- Submissions

Full Text
Modern Approaches in Drug Designing
A Modified Sensitive Fluorescent Assay Demonstrating Varying Inhibitory Effects of Oxidative and Nitrosative Stress on Platelet Surface Protein Disulphide Isomerase
Usman Ali*
Department of Haematology, The Royal London Hospital, UK
*Corresponding author:Usman Ali, Department of Haematology, The Royal London Hospital, 80 Newark St, London E1 2ES, UK
Submission: October 25, 2025;Published: January 21, 2026

ISSN: 2576-9170 Volume5 Issue 1
Abstract
Platelet surface Protein Disulphide-Isomerase (psPDI) is indispensable for thrombus formation at vascular injury sites, primarily through its role in αIIbβ3 integrin activation and is influenced by oxidative and nitrosative stress. Given limitations of traditional assays, a sensitive fluorometric method employing the fluorescence-quenched pseudo-substrate Di-E-GSSG was utilised to determine psPDI reductase activity. The Di-E-GSSG probe was synthesised and validated, exhibiting a greater than 6-fold fluorescence increase with Di-Thio-Threitol (DTT), confirming its suitability. Experiments demonstrated the probe’s resistance to low DTT concentrations, with 5μM DTT deemed appropriate. The assay successfully monitored both bovine liver PDI and psPDI-dependent disulphide reduction in a concentration-dependent manner, confirming psPDI’s contribution to reductase activity and the assay’s efficacy as a simple, single-step tool. Furthermore, psPDI activity was consistently inhibited by known PDI inhibitors: DTNB (100%), bacitracin (80%), PAO (55%) and rutin (14%), serving to confirm the presence of psPDI-contributed surface reductase activity. Most significantly, exposure to oxidative and nitrosative stress agents resulted in a dose-dependent disturbance of psPDI activity. At the highest concentrations, oxidative stress, specifically using hydrogen peroxide and hypochlorous acid, led to complete (100%) ablation of psPDI activity, whereas the nitric oxide donors, nitrosoglutathione and diethylamine nitric oxide, resulted in 97% and 86% inhibition, respectively, thereby retaining 3% and 14% activity with equimolar concentrations. This highlights that oxidative stress exerts a more potent inhibitory effect on psPDI than nitrosative stress. In conclusion, this study demonstrates that the reductive function of psPDI is markedly inhibited within oxidative/nitrosative stress environments, offering insights into its regulation.
Keywords:Blood platelets; Protein disulphide-isomerase; Cell surface; Redox potential; Fluorescent probes
Introduction
Protein Disulphide Isomerase (PDI) is a multifunctional enzyme with ubiquitous expression and wide distribution in eukaryotic cells [1]. It possesses both reductase and oxidase activities, enabling the rearrangement (i.e., isomerisation) of incorrect intramolecular disulphide bonds [2]. Primarily located in the Endoplasmic Reticulum (ER), PDI’s ability to catalyse disulphide bond isomerisation is among its most studied functions [3]. However, despite bearing a KDEL retention signal, PDI is also present in non-ER locations, including the nucleus, cytosol, cell surface and extracellular space [4]. The presence of PDI in these diverse locations has been linked to various pathological conditions, including AIDS [5,6], cancer [7], thrombus formation [8-10] and other pathogenic infections [11,12]. Platelet surface PDI (psPDI) is crucial for platelet activation and secretion [13,14], accounting for approximately 40% of total surface thiols on platelets [15]. Research has demonstrated its involvement in the redox control of platelets through disulphide isomerisation [14,16] and denitrosation [17], both of which influence platelet aggregation. The isomerising activity of psPDI has been shown to modulate αIIbβ3 integrin activation via intricate mechanisms, thereby contributing to thrombus formation [10]. Concurrently, its denitrosating function mediates the delivery of Nitric Oxide (NO) redox species into platelets across the membrane [18], eliciting an antithrombotic response [19].
This study hypothesises that psPDI’s involvement in both pro- and anti-thrombotic states stems from its ability to modulate redox chemistry at the platelet surface. Specifically, it is proposed that psPDI’s influence on thiol redox modifications, which in turn affects its own enzymatic activity, is central to the effects observed following exposure to Oxidative Stress /Nitrosative Stress (OS/ NS) caused by the accumulation of Reactive Oxygen Species/ Reactive Nitrogen Species (ROS/RNS). While ROS are generally considered prothrombotic, RNS tend to inhibit platelet function [20]. Although the modification of thiol proteins by ROS/RNS has been investigated [21], the specific impact on psPDI remains understudied. Nonetheless, the overarching concept of OS/NS mimicking cardiovascular pathologies is well-established [22-24]. Traditional methods for measuring PDI activity have included the reactivation of scrambled RNase (scRNase) and the reduction of insulin. However, a sensitive novel fluorescent assay had been published [25] which can be optimised to measure psPDI reductase activity after exposure to OS/NS. The present study adapts this technique to a 96-well microplate format to investigate the effects of various NO/oxygen reaction products on psPDI activity.
This study was undertaken with the following objectives:
i) To synthesise Di-E-GSSG
ii) To determine the suitability of Di-E-GSSG for quantifying
the activity of PDI from bovine liver, hereafter referred to as an
‘authentic PDI’
iii) To assess psPDI reductase activity using a platelet-based
assay
iv) To confirm the involvement of PDI in catalysing reduction on
the platelet surface through exposure of psPDI to established
thiol isomerase inhibitors; and
v) To examine the effect of oxido-nitrosative stress on psPDI
activity. Ultimately, this research aimed to identify disruptions
in psPDI activity induced by four oxidative/nitrosative stress
agents.
Materials and Reagents
All chemicals and materials were purchased from Sigma- Aldrich or Alexis Biochemicals.
Buffer solutions
Phosphate Buffered Saline (PBS) (100mM potassium phosphate, 2mM EDTA, pH 8.8) was used for Di-E-GSSG preparation. For column equilibration and all subsequent experiments, a Hepes Buffered Saline (HBS) (140mM NaCl, 2.7mM KCl, 5mM Glucose, 10mM HEPES, pH 7.3) was employed.
Preparation of Di-E-GSSG
Di-eosin-oxidised-glutathione was prepared by a modified method of Raturi & Mutus [25], reacting 100μM Glutathione Disulphide (GSSG) with a 10-fold molar excess of eosin isothiocyanate (1mM) in PBS (pH 8.8) overnight at room temperature. The reaction mixture was then purified using a disposable PD-10 desalting column (sephadex G-25 filtration medium) equilibrated with HBS (pH 7.3). Fractions were collected, and their fluorescence was monitored at 550nm (excitation 510nm) before and after treatment with 10 mM Dithiothreitol (DTT). Fractions exhibiting at least a 6-fold increase in fluorescence were pooled and stored at room temperature until use.
Assay to monitor DTT dose response
The DTT dose response assessed whether the Di-E-GSSG probe was reduced by DTT in a dose-dependent manner and if 5μM DTT, as used by Raturi & Mutus, interfered with the fluorescence signal. The probe was treated with serially diluted DTT concentrations (1μM, 10μM, 0.1mM, 1mM and 10mM) and a Milli-QH2O control in HBS (70μl HBS+20μl Di-E-GSSG mixture+10μl ascending DTT concentrations). A standard curve was generated from the resulting fluorescence data.
Isolation of washed human platelets from whole blood
Platelets were isolated following a previously established protocol [18]. After informed consent was secured in accordance with the declaration of Helsinki, whole blood was collected from a healthy volunteer into acid citrate dextrose to prevent coagulation, avoiding EDTA and heparin. Platelet-rich plasma was separated by two consecutive centrifugation steps (300g for 20min and 10min at RT), then acidified to pH 6.1-6.5 with 0.5M citric acid. To prevent aggregation, 1.5μM prostaglandin E1 and 2U/ml apyrase were added before a final centrifugation (1000g for 12min). The platelet pellet was resuspended in 1ml HBS and further purified by gel filtration using a Sephadex G-25 PD-10 desalting column equilibrated with HBS. The washed platelet suspension, manually counted, yielded a concentration of 1.45x109 platelets/ml. For the Di-E-GSSG assay, platelets were diluted to 290x106/ml, with serial dilutions performed to obtain concentrations from 290 to 18x106 platelets/ml.
Assay to measure PDI-dependent disulphide reduction (authentic PDI and psPDI) using Di-E-GSSG
In the presence of HBS containing 5μM DTT, Di-E-GSSG was incubated with serially diluted authentic PDI (10.9nM to 175nM) to assess a dose-related increase in fluorescence, monitored at 510nm excitation and 550nm emission. Any observed doseresponse indicated the probe’s utility for measuring authentic PDI activity. Subsequently, psPDI activity was monitored using the same protocol, substituting authentic PDI with varying concentrations of platelets (290 to 18x106/ml).
Assay to test the effects of oxidative-nitrosative stress agents on psPDI
To investigate the effects of oxidative and nitrosative stress on psPDI activity, platelets (290x106/ml) were incubated for 30min at 37 °C in HBS containing 5μM DTT, with varying concentrations (0.1-1000μM) of oxidative and nitrosative stress agents: Hydrogen Peroxide (H2 O2), Hypochlorous Acid (HOCl), S-Nitrosoglutathione (GSNO) and Di-Ethyl-Amine Nitric Oxide (DEANO). Subsequently, the Di-E-GSSG probe was added to measure psPDI inhibition. The aim was to determine if a concentration-dependent disturbance of psPDI activity occurred in response to these agents.
Three controls were implemented in this experiment:
i) Control 1: 50μl HBS with DTT+10μl Milli-Q H2O (for H2 O2,
HOCl, and GSNO) or NaOH (for DEANO) +20μl platelet
suspension (290x106/ml)
ii) Control 2: Replacement of platelet suspension in Control 1
with basic HBS (pH 7.3, without DTT); and
iii) Control 3: Further substitution of Milli-Q H2O or NaOH in
Control 1 with the stress agents.
Statistical Analysis
Data are presented as mean ± standard error of the mean and analysed using Microsoft Excel. Statistical significance was determined by a two-tailed, two-sample student’s t-test assuming equal variance, with p≤0.05 considered significant.
Result
Preparation of Di-E-GSSG
In an end-point assay, the fluorescence of each eluted fraction was measured (Ex 510nm/Em 550nm) and a fold-increase ratio, corrected against pre-DTT treatment baseline, was calculated. Fractions 3 to 7, exhibiting an intense pink band, showed at least a 6-fold increase in fluorescence upon 10mM DTT treatment (Figure 1), attributed to Eosin-Glutathione (E-GSH) release. These pooled fractions (approximately 100μl) were selected as the probe. While this fold increase was lower than previously described by Raturi & Mutus [25], the DTT treatment still elicited a usable response, indicating Di-E-GSSG’s viability for determining authentic PDI and cell surface thiol levels. Discrepancies in fold increase may be due to differences in experimental conditions, such as HBS concentration during elution, and the absence of E-GSH quantification.
Figure 1:The fold-increase ratio observed for each eluted fraction before and after treatment with 10mM DTT.

Assay to monitor DTT dose response
For the DTT dose response experiment, the reduction of Di- E-GSSG was assessed with variable DTT concentrations (1μM to 10mM) in the absence of PDI, and a standard curve was constructed (Figure 2). No increase in fluorescence signal was observed at minimal DTT concentrations (0 to 10μM). However, fluorescence signals increased dose-dependently at DTT concentrations greater than 10μM, indicating interference (reduction of the Di-E-GSSG disulphide). Conversely, at minimal concentrations, Di-E-GSSG demonstrated resistance to DTT-mediated reduction. Specifically, interpolation showed Di-E-GSSG to be resistant to 5μM DTT, justifying the use of this concentration for subsequent experiments, as it was deemed sufficient for a single enzymatic turnover without causing appreciable blank rates.
Figure 2:DTT dose response in Di-E-GSSG assay.

Reductase activity of authentic PDI
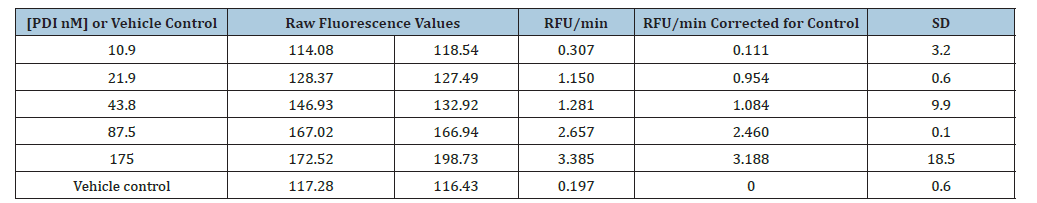
The reductase activity of authentic PDI and psPDI was successfully assessed using a high-throughput assay adapted from the Di-E-GSSG method. In the presence of 5μM DTT, authentic PDI exhibited concentration-dependent reductase activity, evidenced by an increase in Relative Fluorescence Units per minute (RFU/ min) and RFU/min corrected for control (Table 1). RFU values were derived from the mean of duplicate raw fluorescence measurements, corrected for background fluorescence. Subsequently, psPDI activity exhibited a reproducible dose-response relationship (Figure 3) when monitored continuously over time in the presence of 5μM DTT. In control assays without platelet suspension, no increase in fluorescence was observed in the Di-E-GSSG assay. Conversely, the addition of platelet suspension resulted in a stoichiometric and concentration-dependent increase in fluorescence, indicating that psPDI triggered the reduction of Di-E-GSSG. While the exact Di-EGSSG concentration was unknown, the stoichiometric generation of E-GSH confirmed psPDI’s involvement. Authentic PDI induced an immediate fluorescence increase without a lag phase; however, a brief lag phase was observed in psPDI reductase activity, which was eliminated by pre-warming the plate for 30min. Furthermore, increasing psPDI concentrations in the presence of 5μM DTT yielded increasing fluorescence signals, providing evidence that this DTT concentration is sufficient for enzymatic turnover and continuous determination of PDI reductase activity. This suggests the Di-E-GSSG probe’s disulphide bond is accessible to PDI activesite dithiols with 5μM DTT, without appreciable background blank rates. The observed dose-response relationships (Table 1 & Figure 3) strongly indicate that the activity generated by both authentic PDI and psPDI can be attributed to thiol isomerase activity, specifically PDI.
Figure 3:Dose-dependent response of psPDI reductase activity in the Di-E-GSSG assay. Vmax values (expressed as RFU/min), derived from three independent experiments (n=3), are presented as mean ± SEM.

Table 1:The 20min kinetic data for authentic PDI-reductase reactions using Di-E-GSSG assay.
Introducing chemical inhibitors of PDI into psPDI reductase assay
The activity observed on the platelet surface was inhibited by four established thiol isomerase inhibitors: Rutin (100μM), PAO (100μM), DTNB (100μM) and bacitracin (1mM) (Figure 4). A consistent, though variable, degree of inhibition was observed across all inhibitors, none resulting in complete 0% inhibition, thus validating the utility of these inhibition experiments... This consistent inhibition by PDI chemical inhibitors suggests a role for PDI and/or surface thiol isomerases on platelets in generating the activity observed in Figure 3. The mean fluorescence values for bacitracin, DTNB, PAO and rutin were all derived from triplicate experiments (n=3).
Figure 4:Inhibition of psPDI reductase activity by rutin, PAO, DTNB and bacitracin. Data are presented as mean ± SEM from three independent experiments.

The effects of oxidative and nitrosative stress agents on psPDI
Figure 5:Dose-dependent inhibition of psPDI activity following exposure to H2O2, HOCl, GSNO and DEANO (oxidative and nitrosative stress agents), determined using the Di-E-GSSG assay. Data are presented as mean ± SEM from triplicate experiments.

A concentration-dependent inhibition of psPDI activity was observed in response to the oxidative and nitrosative chemical stress agents GSNO, DEANO, H2 O2 and HOCl, with dose-response curves generated (Figure 5). Control experiments, performed in the absence of these agents, exhibited normal PDI activity, confirming the reliability of these results. The percentages of inhibition at 0.1μM, 1μM, 10μM, 0.1mM and 1mM for each stress agent were as follows: H2O2-16%, 33%, 93%, 100% and 100%; HOCl -51%, 92%, 78%, 98% and 100%; GSNO -0%, 6%, 23%, 84% and 97%; DEANO -7%, 10%, 31%, 61% and 86%, respectively. An anomalous result at 1 1μM HOCl, which deviated from the expected dose-response, was disregarded in the interpretation due to potential pipetting errors, though efforts were made to maintain consistent pipetting throughout the experiments.
H2O2 and HOCl demonstrated a more rapid and potent inhibition of psPDI activity compared to GSNO and DEANO. As shown in Figure 5, H2O2 and HOCl significantly inhibited psPDI at lower concentrations (0.1μM, 1μM and 10μM). In contrast, at these same concentrations, inhibition by GSNO and DEANO was less pronounced (0% to 23% for GSNO and 7% to 31% for DEANO). This dose-related disturbance in psPDI activity is likely attributable to thiol modification.
Discussion
Historically, the study of psPDI’s reductive role was challenging due to the limitations of traditional assays like scRNase and insulin turbidity, which suffered from significant non-enzymatic blank rates, hindered accurate estimation of initial rates and lacked precision with crude samples [26]. These issues were addressed by the development of the sensitive, fluorescent self-quenched probe, Di-E-GSSG, by Raturi & Mutus [25]. Fluorescent probes like Di-E-GSSG offer significantly enhanced sensitivity, for example, a 1000-fold increase compared to probes like diabz-GSSG. In present study, the Di-E-GSSG assay was successfully adapted for high-throughput investigation of psPDI activity. A stoichiometric, concentration-dependent increase in fluorescence was observed, indicating that the detected activity was PDI-related. Furthermore, the contribution of psPDI to platelet surface activity was confirmed using established thiol isomerase inhibitors. The Di-E-GSSG assay was also optimised to continuously monitor the effects of OS/NS on PDI reductase activity in platelets. Except for a single anomalous result at 1μM HOCl, a consistent dose-dependent inhibition of psPDI reductase activity was observed when various OS/NS agents were introduced, suggesting that exposure to an OS/NS environment can elicit a concentration-dependent enzyme inhibitory mechanism. The probe’s design allows for continuous monitoring of enzyme activity, including the effects of oxidative and nitrosative stress agents.
The synthesis of Di-E-GSSG involves covalently conjugating two eosin moieties with the two free amino acid groups (i.e., glutamate portion) at the N-termini of GSSG. This reaction yields Di-E-GSSG without by-products. Unlike scRNase and insulin turbidity assays, this purification process is straightforward, requiring only a single step to achieve FSQ of the eosin moieties in the probe. The synthesis process can be straightforward and cost-effective, requiring only a single purification step. Similarly, FSQ peptides have been previously utilised to quantitatively investigate PDI activity [27,28]. Moreover, the concept of FSQ has also been applied in studies of protein dimerization [29], protease activity [30] and protein folding [31]. However, the synthesis of such FSQ peptides often requires a peptide synthesiser and expensive chemicals, which can limit their accessibility for measuring PDI activity [28]. The use of Di-E-GSSG circumvents these limitations, as it is a simple and cost-effective probe.
For FSQ to occur effectively, the eosin molecules in Di-E-GSSG must interact intermolecularly at a distance of no more than approximately 100nm. Effective FSQ is crucial for the probe’s functionality and accurate fluorescence generation upon reduction. To account for this phenomenon, the proximity between amino benzoyl residues in diabz-GSSG was previously determined to be as close as 73nm using molecular dynamic simulations [26]. In the present study, the distance between the two eosins in Di-E-GSSG was not tested using molecular dynamic simulations. Instead, the effect of removing the distance constraint between the two eosin molecules was assessed by adding DTT to abolish the FSQ by spatially separating the two E-GSH molecules. The fluorescence generation after DTT-treatment provided evidence that the eosins in Di-E-GSSG had been adequately self-quenched when the probe was synthesised. Without adequate self-quenching, the eosin molecules would have remained relatively non-fluorescent even after DTT treatment.
Di-E-GSSG was also chosen for its high sensitivity in monitoring PDI reductase activity, exhibiting a 1000-fold increase in sensitivity compared to diabz-GSSG [25]. This enhanced sensitivity makes it a preferred probe. Additionally, PDI demonstrates a relatively higher affinity for Di-E-GSSG than for non-fluorescent GSSG, ensuring low GSSG concentrations do not significantly alter the rate of E-GSH generation. Probes can be designed to have a higher affinity for the target enzyme, ensuring that low substrate concentrations do not significantly alter reaction rates and potentially leading to faster reaction rates due to structural characteristics. This higher affinity may be attributed to hydrophobic interactions near the active site between eosin’s aromatic rings and PDI, supported by hydrophobic regions surrounding active-site dithiols [32]. Alternatively, the two large eosin moieties may distort the disulphide dihedral angle of GSSG, making its disulphide bonds more susceptible to nucleophilic attack by PDI’s active site dithiols, leading to a faster reaction rate [25]. While it is hypothesised that the structural changes in Di-E-GSSG contribute to its higher affinity for PDI, confirming this through structural analysis, such as mass spectrometry to investigate the disulphide dihedral angle, was beyond the scope of the study.
While Di-E-GSSG exhibits low sensitivity for single enzymatic turnover in the absence of DTT, a minimal concentration of DTT was included in this study. DTT is crucial for maintaining and stabilising PDI’s thiol-disulphide exchange activity by restoring activity lost to active site oxidation [25]. External reducing agents (e.g., DTT) are crucial for continuous enzymatic turnover, especially when monitoring reduced states and preventing activity loss due to oxidation. After substrate oxidation, the PDI active site transitions from an oxidised disulphide to a reduced dithiol state, requiring re-oxidation via intra and intermolecular dithiol/disulphide relays for continued catalysis [33]. These redox cycles serve as signalling elements, influencing processes like gene expression [34]. Therefore, DTT was added to the Di-GSSG assays to facilitate continuous PDI turnover to its reduced state. The necessity of DTT was established by Raturi & Mutus [25], who reported that 5μM DTT is required for one enzymatic turnover, as PDI did not turn over to a reduced state without it. In this study, a DTT dose-response also confirmed that 5μM DTT was appropriate and did not cause appreciable blank rates, proving useful for continuously monitoring nanomolar amounts of authentic PDI and psPDI. Di-E-GSSG remained resistant to 5μM DTT but was susceptible to reduction by reduced PDI, further demonstrating its utility. Therefore, 5μM DTT was deemed convenient and appropriate for facilitating continuous PDI turnover in the Di-E-GSSG assays.
To confirm that the activity generated on a platelet surface can be attributed to PDI-like enzymes, four PDI chemical inhibitors (bacitracin, rutin, DTNB and PAO) were utilised as control measures in this study. One limitation was the absence of a specific monoclonal anti-PDI antibody to verify that the observed activity was specifically related to prototypic PDI. A key challenge is the non-specificity of some probes and chemical inhibitors, which may react with a broader population of thiol isomerases rather than being exclusively specific to the target enzyme (e.g., PDI), leading to potential ambiguity in attributing observed activity. Among the chemical PDI inhibitors used, bacitracin has been widely employed, with an IC50 of approximately 150-200mM [35,36]. However, Flaumenhaf [2] screened a small library of approximately 5000- compounds and identified rutin as a selective and most potent PDI inhibitor, with an IC50 of about 6mM as reported by Jasuja et al. [9]. Despite bacitracin’s common use as a specific PDI inhibitor [13,14,35], its inhibitory action is scarcely evidenced, and it’s in vitro mechanisms remain unclear. PAO and DTNB, also used as PDI inhibitors, similarly block the activity of free thiols [37]. In general, all these chemical inhibitors inhibit thiol isomerases, not just prototype PDI specifically, making them non-specific PDI inhibitors. Since PDI accounts for 40% of the overall surface thiols [15], a significant amount of the inhibition can be attributed to prototypic PDI. Nonetheless, these chemical inhibitors impede free thiols and are non-specific to PDI because they also block other thiol isomerases [38]. Karala and Ruddock reported that 1mM bacitracin did not inhibit PDI oxidation and isomerisation activity [38]. However, consistent with the present study, bacitracin demonstrated a partial inhibitory effect on PDI-reductive activity. Observed inhibitory effects on PDI-reductive activity were as follows: Rutin (14% inhibition), PAO (55% inhibition), bacitracin (80% inhibition) and DTNB (100% inhibition). The primary objective of these inhibition experiments was to demonstrate that PDI-like activity on the platelet surface could be partially or totally blocked, a goal achieved by the application of these four chemical inhibitors.
In a subsequent experiment, psPDI was challenged with oxidative stress and nitrosative stress by exposure to four stress agents: Two redox derivatives of nitric oxide -a nitrosonium cation (NO+) (i.e., GSNO) and a NO· radical (i.e., DEANO) -and two OS stress agents -H2O2 and HOCl. The accumulation of ROS/RNS is known to perturb the delicacy of the redox state, leading to OS/NS [39], which consequently alters cellular processes [40], causing events which may play a critical role in the modification of thiols in proteins [41]. The OS environment often co-localises with NS and vice versa. In vivo alteration of NO homeostasis can lead to a rapid conversion into secondary ROS and RNS, implicated in various pathological processes in diseases such as cancer, cystic fibrosis, aging, neurodegenerative and metabolic disorders [42-48]. In biomedical free radical literature, ROS and RNS are used as a collective term for both oxygen/nitrogen-centred radicals such as O2-, OH. and NO. as well as non-radical derivatives of oxygen/nitrogen such as H2O2, HOCl, GSNO and DEANO. These nonradical derivatives are easily converted into free radicals, affecting protein function. For example, many biological systems involve the transferral of a nitrosonium cation (NO+) to a protein S -group from an NO donor, thereby affecting protein function [49]. Accordingly, GSNO can donate NO+ to PDI and inhibit PDI function via S-glutathionylation. NO can directly alter protein biosynthesis through posttranslational modifications, namely nitration or S-nitrosylation or can indirectly alter protein function by interacting with oxygen, thiols, superoxide and heavy metals, all causing protein S-glutathionylation [50]. The ROS/RNS-linked pathogenesis of thrombosis and their involvement in signalling cascades is a recognised phenomenon [40], suggesting the likelihood of psPDI being involved in this process. A review paper indicated that potential targets for ROS/RNS are signalling molecules, such as PDI, that contain critical cysteine residues required to retain their activity [51]. Thus, PDI can be modified by ROS/RNS via thiol chemistry. These findings align with the present study, where OS/NS induced by the four redox derivatives caused dose-dependent inhibition of psPDI activity. Responses like OS/NS can also be triggered by other stress factors such as heat, osmotic shock, and ultraviolet radiation. In the experiments of present study, the sample plate was exposed to 37 °C; therefore, heat stress could have interfered with OS/NS, potentially causing an overemphasised inhibition of psPDI activity. However, exposure to heat to cause such a stress situation was unlikely, as the sample plate was exposed to heat for a very minimal time.
Furthermore, RNS/ROS can chemically modify critical thiols in PDI to form S-nitrosothiols. In theory, this modification can either disrupt or regulate PDI activity [51]. However, the findings of this study have shown that the activity is disrupted dose-dependently. This disruption could be due to a diverse array of systems that are difficult to pinpoint with the results of this study. Additionally, OS appeared to be more rapid than NS. This could be because OS occurs directly, unlike NS. Moreover, ROS are known to be more potent and unstable than NOS, therefore more likely to cause damage and protein dysfunction. H2O2 is a recognised potent thiol oxidant capable of oxidising reduced PDI. In the presence of cystine, treatment of E. coli with H2O2 has previously been shown to increase the level of extracellular GSSG, thereby decreasing the ratio of reduced GSH to oxidised GSH (GSH/GSSG) both by 5-fold, respectively [52]. PDI activity is attenuated at lower ratios of GSH/ GSSG, and this may explain the inhibition of psPDI activity in the Di- E-GSSG assay when exposed to increasing concentrations of H2O2. In contrast to the potential of GSSG in the oxidation of reduced PDI, H2O2 has been found to be relatively slower in oxidising reduced PDI, with half-lives estimated at 2min and 20min, respectively. The difference in the potencies and rapid onset observed in this study requires an in-depth search in the free radical literature, in which many unanswered questions remain as the source material for further investigations into the complete nature of redox signalling.
The Impact and Relevance of This Research
The foundational understanding of platelet disulphide isomerase’s role in thrombosis emerged from early studies on psPDI, which first indicated its link to thrombosis by demonstrating its release from activated platelets [53]. Subsequent investigations into cell surface regulatory mechanisms, particularly those involving redox chemistry, further underscored the significance of studying various effects on psPDI [14,19,54,55]. Among the factors demonstrated to influence platelet function, oxidative stress and nitrosative stress had been notable. These are induced by critical molecular components known to initiate and advance vascular diseases [22-24,56-58], including hypertension and atherosclerosis. While OS/NS-induced platelet dysfunction has been extensively elucidated, the specific impact of OS/NS on psPDI reductase activity itself had not been thoroughly investigated. This research specifically addresses this critical question.
E-GSH/Di-E-GSSG as Thiol Redox Probes
The intracellular ratio of GSH/GSSG serves as a widely accepted measure for assessing cellular toxicity and OS/NS stress [59]. Numerous studies have demonstrated that the catalytic efficiency of PDI is intrinsically linked to the GSH/GSSG ratio, with a more oxidising environment significantly attenuating its isomerase activity [60,61]. These earlier investigations typically involved blocking the activity of reduced PDI with alkylating agents, followed by monitoring its activity after incubation in various redox environments using GSH/GSSG as an indicator [60,61]. Similarly, changes in the reduction of Di-E-GSSG by PDI were monitored to probe the redox state of PDI under variable GSH/GSSG ratios. While it was theoretically possible to estimate the redox state of PDI through the stoichiometric generation of E-GSH monitored after PDI-mediated reduction of Di-E-GSSG, the utility of E-GSH/ Di-E-GSSG as thiol redox probes for investigating the active-site environments of PDI was not explored in the present study.
Limitations of this Research
The primary focus of this project was to elucidate the reductive potential of psPDI and how this potential is modulated under stress conditions such as OS/NS. Extending this investigation to include thiol redox probes E-GSH and Di-E-GSSG would have been highly relevant for a comprehensive understanding of the effects of OS/NS on the psPDI active site redox environment, beyond just its reductase activity. It is also pertinent to note that, in addition to oxygen/NO-induced cellular toxicity, the ratio of GSH/GSSG significantly influences protein folding kinetics [25]. Antioxidant GSH, a tripeptide (L-gamma-glutamyl-Lcysteinyl-glycine), is recognised as the most ubiquitous and abundant non-protein thiol within cells, participating in a multitude of detoxification reactions. In vivo studies have further demonstrated its presence outside cells, where it contributes to protection against OS/NS [62]. A primary limitation of this research stems from the non-specificity of the experimental tools employed. The Di-E-GSSG assay, used to assess PDI reductase activity, and the chemical inhibitors, including rutin, PAO, DTNB, and bacitracin, are not exclusively specific to PDI. Consequently, the observed functional activities (attributable to authentic PDI and psPDI) and inhibited responses (related to thiol isomerase inhibitors and OS/NS may partially derive from a broader population of surface thiol isomerases. While PDI is a prominent and well-characterised member of this family [19], accounting for approximately 40% of total thiols [15], the use of PDI enzyme activity-specific function-blocking antibodies would have provided greater mechanistic resolution. A further intellectual challenge lies in the current understanding of PDI’s localisation. The precise mechanisms by which PDI escapes the ER and appears in non-ER locales, such as the cell surface, are not yet empirically defined. Therefore, conclusions regarding the function of psPDI, despite the pertinence of our findings, must be interpreted with caution [63].
Despite retaining the C-terminal KDEL retention signal, PDI has been consistently documented in extracellular spaces [4]. The exact processes facilitating PDI’s export while maintaining its retention signal remain unclear [19]. However, several mechanisms have been speculated, including: KDEL saturation machinery, sequence cleavage, structural complexing with other macromolecules, leakage from a salvage pathway designed to recycle proteins back to the ER [63]. Once secreted, PDI is understood to bind to the cellular surface through electrostatically induced interactions, as demonstrated in hepatocytes where microtubular activity mediates PDI secretion and electrostatic forces contribute to its attachment to the plasma membrane. Overall, while the findings of this study are pertinent to PDI, these inherent methodological and conceptual considerations should be considered when interpreting the results [64].
Future Directions
The demonstrated inhibitory response of psPDI to OS/NS agents presents a promising avenue for advancing research into platelet-pathway driven thrombosis and atherosclerosis. Future investigations could extend the current findings by incorporating platelet adhesion assays to comprehensively evaluate the functional consequences of OS/NS on platelet activity in vitro and in vivo. Furthermore, the Di-E-GSSG assay, validated in this study, exhibits significant potential for broader application. It could be extended to various cell lines that express PDI on their surface, allowing for a more widespread exploration of OS/NS effects on PDI activity across different cellular contexts. Similarly, optimising the Di-E-GSSG assay to monitor other members of the PDI family would provide valuable insights into their respective roles and susceptibilities to redox modulation. From a therapeutic perspective, integrating various antioxidants into future studies to normalise PDI activity under OS/NS conditions could establish a rational basis for developing novel therapeutic strategies in the field of thrombosis research. This approach could potentially mitigate the pro-thrombotic effects associated with OS/NS-induced psPDI dysfunction.
Conclusion
Investigating psPDI activity has historically been challenging, largely due to the absence of sufficiently sensitive and robust probes. This research is part of a demonstration of an advancement in this area through the successful application of the Di-E-GSSG assay. This probe proved invaluable, enabling a rapid, sensitive, and highly applicable assay for studying psPDI within the platelet samples employed in this study. The utility of the Di-EGSSG assay extends beyond the scope of this research, offering considerable potential for the high-throughput screening of PDI chemical inhibitors. Furthermore, its adaptability allows for straightforward optimisation for fluorescence plate readers, which would significantly enhance its capacity to precisely investigate the intricate effects of various redox buffers and ROS/RNS on psPDI activity. This research contribution provides a way forward for future research into PDI’s role in health and disease.
References
- Goldberger RF, Epstein CJ, Anfinsen CB (1963) Acceleration of reactivation of reduced bovine pancreatic ribonuclease by a microsomal system from rat liver. J Biol Chem 238: 628-635.
- Flaumenhaft R (2013) Protein disulfide isomerase as an antithrombotic target. Trends Cardiovasc Med 23(7): 264-268.
- Bonney W, Gilbert HF (2004) Protein disulfide isomerase. Biochim Biophys Acta 1699(1-2): 35-44.
- Turano C, Coppari S, Altieri F, Ferraro A (2002) Proteins of the PDI family: Unpredicted non-ER locations and functions. J Cell Physiol 193(2): 154-163.
- Abell BA, Brown DT (1993) Sindbis virus membrane fusion is mediated by reduction of glycoprotein disulfide bridges at the cell surface. J Virol 67(9): 5496-5501.
- Ryser HJ, Levy EM, Mandel R, Di-Sciullo GJ (1994) Inhibition of human immunodeficiency virus infection by agents that interfere with thiol-disulfide interchange upon virus-receptor interaction. Proc Natl Acad Sci USA 91(10): 4559-4563.
- Lawrence DA, Song R, Weber P (1996) Surface thiols of human lymphocytes and their changes after in vitro and in vivo J Leukoc Biol 60(5): 611-618.
- Jaehyung C, Daniel RK, Lin L, Huang M, Barbara CF, et al. (2012) Protein disulfide isomerase capture during thrombus formation in vivo depends on the presence of β3 Blood 120(3): 647-655.
- Jasuja R, Freda HP, Daniel RK, Sarah HK, Bruce F, et al. (2012) Protein disulfide isomerase inhibitors constitute a new class of antithrombotic agents. J Clin Invest 122(6): 2104-2113.
- Kim K, Hahm E, Jing L, Ronald G, Lisa MH, et al. (2013) Platelet protein disulfide isomerase is required for thrombus formation but not for hemostasis in mice. Blood 122(6): 1052-1061.
- Naguleswaran A, Christophe G, Nathalie V, Sonda S, Jenoe P, et al. (2005) Neospora caninum protein disulfide isomerase is involved in tachyzoite-host cell interaction. Int J Parasitol 35(13): 1459-1472.
- Conant CG, Stephens RS (2007) Chlamydia attachment to mammalian cells requires protein disulfide isomerase. Cell Microbiol 9(1): 222-232.
- Essex DW, Chen K, Swiatkowska M (1995) Localization of protein disulfide isomerase to the external surface of the platelet plasma membrane. Blood 86(6): 2168-2173.
- Lahav J, Oded H, Michael JB, Jacob L, Beate EK, et al. (2002) Sustained integrin ligation involves extracellular free sulfhydryls and enzymatically catalyzed disulfide exchange. Blood 100(7): 2472-2478.
- Jiang XM, Fitzgerald M, Grant CM, Hogg PJ (1999) Redox control of exofacial protein thiols/disulfides by protein disulfide isomerase. J Biol Chem 274(4): 2416-2423.
- Chen K, Detwiler TC, Essex DW (1995) Characterization of protein disulphide isomerase released from activated platelets. Br J Haematol 90(2): 425-431.
- Root P, Sliskovic I, Mutus B (2004) Platelet cell-surface protein disulphide-isomerase mediated S-nitrosoglutathione consumption. Biochem J 382(Pt 2): 575-580.
- Bell SE, Shah CM, Gordge MP (2007) Protein disulfide-isomerase mediates delivery of nitric oxide redox derivatives into platelets. Biochem J 403(2): 283-288.
- Gordge MP, Xiao F (2010) S-nitrosothiols as selective antithrombotic agents-possible mechanisms. Br J Pharmacol 159(8): 1572-1580.
- Freedman JE (2008) Oxidative stress and platelets. Arterioscler Thromb Vasc Biol 28(3): s11-s16.
- Hurd TR, Prime TA, Harbour ME, Lilley KS, Murphy MP (2007) Detection of reactive oxygen species-sensitive thiol proteins by redox difference gel electrophoresis: Implications for mitochondrial redox signaling. J Biol Chem 282(30): 22040-22051.
- Giugliano D, Ceriello A, Paolisso G (1995) Diabetes mellitus, hypertension and cardiovascular disease: Which role for oxidative stress? Metabolism 44(3): 363-368.
- Griendling KK, Fitz-Gerald GA (2003) Oxidative stress and cardiovascular injury: Part I: Basic mechanisms and in vivo monitoring of ROS. Circulation 108(16): 1912-1916.
- Baeyer H, Hopfenmüller W, Riedel E, Affeld K (2003) Atherosclerosis: Current concepts of pathophysiology and pharmacological intervention based on trial outcomes. Clin Nephrol 60(Suppl 1): S31-S48.
- Raturi A, Mutus B (2007) Characterization of redox state and reductase activity of protein disulfide isomerase under different redox environments using a sensitive fluorescent assay. Free Radic Biol Med 43(1): 62-70.
- Raturi A, Vacratsis PO, Dana S, Lee L, Mutus B (2005) A direct, continuous, sensitive assay for protein disulphide-isomerase based on fluorescence self-quenching. Biochem J 391(Pt 2): 351-357.
- Westphal V, Spetzler JC, Meldal M, Christensen U, Winther JR (1998) Kinetic analysis of the mechanism and specificity of protein-disulfide isomerase using fluorescence-quenched peptides. J Biol Chem 273(39): 24992-24999.
- Camilla, Hilaire PM, Jakob RW (2004) Fluorometric polyethyleneglycol-peptide hybrid substrates for quantitative assay of protein disulfide isomerase. Anal Biochem 333(1): 148-155.
- Wendt H, Berger C, Baici A, Thomas RM, Bosshard HR (1995) Kinetics of folding of leucine zipper domains. Biochemistry 34(12): 4097-4107.
- Jones LJ, Upson RH, Haugland RP, Zhou M, Richard PH, et al. (1997) Quenched BODIPY dye-labeled casein substrates for the assay of protease activity by direct fluorescence measurement. Anal Biochem 251(2): 144-152.
- Zhuang X, Ha T, Kim HD, Labeit S, Chu S, et al. (2000) Fluorescence quenching: A tool for single-molecule protein-folding study. Proc Natl Acad Sci USA 97(26): 14241-14244.
- Lundström L, Krause G, Holmgren A (1992) A Pro to his mutation in active site of thioredoxin increases its disulfide-isomerase activity 10-fold. New refolding systems for reduced or randomly oxidized ribonuclease. J Biol Chem 267(13): 9047-9052.
- Riemer J, Bulleid N, Herrmann JM (2009) Disulfide formation in the ER and mitochondria: Two solutions to a common process. Science 324(5932): 1284-1287.
- Pfeilschifter J, Eberhardt W, Huwiler A (2003) Nitric oxide and mechanisms of redox signaling. J Am Soc Nephrol 14(8-supply 3): S237-S240.
- Mandel R, Ryser HJ, Ghani F, Wu M, Peak D (1993) Inhibition of a reductive function of the plasma membrane by bacitracin and antibodies against protein disulfide-isomerase. Proc Natl Acad Sci USA 90(9): 4112-4116.
- Lovat EP, Marco C, Jane LA, Shaun M, Vittoria P, et al. (2008) Increasing melanoma cell death using inhibitors of protein disulfide isomerases to abrogate survival responses to endoplasmic reticulum stress. Cancer Res 68(13): 5363-5369.
- Manickam N, Ahmad SS, Essex DW (2011) Vicinal thiols are required for activation of the αIIbβ3 platelet integrin. J Thromb Haemost 9(6): 1207-1215.
- Karala AR, Ruddock LW (2010) Bacitracin is not a specific inhibitor of protein disulfide isomerase. FEBS J 277(11): 2454-2462.
- Lisa AR, Jeffery SI, Espey GM, Thomas DD, Wink DA, et al. (2005) Nitric oxide regulates angiogenesis through a functional switch involving thrombospondin-1. Proc Natl Acad Sci USA 102(37): 13147-13152.
- Valko M, Dieter L, Jan M, Telser J, Mazur M, et al. (2007) Free radicals and antioxidants in normal physiological functions and human disease. Int J Biochem Cell Biol 39(1): 44-84.
- Biswas S, Chida AS, Rahman I (2006) Redox modifications of protein-thiols: Emerging roles in cell signaling. Biochem Pharmacol 71(5): 551-564.
- Halliwell B (1993) Free radicals and vascular disease: How much do we know? BMJ 307(6909): 885-886.
- Wiseman H, Halliwell B (1996) Damage to DNA by reactive oxygen and nitrogen species: Role in inflammatory disease and progression to cancer. Biochem J 313(Pt 1): 17-29.
- Lee YJ, Galoforo SS, Davis BH, Sim JE, Corry PM, et al. (1998) Glucose deprivation-induced cytotoxicity and alterations in mitogen-activated protein kinase activation are mediated by oxidative stress in multidrug-resistant human breast carcinoma cells. J Biol Chem 273(9): 5294-5299.
- Ilic TV, Jovanovic M, Jovicic A, Tomovic M (1999) Oxidative stress indicators are elevated in de novo Parkinsons disease patients. Funct Neurol 14(3): 141-147.
- Evans JL, Goldfine ID, Betty AM, Gerold GM (2003) Are oxidative stress-activated signaling pathways mediators of insulin resistance and beta-cell dysfunction? Diabetes 52(1): 1-8.
- Tieu K, Ischiropoulos H, Przedborski S (2003) Nitric oxide and reactive oxygen species in Parkinsons disease. IUBMB Life 55(6): 329-335.
- Townsend D, Tew K (2003) Cancer drugs, genetic variation and the glutathione-S-transferase gene family. Am J Pharmacogenomics 3(3): 157-172.
- Mohr S, Hallak H, Boitte A, Lapetina EG, Brüne B (1999) Nitric oxide-induced S-glutathionylation and inactivation of glyceraldehyde-3-phosphate dehydrogenase. J Biol Chem 274(14): 9427-9430.
- Uys JD, Xiong Y, Townsend DM (2011) Nitrosative stress-induced S-glutathionylation of protein disulfide isomerase. Methods Enzymol 490: 321-332.
- Forman HJ, Fukuto JM, Torres M (2004) Redox signaling: Thiol chemistry defines which reactive oxygen and nitrogen species can act as second messengers. Am J Physiol Cell Physiol 287(2): C246-C256.
- Smirnova GV, Muzyka NG, Oktyabrsky ON (2005) Effects of cystine and hydrogen peroxide on glutathione status and expression of antioxidant genes in Escherichia coli. Biochemistry (Mosc) 70(8): 926-934.
- Chen K, Lin Y, Detwiler TC (1992) Protein disulfide isomerase activity is released by activated platelets. Blood 79(9): 2226-2228.
- Burgess JK, Suter C, Dudman PN, Szöllösi J, Hogg PJ, et al. (2000) Physical proximity and functional association of glycoprotein 1balpha and protein-disulfide isomerase on the platelet plasma membrane. J Biol Chem 275(13): 9758-9766.
- Robinson A, Neill SO, Kiernan A, Donoghue NO, Moran N (2006) Bacitracin reveals a role for multiple thiol isomerases in platelet function. Br J Haematol 132(3): 339-348.
- Harrison DG, Cai H (2003) Endothelial control of vasomotion and nitric oxide production. Cardiol Clin 21(3): 289-302.
- Harrison D, Griendling KK, Landmesser U, Hornig B, Drexler H (2003) Role of oxidative stress in atherosclerosis. Am J Cardiol 91(3A): 7A-11A.
- Weiss N, Heydrick SJ, Postea O, Keller C, Loscalzo J, et al. (2003) Influence of hyper homocysteine mia on the cellular redox state--impact on homocysteine-induced endothelial dysfunction. Clin Chem Lab Med 41(11): 1455-1461.
- Pastore A, Locatelli M, Gaeta LM, Russo AL, G Tozzi, et al. (2001) Determination of blood total, reduced and oxidized glutathione in pediatric subjects. Clin Chem 47(8): 1467-1469.
- Lundström J, Holmgren A (1993) Determination of the reduction-oxidation potential of the thioredoxin-like domains of protein disulfide-isomerase from the equilibrium with glutathione and thioredoxin. Biochemistry 32(26): 6649-6655.
- Hawkin HC, Freed RB (1991) Redox properties and cross-linking of the dithiol/disulphide active sites of mammalian protein disulphide-isomerase. Biochem J 275(Pt 2): 341-348.
- Cho IH, Kim D, Lee JK, Han PL, Kim KS, et al. (2003) Protective effects of extracellular glutathione against Zn2+-induced cell death in vitro and in vivo. J Neurosci Res 74(5): 736-743.
- Johnson S, Michalak M, Opas M, Eggleton P (2001) The ins and outs of calreticulin: From the ER lumen to the extracellular space. Trends Cell Biol 11(3): 122-129.
- Terada K, Noiva R, Jauregui HO, Stockert RJ, Schilsky ML, et al. (1995) Secretion, surface localization, turnover and steady state expression of protein disulfide isomerase in rat hepatocytes. J Biol Chem 270(35): 20410-20416.
© 2025 Usman Ali. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






