- Submissions

Full Text
Novel Techniques in Nutrition and Food Science
Grewia asiatica l. (phalsa): Nutritional Composition, Phytochemistry and Pharmacological Potential-A Comprehensive Review
Sabiha Abbas1, Ali Usman1*, Saqafat Ahmad3, Sana Saadat4, Hafiz Nafees Ul Hassan2*, Fizza Hussain1, Mehak Ali6, Syed Shumaim Ali5 and Samia Fiaz2
1Department of Human Nutrition and Dietetics, Riphah College of Rehabilitation & Allied Health Sciences, Pakistan
2Department of Human Nutrition and Dietetics, Government College University, Pakistan
3Department of Medical Laboratory Technology, Riphah College of Rehabilitation & Allied Health Sciences, Pakistan
4Department of Nutrition School of Allied Health Sciences, CMH Lahore, Pakistan
5Lecturer Gujrat institute of management sciences affiliated with PMAS RWP, Gujrat
6Department of Food Science and Technology, Government College University Faisalabad, Pakistan
*Corresponding author:Ali Usman, Department of Human Nutrition and Dietetics, Riphah College of Rehabilitation & Allied Health Sciences, Pakistan and Hafiz Nafees Ul Hassan, Department of Human Nutrition and Dietetics, Government College University, Pakistan
Submission: February 24, 2026;Published: March 10, 2026

ISSN:2640-9208Volume8 Issue 5
Summary
Grewia asiatica L. commonly known as phalsa, is an underutilized subtropical fruit crop widely cultivated in South Asia and increasingly recognized for its nutritional and pharmacological potential. Traditionally consumed fresh and processed into beverages, syrups and preserves, the fruit has long been used in indigenous medicine for managing gastrointestinal disorders, fever, inflammation and metabolic disturbances. Recent phytochemical and pharmacological investigations have revealed that G. asiatica is a great source of many biologically active compounds including anthocyanins, flavonoid contents, phenolic compounds, tannins, triterpenoids and phytosterols. The fruit pulp is characterized by appreciable levels of dietary fiber, potassium, calcium and polyphenolic antioxidants, while seeds and leaves contain diverse secondary metabolites with demonstrated biological activity. Many studies including in vitro and in vivo indicate significant antioxidant, antimicrobial, anticancer, anti-inflammatory and radioprotective activities, largely attributed to its high total phenolic contents and free radical scavenging capacity. Mechanistic insights suggest involvement of Reactive Oxygen Species (ROS) modulation, lipid peroxidation inhibition and possible regulation of apoptotic pathways. Despite promising preclinical evidence, clinical validation, standardized extract characterization, and bioavailability studies remain limited. This review critically evaluates the current literature on the nutritional profile, phytochemistry and pharmacological properties of G. asiatica, highlights mechanistic perspectives, discusses safety considerations and identifies research gaps necessary for its development as a functional food or nutraceutical. The synthesis presented herein aims to provide a scientific basis for future translational and clinical investigations.
Keywords:Grewia asiatica; Phalsa; Malvaceae; Phytochemicals; Antioxidant activity; Functional Foods; Nutraceuticals; Pharmacological potential
Introduction
Fruits constitute an essential component of human nutrition due to their provision of carbohydrates, dietary fiber, vitamins, minerals and bioactive phytochemicals. Beyond basic nutrition, fruits are increasingly recognized for their protective effects against chronic Non-Communicable Diseases (NCDs), including cardiovascular disorders, diabetes mellitus, neurodegenerative diseases and certain cancers. These protective effects are largely attributed to polyphenols, flavonoids, carotenoids and other antioxidant constituents that modulate oxidative stress and inflammatory pathways [1-14]. Fruits are considered as rich source of valuable nutrients especially carbohydrates, vitamins, minerals and antioxidants with great health benefits which are significant for an active and healthy life. Flavonoids and polyphenolic compounds are found in many types of fruits. They provide several protective effects against chronic diseases; however, their precise mechanisms are not yet defined [15]. The health benefits from eating fruits and vegetables can be attributed to their ability to delay the aging process and provide some degree of protection from the following chronic diseases: cardiovascular disease, certain cancers, rheumatoid arthritis, various lung conditions (including emphysema and chronic obstructive lung disease), cataract formation, Parkinson’s and Alzheimer’s disease [16]. More recent studies indicate that people who eat large amounts of fruits and vegetables have half the risk of developing cancers and have reduced mortality rates from cancers compared to those consuming few servings of either [17]. Several fruits (e.g., ber, phalsa, apple, strawberry) contain antioxidants [3]. The roots of these three fruits are also commonly used to help treat injuries and heal tissues [18]. The scavenging effect of these compounds may be one factor explaining how fruits/ vegetables affect the treatment of injuries caused by oxidative stress [14]. It should be noted that traditional healers have used these same three fruits as medicinal plants for treating injuries throughout history [19].
The use of different plant parts that contain antioxidant chemicals has been linked to a reduction in cancer, cardiovascular and many chronic diseases. The presence of a variety of biofunctional and chemopreventive compounds found within the various parts of plants is also a major factor in the increasing popularity of these foods for health reasons. For thousands of years, medicinal plants have provided the basis for many of today’s medicines and it is widely accepted that the primary reason for their protective properties is due to their phytochemistry and vitamins. Their protective actions result from their antioxidant properties (Rice-Evans, 1995). These properties occur by their ability to scavenge Reactive Oxygen Species (ROS), eg, hydroxyl, peroxide radicals, etc. In recent years, attention has shifted toward underutilized and indigenous fruit species with functional and nutraceutical potential. Among these, Grewia asiatica L., commonly known as phalsa or falsa, has gained renewed interest due to its distinctive nutritional composition and traditional medicinal applications. Native to the Indian subcontinent and cultivated in Pakistan, India, Bangladesh and other tropical and subtropical regions, phalsa is a small berry-like fruit valued for its sweetsour taste and refreshing properties during summer months. In traditional medicine systems, different parts of the plant-including fruit, leaves, bark and roots-have been used for the management of fever, gastrointestinal disorders, inflammation, skin conditions and metabolic disturbances [20]. Although ethnomedicinal claims are abundant, systematic scientific evaluation has only emerged over the past two decades. Available studies suggest that G. asiatica is rich in anthocyanins, flavonoids, phenolic acids and triterpenoids that exhibit diverse biological activities in vitro and in animal models. However, the evidence remains fragmented and requires critical synthesis [21,8].
Medicinal plant: Phalsa
The plants that have been used for medicinal purposes by the ancient cultures have not been studied fully or scientifically and therefore there is limited scientific information available on them. In view of this lack of information and the fact that they were used for the treatment of many common ailments, we have selected the plant Phalsa subinaequalis DC. Syn. G. asiatic. (Phalsa) from the family of Tiliaceae genus Phalsa for an analytical literature review. In addition to having many medicinal properties from its fruit, leaves and bark, Phalsa has an extensive use throughout the world in providing treatment for common ailments. Phalsa is part of the Tiliaceae family and is made up of approximately 150 species; they are classified as small trees or shrubs and are found primarily in the subtropical and tropical regions throughout the world. Phalsa is the only genus within the Tiliaceae family to produce edible fruit. The genus Phalsa received its name from Nehemiah Grew, who was a founding member of modern plant science. Phalsa species generally reach heights of greater than four m and are native to such countries as India, the United States of America, Pakistan, South Africa and Southeast Asia. Generally, all species in the Tiliaceae family are wild and are known to provide fodder, fuel wood, crafting materials, timber and many species also provide therapeutic products, i.e., Phalsa flavescens A Juss, Phalsa villos and Phalsa hirsuta. There are many varieties of plants that have been grown specifically because of the value of their fruit. These plants include Gourd (Gourd, Subinaequalis DC., syn. Gourd, Asiatica) and Phalsa (Phalsaf Tenax) (Frosk. and Youngken 1951). There are also many different medicinal benefits associated with phalsa; due to the fact that they contain a variety of metabolites. These include saponins, coumarin and anthraquinone [19].
Recent scientific evidence suggests that activation of Sirtuin 1 (SIRT1), a NAD⁺-dependent deacetylase associated with longevity and metabolic regulation, plays a critical role in the prevention and management of several chronic diseases including diabetes, cardiovascular disorders, neurodegeneration and age-related metabolic dysfunction. Many plant-derived polyphenols and flavonoids have been reported to influence SIRT1 signaling pathways by improving mitochondrial function, reducing oxidative stress and modulating inflammatory responses [21,3]. Grewia asiatica is rich in bioactive compounds such as anthocyanins, flavonoids, phenolic acids and triterpenoids, which are known to exhibit strong antioxidant and anti-inflammatory activities. These phytochemicals may potentially contribute to mechanisms associated with SIRT1 activation and metabolic homeostasis. For instance, polyphenolic compounds similar to those found in Grewia asiatica have demonstrated the ability to regulate SIRT1- mediated pathways linked with improved insulin sensitivity, cellular protection and delayed cellular aging. However, despite the promising phytochemical profile of Grewia asiatica, direct experimental studies specifically evaluating its role as a SIRT1 activator remain limited. Therefore, further molecular, biochemical and clinical investigations are required to assess whether the phytoconstituents of Grewia asiatica can modulate SIRT1 activity and contribute to therapeutic strategies for chronic disease prevention and management [5,6]. Many edible types of Grewia have been used to relieve indigestion in addition to treating some skin and intestinal conditions, cough, fever, diarrhea, dysentery, jaundice, rheumatism and mild infections due to their mild antibacterial properties as well. Supplements made from these plants have been used to treat the healing of a bone fracture and for bone strengthening. Other written accounts also list these edible plants as being known for their medicinal ability for the healing of wounds [22]. These vegetables are also known to assist in treating iron deficiency anaemia [23]. Despite being used for so many different reasons, the use of these plants continues to be neglected; therefore, documenting the biological and chemical characteristics of Grewia subinaequalis DC. (syn. Grewia asiatic) (Phalsa) will help bring attention to and encourage the study of this indigenous plant of India. This study will be used as a point of reference for future studies pertaining to this important medicinal source. Grewia (Tiliaceae) is a group consisting primarily (over) 150 shrub and small tree (unone small tree and shrub) species found throughout tropical and subtropical parts-of-the-world. Grewia is the only Genus within this Family that has Edible Fruit. In Pakistan there are 10 identified Species of Grewia: G. damine Gaertn. (Syrian Knotgrass), G. elastica Royle (Elastic Grewia), G. glabra Blume (Smooth Grewia), G. helicterifolia Wall (Helicterus), Grewia microcos L. (Small Grewia), G. optiva J. R. Drumm. ex Burret, G. sapida Roxb (Nectar), G. tenax (Forssk.) Fiori (Waiting) and G. villosa Willd (Central Asian) [24]. G. asiatica L. (Phalsa) grows Wild and is Cultivated in most of South Asia. The Genus “Grewia” was named after the “father” of Plant Physiology (Nehemiah Grew), which represents the Asian origin of this Species. G. asiatica (Phalsa) not only is One of the most Nutrient-Rich Foods we have, but it also is an effective Traditional Medicine. Even though G. asiatica (Phalsa) has many Uses the lack of adequate Literature demonstrates it has been largely Neglected. We have completed a study of the native phytochemistry, food value and therapeutic value of certain indigenous plants found in Pakistan [25] and made it publicly available to assist in additional studies for these and other plants of importance in Pakistan (Figure 1).
Figure 1:
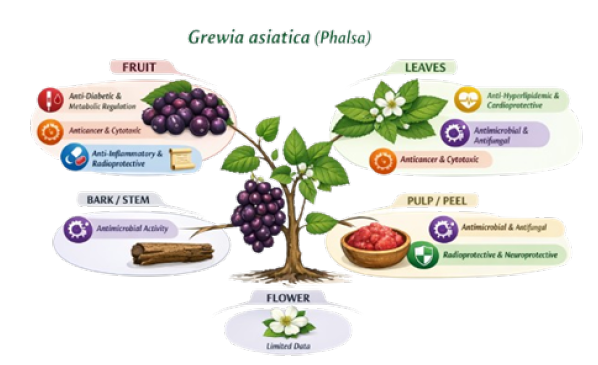
Traditional uses of phalsa
Flores has flowers usually in Jan-Feb and then the fruit starts growing between May and June. When the fruit is ripening, the skin starts out as a light green colour and then it changes to cherry red or purplish red; once the fruit is fully matured its colour will have turned dark purple, almost black [26]. The skin of ripe fruit is soft and delicate, but has a thin, whitish “blush” on the outside [26]. The flavor of Phalsa fruits can be described as sweet with some sourness and an acidic flavour. It has a grape-like flavour. Phalsa fruits are very well-priced, at about $1/kg in local markets and they are commonly eaten fresh with some salt to make them taste better. They can also be preserved or processed into jams, pies, beverages (such as squashes) or chutney [27]. It has a short shelf life, making it more suitable for selling locally [6]. Different disorders, such as heart disorders, blood disorders, and liver disorders, as well as anorexia, indigestion, thirst, toxemia, stomatitis, hiccough, asthma, spermatorrhoea, fevers and diarrhea, have all been attributed to the beneficial effects of the fruit of the plant. It is used to treat throat problems, tuberculosis and sexual debility [9,10]. The root bark of the plant is used for treating rheumatism and urinary tract issues [10,11]. In sugar purification, the stem bark of this plant is used [12]. The leaves of this plant are used for treating wounds or cuts to the skin, and for irritation from or pain caused by rashes on the skin [13]. The leaves are also believed to be antibiotic in nature and can be used as fodder for livestock. The stems and bark of the plant can also be used to produce rope, baskets and as a source of fuel. The bark can be used as a soap substitute [14].
Compositional studies
Fruits of G. asiatica are low in calories and fat, and high in vitamins, minerals and fiber. It contains carbohydrates, dietary fibre, vitamin C, iron, calcium and potassium, making it nutritionally valuable [28]. Its high anthocyanin content contributes significantly to antioxidant potential [19]. Mineral bioavailability studies indicate that phalsa may serve as a supplementary dietary source in micronutrient-deficient populations [22]. Post-harvest processing influences nutrient retention and antioxidant stability [29] (Figure 2).
Figure 2:Nutritional Values of Fruit (g).
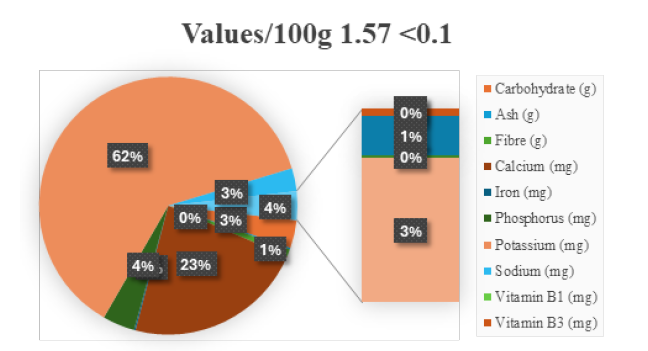
Indigenous people have long regarded fruit juices and squash as nutritious beverages. A summer fruit drink called phalsay ka sharbat (made from fruits) can be found in food stores. This drink is considered to be a cardiac tonic. When combined with other nutrients, the juice may serve as a source of nutrition for the diet. Because the carbohydrates in low glycemic index foods are broken down more slowly than the carbohydrates found in higher glycemic index foods, they are a good option to help with diabetes. Low glycemic index foods may also reduce the risk of heart disease and obesity. Essential amino acids considered nutritionally necessary (ex. threonine and methionine) can be found in the pulp of the fruit and in the seeds of the fruit respectively, whereas predominately found in juice are the following amino acids: Phosphor-serine, serine and taurine. The concentration of phosphor-serine is greater in the pulp compared to the other free amino acids and a high concentration of aspartic acid, glycine and tyrosine was found in the hydrolyzed product [18]. Additionally, pigments and total soluble solids can be extracted from pomace. The highest yield of pigments and total soluble solids has been achieved when adding 75% water to the pomace [30].
Fruit juice formulated and standardized to make Ready-To- Drink beverage (RTD) contained 25% juice with a Brix-acid ratio of 45:1 [15]. A syrup from fruit juice was made by combining the clear juice with an equal quantity of sugar before adding sodium benzoate [17]. Both hydrolyzed and unhydrolyzed (free) pulp and seed were evaluated for amino acids, which were also used to determine extent of adulteration of fruit juice. Threonine existed in pulp while it was absent in seed extract (locally), whereas methionine was only found in seeds; therefore, threonine presence in fruit juice can be attributed to adulteration. Phosphoserine and serine were the two primary amino acids vast in number present in juice [31]. Evaluation of G. asiatica fruits for six trace minerals (Co, Cr, Cu, Ni, Zn and Fe) using both Fresh Weight (FW) and Dried Weight (DW) provided some information regarding their concentrations [32]. The highest concentration of these essential nutrients was iron and the lowest concentration was cobalt. Trace minerals play major roles in many chemical and biological functions in human body. The seeds’ chemical composition shows they’re 5% oil and are a bright yellow colour. There is different composition of fatty acid present such as palmitic (8%), stearic (11%), oleic (13.5%) and linoleic acids (64.5%) while small amount of unsaponifiable matter (3%) was also detected [6].
Phytochemistry
Preliminary phytochemical screening and primary metabolites
Phytochemical screening reveals the presence of flavonoids, tannins, saponins, glycosides and alkaloids [33]. High-resolution LC-MS profiling has identified quercetin, kaempferol and cyanidin derivatives [20]. Flavonoid derivatives isolated from G. asiatica have shown strong radical-scavenging activity [24,20]. Phenolic profiling confirms significant antioxidant potential compared to other berry fruits [21].
Fruits
Preliminary phytochemical screening of the fruits revealed that methanolic extracts contained carbohydrates, tannins, phenolic compounds, flavonoids and vitamin C; petroleum ether extracts contained flavonoids and fixed oil; benzene extract contained steroids; ethyl acetate extracts contained carbohydrates, tannins, flavonoids and phenolic compounds; and aqueous extracts contained carbohydrates, tannins, phenolic compounds and protein [34]. Paper chromatography analysis of the fruit’s ethanol extract revealed amino acids (proline, glutaric acid, lysine and phenylalanine) and carbohydrates (glucose, xylose and arabinose) present [1].
Leaves
Diterpenes, glycosides and fats are contained within the petroleum ether extract of leaves as identified through the phytochemical screening of leaves; chloroform extract contains alkaloids and glycosides; whilst the ethanolic extract consists of triterpenoids, sterols, flavonoids, saponins and tannins [2]. The pharmacognostic evaluation of leaves revealed that they have a total ash content of 5% comprising of water-soluble ash (2.5%) and acid-insoluble ash (2.1%) [3]. The phytochemical evaluation studies of roots and barks have yet to take place.
Pharmacological Activities
Antioxidant activity
Invitro antioxidant assays demonstrate strong DPPH and FRAP activity of phalsa extracts [4]. Anthocyanins in phalsa reduce oxidative stress biomarkers and modulate endogenous antioxidant enzymes [5]. Comparative studies show that phalsa exhibits higher antioxidant capacity than several common tropical fruits [6].
Fruits
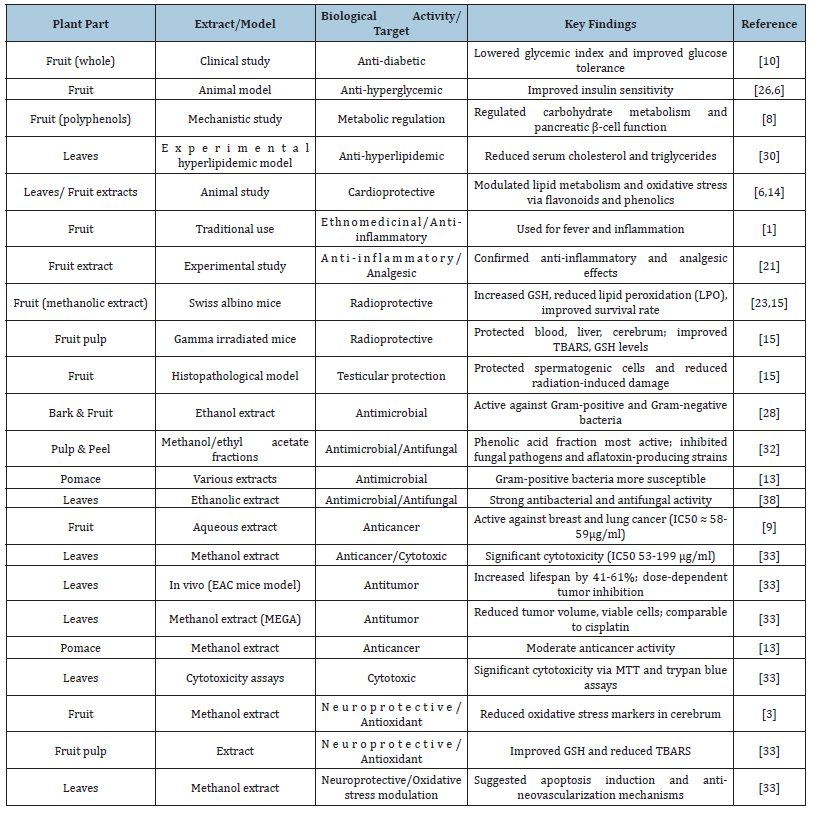
Table 1:Pharmacological and biological activities of different parts of Grewia asiatica.
This table summarizes the pharmacological and biological activities of various parts of Grewia asiatica, including fruits,
leaves, bark, pulp, peel and pomace. Activities reported include anti-diabetic, anti-hyperlipidemic, cardioprotective, antiinflammatory,
analgesic, radioprotective, antimicrobial, antifungal, anticancer, antitumor, cytotoxic, neuroprotective,
and antioxidant effects. Extract type and experimental models are indicated where applicable. IC50 values, GSH
(reduced glutathione), TBARS (thiobarbituric acid reactive substances), and other markers are reported as per the
original studies.
Grewia asiatica (known as Phalsa) contains considerable levels of antioxidants such as vitamin C, total phenolics, flavonoids, tannins and anthocyanins in fresh fruit. The four sub-fractions, non-anthocyanin fraction into flavanols (sub-fraction I(a)), anthocyanins (sub-fraction II), phenolic acids (sub-fraction I(c)) and flavonols (sub-fraction I(b)), were evaluated for their ability to scavenge free radicals using the DPPH method (see Table 1). All the sub-fractions exhibited significant free radical scavenging abilities, which increased with increasing concentration (p<0.05). The highest percentage of scavenging activity was found within the range of 62-85% for all sub-fractions combined at the 20ppm concentration of each compound (p<0.05). The greatest degree of free radical scavenging activities occurred in the order of subfraction I(b) > sub-fraction I(c) > sub-fraction II > sub-fraction I(a). Numerous studies performed in vitro have shown that polyphenols are capable of acting as free radical scavengers and prevent the oxidative modification of LDL (low density lipoproteins) that contribute to heart disease [7].
Total phenolic content, flavonoid and anthocyanin concentrations [20] are the main determinants of the antioxidant activity of the G. asiatica (Phalsa) fruit. The order of antioxidant activity among the different fractions was as follows: Fraction Ib > Fraction Ic > Fraction II > Fraction Ia. Fraction Ia had the lowest total phenolic content. In Fraction Ib, approx. 72+/-6.0mg of anthocyanins were present/100gm. Polyphenolic content of foods provides more effective antioxidants than ascorbic acid, vitamin E and β-carotene (Vinson et al, 1995). Flavonoid distribution was as follows - Fraction II > Fraction Ic > Fraction Ib. Flavonoids are colour pigments, which are part of the polyphenol family, are found throughout the plant world and are also characterised by their antiradical and chelating activity. Flavonoids will either scavenge free hydroxyl and peroxy radicals or they will extract iron to decrease the superoxide Fenton reaction (Afanasev et al, 1989). There is a direct correlation between the antioxidant capacity of fruits and their flavonoid content/Wang, 1996). All fractions of G. asiatica (fruit) were shown to effectively oxidise β-carotene in an aqueous linoleic acid emulsion (p < 0.05) [8].
The oxidation of β-carotene was demonstrated to be inhibited by the 58-89% fractions (i.e. Fraction Ib contained approx. 89% inhibition of oxidation, whereas Fraction Ia contained 58% inhibition) of the various fractions of G. asiatica. There are several definitions for the term “antioxidant,” but one basic quality of an antioxidant is its ability to delay or prevent the oxidation of a substrate (i.e., the oxidation of β-carotene). The antioxidant activity of the total G. asiatica fruit extract is determined by the degree of reduction (i.e., the amount of hydrogen it donates) [9]. Table 1 presents the reducing abilities of each fraction of G. asiatica fruit extract (Phalsa) in terms of the potassium ferricyanide reduction assay. The level of absorbance (A) of the fractions was measured according to a concentration of 50ppm at 700 nanometers (nm) and the A values ranged from 1.53-3.1. The reducing ability of the various fractions relates to the presence of reductones [10]. Reductones not only stop chain reactions of free radicals chains by donating a single hydrogen atom, but also terminate (i.e., quench/ enzyme) the formation of peroxides by reacting with the precursors of peroxides [11].
Antioxidant activity of methanol extract from fruit of G. asiatica evaluated using different assays showed antioxidant activities significantly lower than other fruits. Total flavonoid content (4.608QEmg/g), total phenolic content (144.11mg GAE/g), and total anthocyanin content (4.892mg/kg) were found at significant rates. Antiradical activity against DPPH was significant (84.83) and against peroxyradicals (37%). The TEAC (269.038TEACmM) and FRAP (4.14GAE/g) values were also comparatively higher than several other documented plant naturally occurring sources (Srivastava and al. 2012).
Fruit pomace
Total flavonoids, alkaloids, saponins and tannins were determined from fruit pomace and were measured as dry matter. The amount in dry matter for flavonoids was 12.42±0.56(CEmg/g) on average, alkaloids were 1.56±1.2(g/100g) on average, saponins were 1.05±0.96(g/100g) on average and tannins were 0.52± 1.25 (g/100g) on average [12]. Therefore, it can be stated as fruit pomace being waste material, there is still a significant amount of antioxidant present in this material. The aqueous extract of the fruit contained 5.25 total phenols (CE) and 0.13 total flavonoids (GAE). The extract of the fruit had a higher concentration of both phenols and flavonoids compared to 21 other extracts evaluated at the same time [26].
Leaves
The antioxidant ability of the leaf extracts of G. asiatica was evaluated based on their capacity to scavenge free radicals as measured by the DPPH method and by measuring the inhibition of the nitric oxide radical [13]. The extracts of G. asiatica demonstrated significant levels of antioxidant activity by exhibiting low IC50 values when evaluated using both the DPPH and nitric oxide radical inhibition assay (Table 1) [35]. The sequentially extracted leaves, including petroleum ether, benzene, ethyl acetate, methanol, water and 50% crude methanol, produced IC50 values of 249.60±7.37, 16.19±2.132, 26.17±1.49, 27.38±1.80, 176.14 ±5.53 and 56.40±3.98μg/ml, respectively in the DPPH assay and of 22.12±02.65, 27.00±01.62, 47.38± 05.88, 56.85±06.16, 152.75±5.76 and 72.75±13.76μg/ml in the nitric oxide radical inhibition assay. These IC50 values were lower than those obtained from ascorbic acid and quercetin when assayed as controls. Free radical induced oxidative stress has been implicated in the pathogenesis of numerous disease states and most often is a result of impaired endogenous antioxidant defenses (Halliwell and Gutteridge, 1989). Six extracts of G. asiatica (Phalsa) leaves and two reference compounds were assessed for their antioxidant properties using the DPPH method. The successive extracts of benzene and ethyl acetate exhibited the highest antioxidant activities with IC50 values of 16.19±2.13 and 26.17±1.49μg/ml, respectively, followed by the methanol extract, which had an IC50 value of 27.38±1.80μg/ml. The 50% methanol and distilled water crude extracts of G. asiatica (Phalsa) exhibited IC50 values of 56.40±3.98 and 176.14±5.53μg/ml, respectively. Conversely, the petroleum ether extract demonstrated the lowest antioxidant activity with an IC50 value of 249.60±7.37μg/ml. The known antioxidants ascorbic acid and quercetin had IC50 values of 78.17±4.05 and 53.60±1.79μg/ml, respectively. Therefore, the antioxidant capacity of G. asiatica (Phalsa) may be attributed to the presence of total phenolics, flavonoids, tannins and the anthocyanins, which are affected by a number of factors, including genotype, growth conditions, agronomic practices; seasonal effects, maturity, post-harvest storage, processing conditions and extraction solvents.
Antimicrobial Activity
Fruit and bark
Ethanol extracts from G. asiatica’s bark and fruit have presented antimicrobial activity against Bacillus subtilis, Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus pneumoniae and six different gram-negative bacteria (Escherichia coli, Proteus vulgaris, Proteus mirabilis, Salmonella typhi para-A, Salmonella typhi para-B and Shigella dysenteriae), with S. aureus, E. coli and P. vulgaris showing activity (Israr et al. 2012).
Pulp and peel
Extracts of G. asiatica fruit and skin found to contain three different types of polyphenols: Ethyl acetate extracts consist of: Neutral polypenolic fractions (comprised of flavanols and other polyphenols); neutral flavonoid bc fractions; acidic polyphenolics & anthocyanin fractions. All major fraction samples were evaluated for antibacterial activity; all feline samples exhibited activity, while anthocyanin of those. The most susceptible species was Staphylococcus aureus (amongst the Gram-positive strains); Salmonella typhi was the most susceptible among the Gramnegative strains. Bacillus subtilis was the most resistant of the Gram-positive and E. coli the most resistant of the Gram-negative strains; however, both Aspergillus spp. was completely inhibited (by any of the major fractions). Flavanol-fractions as one other polyphenol have been tested for antifungal potential, with no growth of Trichophyton mentagrophytes or Trichophyton rubrum. The inhibition of the Aspergillus spp. by the flavanol (and other) fractions gives evidence of the possibility that the compounds contained in those fractions may be responsible for preventing the development of aflatoxins in food products. As the most active one of the five types of fractions identified in this study (phenolic), phenolic acid was tested for antifungal activity using six different types of fungi: Penicillium notatum (a type of mold), Aspergillus niger, Aspergillus flavus, Microsporum Gypseum, Trichophyton mentagrophytes and Trichophyton rubrum. All six species were significantly inhibited by phenolic acid [36].
Pomace
The different extracts of G. asiatica pomace were tested for their effectiveness against Gram positive (Bacillus subtilis, B. cereus, Staphylococcus aureus, Enterococcus faecalis) and Gram negative (Escherichia coli, Listeria Mon cytogeneses, Salmonella typhimurium, Shigella flexneri and Pseudomonas aerugenosa) strains of bacteria. The results showed that the Gram-positive organisms were significantly more affected by the extracts than were the Gram negative. According to Gupta et al. (2012), the Grampositive bacteria were much more sensitive to the crude extracts and the antibacterial compounds isolated from the pomace than were the Gram-negative organisms, which is likely due to the structure of their cell walls.
Leaves
The leaves of G. asiatica contain extracts that have shown activity against bacteria and fungus; the bacterium Proteus mirabilis and the fungi Aspergillus effusus, A. parasiticus, A. niger, Saccharomyces cerevisiae, Candida albicans, Yersinia aldovae, Fusarium solani, Macrophomina phaseolina and Trichophyton rubrum demonstrate significant susceptibility to the ethanolic extract.
Anticancer Activity
Fruits
Fruits’ aqueous extracts exhibited notable anticancer effects against both liver and breast tumors. To gauge the extract’s in vitro cytotoxicity, we employed the MTT assay and tested the following human cancer cell lines: HEC (HEK-293), MCF (MCF-7), HELA (HELA), NCI (NCI-H522) and Hep-2 (Hep-2). The aqueous extracts of the tested fruits had IC50 values of 59.03(lung) and 58.65 (breast)μg/mL, indicating their efficacy against HEC and MCF human tumor cell lines, respectively. Therefore, the tested fruits could be an important tool in treating human cancer [37].
Leaves
The MTT assay was used to evaluate the in vitro anticancer and cytotoxicity activity of a methanol extract of G. asiatica leaves against 4 different human cancer cell lines: Acute myeloblastic leukaemia (HL-60), chronic myelogenous leukaemia (K-562), breast adenocarcinoma (MCF-7) and cervical epithelial carcinoma (Hela) with IC50 values of 53.70, 54.90, 199.5 and 177.8, 89.12 respectively. The intraperitoneal administration of 250 and 500mg/ kg of extract to male Swiss albino mice resulted in an increase in the life span of Ehrlich’s Ascites Carcinoma (EAC) tumour-bearing mice by 41.22% and 61.06% respectively. The methanol extract of leaves provided protection against EAC development in mice in a dose-dependent manner [18]. Aqueous leaf extracts showed significant anticancer activity against liver and breast cancer. The leaf extract showed activity against breast (IC50=50.37μg/mL) and Hep-2 (IC50=61.23μg/mL) cancer cell lines. These results provide evidence to support the use of leaf extracts as a potential therapeutic agent in the treatment of human cancers [30].
Pomace
Pomace methanol extract of G. asiatica is an effective treatment in vitro against certain types of cancer (HeLa cervical cancer cells), MCF-7 breast cancer cells and HepG-2 liver cancer cells but requires further testing for cytotoxicity against those same cell lines in vivo before confirming the potential medicinal value of this extract. In conclusion, results from this research could lead to developing new anticancer agents derived from the natural phytochemicals found in G. asiatica pomace according to Gupta et al., 2013.
Antitumor Activity
Leaves
The effects of methanol extracts of Grewia Asiatica (Phalsa) (MEGA) leaves on cancer cells in mice were examined using an ascitic tumour model (as per Nair and Panikkar, 1990). Five mice were placed into four groups. Each animal was given an intraperitoneal injection of 2×10^6 viable EAC cells/ml in phosphate buffered saline from a mouse with a 15day old EAC ascitic tumor. After 24 hours of EAC inoculation, MEGA was given orally at either 250 or 500mg/kg body weight for 10 consecutive days. The group that received only the vehicle (0.9% w/v NaCl) served as the control. Cisplatin (2mg/ kg b.w.) was also used as a standard reference drug administered intraperitoneally. Blood parameter tests, increase in life span (ILS), tumour volume, number of tumour cells per millilitre, number of viable and non-viable cells per millilitre and mean survival time of control and treatment groups were measured and compared with that of standard Cisplatin. The ILS was calculated using the formula for % ILS=(1-T/C)×100, where T is the mean survival time in the treated group and C is the mean survival time in the control group (Nair and Panikkar, 1990). The results demonstrated that all the leaf extracts of G. asatica (Phalsa) exhibited significant levels of cytotoxicity towards various types of tumour cells and displayed antitumour activity. The life span of tumour bearing mice subjected to MEGA treatment was extended and is strongly indicative of possible anticancer properties. The body masses of all the tumour bearing mice used in this study were reduced with the administration of MEGA. The choice of the doses of MEGA (250mg/kg and 500mg/kg) used in the present study was based on the results derived from preliminary studies. The antitumour properties of this plant may be attributed to inducement of apoptosis or inhibition of neovascularization. Many phytoconstituents are present in this plant which may explain the reported antitumour activity. Currently further studies are being undertaken in order to identify the individual phytochemical entity/ies that exhibit these properties, and to determine the overall chemistry of G. asiaatica (Phalsa) [15]. All antitumour activity was determined exclusively from the leaves of the G. asatica (Phalsa) plant.
Cytotoxicity Activity
Leaves
The effects of methanol extracts of Grewia Asiatica (Phalsa) (MEGA) leaves on cancer cells in mice were examined using an ascitic tumour model (as per Nair and Panikkar, 1990). Five mice were placed into four groups. Each animal was given an intraperitoneal injection of 2×10^6 viable EAC cells/ml in phosphate buffered saline from a mouse with a 15day old EAC ascitic tumor. After 24 hours of EAC inoculation, MEGA was given orally at either 250 or 500mg/kg body weight for 10 consecutive days. The group that received only the vehicle (0.9% w/v NaCl) served as the control. Cisplatin (2mg/ kg b.w.) was also used as a standard reference drug administered intraperitoneally. Blood parameter tests, increase in life span (ILS), tumour volume, number of tumour cells per millilitre, number of viable and non-viable cells per millilitre and mean survival time of control and treatment groups were measured and compared with that of standard Cisplatin. The ILS was calculated using the formula for % ILS=(1-T/C)×100, where T is the mean survival time in the treated group and C is the mean survival time in the control group (Nair and Panikkar, 1990). The results demonstrated that all the leaf extracts of G. asatica (Phalsa) exhibited significant levels of cytotoxicity towards various types of tumour cells and displayed antitumour activity. The life span of tumour bearing mice subjected to MEGA treatment was extended and is strongly indicative of possible anticancer properties. The body masses of all the tumour bearing mice used in this study were reduced with the administration of MEGA. The choice of the doses of MEGA (250mg/kg and 500mg/kg) used in the present study was based on the results derived from preliminary studies. The antitumour properties of this plant may be attributed to inducement of apoptosis or inhibition of neovascularization. Many phytoconstituents are present in this plant which may explain the reported antitumour activity. Currently further studies are being undertaken in order to identify the individual phytochemical entity/ies that exhibit these properties, and to determine the overall chemistry of G. asiaatica (Phalsa) [15]. All antitumour activity was determined exclusively from the leaves of the G. asatica (Phalsa) plant.
Radioprotective activity
The increasing use of nuclear radiation for human welfare despite its beneficial effects has some undesirable side effects so there is need to check the side effects for the same. Search for the chemical agents that are able to protect human beings from the ionizing radiation is a key issue in radiation biology [17].
Fruit
Radioprotective effects of the methanolic extract of G. asiaica (Phalsa) fruit were evaluated in the brains of Swiss albino mice; mice were split into four groups, with group I being the untreated control, group II receiving the extract by mouth at 700mg/kg once daily for 15 days (treated), group III receiving distilled water as the control for 15 days, and group IV receiving the extract by mouth for 15 days before being exposed to a single dose of 5 Gy gamma radiation. Mice were sacrificed at 1, 3, 7, 15 and 30 days after exposure to radiation. Brains were removed to estimate brain glutathione (GSH) concentrations and levels of Lipid Peroxidation (LPO). Extract supplementation reduced radiation-induced increases in LPO by approximately 5% at 30 days after exposure (post irradiation). In addition, although GSH levels were depleted by radiation, supplementation raised levels of GSH by 14.57% at 30 days after exposure (post irradiation). This suggests that the extracts have the potential to protect against radiation-induced disturbances (Ahaskar & Sharma, 2006).
Researchers studied the protective effects of an extract against X-ray radiation exposure in mice. The extract was determined to be safe to the mice and provided the maximum amount of protection when the mice received 700mg/kg/day for 15 days before being exposed to 10 Gy of gamma rays. There were significantly more survivors in the group of mice that received the extract prior to irradiation; therefore, the LD50/30 value for the irradiated control group was 6.21 while the LD50/30 value for the extracted irradiated group was 9.53, providing a dose reduction factor of 1.53. Mice receiving the extract demonstrated significantly increased levels of reduced glutathione and significantly decreased levels of lipid peroxidation in both their brain and liver tissue samples at 24 hours following exposure to radiation. Researchers also studied the protective effects of the extract against changes to the cerebellum of mice that were subjected to radiation exposure at (1, 3, 7, 15 and 30-day intervals). Following radiation exposure to the cerebellum, researchers observed an increase in lipid peroxidation levels; however, the addition of the extracted provided protection against those increases. Similarly, radiation-induced depletion in proteins was significantly controlled by extract administration [31].
An investigation was done to determine the radioprotective benefits of an extract from Sutherlandia frutescens in male Swiss mice using a model that compared radiated and non-radiated groups. Five groups of animals were evaluated: controls; animals receiving the extract at a dose of 700mg/kg for fifteen days followed by radiation; animals receiving 5 Gy of radiation but no extract; animals receiving extract and radiation; and animals receiving extract prior to and after radiation. Following exposure to radiation, there was significant lipid peroxidation (as indicated by an increased level of thiobarbituric acid reactive substances (TBARS), decreased levels of glutathione (GSH) and total protein, in comparison with controls). The use of the extract prior to and after radiation resulted in decreased TBARS levels and increased concentrations of both GSH and total protein in the intestine and testis for all time points following irradiation, compared with irradiated animals. In addition, the extract provided significant protection to the DNA and RNA of the testis. The extract exhibited a high degree of radical scavenging ability (in both the DPPH and O2 assays) and demonstrated in vitro radioprotective capability, using the protein carbonyl assay, in a dose-dependent manner [31]. Through histopathological analysis, the effect of the fruit extract on radioprotection was tested in the testis of mice. The irradiation of the animals resulted in a significant reduction in testis weight, whilst the fruit extract treated group had testis weights which were significantly increased when compared to the irradiated only group. The histopathological analysis of testicular tissue of the irradiated group was found to contain a significantly reduced number of spermatogonia “A”, spermatogonia “B”, spermatocytes, and spermatid, when compared to the irradiated group treated with the fruit extract pre- and post-irritation, all the way until the final autopsy interval at 30 days post irradiation. This indicates that the fruit extract offers protection against the damaging effects of irradiation on the testis (Sharma and Sisodia, 2010).
Fruit pulp
The study assessed the radioprotective properties of the fruit pulp extract of G. Asiatica on blood cells and plasma levels of different hematological and biochemical parameters, compared with unexposed controls, in Swiss albino mice exposed to 5 Gy of gamma irradiation. This study was performed over the course of 15 days, during which G. Asiatica was provided (=700mg/ kg/day orally + 5 Gy gamma irradiation) to irradiated animals before they were sacrificed 0, 24 and 72 h post-evaluation. G. Asiatica significantly increased gsh, gsphx, and serum sugar. G. Asiatica was also able to improve the hematological parameters of irradiated mice compared to those in the control group (with only 5 Gy of gamma irradiation), while at the same time attenuating the elevation of serum cholesterol and lipid peroxidation due to gamma irradiation [32]. A study investigated the effect of a fruit pulp extract on the brains of Swiss albino mice exposed to 5 Gy of gamma radiation. After the mice were sacrificed at different intervals (1-30 days) post-exposure, biochemical metrics were measured from their brains. Results showed that extract-pretreatment significantly reduced the increase in lipid peroxidation in the brains of irradiated mice. The same assay was also performed with blood and livers of irradiated mice pre-treated with the same extract by measuring reductions of glutathione (GSH) and lipid peroxidation. The results indicated that extract post-treatment protected liver and blood from damage caused by radiation; this was achieved through an inhibition of glutathione depletion and by lowering lipid peroxidation levels back to near normal by 30 days post-treatment. The degree of recovery from oxidative damage measured by thiobarbituric acid reactive substance (TBARS) and GSH content was significantly greater (p<0.001) in the irradiated and extract treated group [16]. Given the biochemical effects of G. asiaica (Phalsa) in the liver and brain of this study it could be possible to use such dietary compounds as radioprotectors in clinical applications [38-42].
Conclusion
Grewia asiatica represents a nutritionally dense and pharmacologically promising underutilized fruit. Its antioxidant, antidiabetic, cardioprotective and anti-inflammatory properties are supported by growing experimental evidence. Although promising pharmacological properties have been demonstrated, largescale randomized controlled trials are lacking. Standardization of extraction methods and dosage optimization remain critical challenges. Future research should focus on clinical validation, bioavailability studies and molecular mechanisms of action.
Acknowledgement
The authors would like to thank Colima University and the National Autonomous University of Mexico for their support.
References
- Goswami S, Jain R, Masih H (2018) Antifungal, antioxidant and DNA protection potential of Grewia asiatica L. leaves acetone extract. Journal of Pharmacognosy and Phytochemistry 7(1S): 212-217.
- Muhammad Zia-Ul-Haq, Milan S Stanković, Komal Rizwan, Vincenzo De Feo (2013) Grewia asiatica L. a food plant with multiple uses. Molecules 18(3): 2663-2682.
- Hubbard BP, Sinclair DA (2014) Small molecule SIRT1 activators for the treatment of aging and age-related diseases. Trends in Pharmacological Sciences 35(3): 146-154.
- Joshi N Sharma R (2018) Anthocyanin quantification in phalsa cultivars. International Journal of Fruit Science.
- Kitada M, Koya D (2016) Sirtuins in type 2 diabetes: Mechanisms and therapeutic potential. Diabetes & Metabolism Journal 40(4): 285-296.
- Kaeberlein M, McDonagh T, Heltweg B (2015) Substrate-specific activation of sirtuins by resveratrol. Journal of Biological Chemistry 290(12): 7597-7605.
- Kaur K, Singh P (2017) Phenolic and flavonoid profile analysis of Grewia asiatica. Journal of Food Biochemistry.
- Khangarot K, Mishra A, Bhardwaj R, Sharma RA (2024) Phytochemical and in vitro antioxidant screening of Grewia asiatica L. Journal of Pharmacognosy and Phytochemistry 13(1): 230-232.
- Khan A, Verma S (2019) Functional characterization of Grewia asiatica bioactive. Journal of Agricultural and Food Chemistry.
- Khattab K, Ahmed S (2015) Antihyperglycemic effects of Grewia asiatica in diabetic models. Journal of Medicinal Food.
- Khattab K, Ahmed S (2019) Nutritional and antioxidant profiling of phalsa. Food Chemistry.
- Kumar A Singh R (2022) Mechanisms of polyphenols in phalsa. Molecular Nutrition & Food Research.
- Marya CM, Gupta R (2011) Anticancer evaluation of phalsa extracts. Cancer Letters.
- Mesaik MA, Khan M (2013) Effect of Grewia asiatica fruit on glycemic index and phagocytosis in healthy human subjects. Pakistan Journal of Pharmaceutical Sciences 26(1): 85-89.
- Rajput M, Sharma R (2019) Effects of storage on antioxidant retention in phalsa. Food Chemistry.
- Sharma KV, Sisodia R (2009) Radioprotective effect of Grewia asiatica in mice models. Journal Name.
- Rajput R, Kumar S (2018) Surrounding soil and metabolite variations in Grewia asiatica. Plant Science Today.
- Qamar M, Khan A (2021) The chemical composition and health-promoting effects of the Grewia species-A systematic review and meta-analysis. Nutrients 13(12): 4565.
- Ali S, Khan M (2021) Post-harvest handling and processing of underutilized fruits including phalsa. Food Control.
- Agarwal S, Mishra K (1980) Flavonoid derivatives in Grewia asiatica. Phytochemistry.
- Baur JA, Pearson KJ, Price NL, Jamieson HA, Lerin C, et al. (2006) Resveratrol improves health and survival of mice on a high-calorie diet. Nature 444(7117): 337-342.
- Asghar MN, Ullah Khan I, Sherin L, Ashfaq M (2010) Evaluation of antioxidant activity of Grewia asiatica berry. Asian Journal of Chemistry 20(7): 5123-5132.
- Ahaskar M, Sharma S (2007) Post-treatment effect of Grewia asiatica against radiation-induced biochemical changes. International Journal of Radiation Research.
- Agarwal S, Mishra K (1979) New flavonoids from the Grewia asiatica. Journal of the Indian Chemical Society 56: 649.
- Akand JC (1960) Efficacy of sodium benzoate to control yeast fermentation in phalsa juice. Indian Journal of Horticulture 17(2): 138-141.
- Ahmed A, Siddiqui AA (2015) Effects of phalsa on metabolic indices. Journal of Functional Foods.
- Ahmed K, Khan M (2014) Antioxidant enzyme modulation by phalsa extracts. Journal of Nutritional Biochemistry.
- Ahmed S, Ali R (2020) Antibacterial activity of Grewia asiatica extracts against clinical strains. Journal of Applied Microbiology.
- Baliga MS, Dsouza J (2011) Medicinal potential of selected berries including phalsa. Nutrition Journal.
- Rani S, Singh P (2016) Grain and berry polyphenol comparison including phalsa. Journal of Agricultural Chemistry.
- Sharma A, Singh R (2015) Phytochemical variability in phalsa fruit parts. Journal of Food Composition and Analysis.
- Sharma D, Singh S (2019) Nutraceutical potential of phalsa phenolics. Journal of Functional Foods.
- Bibhuti P, Sharma D (2011) Methanol extract (MEGA) reduces tumor volume and viable cells in tumor ascites model. Journal Name.
- Das S, Singh R (2012) Analgesic and anti-pyretic activities in Grewia asiatica. Journal of Ethnopharmacology.
- Mishra R, Sharma R (2021) Impact of Grewia asiatica extracts on neural oxidative biomarkers. Brain Research Bulletin.
- Nawaz S, Naz A, Shafi A, Amin M, Farooq U, et al. (2020) Product development of frozen and dehydrated phalsa (Grewia asiatica). Agricultural Sciences Journal 2(1): 16-22.
- Pandey V, Singh G (2016) Phytoconstituents and antioxidant activities of phalsa pomace. Food Science and Nutrition.
- Sharma P, Verma S (2019) Functional beverages from Grewia asiatica. Food Science & Technology International.
- Singh D, Singh P (2017) Role of phalsa anthocyanins in oxidative stress mitigation. Oxidative Medicine and Cellular Longevity.
- Siddiqi A, Khan M (2011) Profiling of polyphenols in phalsa (Grewia asiatica L.) fruits using high-resolution LC-MS. PMC.
- Verma P, Singh R (2020) Dietary fiber and mineral bioavailability in phalsa. Journal of Trace Elements in Medicine and Biology.
- Zia-ul-Haq M, Riaz M (2012) Antimicrobial and antifungal activity of Grewia asiatica leaves ethanolic extract. Journal Name.
© 2026 Ali Usman & Hafiz Nafees Ul Hassan. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






