- Submissions

Full Text
Intervention in Obesity & Diabetes
Innovations in Insulin Delivery for Diabetes Management
Borish Loushambam1 and Sivakumar Vijayaraghavalu2*
1Department of Life Sciences (Zoology), Manipur University (A Central University), India
2Department of Medical Science & Technology, Indian Institute of Technology Madras, India
*Corresponding author:Sivakumar Vijayaraghavalu, Department of Medical Science & Technology, Indian Institute of Technology Madras, Chennai 600036, TN, India
Submission:January 29, 2026;Published: March 10, 2026

ISSN 2578-0263Volume7 Issue2
Abstract
Diabetes mellitus is also one of the most pressing health problems in the world today, with both type 1 and type 2 diabetes increasing morbidity and other chronic complications. Although insulin therapy is lifesaving, existing delivery methods, including multiple daily injections, insulin pens, etc., have limitations in terms of invasiveness, poor adherence, and poor glycaemic control. Such restrictions have necessitated rapid innovation in insulin-delivery technologies to enhance physiological mimicry and improve care for people. The current mini-review summarizes insulin delivery system development, focusing on the evolution from the earliest injection-based devices to modern Automatic Insulin Delivery (AID) and hybrid closed-loop platforms. Most recent trends in control systems based on algorithms, wearable and patch-capable devices, glucose-responsive (innovative) insulin formulations, and alternative non-invasive delivery methods, such as oral, transdermal, and inhalable medications, are critically addressed. Special focus is being made on carriers enabled by nanotechnology, biocompatible materials, and integrated biosensors, which make it stable, responsive, and capable of sustained insulin release. Existing clinical evidence, regulatory do-not-resuscitate orders, and practical issues related to safety, usability, cost, and fair access are also discussed. Lastly, the novel trends combining AI, individualized dosing plans, and intelligent biomaterials are highlighted as the most effective forces shaping the future of insulin treatment. Today, these developments indicate a shift toward safer, more reliable, and patient-friendly insulin delivery systems that could change the way diabetes is managed..
Keywords:Insulin delivery systems; Automated insulin delivery; Glucose-responsive insulin; Nanotechnology; Microneedle-based delivery; Diabetes mellitus
Abbreviations: HCL: Hybrid Closed-Loop; MDI: Multiple Daily Injections; CSII: Continuous Subcutaneous Insulin Infusion; AID: Automated Insulin Delivery; Gox: Glucose Oxidase; MN: Microneedle; CGM: Continuous Glucose Monitoring; T2D: Type 2 Diabetes; T1D: Type 1 Diabetes; HbA1c: Glycated Hemoglobin; PBA: Phenylboronic Acid; SLNs: Solid Lipid Nanoparticles; NLCs: Nanostructured Lipid Carriers; NPs: Nanoparticles; GI: Gastrointestinal; PNPs: Polymeric Nanoparticles
Introduction
Diabetes mellitus is a fast-growing global health issue of the population. The current prevalence of diabetes in the older adult population is about 11% (1 in 9 adults) with 589 million adults (20-79 years) living with diabetes worldwide according to the International Diabetes Federation (IDF) Diabetes Atlas 2025 (11th edition). Almost 43% (about 250 million) of the cases are undiagnosed, and almost 80% of all people with the condition are in the low- and middle-income countries. Unless the current trends change, the number of diabetics will have increased to about 850 million around the world by the year 2050, and more effective prevention and management strategies are required to manage the situation [1]. Type 2 Diabetes (T2D) is more prevalent, while Type 1 Diabetes (T1D) is a significant and increasing clinical burden necessitating vital insulin treatment and high chances of acute and chronic morbidity. Diabetes as a pathophysiological condition is a persistent hyperglycaemia that occurs due to inadequate insulin secretion, insulin dysfunction, and both. T1D is a result of pancreatic 2-cells autoimmune destruction causing absolute insulin deficiency, and T2D is a result of insulin resistance with subsequent 2-cell dysfunction [2,3] Insulin is a peptide hormone with a weight of approximately 5.8kDa that is synthesized only in pancreatic β-cells in the form of preproinsulin, cleaved to form proinsulin in the endoplasmic reticulum, and finally cleaved in secretory granules to release biologically active insulin and equimolar C-peptide before being released in a regulated manner [4].
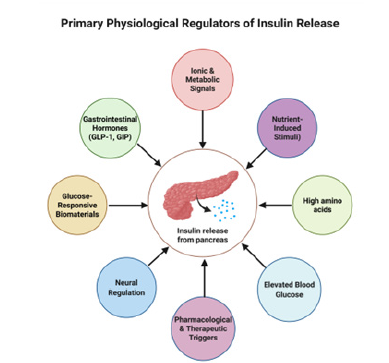
The major physiological factors initiating insulin release are summarized in Figure 1. On the physiological side, insulin plays a key role in glucose homeostasis by facilitating uptake of glucose in skeletal muscle and fat tissue, and coordination of lipid and protein metabolism via receptor mediated signaling pathways [2]. The destruction of such processes due to of β-cell loss, maladjusted insulin production, or secretion, and/or insulin resistance results in persistent hyperglycaemia and further microvascular and macrovascular complications and thus, protection of β-cell functioning and the utilization of insulin becomes [3,5]. In people with T1D, as well as in a significant proportion of those with advanced T2D, insulin is usually injected into the body by Multiple Daily Injections (MDI) by syringes or insulin pumps. Though effective, injection-based regimens have been linked to high levels of treatment burden, behavioral barrier (e.g., needle fear, stigma, and regimen fatigue), and clinical constraints (e.g. hypoglycaemia and glycaemic variability) because of the inability to dynamically adjust insulin delivery to changing physiological needs [6,7]. These barriers tend to lead to failure to get the right dose, suboptimal and insufficient glycaemic control despite intensification of therapy. To overcome these limiting factors, superior insulin delivery technologies have been designed to make them less invasive and enhance pharmacodynamic harmony between the insulin supply and the glucose swings. Closed-loop and automated systems with continuous glucose monitoring, and new, so-called smart delivery systems, such as glucose-responsive Microneedle (MN) patches and stimulus-responsive biomaterials, are expected to provide more time in target range, lessen the incidence of hypoglycaemia, and reduce the dependence on user-specific dosing decisions [8-10].
Figure 1:Major physiological regulators triggering pancreatic β-cell insulin secretion.
Evolution of Insulin Delivery Technologies
Early modes of administering insulin were manual and quite hostile necessitating the development of reusable syringes to advanced pens. The conventional methods of insulin delivery have been based previously through subcutaneous insulin administration, with a MDI dosage using syringes or insulin pens, which are still extensively utilized to this day in the management of both T1D and T2D. Traditional syringes have the flexibility of dosing but are believed to have a lack of convenience and adherence and insulin pens have a higher dose-precision in the focus of lasting glycaemic management addition to allowance to portability and patient acceptance than traditional syringes yet with a comparable pharmacokinetic profile [11]. They are usually based on the use of basal (long-acting) and bolus (rapid or short-acting) insulin preparations in varying ratios to induce approximation of endogenous insulin secretion; but they do not provide full replication of the endogenous insulin secretion and are frequently associated with glycaemic variability and elevated risk of hypoglycaemia [12]. Irrespective of these shortcomings, MDI based insulin therapy is still a basic and viable intervention in glycaemic regulation, especially in a resource constrained environment, and at the commencement phase of insulin administration. [12]. Moreover, recent advancements such as smart insulin pens have integrated memory functions, bolus calculators, and reminders into insulin pens and the ability to add Continuous Glucose Monitoring (CGM) systems to assist in real time decision-making and data review. These innovations aim to maintain simplicity of most of these injections on a daily routine while including digital features so that the user burden can be reduced, and glycaemic outcomes are improved [13,14].
Advanced Insulin Delivery Methods
Continuous subcutaneous insulin infusion (CSII)
Insulin pump technology was the beginning of a paradigm shift towards programming of constant insulin pump delivery. These implanted systems inject insulin subcutaneously at controlled basal rates together with meal boluses that are initiated by the user, which is more closely mimick physiological delivery as compared to intermittent injections. Recent developments include patch pumps, which are small, tubeless and more discrete and convenient, with wider accessibility to digital health platforms. These systems have been found to provide better glycaemic control and lessened the glycaemic variability in contrast to usual regimens, but the cost and accessibility are still a hindrance in most areas [13].
Hybrid closed-loop systems
Traditional CSII in which the user must continuously enter basal and bolus insulin has necessitated the creation of Automated Insulin Delivery (AID) systems incorporating CGM, insulin pump, and control software. This development has enhanced the sensoraugmented progressively to Hybrid Closed-Loop (HCL) and, most recently, toward full or near-full automation of closed-loop systems, commonly called artificial pancreas systems [15]. The concept of HCLs represents a major transitional step towards the process of automatizing the delivery of basal insulin without the need for user initiated meal boluses. A 6-month randomized multicenter trial found that the application of an HCL system significantly increased time spent in the target glucose range (70-180mg/dL) by 11%, compared to sensor augmented pump therapy, with reductions in the mean glucose levels and Glycated Hemoglobin (HbA1c) and no increase of severe hypoglycemia [16]. These results defined the clinical superiority of the HCL systems to previous pump-based strategies in adolescents and adults with T1D. A randomized crossover trial in very young children (1 to 7 years) indicated that HCL therapy was associated with more time in range by 8.7% than sensor-augmented pump therapy and less hyperglycemia, with equal exposure to hypoglycemia [17]. These findings have major implications based on the high glycemic variability and high caregiver burden of this age group. Likewise, HCL therapy was beneficial in controlling glycemic levels in mothers with T1D during pregnancy. The time-in-range of pregnant women receiving HCL systems was 68.2% in the AiDAPT trial expected control against 55.6% using standard insulin therapy, accompanied by improvements in hyperglycemia and HbA1c, and with no additional severe hypoglycemic or ketoacidosis occurrences [18].
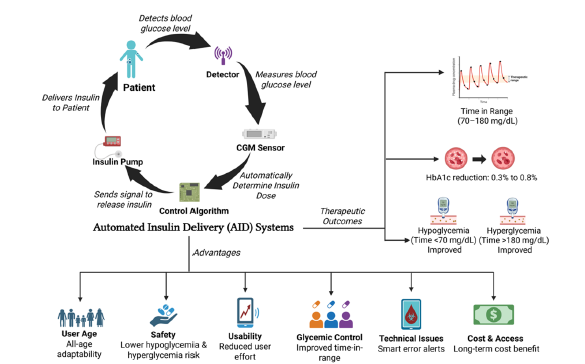
Recent innovations have resulted in superior closed-loop hybrid systems which can use automated correction boluses as well as adaptive basal modulation. Such systems also lessen burdens on the users and enhance glycemic effectiveness because they react quicker towards an escalation in glucose [15]. In parallel, the approach to do away with user-specific insulin dosing has yielded fully automated or near-fully automated closed-loop systems, such as the bionic pancreas. The insulin only bionic pancreas which initialized on body weight and didn’t require carbohydrate counting, reduced the level of HbA1c by about 0.5% in 13 weeks relative to conventional care, and did not raise the frequency of time spent in clinically significant hypoglycemia (below 54mg/dL), or diabetic ketoacidosis [19]. These results emphasize the likelihood of selfdelivering insulin actions with minimum patient involvement and portray a paradigm shift in the direction of adaptive, simplified glucose management. Figure 2 illustrates the working mechanism of AID systems, highlighting real-time glucose sensing, algorithmdriven insulin modulation, and the resulting therapeutic outcomes and advantages.
Figure 2:Mechanism, therapeutic outcomes, and advantages of Automated Insulin Delivery (AID) systems.
Smart and responsive glucose insulin delivery systems
By interlinking glucose sensing to insulin release or activity, glucose-responsive insulin delivery systems seek to mimic physiological functions of pancreatic β-cells, while addressing the risks of hypoglycemia and enhancing glycaemic control. In contrast to open approaches based on algorithms, closed-loop systems are dependent on inherent material or molecular sensitivity to glucose. The systems can autonomously regulate themselves without electronic or off-the-chip computation [20,21]. Enzymebased systems that use Glucose Oxidase (GOx) are among the most studied and earliest strategies available. Gox triggers glucose oxidation to gluconic acid and hydrogen peroxide resulting in local pH decrease, hypoxia, and reactive oxygen species which can be utilized to cause insulin release by polymer swelling degradation, membrane gating, or vesicle fusion [21]. GOxbased vesicles, microgels, membranes and hydrogel devices with glucose dependence have been found to release insulin in vitro and extended normoglycaemia in streptozotocin-induced diabetic rodent systems, in some cases over a prolonged duration and in a single dose [21]. An enzyme free alternative is represented by synthetic glucose-responsive polymers that are functionalized with Phenylboronic Acid (PBA) moieties. PBA reversibly reacts with cisdiol groups of glucose creating boronate esters that change polymer charge density, swelling behaviour or molecular interactions and thus regulates insulin release [22]. One such improvement has been the creation of glucose-sensitive MN patches, produced using in situ photopolymerization of PBA-based polymers, which allows a large insulin concentration (approximately 20 w%) and a quicker glucose-dependent release [22]. A PBA-based MN patch to control plasma glucose levels in insulin-deficient diabetic minipigs over 20 hours in a large-animal model proved that it was a highly responsive quick patch with significant application potential in medicine [22].
However, issues with glucose selectivity, chemical stability and toxicity of boronic acid derivatives in the long term in aqueous conditions, regardless of promising results, make PBA-based systems subject to comprehensive preclinical safety testing [20]. Competitive glucose binding has been used to prepare glucoseresponsive hydrogels and insulin conjugates using glucose-binding proteins and lectins, including concanavalin A. Although these systems can include reversible glucose-dependent insulin release, their practical use is restricted by immunogenicity, toxicity, and instabilities, especially with regard to plant-based or bacterial proteins [20]. This has rendered the majority of lectin-based systems just a proof-of-concept platform, but not therapeutically viable. Along with depot-based formulations, glucose-responsive behaviour has been engineered onto insulin molecules. Such strategies involve insulin conjugates or analogues that react to the level of glucose by modifying receptor binding, clearance, or selfassembly. Among them, mannose receptor-mediated clearance principles and carbohydrate-modified insulin conjugates are advanced to early clinical trials. However, with less insulin strength and lack of glucose sensitivity, they proceeded no further [20].
Transdermal platforms
The use of advanced transdermal and minimally invasive means of insulin delivery has received a lot of attention as an alternative to traditional subcutaneous injections because it aims at reduction of injection burden, providing better adherence and more physiological insulin pharmacokinetics. MN arrays and wearable patch pumps are two different yet complementary technological directions among them. MN arrays are minimally invasive devices that typically include micro-scale projections that are able to permeate the stratum corneum and inject insulin into the viable epidermis or superficial dermis, avoiding the pain and tissue injury caused by hypodermic needles [23]. In contrast to subcutaneous injections with depths of several millimeters, MNs normally have a depth of less than 1 mm which minimizes risks of bereaving and bleeding as well as lipodystrophy and allows fast insulin absorption due to the dense dermal microvasculature [24]. According to recent literature, there are insulin MN systems of hollow, dissolving, hydrogel-forming, biodegradable, and glucoseresponsive systems, each with different delivery features [23,24]. Hollow MNs are microinjection channels which are linked with external reservoirs or micropumps, providing a relatively high dose accuracy and delivery rate but with more complicated devices and dependence on auxiliary actuation systems [23]. Conversely, dissolving and biodegradable polymeric MNs entrap insulin into water-soluble or bio erodible acids enabling bolus or short-term sustained release as the needles dissolve in interstitial fluid. These systems facilitate the design of devices and remove sharp waste, but the clinical relevance of these typically had impediments of insulin loading and ability to scale the dose [25]. In order to resolve these shortcomings, more recent developments have been directed towards polymer engineering and the MN architecture.
Long-acting polymeric MNs use extended-release polymers, nano- or micro-particle reservoirs, or apply diffusion through a back-layer to adjust the delivery of insulin release over a long duration, decrease the dosing frequency, and minimize plasma concentration variation [24]. These types of design have shown to be able to provide sustained systemic exposure and have better PK profiles in preclinical models, but the reproducibility of dose across individuals is difficult. One of the biggest translational advances of the last three years has been the creation of high-insulin-loading MN patches. In more traditional solvent-cast MNs, insulin usable concentrations are only 5-20w% insulin, which is not enough to treat humans [21]. More recent designs with solid insulin powder cores with glucose-responsive shells of polymer have reached insulin loading of over 70w% enabling clinically relevant dosing in practicable patch sizes [21]. Such patches were also able to sustain normoglycaemia for up to 48 hours in streptozotocin-induced diabetic minipigs (>25kg), utilizing patches with an area of about 3.5cm2 [21]. Glucose-responsive MNs enable a single material platform to autonomously adjust insulin release in response to local glucose concentrations. In recent systems, the use of phenylboronic acid-based polymers or enzyme-free glucose-binding chemistries in MN shells or hydrogel matrices are used [25].
When hyperglycaemia occurs, the binding of glucose increases polymer charge density and swelling resulting in faster diffusion facilitated by insulin and suppressed release by normoglycaemic or hypoglycaemic conditions. Advanced multi-layer or triple-structure MN designs have resolved critical drawbacks of the previous smart MNs, such as imprecise drug delivery and lack of predictability during dosing. For instance, MNs that are glucose reactive and have an outer shell that is responsive to glucose, a solid insulin powder reservoir and a solid or osmotic propelling core resulted in almost complete insulin release and glycaemic regulation in diabetic miceup to 24 hours with patches the size of 0.3cm2. In addition, to enhance patient guidance and compliance, incorporation of visual indicators on MN backings have been suggested to indicate completion of drug release [26]. Even with these developments, MN systems that are glucose responsive are largely preclinical. The major issues have been identified to be interindividual differences in skin characteristics, potential chronic toxicity of glucoseresponsive chemistries, consistency in the production of goods, and regulatory complexity in drug- device combinations [23,25]. In parallel, wearable patch pumps have also been developed, which are minimally invasive insulin delivery pumps, which do not involve an external set of tubes, but provide continuous subcutaneous infusion. Patch-sized pumps are body-worn, compact, modern insulin pumps that incorporate insulin reservoirs, cannulas to deliver the insulin, and electronic components of control, and which are becoming more compatible with CGM and AID algorithms [27].
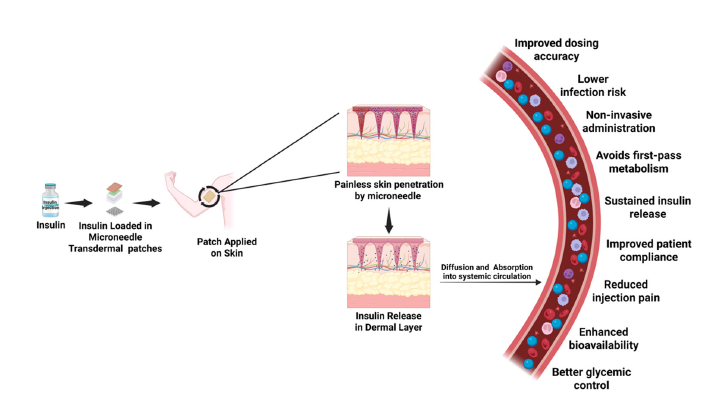
Patch pumps in comparison to traditional pumps are more discrete, mobile and easy to operate, thus leading to patient acceptance. Nonetheless, patch pumps, however, are still being essentially reliant on subcutaneous cannulas and electronically controlled, as opposed to being material-based glucose responsive. Therefore, they have restrictions, such as the failure of insulin infusion sites, delayed absorption of insulin, and the necessity of changing the sites of insulin administration regularly [27]. In terms of translation application, patch pumps are relatively mature and broadly used technology in the clinic, but MN platforms are only going through first-in-human validation. Figure 3 summarizes the mechanistic pathway and clinical advantages of transdermal insulin delivery systems.
Figure 3:Mechanism and therapeutic advantages of transdermal insulin delivery systems.
Nanotechnology based Insulin Delivery Systems
Nanotechnology-enabled oral delivery of insulin has long been pursued as a non-invasive alternative to subcutaneous injections not only for increasing patient compliance but also in recreating a more physiological hepatic-first distribution of insulin. Upon oral intake, insulin is subjected to acids and gastric digestive enzymes such as pepsin, trypsin, and chymotrypsin, which results in its immediate denaturation and enzymatic denaturation [28]. Moreover, the layer of intestinal mucus and the highly controlled epithelial junctions restrict the transport of macromolecules, thereby limiting transcellular and paracellular absorption [29]. To overcome these barriers nanocarriers have been developed with tightly controlled size, surface charge, composition and functionalization. Nanoscale insulin encapsulation protects insulin against chemical and enzymatic degradation, whereas pH-responsive release, muco adhesion, receptor targeting, or transient control of epithelial permeability are achieved by surface modifications [28]. Among the oral insulin carriers mostly studied are lipid-based nanoparticles, namely, Solid Lipid Nanoparticles (SLNs), Nanostructured Lipid Carriers (NLCs), liposomes, and nano emulsions. Such systems use the lipid-rich structure of biological membranes that allow cellular intake and also offer an absorbing framework to insulin [29].
Compared to liposomes and emulsions, the SLNs and NLCs are more stable, but the solid lipid cores can be restrictive in regard to insulin loading and uptake by epithelial cells. The relative bioavailability reported in preclinical studies was generally lower than 10% with glucose-lowering effects reported as being maintained but inconsistent across models [28]. To increase intestinal delivery through receptor-mediated absorption or mucoadhesion, liposomes and noisome have been designed to be coated with bile salts, vitamins (e.g., biotin) or chitosan. Surfacemodified vehicles such as these have shown modest increases in bioavailability, often 5-10% and are limited by poor stability in the GI tract and batch-to-batch inconsistency [29]. Another major category of oral insulin Nanocarriers is Polymeric Nanoparticles (PNPs) that are made out of different materials including chitosan, PLGA, alginate, and pH-sensitive methacrylate copolymers. These systems offer controllable degradation patterns and may be programmed to be released selectively in the intestine and not the stomach. Chitosan and its derivatives stand out especially because of their mucoadhesive characteristics, as well as their capability to open tight junctions, thus improving transient paracellular transport [29]. Nevertheless, long-term regulation of epithelial barriers provokes concerns about mucosal irritation and immune response activation. Hybrid nanocarriers are polymeric-inorganic or lipid nanoparticles that are used to enhance targeting and stability.
A number of formulations take advantage of bile acid receptors, vitamin B12-linked routes or glucose-sensing connections to be absorbed in the intestine and targeted to the liver. Although various systems have demonstrated pharmacological bioavailability of over 20% in rodent models, these findings usually depend on the supraphysiological dose of insulin, making them difficult to translate to humans [28]. One of the recent improvements is the fabrication of insulin-conjugated Silver Sulfide Quantum Dots (Ag2S QDs) wrapped in enzyme responsive chitosan-glucose copolymers. This formulation is insoluble in acidic gastric conditions, protects insulin against degradation by enzymes, and the insulin-containing payload becomes free under the activity of glucosidase in neutral pH [30]. Oral delivery of this nanotherapeutic in mice and rats led to hepatic preferential distribution and dose-dependent cholesterol lowering and a significantly lower probability of hypoglycaemia than injected insulin. Notably, glucose reductions were also observed in non-diabetic baboons with no biochemical and hematological toxicity, which is a rare instance of an oral insulin nanotechnology validation in large animals [30].
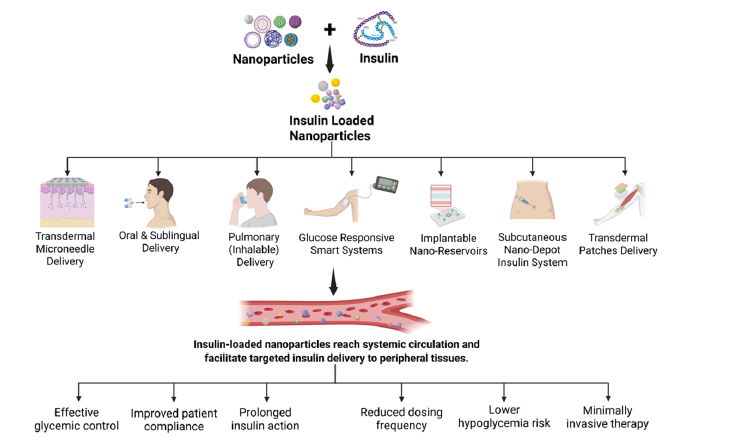
Pharmacokinetics showed decreased peak system insulin levels yet prolonged pharmacodynamic responses, which is exhibited by prevailing hepatic insulin action and decreased peripheral hyper insulinaemia. These data highlight the need to potentially transform the paradigm whereby oral nanocarriers focus on physiological nanocarrier insulin signaling and not on the highest systemic exposure [28,30]. Figure 4 illustrates the mechanistic pathways of nanotechnology-based insulin delivery, highlighting nanoencapsulation, targeted transport via multiple delivery routes, and the resulting therapeutic advantages.
Figure 4:Mechanistic pathways, delivery strategies, and therapeutic advantages of nanotechnology-based insulin delivery systems.
Advanced Insulin Delivery and Critical Gene Expression
There is currently no direct evidence that advanced technologies such as CSII, AID, and hybrid closed-loop systems enhance the expression of critical survival or longevity-associated genes, including Sirtuin 1 (SIRT1), independently. SIRT1 is an NAD+-dependent deacetylase that can centralize systemic insulin sensitivity, mitochondrial performance, and control oxidative stress and inflammation. It has been widely examined as a metabolic regulator in relation to insulin resistance and type 2 diabetes [31,32]. SIRT1 is associated with components of insulin signaling, such as insulin receptor substrates and insulin-AMPK signaling, and regulates glucose and lipid metabolism networks [31,32]. Notably, the activity of SIRT1 is not directly affected by the exposure to exogenous insulin. In chronic hyperglycemic conditions, SIRT1 expression and activity are frequently impaired, and interventions that improve metabolic homeostasis have been reported to increase SIRT1 activity and metabolic signaling [31,32]. Thus, although glycemic control with an optimized insulin delivery can perhaps decrease glucotoxicity and oxidative stress; to establish a more conducive metabolic environment in which gene regulation is normalized, the therapeutic effects of these devices must be envisioned in terms of physiological effects on blood glucose and tissue health instead of being considered direct modulators of gene regulation. Future research with longitudinal molecular profiling in individuals on AID systems can help determine whether long-term metabolic stabilization is associated with positive modifications in gene regulatory networks, in addition to the established physiological advantages.
Translational and Safety Issues
Although significant progress has been made over time in the development of insulin delivery systems based upon glucoseresponsiveness, enzyme-based systems still have significant safety and translational issues that have hitherto prevented such systems being routinely used in clinical practice. A major limitation is due to the fact that the release of insulin from subcutaneous depots is usually irreversible and limits active dose adjustment upon injection. This limitation is further exacerbated by inherent delays due to glucose diffusion to the sensing component, and further insulin uptake into the blood, which means that the response kinetics are frequently not in harmony with physiological dynamics of glucose [20]. Other issues are that the enzymes may not survive during the long periods of implantation or storage, and immunogenicity may occur and local tissue may produce hydrogen peroxide in the process of glucose oxidation. Co-immobilization of catalase, integration of oxygen-generating nanoparticles, or implementation of antioxidant matrices are mitigation strategies that have already shown a partial means of reducing oxidative stress and enhancing enzymatic activity in preclinical conditions. Nevertheless, these designs bring further material and compositional complexity, posing unresolved questions for long-term biocompatibility, safety in the use of chronic exposure and reproducibility at the manufacturing scale [21].
Consequently, there is no glucose-responsive insulin formulation, so far, that has been developed for use in the routine clinical application. In a larger context, understanding translational challenges should not be confined to the chemistry of enzymes but should consider basic system-level constraints. The major obstacles are a lack of response rate compared with endogenous glucose excursions, the inability to establish steep and predictable associations between glucose and insulin dose and response in the tight normal glycaemic range, material and functional stability in the long-term, and questions about chronic toxicity, scalability, and manufacturability [20]. The above constraints highlight the shortcomings of entirely material-based responsiveness which are in use in complex and changing physiological settings. There are some early indications that clinical success in the future will be attainable by incorporating optimized glucose-sensing chemistries into more complicated delivery architectures which offer more control layers. It has been suggested that MN arrays and hybrid systems that integrate material responsiveness with devices-level control could be the promising way to address this divide [22].
Here, progressive transdermal and minimally invasive technologies indicate a greater paradigm shift in a mechanicalonly method of delivering insulin to a patient-centered approach to material delivery. Premises carried by MNs have different benefits such as less pain, higher patient acceptance, quicker insulin absorption, and the possibility of autonomous glucose responsiveness. Patch pumps, in turn, offer high capacity, dependable system of insulin delivery based on existing clinical infrastructure. Translational initiatives in the future could thus need to be approached in a combinatorial manner, such as prandial or adaptive dosing slow to high loading MN patches being paired with wearable pumps or smart pens to deliver basal insulin, thus, balancing dose capacity, responsiveness and usability. In parallel, the delivery of oral insulin by nanotechnology has demonstrated positive preclinical advances but is yet to be demonstrated in a clinical sense. Low and highly variable bioavailability, high dependence on the use of animal models that do not exhibit much promiscuity to the physiology of the human gastrointestinal system, difficulty decoupling the pharmacodynamics of nanocarriers and pharmacodynamics of insulin, and regulatory uncertainty linked to complex nanomedicine formulations are major barriers to translational efficacy [28].
Furthermore, most of the reported systems use substantially higher insulin doses than those clinically that have cast serious doubts on scalability, cost-efficiency and safety in the long run. All of these safety and translational issues bring up collective requirements of integrative design approaches that enable material innovation to match physiological realism, manufacturability and regulatory viability. These interdependent limitations need to be addressed to transform next-generation insulin delivery systems based on glucose responsiveness and other insulin delivery systems into clinically feasible therapies.
Conclusion
Recently, new technologies on insulin delivery have led to the apparent change in traditional, user-based dosing to more adaptable, physiologically meta-cognitive and patientfocused systems. The range of innovations that have enhanced the conceptual and technological horizons of insulin therapy include glucose-responsive materials, and MN based transdermal systems, wearable pumps and the use of nanotechnology in the development of oral formulations. These methods will overcome historical shortcomings of subcutaneous injections such as slow pharmacokinetic properties, fixed doses, and the high costs of treatment, but also enhance better glycaemic control and adherence to treatment. Simultaneously, preclinical development has shown some essential safety, stability, and translational limitations that might still not be overcome, especially in response rate, dose accuracy within the normal glycaemic scope, protracted biocompatibility, and mass-producible production.
It is noteworthy that the cumulating evidence indicates that there will be no chance of a delivery modality based on which a universal answer can be obtained. Rather, future clinical impact will probably be provided as integrative strategies of combining optimized glucose responsive chemistries with enhanced delivery architecture and device level regulation thus adjusting responsiveness, dose ability and usability. Hybrid systems using material-enabled autonomy with known infrastructure of insulin delivery is a promising way to go. A combination of these initiatives can help to make insulin therapy more proactive, predictive, and sustainable glycaemic control rather than a responsive approach to glucose management.
Acknowledgement
The author gratefully acknowledges the Department of Life Sciences (Zoology), Manipur University, for providing laboratory facilities and academic support. The author also sincerely thanks the DST INSPIRE (Innovation in Science Pursuit for Inspired Research) Fellowship, Department of Science and Technology, Ministry of Science and Technology, Government of India, for financial support.
Author Contributions
Borish Loushambam, as the first author, was responsible for data collection, data analysis, and drafting the manuscript. Sivakumar Vijayaraghavalu, the corresponding author, guided manuscript preparation, conceptualized the review, supervised the study, and finalized the manuscript. The authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not Applicable.
Informed Consent Statement
Not Applicable.
Data Availability Statement
This narrative review synthesizes information from previously published studies, which are appropriately cited within the manuscript. No new data were generated or analyzed in this study. Therefore, data sharing is not applicable.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Diabetes Atlas (2025) IDF Diabetes Atlas | Global Diabetes Data & Statistics. Diabetes Atlas.
- Khin PP, Lee JH, Jun HS (2023) Pancreatic beta-cell dysfunction in type 2 diabetes. European Journal of Inflammation 21: 1721727X231154152.
- Lu X, Xie Q, Pan X, Zhang R, Zhang X, et al. (2024) Type 2 diabetes mellitus in adults: Pathogenesis, prevention and therapy. Signal Transduction and Targeted Therapy 9(1): 262.
- Ataie-Ashtiani S, Forbes B (2023) A review of the biosynthesis and structural implications of insulin gene mutations linked to human disease. Cells 12(7): 1008.
- Hill TG, Gao R, Benrick A, Kothegala L, Rorsman N, et al. (2024) Loss of electrical β-cell to δ-cell coupling underlies impaired hypoglycaemia-induced glucagon secretion in type 1 diabetes. Nature Metabolism 6(11): 2070-2081.
- Brixner D, Edelman SV, Sieradzan R, Gavin JR (2024) Addressing the burden of multiple daily insulin injections in type 2 diabetes with insulin pump technology: A narrative review. Diabetes Therapy 15(7): 1525-1534.
- Galdón Sanz-Pastor A, Justel Enríquez A, Sánchez Bao A, Ampudia-Blasco FJ (2024) Current barriers to initiating insulin therapy in individuals with type 2 diabetes. Frontiers in Endocrinology 15: 1366368.
- Kwon SY, Moon JS (2025) Advances in continuous glucose monitoring: Clinical applications. Endocrinology and Metabolism 40(2): 161-173.
- Pal S, Rakshit T, Saha S, Jinagal D (2025) Glucose-responsive materials for smart insulin delivery: From protein-based to protein-free design. ACS Materials Au 5(2): 239-252.
- Martínez-Navarrete M, Pérez-López A, Guillot AJ, Cordeiro AS, Melero A, et al. (2024) Latest advances in glucose-responsive microneedle-based systems for transdermal insulin delivery. International Journal of Biological Macromolecules 263: 130301.
- Shah R, Patel M, Maahs D, Shah V (2016) Insulin delivery methods: Past, present and future. International Journal of Pharmaceutical Investigation 6(1): 1-9.
- Candido R, Wyne K, Romoli E (2018) A review of basal-bolus therapy using insulin glargine and insulin lispro in the management of diabetes mellitus. Diabetes Therapy 9(3): 927-949.
- Yang R, Yang Z, Chi J, Zhu Y (2025) Insulin delivery devices in diabetes management: Applications and advancements. Intelligent Pharmacy 3(3): 235-242.
- Singh LG, Gothong C, Ash GI, Hernandez R, Spanakis EK (2025) Advancing telemedicine using smart insulin pens with continuous glucose monitoring and telecommunication systems: A case series. Journal of Clinical Medicine 14(6): 1794.
- Moon SJ, Jung I, Park CY (2021) Current advances of artificial pancreas systems: A comprehensive review of the clinical evidence. Diabetes & Metabolism Journal 45(6): 813-839.
- Brown SA, Kovatchev BP, Raghinaru D, Lum JW, Buckingham BA, et al. (2019) Six-month randomized, multicenter trial of closed-loop control in type 1 diabetes. New England Journal of Medicine 381(18): 1707-1717.
- Ware J, Allen JM, Boughton CK, Wilinska ME, Hartnell S, et al. (2022) Randomized trial of closed-loop control in very young children with type 1 diabetes. New England Journal of Medicine 386(3): 209-219.
- Lee TTM, Collett C, Bergford S, Murphy HR (2023) Automated insulin delivery in women with pregnancy complicated by type 1 diabetes. New England Journal of Medicine 389(17): 1566-1578.
- Bionic Pancreas Research Group (2022) Multicenter, randomized trial of a bionic pancreas in type 1 diabetes. New England Journal of Medicine 387(13): 1161-1172.
- Hoeg-Jensen T (2021) Review: Glucose-sensitive insulin. Molecular Metabolism 46: 101107.
- Wang S, Yang C, Zhang W, Zhao S, You J, et al. (2024) Glucose-responsive microneedle patch with high insulin loading capacity for prolonged glycemic control in mice and minipigs. ACS Nano 18(38): 26056-26065.
- Yu J, Wang J, Zhang Y, Chen G, Mao W, et al. (2020) Glucose-responsive insulin patch for the regulation of blood glucose in mice and minipigs. Nature Biomedical Engineering 4(5): 499-506.
- Zhao J, Xu G, Yao X, Zhou H, Lyu B, et al. (2022) Microneedle-based insulin transdermal delivery system: Current status and translation challenges. Drug Delivery and Translational Research 12(10): 2403-2427.
- Meng F, Qiao X, Xin C, Ju X, He M (2024) Recent progress of polymeric microneedle-assisted long-acting transdermal drug delivery. Journal of Pharmacy& Pharmaceutical Sciences 27: 12434.
- Starlin Chellathurai M, Mahmood S, Mohamed Sofian Z, Janakiraman AK (2024) Biodegradable polymeric insulin microneedles: A design and materials perspective review. Drug Delivery 31(1): 2296350.
- He Y, Chen N, Zang M, Zhang J, Zhang Y, et al. (2024) Glucose-responsive insulin microneedle patches for long-acting delivery and release visualization. Journal of Controlled Release 368: 430-443.
- Rimon MTI, Hasan MW, Hassan MF, Cesmeci S (2024) Advancements in insulin pumps: A comprehensive exploration of insulin pump systems, technologies, and future directions. Pharmaceutics 16(7): 944.
- Fontana G, Innamorati G, Giacomello L (2025) Nanoparticle-based oral insulin delivery: Challenges, advances, and future directions. Pharmaceutics 17(12): 1563.
- Han Y, Spicer J, Huang Y, Bunt C, Liu M, et al. (2024) Advancements in oral insulin: A century of research and the emergence of targeted nanoparticle strategies. European Journal of Lipid Science and Technology 126(7): 2300271.
- Hunt NJ, Lockwood GP, Heffernan SJ, Cogger VC (2024) Oral nanotherapeutic formulation of insulin with reduced episodes of hypoglycaemia. Nature Nanotechnology 19(4): 534-544.
- Liang F, Kume S, Koya D (2009) SIRT1 and insulin resistance. Nature Reviews Endocrinology 5: 367-373.
- Martins IJ (2019) Insulin therapy and autoimmune disease with relevance to non-alcoholic fatty liver disease. In: Non-alcoholic Fatty Liver Disease-An Update. Intech Open.
© 2026 Sivakumar Vijayaraghavalu. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






