- Submissions

Full Text
Environmental Analysis & Ecology Studies
Health Status Assessment of Tabarka Port, Tunisia (Western Mediterranean Sea) Using Oxidative Stress and Neurotoxicity Marker in Golden Grey Mullet
Ayari-Kliti Rakia1*, Rjeibi Okbi2 and Sellami Badreddine1
1Laboratory of Marine Biodiversity, National Institute of Marine Science and Technology, Carthage University, Tunisia
2Lobaratory of Marine Living resources, National Institute of Marine Science and Technology, Carthage University, Tunisia
*Corresponding author:Ayari-Kliti Rakia, Laboratory of Marine Biodiversity, National Institute of Marine Science and Technology, Carthage University, Tunisia
Submission: January 28, 2026; Published: March 10, 2026

ISSN 2578-0336 Volume13 Issue 3
Abstract
Ports are highly anthropized areas that receive multiple sources of pollution, requiring monitoring plans to improve environmental status. In this context, we suggest detecting the combined effects of different types of pollutants discharged into the port of Tabarka on the golden grey mullet Chelon auratus (Risso, 1810). We focused on comparing the biochemical responses of the Tabarka port population and another sample from a nearby non-anthropized marine area. Biochemical responses were assessed through enzymatic activities (CAT and AChE) and lipid peroxidation levels (MDA). Our results revealed variations in CAT enzymatic activity and MDA levels between the port and marine fish populations, confirming a general state of oxidative stress in the former. Additionally, neurotoxicity due to neurotoxins in the port fish was demonstrated. Overall, the biochemical responses approach appears to be valuable for harbor water biomonitoring, as they collectively highlight the effects of different pollutants on mullet fish physiology and could provide guidance for future adapted environmental monitoring.
Keywords:Chelon auratus; Biomarkers; Water biomonitoring; Port of Tabarka
Introduction
Ports are remarkable ecosystems due firstly to their generally semi-closed structure, and to the anthropogenic pressure they undergo. These mainly concern domestic discharges and hydrocarbon leaks from boats. Several threats could increasingly occur in Tunisian ports and elsewhere, affecting the water quality as a consequence of organic matter and toxic products accumulation. This leads us to evaluate the pollution impacts in these ecosystems, which is imperative to protect them from additional deterioration. Marine organisms living in such an environment bioconcentrate pollutants that affect their state of health. Mugilidae, known for their wide ecological distribution mainly in ports, are gregarious fish widely used as sentinel organisms in biomonitoring programs of aquatic pollution [1-9]. Despite its extensive presence in the contaminated coastal environment, the Mugilidae family has commercial importance, which could have a health impact on consumers.
Deleterious pollutants’ effects on living organisms are difficult to detect since many of them will be visible only after long periods beyond a destructive and non-return point, which can’t be reversed by remedial actions. This triggered the establishment of early-warning signals called biomarkers, known as measured signs indicating modifications due to the presence and the magnitude of pollutants. In this respect, we are called to apply sensitive laboratory bioassays, based upon biomarker responses, to assess the toxicological impact of pollutants on mugilids population of the port of Tabarka, receiving some uncontrolled discharges from some neighboring hotel units. In the absence of any monitoring programme concerning the ecological status of this area, this contribution assesses for the first time a reference health status of the port of Tabarka by detecting the combined effects of pollutants on the mugilid health, which could guide future adapted environmental monitoring.
Materials and Methods
Biological material
The golden grey mullet, Chelon auratus (Risso, 1810), was the most abundant fish in the port of Tabarka and was represented by another control marine population spared from sources of pollution. The choice of this fish could better visualize the effect of port pollution on the concerned population. The main morphological criteria that distinguish Chelon auratus from other species of the same family are the presence of a golden spot on each operculum.
Sampling and study area
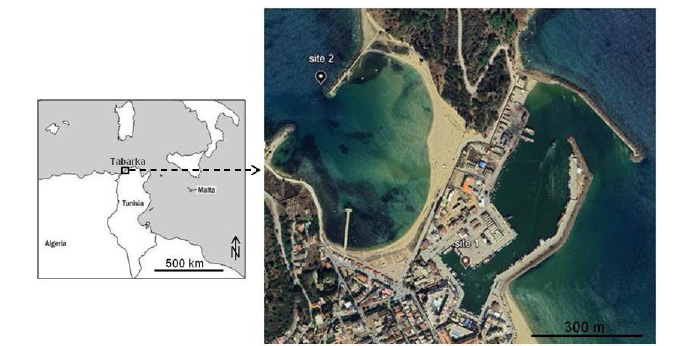
During spring, a dozen individuals of Chelon auratus were collected from each site using a 35/50 gill net, then kept alive in seawater. The two concerned sites were located in Tabarka region at the north-western Tunisian coast; site 1 in the port (36°57’26’’N 8°45’33’’E) and the site 2 in a nearby coastal sea area (36°57’42’’N 8°45’17’’E) (Figure 1). The port is characterized by touristic and fisheries activities, as well as it collects water from rainwater canalizations connected to sewage sewers of some neighboring hotel units.
Figure 1:The sampling sites of the mullet Chelon auratus from Tabarka region (modified from google earth).
Biochemical analysis
After weighing and measuring, organisms were dissected. Gills, liver, and brain were excised, crushed using an ultra Turax (IKA), then homogenized in three volumes of TBS buffer (Tris 50mM, NaCl 150mM, pH 7,4). The homogenate was centrifuged at 9000 x g for 30min to obtain S9 fractions (the post-mitochondrial aqueous fraction) that were stored at -80 °C to be used for the determination of enzyme activities. Total Protein content essential to estimate and to calculate enzyme activities was carried out according to Bradford’s method [10] using serum albumin as a standard.
Determining catalase activity was carried out according to Claiborne’s method [11]. Reaction mixture of a final volume of 1mL, contained S9 fraction, phosphate buffer (pH 7,4), and Hydrogen Peroxide (H2O2). Another reaction, which contains the same components except the fraction S9, was then optical measurement to measure catalase activity by the disappearance of hydrogen peroxide. Results were expressed as μmoles of H2O2 transformed per min and per mg protein.
Acetylcholinesterase activity assay was carried out according to the colorimetric Ellman‘s method [12]. Reaction mixture of a final volume of 1mL, containing 50μl of the S9 fraction, phosphate buffer (pH 7,4), and acetylcholine in the presence of the DNTN reagent. Optical density was monitored in order to measure the enzymatic activity, expressed in μmoles of thiocholine per min and per mg protein.
Malondialdehyde (MDA), toxic aldehyde end product of lipid peroxidation. Its presence marks oxidative stress due to free radicals. The amount of peroxidized lipids is estimated by the amount of MDA formed, which was expressed in nmol.ml-1 of S9, and the relationship between its concentration and the absorbance (optical density or OD) is based on the Beer-Lambert law.
Statistical analysis
For each biomarker, the determination of the average activity for individuals and the standard deviation was necessary to describe their state. The first-order ANOVA test was used to check the homogeneity of variances in order to highlight an overall significant difference between fish collected from the port and those collected from the sea. The Tukey significant difference test, also called the highly significant difference test, was used in this study for multiple comparisons between sites. Results were expressed as mean values ± significant deviation.
Ethical statement and anesthesia agent
All experimental procedures involving fish were conducted in accordance with internationally accepted guidelines for the care and use of laboratory animals in research. The study protocol was approved by the relevant Institutional Animal Care and Use Committee/Ethics Committee of the National Institute of Marine Sciences and Technologies (approval number: 1592023, dated September 15, 2023). All efforts were made to minimize animal suffering and to reduce the number of animals used. All experimental procedures are reported in accordance with ARRIVE guidelines.
Before dissection, fish (Chelon auratus) were anesthetized using tricaine methanesulfonate (MS-222) at a concentration of 150mg/L, buffered with sodium bicarbonate to neutral pH, following established guidelines for fish anesthesia. Adequate anesthesia was confirmed by loss of equilibrium and absence of reflex responses. Fish were then humanely euthanized by overdose of MS-222 before tissue collection for biochemical and histological analyses.
Data Availability Statement
The authors confirm that the data supporting the findings of this study are available from the corresponding author upon reasonable request.
Results
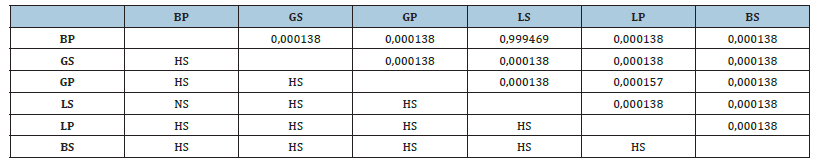
Considering the two sites and whatever the organ, catalase activity is always high in the port mugilid compared to the marine mugilid (Figure 2), and this with highly significant differences (Table 1). This can only be linked to the oxidative stress generated by ROS (reactive oxygen species) since its level is maintained by the antioxidant system, where catalase is the main participant. This confirms the results of many previous works by Schieber & Chandel [13,14]. Regarding the catalase activity at the same site (Table 1), the highest value was recorded at the gills compared to the brain and liver. The average catalase activity in sea mugilid did not show a significant difference between gills and brain (p>0.05). However, a significant difference was recorded in the liver.
Figure 2:Average variation of catalase activity in Chelon auratus collected at the port and the sea of Tabarka. a, b, c, d and e: different activity at the 5% threshold (ANOVA: Tukey’s HSD test).
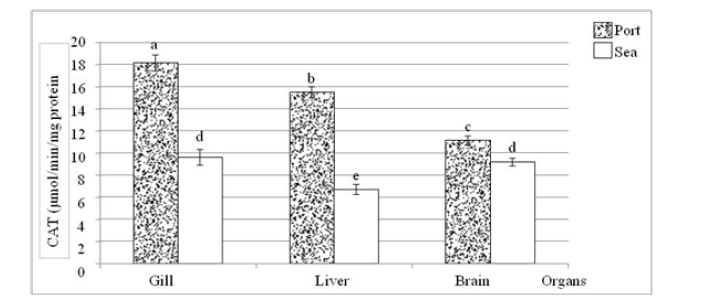
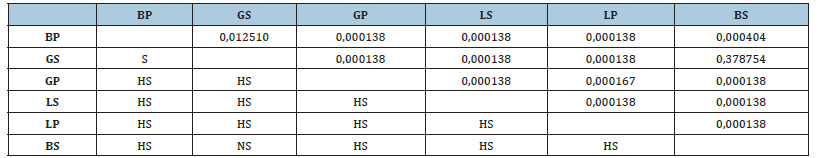
Table 1:Difference in catalase activity between organs in the two sites (ANOVA 1, Tukey’s HSD test, difference marked at p<0.05). BP: brain/port; BS: brain/sea; GS: gill/sea; GP: gill/ port, LS: liver/ sea, LP: liver/port, NS: not significant, HS: highly significant.
Regardless of the same organ in the two sites, the MDA level is elevated in the port sample compared to the marine sample (Figure 3). The MDA level varies, for example, from 1.30±0.04 nmol/min/mg of protein in the gill of fish collected from the sea to 2.14±0.14nmol/min/mg of protein in the gill of the port mugilid with a significant difference (Figure 3 & Table 2).
Figure 3:MDA levels in the gills, liver and brain of the fish from the Port and Tabarka Sea. a, b, c: different activity at the 5% threshold (ANOVA: Tukey’s HSD test).
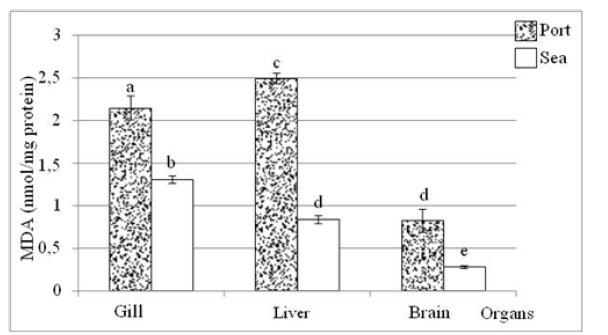
Table 2:Difference in MDA activity between organs in the two sites (ANOVA 1, Tukey’s HSD test, difference marked at p<0.05). BP: brain/port; BS: brain/sea; GS: gill/sea; GP: gill/ port, LS: liver/ sea, LP: liver/port, NS: not significant, HS: highly significant.
In the port fish, the average acetylcholinesterase activity is slightly higher in the brain than in the gills and liver, while in the sea mullet, it is much higher in the gills and brain than in the liver (Figure 4). Considering the two sites, the AChE activity in port fish is always lower than that in marine fish, with highly significant differences (Table 3).
Figure 4:Variation in the average activity of acetylcholinesterase in the gills, liver and brain of port fish and marine fish. a, b, c and d: different activity at the 5% threshold (ANOVA: Tukey’s HSD test).
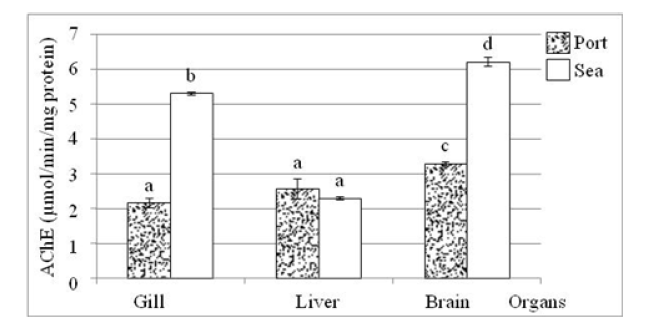
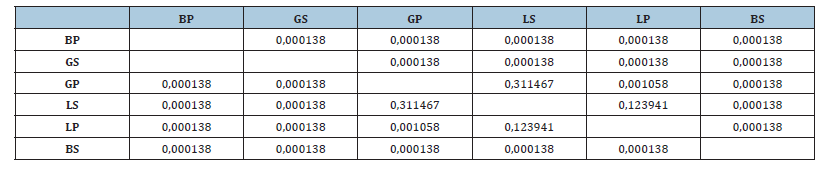
Table 3:Difference in acetylcholinesterase activity between organs in the two sites (ANOVA 1, Tukey’s HSD test, difference marked at p<0.05). BP: brain/port; BS: brain/sea; GS: gill/sea; GP: gill/ port, LS: liver/ sea, LP: liver/port.
Discussion
The results of the current work highlight a clear difference between fish collected from the port and those collected from the sea. The formers show an oxidative stress confirmed by the high activity of catalase and the high level of MDA in the different organs. Several other studies carried out on aquatic ecosystems subject to global changes and essentially to anthropogenic pollution show a close relationship between oxidative stress and the production of reactive oxygen species ROS. The high catalase activity in the port fish is probably related to oxidative stress generated by ROS, whose levels are maintained by the antioxidant system, where catalase is the main participant [13]. Our results are close to those previously observed in other marine organisms exposed to different environmental stressors [15,16]. The high catalase activity in the port mugilid also showed a specific response for each tissue. Thus, the highest value was recorded at the gill level compared to the brain and liver, and this may be related to the function and biological composition of these organs [17]. Gills of aquatic organisms play an essential role in internal circulation [18] 9*/, bioconcentration, accumulation, and detoxification of contaminants, making them the first barrier of defense against environmental pollution. The gills are in direct contact with several harbor pollutants (organic matter, heavy metals, hydrocarbons, etc). Moreover, the specific response of each organ in response to environmental stress can also be attributed to the different pathways of transport and accumulation of stress factors, speciation, and tissue sequestration in marine organisms [19-21]..
The liver is an organ responsible for the metabolism and detoxification of pollutants in aquatic organisms [22]. The catalase activity in the liver of Chelon auratus collected from the port was higher than that in mugilid collected from the sea. This result confirms the presence of a defense response induced against oxidative stress. It is important to note that the difference in CAT activity between organs may be within the norm or also induced.
When oxidative stress exceeds the capacity of antioxidant enzymes, cellular damage affecting cells and membrane structure occurs [23]. Reactive oxygen species accumulated in the presence of stress can attack polyunsaturated fatty acids (PUFAs) in the cell membrane. Malondialdehyde is the metabolite derived from lipid peroxidation and has been widely used as an indicator of oxidative damage to membranes.
In this study, the MDA level was elevated in the Chelon auratus population of the port compared to that collected from the sea. The importance of PUFAs oxidation in port fish indicates the inability to detect and eliminate the harmful effects of foreign substances. These may be partly metallic in nature because heavy metals are responsible for the increase in MDA levels [24,25].
Triglycerides are stored in different muscle tissues and mainly in the liver, which is more affected by lipid peroxidation. The existence of an endodermal barrier, the first to be affected by environmental stressors, explains the higher MDA level in the gills compared to that recorded in the liver, and this for both sites. The gill, therefore, remains the first target organ for stressors. Our results are comparable to those found by Pytharopoulou et al. [26], who showed that MDA levels are elevated in the gills of organisms from a polluted site compared to organisms from a healthy site.
The higher levels of MDA in Chelon auratus from the port of Tabarka may also be partly due to its possibly higher fat content than in marine species [27], as well as to the rate of exposure of the fish to different pollutants, urban and industrial [28].
The Acetylcholinesterase (AChE), widely used to assess the biological effects of chemicals, including pesticides, on marine organisms, represents a recent tool in the fields of biomonitoring of coastal environments [29-31]. This enzyme is very sensitive to exposure to organophosphate, carbamate, and pyrethroid pesticides due to their essential role in the transmission of nerve impulses in vertebrates, including fish [32,33]. It is an excellent neurotoxicity biomarker, whose presence mainly at the synapses in nervous tissue and neuromuscular junction explains the importance of its activity in the brain and gills of mugilids, whether from the port or the sea. The AChE activity in port fish, which is always lower than that in marine fish with highly significant differences, indicates the presence of contaminants at the port that inhibit AChE and therefore affect the neuromuscular system of mugilids. Our results confirm those already found by Dellali et al. [30], reported a spatial variation in AChE activity characterized by low values recorded at polluted sites.
Conclusion
Current assessment of water quality in Tunisian ports suffers from data gaps and requires the choice of an appropriate biological model to assess their health status and obtain information to prevent pollution sources and to plan remediation programs. The current study proves that the biochemical analyses could be a reliable tool for studying the health status of the port of Tabarka. It characterises the differences between biochemical responses of mugilids from the port of Tabarka, an alarmingly polluted area, and from a natural coastal area in the same region but spared from sources of disturbance. High catalase activity and MDA level were observed in mugilids collected from the port compared to those collected from the sea, unlike acetylcholinesterase activity. This highlights the oxidative stress and the cellular damage in the port mugilid, and the presence of neurotoxins in the port water. In addition, the oxidative stress at the gill leads to the use of this organ in aquatic environment biomonitoring programs.
Funding Declarations
Authors declare that they did not receive any funding related to the current research paper.
Acknowledgments
This study was supported by the National Institute of Marine Sciences and Technologies and the Faculty of Sciences of Bizerte, University of Carthage. The authors are grateful to all technician’s team of the Tabarka Center for their help.
References
- Chambers JE (1979) Induction of microsomal mixed-function oxidase system components in striped mullet by short-term exposure to crude oil. Toxicology Letters 4: 227-230.
- Kurelec B, Garg A, Krca S, Chacko M, Gupta RC (1989) Natural environment surpasses polluted environment in inducing DNA damage in fish. Carcinogenesis 10: 1337-1339.
- Rodríguez-Ariza A, Dorado G, Navas JI, Pueyo C, López Barea J (1994) Promutagen activation by fish liver as a biomarker of littoral pollution. Environmental and Molecular Mutagenesis 24: 116-123.
- Marcovecchio J (2004) The use of Micropogonias furnieri and Mugil liza as bioindicators of heavy metals pollution in La Plata River estuary, Argentina. Sciences of The Total Environment 323(1-3): 219-226.
- Bahnasawy M, Khidr AA, Dheina N (2009) Seasonal variations of heavy metals concentrations in Mullet, Mugil cephalus and Liza ramada (Mugilidae) from Lake Manzala, Egypt. Egyptian Journal of Aquatic Biology and Fisheries 13(2): 81-100.
- Ben Ameur W, de Lapuente J, El Megdiche Y, Barhoumi B, Trabelsi S, et al. (2012) Oxidative stress, genotoxicity, and histopathology biomarker responses in mullet (Mugil cephalus) and sea bass (Dicentrarchus labrax) liver from Bizerte Lagoon (Tunisia). Marine Pollution Bulletin 64(2): 241-251.
- Ben Ameur W, El Megdiche Y, de Lapuente J, Barhoumi B, Trabelsi S, et al. (2015) Oxidative stress, genotoxicity, and histopathology biomarker responses in Mugil cephalus and Dicentrarchus labrax gill exposed to persistent pollutants. A field study in the Bizerte Lagoon: Tunisia. Chemosphere 135: 67-74.
- Arockia Vasanthi L, Revathi P, Mini J, Munuswamy N (2013) Integrated use of histological and ultrastructural biomarkers in Mugil cephalus for assessing heavy metal pollution in Ennore estuary, Chennai. Chemosphere 91(8): 1156-1164.
- Waltham NJ, Teasdale PR, Connolly RM (2013) Use of flathead mullet (Mugil cephalus) in coastal biomonitor studies: Review and recommendations for future studies. Marine Pollution Bulletin 69 (1-2): 195-205.
- Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry 72(1-2): 248-254.
- Claiborne A (1985) Catalase activity. In: Greenwald RA (Ed,). CRC Handbook of Methods for Oxygen Radical Research. CRC Press, Boca Raton, Florida, USA, pp. 283-284.
- Ellman GL, Courtney KD, Andres V, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochemical Pharmacology 7:88-95.
- Schieber M, Chandel NS (2014) ROS function in redox signaling and oxidative stress. Current Biology May 24(10): 453-462.
- Kim J, Perera NCN, Godahewa GI, Priyathilaka TT, Lee J (2019) Characterization of a catalase from red-lip mullet (Liza haematocheila): Demonstration of antioxidative activity and mRNA upregulation in response to immunostimulants. Gene 712: 143945.
- Lewis C, Ellis RP, Vernon E, Elliot K, Newbatt S, et al. (2016) Ocean acidification increases copper toxicity differentially in two key marine invertebrates with distinct acid-base responses. Scientific Reports 6: 21554.
- Juhel G, Bayen S, Goh C, Lee WK, Kelly BC (2017) Use of a suite of biomarkers to assess the 560 effects of carbamazepine, bisphenol A, atrazine, and their mixtures on green mussels, Perna viridis. Environmental Toxicology and Chemistry 36(2): 429-441.
- Chen S, Qu M, Ding J, Zhang Y, Wang Y, et al. (2018) BaP-metals co-exposure induced tissue-specific antioxidant defense in marine mussels Mytilus coruscus. Chemosphere 205: 286-296.
- Gómez-Mendikute A, Elizondo M, Venier P, Cajaraville MP (2005) Characterization of mussel gill 543 cells in vivo and in vitro. Cell Tissue Research 321(1): 131-140.
- Canesi L, Viarengo A, Leonzio C, Filippelli M, Gallo G (1999) Heavy metals and glutathione metabolism in mussel tissues. Aquatic Toxicology 46(1): 67-76.
- Navarro A, Faria M, Barata C, Piña B (2011) Transcriptional response of stress genes to metal exposure in zebra mussel larvae and adults. Environmental Pollution 159(1): 100-107.
- Shenai-Tirodkar PS, Gauns MU, Mujawar MWA, Ansari ZA (2017) Antioxidant responses in gills and digestive gland of oyster Crassostrea madrasensis (Preston) under lead exposure. Ecotoxicology and Environmental Safety 142: 87-94.
- Canesi L, Ciacci C, Balbi T (2015) Interactive effects of nanoparticles with other contaminants in aquatic organisms: Friend or foe? Marine Environment Research 111: 128-134.
- Blier PW, Vandenberg G, Le François NR, Proulx E, Dupuis F, et al. (2025) Oxidative stress and growth performance are modulated by polyunsaturated fatty acids in Arctic charr, brook charr, and their reciprocal hybrids. Conservation Physiology 13(1): coaf032.
- Sunderman FW, Marzouk A, Hopfer SM, Zaharia O, Reid MC (1985) Increased lipid peroxidation in tissues of nickel chloride-treated rats. Annals of Clinical and Laboratory Science 15: 229-236.
- Vlahogianni TH, Valavanidis A (2007) Heavy-metal effects on lipid peroxidation and antioxidant defence enzymes in mussels Mytilus galloprovincialis. Chemistry and Ecology 23(5): 361-371.
- Pytharopoulou S, Sazakli E, Grintzalis K, Georgiou CD, Leotsinidis M, et al. (2008) Translational responses of Mytilus galloprovincialis to environmental pollution: Integrating the responses to oxidative stress and other biomarker responses into a general stress index. Aquatic Toxicology 89(1): 18-27.
- Okolie NP, Akioyamen MO, Okpoba N, Okonkwo C (2009) Malondialdehyde levels of frozen fish, chicken, and turkey on sale in Benin City markets. African Journal of Biotechnology 8(23): 6638-6640.
- Ajayi T, Ajasin F, Ajayi Y, Sonibare F, Ojezele M, et al. (2014) Analysis of Malondialdehyde (MDA) contents in frozen fish Sold in two fish markets in Ibadan, Southwest Nigeria. International Journal of Scientific & Engineering Research 5(7): 767-769.
- Moulton CA, Fleming WJ, Purnell CE (1996) Effects of two cholinesterase-inhibiting pesticides on freshwater mussels. Environmental Toxicology and Chemistry 15(2): 131-137.
- Dellali M, Gnassia Barelli M, Roméo M, Aissa P (2001) The use of acétylcholinestérase activity in Ruditapes decussatus and Mytilus galloprovincialis in the biomonitoring of Bizerta lagoon. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology 130(2): 227-235.
- Hernández-Moreno D, Soler F, Míguez MP, Pérez-López M (2010) Brain acetylcholinesterase, malondialdehyde, and reduced glutathione as biomarkers of continuous exposure of tench, Tinca tinca, to carbofuran or deltamethrin. Science of the Total Environment 408(21): 4976-4983.
- Bocquené G, Galgani E, Truquet P (1990) Characterisation and assay conditions for the use of ACHe activity from several marine species in pollution monitoring. Marine Environment Research 30(2): 75-89.
- Walker CH (1998) The use of biomarkers to measure the interactive effects of chemicals. Ecotoxicology and Environmental Safety 40(1-2): 65-70.
© 2026 © Ayari-Kliti Rakia. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






