- Submissions

Full Text
COJ Biomedical Science & Research
Initial Milestones of Covid-19 As Triggers for Effective and Safe Immunization Updates
Bernardo Montesanti Machado de Almeida1, Marileia Scartezini1, Caio Corsi Klosovski1, Anita L R Saldanha1, Ana Paula Pantoja Margeotto1, André Luis Varela Gasparoto2 and Tania Leme da Rocha Martinez1*
1Nephrology Department, BP - A Beneficência Portuguesa de São Paulo, São Paulo, Brazil
2Intensive Care Unit, BP - A Beneficência Portuguesa de São Paulo, São Paulo, Brazil
*Corresponding author:Tania Leme da Rocha Martinez, BP - A Beneficência Portuguesa de São Paulo, São Paulo, Brazil, Comandante Ismael Guilherme Street, 358-Lusitania Garden 04031-120 - São Paulo-SP, Brazil
Submission: May 30, 2025; Published: June 10, 2025

Volume2 Issue3June 10, 2025
Abstract
Due to the fact that there are still many projects regarding prevention and immunization to avoid new COVID-19 pandemia episodes the focus on data of the first years can provide new lines of investigation. This research presents weekly results of patients as to their tests, symptoms and immunization status. The results were registered pre and very early vaccination. They show the increase in all numbers as from the start of the pandemia. Due to this information the preventive programs have to be expanded and updated, which includes public awareness attention effective and safe vaccines.
Keywords:COVID-19; Vaccines; Immunization; Pandemia; Prevention
Abbreviations: POCT: Point-of-Care Testing
Introduction
The world can still be living with the perspective of a new COVID-19 pandemia, and so all efforts are important to prevent the lower possibility of health damages. It is of paramount importance to review the development of COVID-19 vaccines, addressing their milestones, lessons learned, and future prospects. The SARS-CoV-2 virus, responsible for COVID-19, has led to an unprecedented global effort to develop effective and safe vaccines. The rapid evolution of Variants of Concern (VOCs) has challenged the efficacy of immunizers, making continued genomic surveillance and vaccine adaptation essential. COVID-19 vaccines have been developed based on several technology platforms: Inactivated, attenuated, viral vector, protein subunit, RNA, DNA, and Virus-Like Particle (VLP) vaccines. Each has advantages and disadvantages. RNA and viral vector vaccines, for example, demonstrate a strong ability to induce cellular and humoral immune responses, but they can also be associated with adverse effects such as myocarditis or thrombocytopenia.
It is worth noting the immune response induced by vaccines, including the activation of T helper type 1 (Th1) cells, Cytotoxic T cells (CTLs), T follicular helper (Tfh) cells, and the formation of Germinal Centers (GCs), which are essential for the production of high-affinity antibodies and long-lasting immunological memory. Vaccines such as BNT162b2 (Pfizer- BioNTech) and mRNA-1273 (Moderna) are observed to induce robust responses in terms of both antibodies and memory cells. Efficacy and safety data from different vaccine platforms, with emphasis on preclinical trials in animal models and clinical studies in humans. Inactivated vaccines, such as BBIBP-CorV and CoronaVac, have shown good safety, especially in broader populations, although they induce less robust immune responses compared to RNA or viral vector vaccines. Viral vector vaccines, such as AZD1222 (Oxford/AstraZeneca) and Ad26. COV2. S (Johnson & Johnson), have good immune responses, but with a risk of adverse effects in specific cases. Protein subunit vaccines, such as NVX-CoV2373 (Novavax), have also shown promise, inducing high levels of neutralizing antibodies and effective cellular responses. DNA and Virus-Like Particle (VLP) vaccines are still in preliminary stages, with challenges mainly related to low immunogenicity and production complexity, respectively. Faced with the emergence of variants such as Beta, Gamma, and Omicron, the study emphasizes the need to update vaccine formulations and adopt strategies such as heterologous booster vaccines, the use of optimized adjuvants, and the development of next-generation vaccines, such as nanoparticulates and multivalents [1-12].
Objective
To examine data from the first months of the COVID-19 pandemia collected in all Brazilian regions as to positive results for COVID-19, symptoms and immunization status.
Method
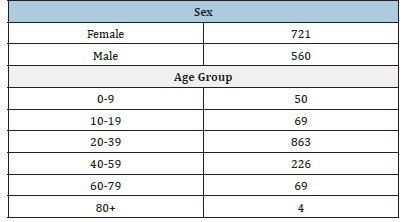
The study consisted of 19.949 participants, all testing spontaneously for COVID-19, divided by sex and age groups from zero to over 80 years old, followed weekly from April 2021 to January 2022 (Table 1). The COVID test was performed on the Point-of-Care Testing (POCT) platform, using a colorimetric test strip and the result is obtained in a few minutes. The POCT equipment Hilab (Hi Technologies, Curitiba/PR) uses internet Of Things (iOT) technology, which recognizes the unique QR code and sends the reaction information via cloud to the company’s laboratory in Curitiba, where a trained professional validates the reaction. The analysis of the reaction that arrives, regardless of the collection location, will ensure the absence of interferences to issue the signed report to those who made the registration and sample collection, at the health care service location, as well as to the patient’s smartphone, in up to 10 minutes. From each outpatient presenting at the health service location, forty microliters (40μl) of whole blood fingerstick sample were collected in capillary pipette, according to the instructions to avoid pre-analytical interference. The system of information exchange is encrypted in several layers. Personal information is dissociated from the results (different databases), making breach of confidentiality extremely difficult.
Table 1: Participants by sex and age.
The system of information exchange is encrypted in several layers. Personal information is dissociated from the results (different databases), making breach of confidentiality extremely difficult.
Result
All the collected results are presented in the form of table and Figure 1-6.
Figure 1:Distribution of participants per week.
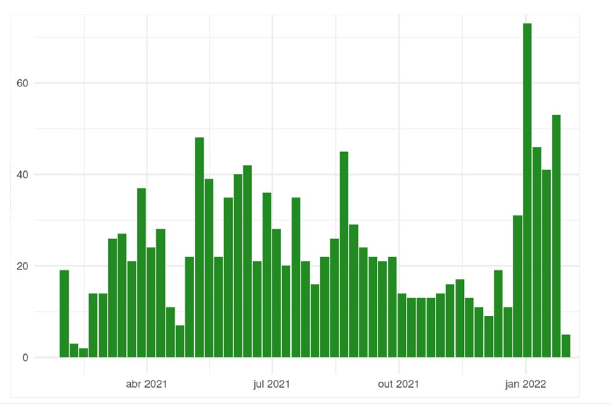
Figure 2:Distribution of participants and results by week.
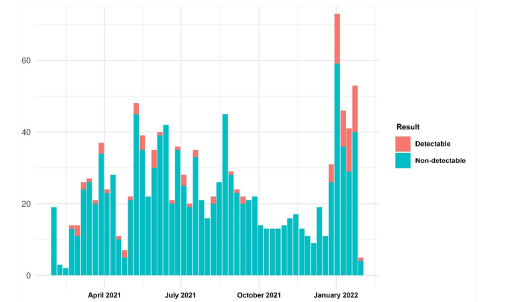
Figure 3:Distribution of positivity by week (%).
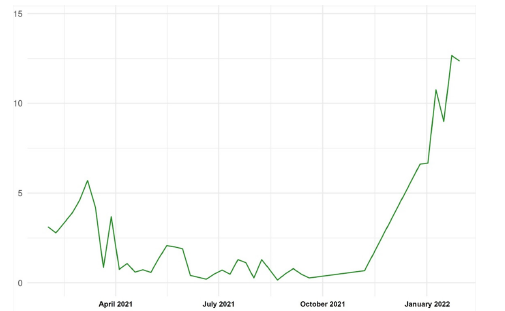
Figure 4:Vaccination distribution per week (%).
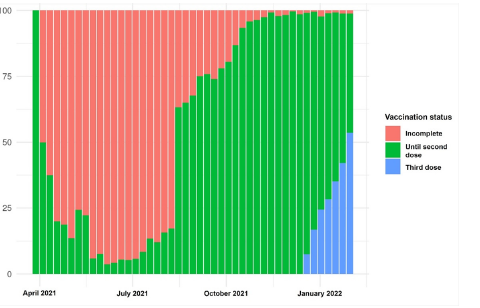
Figure 5:Distribution of patients’ symptoms per week (%).
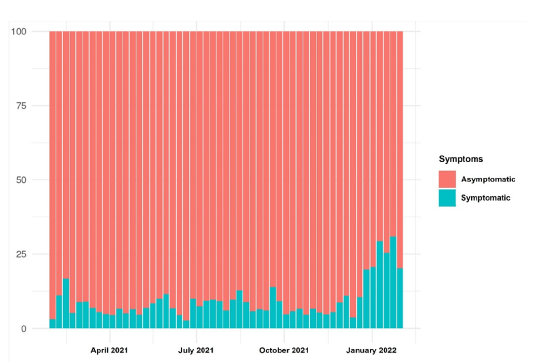
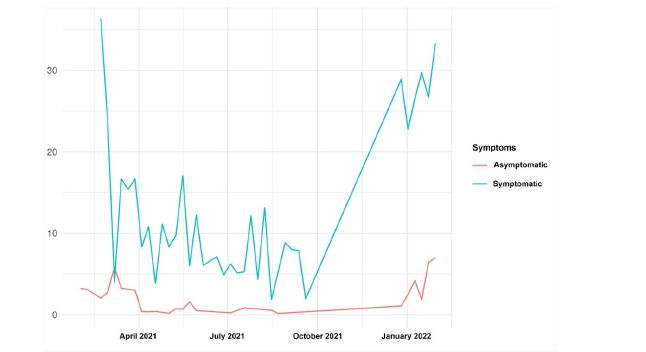
Figure 6:Distribution of positivity by symptoms per week (%).
Discussion
The data presented points to the very beginning of the pandemia. These data may give clues to designs of protocols for new vaccines. Health professionals do not have as yet an approved vaccine tested worldwide [13-18].
Conclusions
While current vaccines have played an essential role in controlling the pandemic, it is crucial to continue investing in genomic surveillance, technological innovation, and adaptive immunization strategies to ensure effective and long-lasting protection against SARS-CoV-2 and its emerging variants.
AcknowledgementS
To Mr. Marcus Vinícius Mazega Figueredo, PhD in Information Technology, CEO at Hi Technologies SA, for letting us make use of his database so as to compare to those of our hospital that’s been receiving patients from parts of all over Brazil.
References
- Martinez TLR, Saldanha ALR, Margeotto APP, Gasparoto AL, Guarnieri FH, et al. (2021) Outcome of dyslipidemic covid 19 patients in intensive care unit submitted to hemodialysis. Atherosclerosis 331: e212.
- Christodouleas DC, Kaur B, Chorti P (2018) From point-of-care testing to eHealth diagnostic devices (eDiagnostics). ACS Cent Sci 4(12): 1600-1616.
- Chen YT, Lee YC, Lai YH, Lim JC, Huang NT, et al. (2020) Review of integrated optical biosensors for point-of-care applications. Biosensors 10(12): 209.
- Jain S, Nehra M, Kumar R, Dilbaghi N, Hu TY, et al. (2021) Internet of medical things (IoMT)-integrated biosensors for point-of-care testing of infectious diseases. Biosens Bioelectron 179: 113074.
- Matteucci E, Della BL, Rossi L, Pellegrini G, Giampietro O (2014) Improving cardiocheck PA analytical performance: Three-year study. Clin Chem Lab Med 52(9): 1291-1296.
- Ferreira CES, Carolina NF, Correr CJ, Zucker ML, Andriolo A, et al. (2015) Clinical correlation between a point-of-care testing system and laboratory automation for lipid profile. Clin Chim Acta 446: 263-266.
- Lippi G, Plebani M, Favaloro EJ, Trenti T (2010) Laboratory testing in pharmacies. Clin Chem Lab Med 48(7): 943-953.
- Zucker ML, Anderson R, Carrara J, Coffman SK, Cursio C, et al. (2010) Selection criteria for point-of-care testing devices; approved guideline. POCT09-A. Clinical and Laboratory Standards Institute 30(8).
- Sumita NM. Guidelines for the management and quality assurance of Remote Laboratory Tests (RLT) of the Brazilian Society of Clinical Pathology/Laboratory Medicine (BSCP/ML) (2016). 2. ed. Barueri: Manole, 534 p. ISBN 978-85-7868-231-6.
- Ferreira CES, Guerra JCC, Slhessarenko N, Scartezini M, Franca CN, et al. (2018) Point-of-care testing: General aspects. Clin Lab 64(1): 1-9.
- Sumita NM, Ferreira CES, Martino MDV, Franca CN, Faulhaber ACL, et al. (2018) Clinical applications of point-of-care testing in different conditions. Clin Lab 64(7): 1105-1112.
- Price CP, Smith I, Bruel AVD (2018) Improving the quality of point-of-care testing. Fam Pract 35(4): 358-364.
- Tregoning JS, Flight KE, Higham SL, Wang Z, Pierce BF (2021) Progress of the COVID-19 vaccine effort: Viruses, vaccines and variants versus efficacy, effectiveness and escape. Nat Rev Immunol 21(10): 626-636.
- Barbari A (2021) COVID-19 vaccine concerns: Fact or fiction? Exp Clin Transplant 19(7): 627-634.
- Hassine HI (2022) Covid-19 vaccines and variants of concern: A review. Rev Med Virol 32(4): e2313.
- Li M, Wang H, Tian L, Pang Z, Yang Q, Huang T, et al. (2022) COVID-19 vaccine development: Milestones, lessons and prospects. Signal Transduct Target Ther 7(1): 146.
- Zhou Z, Zhu Y, Chu M (2022) Role of COVID-19 vaccines in SARS-CoV-2 variants. Front Immunol 13: 898192.
- Baer SL, Tran S (2022) COVID-19 vaccine efficacy in a rapidly changing landscape. J Investig Med 70(6): 1327-1328.
© 2025 Tania Leme da Rocha Martinez. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






