- Submissions

Full Text
Approaches in Poultry, Dairy & Veterinary Sciences
Establishment of Rapid Detection Method for Monkeypox Virus in Captive Primates
Yuyan YOU1*, Xinyi FU2, Chao Bai1, Dongxia WU2, Xin Hu1, Feier HAO1, Long CHEN1, Ning ZHANG1, Yan FENG1, Zhong WU1, Zengshuai ZHANG1, Wenlong WANG1, Jinxin GENG1 and Yanping Lu1*
1 Beijing Key Laboratory of Captive Wildlife Technologies, China
2 Thrive Bioresearch, China
*Corresponding author: Yuyan YOU & Yanping Lu, Beijing Key Laboratory of Captive Wildlife Technologies, Beijing Zoo, Beijing 100044, China
Submission: January 12, 2026;Published: February 18, 2026

ISSN: 2576-9162 Volume 10 Issue 1
Abstract
In this study, we focus on the zoo scenarios where frequent human-animal contact occurs. we aim to establish an efficient, accurate, convenient, and animal-friendly detection method for monkeypox virus. By adopting Colloidal Gold Immunochromatography (CGIA) combined with non-invasive sampling strategies, effective monitoring of monkeypox virus was achieved. The study prepared a colloidal gold immunochromatographic test strip and comprehensively validated its performance. The results showed that the test strip exhibited excellent specificity, high sensitivity (0.5ng/ml), good intra-batch and interbatch repeatability, strong stability, and was unaffected by faecal sample matrices. The test strip can rapidly complete detection within 10-15 minutes. This research provides a powerful support for reducing the risk of zoonotic diseases in captive primates, safeguarding the health of relevant staff and the public, and strengthening infectious disease prevention and control.
Keywords:Monkey pox virus; Colloidal gold immunochromatography; Primates; Feces
Introduction
Monkeypox was first documented in 1958 in laboratory monkeys in Denmark [1], while the first human case of infection was reported in the Democratic Republic of the Congo in 1970 [2]. A 2021 study conducted long-term monitoring on pathogens of 887 zoonoses and ranked them according to risk levels, with the results showing that Monkeypox virus (MPXV) and vaccinia virus in the genus Ortho poxvirus ranked 24th and 28th respectively [3]. Monkeypox, a zoonotic ortho poxvirus disease caused by MPXV, was designated as a “Public Health Emergency of International Concern (PHEIC)” by the World Health Organization (WHO) on 2022 [4,5]. At present, the complete host range of MPXV has not been fully identified. It is known that squirrels, Gambian pouched rats, groundhogs, non-human primates (e.g., chimpanzees, cynomolgus monkeys, vervet monkeys, red colobus monkeys, etc.) [6,7] and humans can all be infected with MPXV and serve as sources of infection [8-10]. In recent years, this disease has attracted widespread attention worldwide, as it not only threatens human health but also poses challenges to wildlife conservation and ecological balance.
Monkeypox Virus (MPXV) infection can cause skin rashes in non-human primates; in more severe cases, symptoms such as coughing, dyspnoea, anorexia, and facial edema may also occur [11]. MPXV can be detected in the damaged skin, secretions, and excretions of infected animals, and it can be transmitted via the respiratory tract, direct contact with broken skin, and ingestion of infected tissues [11]. Additionally, the virus can spread through mother-to-child transmission and sexual contact, and it is also transmissible during the incubation period [12- 17]. Since May 2022, monkeypox outbreaks have occurred in multiple non-endemic regions [18] and spread rapidly. From 2022 to August 31, 2023, a total of 1,099 confirmed monkeypox cases have been reported in China [19]. The number of infections has been on the rise year by year, making it a public health concern that requires special attention. Given the large variety, high population density and frequent human contact of animals in zoos, an outbreak of Monkeypox Virus (MPXV) infection would lead to extremely rapid transmission. This could result in collective infection among animals in the same breeding environment, as well as infections in keepers, tourists and other related personnel, thereby triggering an epidemic spread. Moreover, if MPXV circulates within animal populations, it may facilitate the emergence of new viral variants. Therefore, surveillance and rapid identification of MPXV infection are crucial for disease control [20].
At present, there are numerous methods for detecting monkeypox virus, mainly including virus isolation and culture, Electron Microscopy (EM) detection [21], and genetic detection methods such as Nucleic Acid Amplification Test (NAAT), Polymerase Chain Reaction (PCR), quantitative real-time PCR, Recombinase Polymerase Amplification (RPA), Next Generation Sequencing (NGS) and Restriction Fragment Length Polymorphism (RFLP). In addition, immunoassay methods such as Western blot, immunohistochemistry (IHC), Enzyme-Linked Immunosorbent Assay (ELISA) and colloidal gold enhanced immunochromatography assay (CGIA) are also available. Each of these methods has its own advantages and disadvantages in practical application [22]. The virus isolation and culture method was first applied to vaccinia virus culture in 1913. It can also be used for the detection of Monkeypox Virus (MPXV) with high accuracy but requires a Biosafety Level 3 (BSL-3) or higher laboratory and relies on experienced technicians [23]. Electron microscopy detection methods include Transmission Electron Microscope (TEM) and Scanning Electron Microscope (SEM) [24]. However, these methods have drawbacks such as complex sample preparation, long detection cycles, the need for professional operators, and high cost of equipment [25]. In terms of genetic testing, the Restriction Fragment Length Polymorphism (RFLP) method is time-consuming and requires prior virus isolation and culture. Polymerase Chain Reaction (PCR) and Quantitative Real-Time PCR (qPCR) are widely used due to their high accuracy and sensitivity [26], but they demand strict testing environments, professional technicians, and dedicated laboratory instruments [21-22].
Next-Generation Sequencing (NGS) technology is based on the principle of parallel sequencing. It can cover tens of thousands to millions of gene fragments in a single test and analyse hundreds of thousands to millions of DNA molecules simultaneously. Nevertheless, this technology has high costs and involves complex data analysis. For immunological detection, Western Blot and Immunohistochemistry (IHC) involve cumbersome procedures, are time-consuming, and have high requirements for venues, equipment, and personnel. Enzyme-Linked Immunosorbent Assay (ELISA) shows relatively good performance in detecting specific IgM and IgG antibodies in serum at 5 days and 8 days post monkeypox virus infection. Although the operation is relatively simple, laboratory technicians need to control factors that may affect results, such as antibody concentration, incubation time, and ambient temperature [21], which requires a certain level of professional expertise. Colloidal Gold Immunochromatographic Assay (CGIA) integrates colloidal gold technology with immunochromatography, serving as a rapid antigen-antibody detection method using colloidal gold as a tracer. Studies have shown that the application of CGIA in Escherichia coli has established it as a novel immunoassay technique following fluorescence labeling, enzyme labeling, and radioimmunoassay [27]. CGIA has advantages including simple operation, adaptability to most testing environments, low detection cost, no requirement for specialized equipment, and no need for chromogenic reagents. Compared with ELISA, it eliminates the steps involving toxic enzyme substrates and stop solutions, thus posing no toxic hazards to the human body [22].
This study focuses on zoos with frequent human-animal contact. It will establish a colloidal gold immunochromatographic assay for detecting monkeypox virus, combined with a non-invasive sampling strategy, to achieve efficient, accurate, convenient and animal-friendly detection of monkeypox virus. In view of this, the present study focuses on the Original West African Clade II of Monkeypox Virus (MPXV), which is characterized by a broader transmission range. In the context of zoo settings, we aim to establish a MPXV detection method based on the colloidal gold immunochromatographic assay, and further integrate this method with a non-invasive sampling strategy. The proposed approach is designed to enable efficient, accurate, convenient and animalfriendly detection of MPXV, thereby reducing the risk of potential zoonotic transmission from captive primates to relevant staff and susceptible populations, and providing robust technical support for public health security and infectious disease prevention and control.
Materials and Methods
Main materials and reagents
Nitrocellulose (NC) membrane, PVC backing plate, sample pad, gold conjugate pad, absorbent paper, plastic cassette, MPXV A29 (Original West African Clade Ⅱ) recombinant protein, anti-MPXV A29 labeled antibody, anti-MPXV A29 coated antibody, goat antimouse IgG antibody, disodium hydrogen phosphate dodecahydrate, Polyethylene Glycol (PEG 20000), potassium dihydrogen phosphate, anhydrous potassium carbonate, Proclin 300, β-mercaptoethanol, sodium hydroxide (NaOH), polyvinylpyrrolidone (PVP 10000), sodium caseinate, Tween 20, trisodium citrate dihydrate, chloroauric acid, sucrose, Bovine Serum Albumin (BSA), S9 surfactant, glycerol.
Establishment of colloidal gold immunochromatographic test strip
Preparation of Colloidal Gold Solution: Add ultrapure water accounting for approximately two-thirds of the volume into a 250ml round-bottom flask, then place the flask on a magnetic stirring heating mantle. Set the temperature to 150 °C and start slow stirring. When the ultrapure water in the flask boils, start timing for 5 minutes, then discard the ultrapure water inside the flask. Re-add 150ml of ultrapure water and wait for it to boil for 5 minutes. Next, add 150μl of 10% chloroauric acid solution into the round-bottom flask, and heat with stirring for 5 minutes. Quickly add 225μl of 10% trisodium citrate solution into the flask; the colour of the solution will change sequentially from yellow to gray, then to black, and finally to purple. After the colour of the solution stabilizes for 15 minutes, the purple-red colloidal gold solution is obtained. Take 1ml of the colloidal gold solution and place it into a quartz cuvette. Use an ultraviolet-visible spectrophotometer to measure the absorption spectrum in the visible light region, and judge the quality and particle size of the colloidal gold solution according to the maximum absorption peak and the width of the main peak.
Treatment of gold conjugate pad and sample pad: Mix disodium hydrogen phosphate dodecahydrate, potassium dihydrogen phosphate, PVP10000, BSA, Tween 20, sucrose, sodium caseinate and S9 surfactant in appropriate proportions, then add ultrapure water and mix well to prepare the gold conjugate pad treatment solution. Mix disodium hydrogen phosphate dodecahydrate, potassium dihydrogen phosphate, PVP10000, PEG, BSA, sodium caseinate and S9 surfactant in appropriate proportions, then add ultrapure water and mix well to prepare the sample pad treatment solution. Immerse the gold conjugate pad and sample pad in their corresponding treatment solutions respectively for 30 minutes, squeeze out the excess water, then place them in a blast drying oven and dry them overnight at 37 °C.
Antibody coating: Using the XYZ three-dimensional largeplane membrane scribing and gold spraying instrument, coat the Nitrocellulose (NC) membrane with goat anti-mouse IgG antibody at a mass concentration of 0.5mg/mL and anti-MPXV A29 coating antibody at a mass concentration of 0.8mg/mL at a scribing volume of 1μL/cm, which serve as the control line (C-line) and test line (T-line), respectively. The final coating amounts are 0.5mg/cm and 0.8mg/cm, correspondingly. Then place the membrane in a blast drying oven at 37 °C and dry it overnight.
Preparation of gold conjugate solution: Mix disodium hydrogen phosphate dodecahydrate, potassium dihydrogen phosphate, Bovine Serum Albumin (BSA), and sucrose in a certain ratio, add ultrapure water to homogenize the mixture, and thus obtain the gold conjugate solution.
Labeling of gold-labeled antibody complex: Add 1mL of the above-prepared colloidal gold solution, 100μL of 0.1% potassium carbonate solution, and 8μg of anti-MPXV A29 labeling antibody into a 1.5mL centrifuge tube. Place the tube on a rotary mixer and incubate slowly in the dark for 40min. Then add 100μL of 10% Bovine Serum Albumin (BSA) solution and incubate slowly in the dark for another 40min under the same conditions. Centrifuge the solution at 13,000 rpm and 4 °C for 30min, then discard the supernatant. Finally, resuspend the gold-labeled antibody complex to a volume of 100μL with the gold conjugate solution, spray it onto the gold conjugate pad using the XYZ three-dimensional largeplane membrane scribing and gold spraying instrument, and dry it at 37 °C for later use.
Assembly of test strips
Adhere the absorbent paper, the aforementioned gold conjugate pad, and the aforementioned sample pad to the PVC backing plate with the antibody-coated Nitrocellulose (NC) membrane in sequence. Cut the assembled plate into test strips with a width of 3mm, and place the strips into plastic cassettes for use.
Sample processing method
Add 0.5mL of mercaptoethanol, 3mL of glycerol, 1g of polyethylene glycol, 1g of sodium caseinate, and 1mL of Proclin 300 to 0.02M phosphate buffer. Adjust the pH of the solution to 7.0 with sodium hydroxide and dilute it to a final volume of 100mL to obtain the sample treatment solution. Dip a cotton swab into the feces of primates, with the feces covering approximately 1/3-1/2 of the swab. Insert the swab into a preservation tube containing 1mL of the sample treatment solution, stir to mix thoroughly, and then let it stand for 3-5min to obtain the sample supernatant.
Detection method using test cassettes
Pipette 100μL of the sample supernatant and add it to the sample well of the test cassette. Wait for 10-15min and observe the line development to determine the test result. The C-line serves as the control line; the appearance of this line indicates a valid test result. The T-line serves as the test line: a visible band denotes a positive result, while no visible band denotes a negative result. The higher the viral load in the sample, the darker the colour of the developed line.
Performance verification of test cassettes
Prepare three batches of test cassettes in accordance with the established test cassette system, with no fewer than 200 test cassettes per batch. Conduct specificity tests, sensitivity tests, repeatability tests, and stability tests on the prepared test cassettes, and perform clinical application verification using different sample matrices to complete the performance evaluation and verification of the test cassettes.
Result
Quality detection of colloidal gold solution
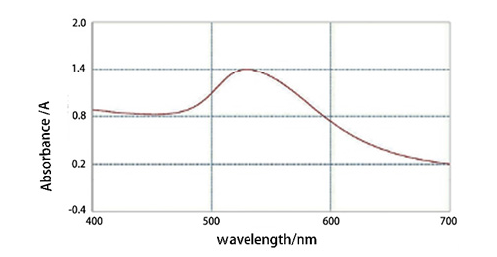
The maximum absorption peak of the colloidal gold solution used in this study was at 530nm with an extremely narrow peak width (Figure 1). According to the conclusion proposed by Peng Jianchun (2000) [28] that a narrower main peak width indicates more uniform colloidal gold particles, combined with the regression equation between maximum absorption peak and particle size: Y=0.4271X+514.56, it was confirmed that the colloidal gold solution had uniform particles with a particle size range of approximately 35-40nm.
Figure 1:Absorption spectrum of colloidal gold in visible region (400-700nm).
Specificity test
Figure 2:Absorption spectrum of colloidal gold in visible region (400-700nm).
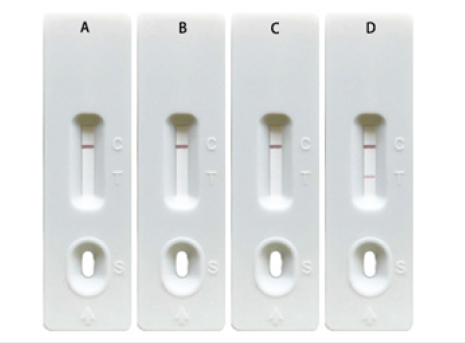
The inactivated SARS-CoV-2, inactivated rabies virus, negative control (sample diluent) and positive control (10ng/mL MPXV A29 recombinant protein) were added to the sample well of the test card. Except for the positive control, no magenta bands appeared at the T-line in the test results of the inactivated SARS-CoV-2, inactivated rabies virus and negative control (Figure 2). This result demonstrates that the test card has good specificity.
Sensitivity test
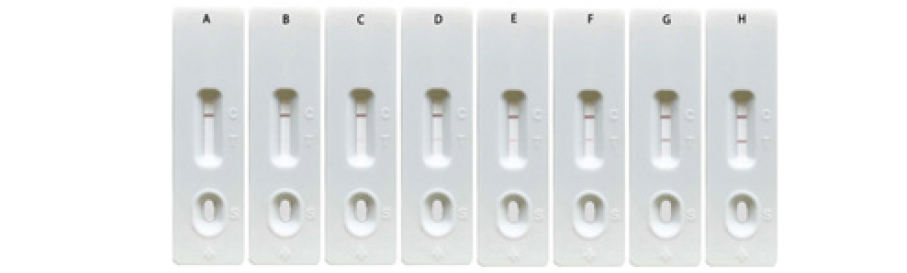
A concentration gradient of MPXV A29 recombinant protein was prepared at 100ng/mL, 50ng/mL, 10ng/mL, 5ng/mL, 1ng/ mL, 0.5ng/mL, and 0ng/mL. Then 100μL of the diluted MPXV A29 recombinant protein was added to each sample well. The results showed that there was a significant difference in the colour intensity of the test line between the 0.5ng/mL MPXV A29 recombinant protein group and the 0.25ng/mL group. No colour appeared on the test line when the concentration of MPXV A29 recombinant protein was lower than 0.5ng/mL (Figure 3). The sensitivity of the test card was determined to be 0.5ng/mL.
Figure 3:Sensitivity test of detection card. (A.0ng/ml, B.0.25ng/ml, C.0.5ng/ml, D.1ng/ml, E.5ng/ml, F.10ng/ml, G.50ng/ml, H.100ng/ml).
Repeatability test
Figure 4:Repeatability test in test card batch. (I, II and III are three groups of repeated experiments; A. 0ng/ml, B. 0.5ng/ml, C.1ng/ml, D.10ng/ml).

Intra-batch repeatability: A series of MPXV A29 recombinant protein solutions with mass concentrations of 10ng/mL, 1ng/mL, 0.5 ng/mL and 0 ng/mL were prepared by dilution. Twelve test strips from the same batch were selected and divided into 4 groups with 3 strips per group. Samples of the same concentration were added to the sample wells of each group, and the band development was observed. The colour intensities of both the Control Line (C line) and Test Line (T line) were uniform across all test strips, and the detection results were completely consistent (Figure 4). These findings demonstrate that the test strips possess excellent intrabatch repeatability.
Batch-to-batch reproducibility: The MPXV A29 recombinant protein was serially diluted to a concentration gradient of 10ng/ mL, 1ng/mL, 0.5ng/mL, and 0ng/mL. Three different batches of test strips were selected, with 4 strips per batch. Samples of the same concentration were added to the sample wells of the test strips from the three batches, respectively, and the line development was observed. The colour intensities of both the C and T lines on each test strip were uniform, and the test results were completely consistent (Figure 5). These findings demonstrate that the test strips have excellent batch-to-batch reproducibility.
Figure 5:Repeatability test between test cards. (I, II and III were repeated experiments in three batches respectively; A.0ng/ml, B.0.5ng/ml, C.1ng/ml, D.10ng/ml).

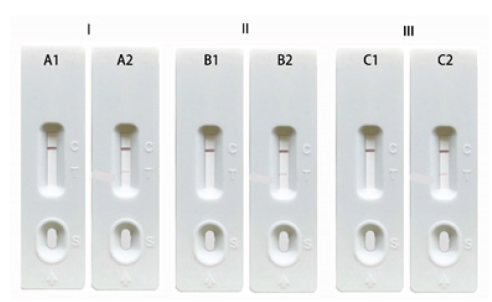
Stability test: The prepared test strips were placed in an oven at 37 °C. At 7d, 15d and 30d, 2 test strips were taken out respectively. 0ng/mL and 1ng/mL MPXV A29 recombinant protein samples were added to the sample wells, and the line development was observed. The colour intensities of both the C and T lines on each test strip were uniform, and the test results were completely consistent (Figure 6). These results demonstrate that the test strips have good stability.
Figure 6:Test card stability test. (A1.7d, 0ng/ml; A2. 7d, 1ng/ml; B1.15d, 0ng/ml; B2. 15d, 1ng/ml; C1. 30d, 0ng/ml; C2. 30d, 1ng/ml).
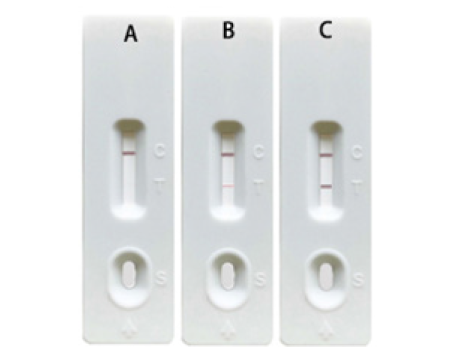
Clinical application of the method: A total of 80 fecal samples were collected from primates, including Yunnan snub-nosed monkeys, Sichuan snub-nosed monkeys, rhesus monkeys, longtailed monkeys, François’ langurs, and ring-tailed lemurs. The fecal samples were placed into sample diluent and processed according to the method described in section 1.4 to obtain sample supernatants. MPXV A29 was then added to the supernatants as clinical samples to prepare spiked samples with final mass concentrations of 50ng/ mL, 1ng/mL, and 0ng/mL. After detection using the test strips, it was found that the T-line colour development of samples from different primate species showed consistent results, and the colour intensity (Figure 7) was identical to that shown in Figure 1. These findings indicate that the test strips are not affected by sample matrix interference and can effectively detect monkeypox virus in clinical samples.
Figure 7:Sample addition test. A.0ng/ml spiked samples, B.1ng/ml spiked samples, C.50ng/ml spiked samples.
Discussion
In the field of Monkeypox Virus (MPXV) prevention and control research, the indirect Enzyme-Linked Immunosorbent Assay (ELISA) established by Zhu Junda et al. [29] for MPXV antibody detection and the throat swab viral antigen detection method developed by Chen Guoqiang et al. [30] both rely on professional technicians, dedicated laboratory spaces and specialized equipment [31], making them difficult to meet the requirements of on-site rapid detection. The semi-quantitative method for MPXV antigen detection using immunochromatographic test strips established by Lai Xiangde et al. [32] is also not suitable for the rapid screening of animal diseases due to its reliance on serum samples and complex operational procedures. Against this backdrop, this study focused on addressing the challenges of MPXV detection in captive primates. By using fecal samples and the colloidal gold immunochromatographic assay, the study achieved non-invasive sampling, reduced the detection threshold and lowered the associated costs.
Technical advantages of colloidal gold assay
Specificity and sensitivity of colloidal gold assay: The colloidal Gold Immunochromatographic Assay (CGIA) detection system established in this study exhibits excellent technical characteristics in Monkeypox Virus (MPXV) detection, and a rapid MPXV detection system based on CGIA has been successfully constructed. With the MPXV A29 protein as the target analyte, the optimal system conditions were determined as follows: The antibody coating concentration was set at 1mg/mL, and the antibody labeling amount was 10μg. This assay achieves a detection sensitivity of 0.5ng/mL and shows no cross-reactivity with severe acute respiratory Syndrome Coronavirus 2 (SARS-CoV-2) or rabies virus, demonstrating high specificity. It enables accurate identification of MPXV antigens and thus reduces the risk of misdiagnosis. Compared with other rapid detection technologies of the same category, this sensitivity level stands in a leading position. The assay is capable of capturing signals of low-concentration viral infection in a timely manner, which gains valuable time for early epidemic warning, prevention and control, and facilitates the achievement of the prevention and control goal of early detection and early intervention.
Stability and repeatability of the colloidal gold detection method: In the intra-batch repeatability test of the colloidal gold detection method, the same batch of test strips was used for repeated detection of samples with different concentrations. The colour development intensity of both the Control Line (C line) and Test Line (T line) of all test strips remained highly consistent, indicating stable and reliable detection results. In the inter-batch repeatability test, test strips from different batches were applied to detect the same samples, and good consistency was also achieved. These results demonstrate that the detection method is not affected by differences in laboratory environments or changes in operators and possesses high stability and repeatability. The detection performance of the test strips showed no significant changes after 7 days of storage at 37 ℃, exhibiting strong stability which facilitates storage, transportation and popularization. Moreover, the fecal sample matrix did not interfere with the detection results. To simulate extreme storage conditions, the test strips were placed in an accelerated aging environment at 37 ℃ and continuously observed for 30 days. The results showed that the test strips maintained stable detection performance throughout the observation period, with no obvious attenuation in the colour development effect of the C line and T line. This characteristic greatly facilitates the storage and transportation of the test strips, laying a solid foundation for their wide application in different regions and under various conditions.
Innovation and significance of non-invasive sampling strategy
The non-invasive sampling strategy constitutes a major innovation of this study. Given practical challenges such as the high difficulty in capturing captive wild animals and their high sensitivity to stress responses, this study innovatively adopts fecal samples as detection materials. Using a self-developed special sample treatment solution, fecal samples were efficiently processed to successfully obtain sample supernatants suitable for detection. Clinical application results show that this detection system is not interfered by the complex matrix of fecal samples and can stably and accurately identify monkeypox virus antigens in fecal samples from different primate species. This non-invasive sampling strategy minimizes harm and stress responses to animals during sampling, fully embodies the concept of animal welfare protection, and is conducive to maintaining the health status of captive primates. On the other hand, fecal sample collection is simple and easy to implement, requiring no professional capture equipment or techniques. It enables large-scale and periodic sample collection and detection, significantly improving the efficiency and feasibility of animal health monitoring, and providing an innovative solution for the prevention and control of diseases in captive primates.
Application value and public health significance of the colloidal gold detection method
In animal-intensive venues with frequent human-animal contact such as zoos and wildlife breeding bases, the introduction of monkeypox virus could easily trigger mass infection incidents. Infected animals are not only threatened by health risks, but may also transmit the virus to breeders, tourists and other groups through direct contact, respiratory transmission and other routes, resulting in cross-species transmission and spread of the epidemic. The detection method established in this study perfectly meets the demand for large-scale screening with its prominent advantages such as low cost, simple operation, and no need for professional instruments, equipment or technical personnel. A single test can yield results within only 10-15 minutes, enabling rapid and efficient virus detection in a large number of captive primates. Regular screening can help detect occult infected individuals in a timely manner, promptly implement prevention and control measures such as isolation and treatment, effectively cut off the virus transmission chain, and reduce the risk of zoonotic disease transmission. This method not only protects animal health and maintains the normal operation order of zoos but also plays an important role in safeguarding the health and safety of staff and the public, consolidating the public health defence line, and providing strong technical support for infectious disease prevention and control work.
Fund Project
National Natural Science Foundation of China (No. 31872257); Beijing Zoo Project (No. KGBZ202301).
References
- Von Magnus P, Andersen EK, Peteraen KB (1959) A pox-like disease in cynomolgus monkeys. Acta Pathol Microbiol Scand 46(2): 156-176.
- Ladnyj ID, Ziegler P, Kima E (1972) A human infection caused by monkeypox virus in Basankusu Territory. Democratic Republic of Congo. Bull World Health Organ 46(5): 593-597.
- Grange Z, Goldstein T, Johnson CK, Simon A, Kirsten G, et al. (2021) Ranking the risk of animal-to-human spillover for newly discovered viruses. PNAS 118(15): e2002324118.
- Wang Z, Zhang Z, Wu W (2023) Diagnosis, prevention and treatment of monkeypox. Acta Veterinaria et Zootechnica Sinica 54(8): 3195-3205.
- WHO (2005) Second meeting of the International Health Regulations (IHR) emergency committee regarding the multi-country outbreak of monkeypox EB/0L.
- Zhou T, Jia HJ, He XB (2012) Research progress on epidemic status and host spectrum of zoonotic poxvirus diseases. Chinese Journal of Zoonoses 28(2): 159-163.
- Breman JG, Bernadou J, Nakano JH (1977) Poxvirus in West African nonhuman primates: Serological survey results. Bull World Health Organ 55(5): 605-612.
- Parker S, Buller RM (2013) A review of experimental and natural infections of animals with monkeypox virus between 1958 and 2012. Future Virology 8(2): 129-157.
- Sklenovská N, Van Ranst M (2018) Emergence of monkeypox as the most important Orthopoxvirus infection in humans. Front Public Health 6: 241.
- Li X, Yang J, Zhao HP (2023) Etiology, epidemiological characteristics, detection, prevention and control technologies of monkeypox. Chinese Journal of Animal Health Inspection 40(2): 51-57.
- Jing W, Fang YX, He XB (2024) Epidemic status of monkeypox, risk of animal spillover and prevention and control strategies and measures in China. Chinese Journal of Veterinary Science 54(01): 101-108.
- Ogoina D, Izibewule JH, Ogunleye A, Ebi E, Uchenna A, et al. (2019) The 2017 human monkeypox outbreak in Nigeria-report of outbreak experience and response in the Niger Delta University Teaching Hospital, Bayelsa State, Nigeria. Public Library of Science ONE 14(4): e0214229.
- World Health Organization. Multi-country monkeypox outbreak: Situation update EB/OL (2022-06-27)2022-11-21.
- Kozlov M (2022) Monkeypox outbreaks: 4 key questions researchers have. Nature 606(7913): 238- 239.
- Parker S, Buller RM (2013) A review of experimental and natural infections of animals with monkeypox virus between 1958 and 2012. Future Virol 8(2): 129-157.
- Kozlov M (2022) Monkeypox outbreaks: 4 key questions researchers have. Nature 606(7913): 238-239.
- Grant R, Nguyen LBL, Breban R (2020) Modelling human-to-human transmission of monkeypox. Bull World Health Organ 98(9): 638-640.
- Bhagavathula AS, Khubchandani J (2022) Monkeypox outbreaks and global health emergency declaration: Can such declarations influence public interest in the disease. Brain Behav Immun 106: 113-114.
- Han H, Wu B, Jia JJ (2023) Risk assessment of the global monkeypox epidemic in September 2023. Port Health Control 28(6): 52-55.
- Wang Y, Han SY, Wei FH (2023) The role of wild animals in monkeypox virus transmission. Chinese Journal of Veterinary Medicine 59(10): 100-104.
- Zhou P, Zhu H, Zhao XN (2025) Research progress on monkeypox virus evolution and detection technology. New Farmers 5: 78-80.
- Wang MT, Cui FQ (2023) Epidemiological characteristics and current status of prevention and control of monkeypox. Jiangsu Journal of Preventive Medicine 34 (1): 8-11.
- Kong FD, Gong YQ, Chen Q (2025) Research progress on laboratory detection methods for monkeypox virus. China Port Science and Technology 7(2): 26-33.
- Sun Y, Zhou LH (2020) Application of electron microscopy technology in medicine: A review of application of electron microscopy technology in the medical field. Journal of Chinese Electron Microscopy Society 39(2): 232-233.
- Reed KD, Melski JW, Graham MB, Russell LR, Mark JS, et al. (2004) The detection of monkey-pox in humans in the Western Hemisphere. New England Journal of Medicine 350(4): 342-350.
- Jiangsu (2013) Entry-exit inspection and quarantine bureau of the people's Republic of China. Quarantine Protocol for Monkeypox: SN/T 3487-2013 S. Jiangsu: General Administration of Quality Supervision, Inspection and Quarantine of the People's Republic of China, p. 1.
- Faulk WP, Taylor GM (1971) An immuno colloid method for the electron microscope. Immunochemistry 8(11): 1081-1083.
- Peng JC, Liu XD, Ding XP (2000) Evaluation of colloidal gold particle size and distribution by visible light spectroscopy. Bulletin of the Academy of Military Medical Sciences 24(3): 211-212.
- Zhu JD, Wang S, Ren SN (2022) Establishment of an indirect ELISA antibody detection method based on monkeypox virus A27L protein. Chinese Journal of Veterinary Medicine 58(10): 1-7.
- Chen GQ, Chen L, Zhang JY (2011) Development and application of a PCR detection kit for monkeypox virus. Animal Husbandry and Veterinary Medicine 43(10): 66-68.
- Zhou MJ, Li XP, Dai RY (2019) Research progress on immunochromatographic strip detection technology. Laboratory Medicine and Clinic 16(22): 5.
- Lai XD, Cao PP, Peng YN (2023) Establishment of a semi-quantitative method for monkeypox virus antigen detection by immunochromatographic strip based on rapid colloidal gold synthesis via microwave method. Chinese Journal of Zoonoses 39(7): 617-621.
© 2026 Yuyan YOU & Yanping Lu. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






