Bhoite SP, Bansode AH, Burate PA, Suryavanshi G (2019) AgNO3-catalysed intramolecular cyclization: Access to functionalized cyclopentanones and spiro-cyclopentanones. Asian J Org Chem 8(10): 1907-1911.
- Katiyar S, Jaiswal AK, Kushawaha AK, Bhatt H, Kant R, et al. (2025) Unraveling the straightforward synthesis of highly functionalized m-terphenyls via [4 +2] annulations of ynones and α, α-dicyanoalkenes. J Org Chem 90(40): 14094-14099.
- Frisch MJ, Trucks GW, Schlegel HB (2010) Gaussian 09, Revision B. 01. Gaussian, Inc., Wallingford, England.
- Hay PJ, Wadt WR (1985) Ab initio effective core potentials for molecular calculations-potentials for the transition-metal atoms Sc to Hg. J Chem Phys 82(1): 270-283.
- Lv H, Han F, Wang N, Lu N, Song Z, et al. (2022) Ionic liquid catalyzed C-C bond formation for the synthesis of polysubstituted olefins. Eur J Org Chem 2022(45): e202201222.
- Zhuang H, Lu N, Ji N, Han F, Miao C (2021) Bu4NHSO4‐catalyzed direct N‐Allylation of pyrazole and its derivatives with allylic alcohols in water: A metal‐free, recyclable and sustainable system. Advanced Synthesis & Catalysis 363(24): 5461-5472.
- Lu N, Lan X, Miao C, Qian P (2020) Theoretical investigation on transformation of Cr(II) to Cr(V) complexes bearing tetra-N-heterocyclic carbene and group transfer reactivity. Int J Quantum Chem 120(18): e26340.
- Lu N, Liang H, Qian P, Lan X, Miao C (2020) Theoretical investigation on the mechanism and enantioselectivity of organocatalytic asymmetric povarov reactions of anilines and aldehydes. Int J Quantum Chem 121(8): e26574.
- Lu N, Wang Y (2023) Alloy and media effects on the ethanol partial oxidation catalyzed by bimetallic Pt6M (M= Co, Ni, Cu, Zn, Ru, Rh, Pd, Sn, Re, Ir and Pt). Computational and Theoretical Chemistry 1228: 114252.
- Catellani M, Mealli C, Motti E, Paoli P, Perez CE, et al. (2002) Palladium-arene interactions in catalytic intermediates: An experimental and theoretical investigation of the soft rearrangement between η1 and η2 coordination modes. J Am Chem Soc 124(16): 4336-4346.
- Marenich AV, Cramer CJ, Truhlar DG (2009) Universal solvation model based on solute electron density and on a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions. J Phys Chem B 113(18): 6378-6396.
- Tapia O (1992) Solvent effect theories: Quantum and classical formalisms and their applications in chemistry and biochemistry. J Math Chem 10: 139-181.
- Tomasi J, Persico M (1994) Molecular interactions in solution: An overview of methods based on continuous distributions of the solvent. Chem Rev 94(7): 2027-2094.
- Tomasi J, Mennucci B, Cammi R (2005) Quantum mechanical continuum solvation models. Chem Rev 105(8): 2999-3093.
- Reed AE, Weinstock RB, Weinhold F (1985) Natural population analysis. J Chem Phys 83(2): 735-746.
- Reed AE, Curtiss LA, Weinhold F (1988) Intermolecular interactions from a natural bond orbital donor-acceptor viewpoint. Chem Rev 88(6): 899-926.
- Lu T, Chen F (2012) Multiwfn: A multifunctional wavefunction analyzer. J Comput Chem 33(5): 580-592.
© 2025 Nan Lu. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






