- Submissions

Full Text
Aspects in Mining & Mineral Science
A Mini-Review on the Conversion of Biomass-Derived Compounds to Fuel-additives and Fine Chemicals
Boggala Sasikumar1,2*, Patola Gandhi1 and Akula Venugopal1,2
1 Department of Catalysis and Fine Chemicals, CSIR-Indian Institute of Chemical Technology, India
2 Academy of Scientific and Innovative Research, India
*Corresponding author:Boggala Sasikumar, Department of Catalysis and Fine Chemicals, CSIR-Indian Institute of Chemical Technology, Hyderabad, 500007, Telangana state and Academy of Scientific and Innovative Research, Ghaziabad, Uttar Pradesh, 201002, India
Submission: February 11, 2026: Published: April 08, 2026

ISSN 2578-0255Volume14 Issue 5
Abstract
The progressive depletion of fossil resources and the severe environmental impacts associated with their utilization have accelerated the development of renewable and sustainable pathways for the production of fuels and chemicals. Lignocellulosic biomass has emerged as an abundant, low-cost, and carbon-neutral feedstock that can be efficiently converted into versatile platform molecules through catalytic processing. This review provides a comprehensive overview of biomass feedstocks, their extraction and upgrading strategies, and the catalytic synthesis of key platform chemicals such as levulinic acid and furfural. Particular emphasis is placed on the subsequent valorization of these intermediates into sustainable fuel additives and value-added chemicals. The role of heterogeneous catalytic systems, especially Ni-based catalysts, in the biomass conversion reactions.
Keywords:Biomass; Fuel-additives; Heterogeneous catalysts; Levulinic acid; Furfural
Introduction
Modern society is strongly dependent on fossil-based fuels and chemicals, leading to substantial greenhouse gas emissions, environmental degradation, and growing concerns regarding long-term resource sustainability. In response, the refinery and chemical industries are increasingly transitioning toward renewable feedstocks and environmentally benign catalytic processes. The rising global demand for clean energy and sustainable chemicals has accelerated research into the catalytic upgrading of biomass-derived resources for the production of fuels and value-added chemicals [1,2]. Biomass represents a viable alternative to fossil resources, as it is the only renewable and sustainable source for the large-scale production of fuel and chemicals. Biomass feedstocks are generally classified into sugars, triglycerides, and lignocellulosic materials. Conversion of biomass into useful chemicals is typically achieved through three major catalytic pathways: gasification, pyrolysis, and hydrolysis [3]. Among these, lignocellulosic biomass owing to its widespread availability, economic viability, and sustainability advantages, lignocellulosic biomass has emerged as the most promising renewable resource for future fuel and chemical production [1,4]. Fuel additives derived from biomass are particularly attractive because they can be blended with conventional fuels to improve combustion efficiency and reduce emissions without major modifications to engines or distribution systems. Levulinic acid can be efficiently valorized into a spectrum of products, including γ-valerolactone, valeric acid, levulinate esters, and other fuel-relevant oxygenates, while furfural serves as a key precursor for furfuryl alcohol, tetrahydrofuran derivatives, and related fine chemicals and these have demonstrated favorable physicochemical properties, including high octane or cetane numbers, low vapor pressure, and improved soot reduction [5].
Extraction Biomass Feedstocks and their Conversions
Agricultural residues (e.g., corn stover, wheat straw, and rice husk), forestry residues, and dedicated energy crops are widely studied owing to their high carbohydrate content and broad availability. The catalytic conversion of biomass into sustainable production of fuel-additives and value-added chemicals. As illustrated in Figure 1, the three principal biomass components includes cellulose, hemicellulose, and triglycerides undergo distinct catalytic pathways through sequential chemical transformations generated the fine-chemicals and fuel-additives [3]. Lignocellulosic biomass is initially depolymerized via acid-catalyzed hydrolysis to glucose, which can be isomerized to fructose. These hexose sugars are readily dehydrated to form 5-Hydroxymethylfurfural (HMF), a pivotal platform molecule in biomass valorization. HMF and its derivatives undergo a variety of reactions, including hydrogenation, etherification, acetalization, and carbon–carbon coupling, producing oxygenated intermediates [6].
Figure 1:Process of convert biomass into valuable chemicals and fuel additives.
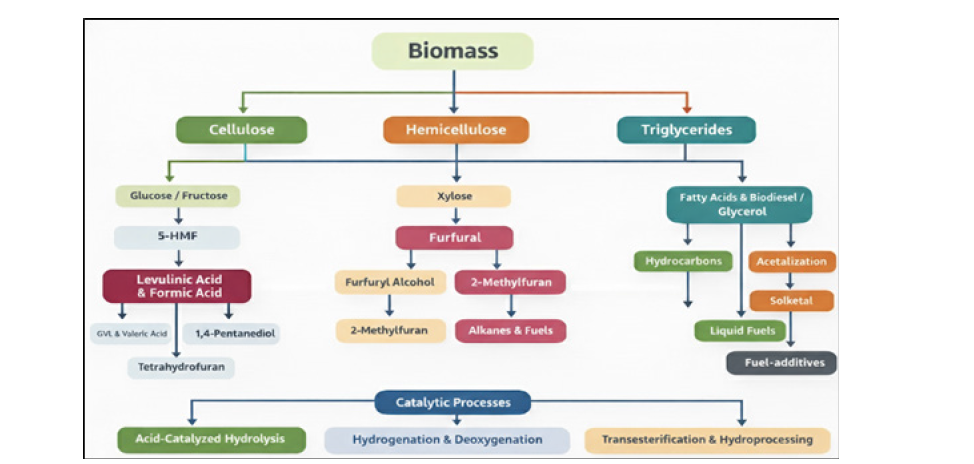
Levulinic acid is typically produced via acid-catalyzed conversion of cellulose or hexose sugars, involving cellulose depolymerization, dehydration to HMF, and subsequent rehydration to levulinic acid and formic acid [6]. Although mineral acids offer high catalytic activity, issues related to corrosion, catalyst recovery, and waste generation have driven the development of solid acid catalysts and solvent-assisted systems. Hydrogenation and hydrodeoxygenation of levulinic acid produce γ-Valerolactone (GVL), velaric acid, 1,4-penatnediol and tetrahydrofuran derivatives. These transformations required the critical role of bifunctional acid-metal catalysts in facilitating tandem dehydration-hydrogenation processes [7-9]. In contrast, hemicellulose is more readily hydrolyzed and predominantly generates pentose sugars, particularly xylose. Dehydration of xylose leads to furfural, which is a key platform molecule for hemicellulose upgrading. Furfural can be selectively hydrogenated to furfuryl alcohol, transformed into alkylated furans such as 2-methylfuran, or subjected to aldol condensation and oligomerization to build larger carbon frameworks [10]. These intermediates are subsequently hydrogenated and hydro-deoxygenated to produce higher hydrocarbons and liquid alkanes.
The hemicellulose-derived pathway is characterized by efficient carbon utilization and relatively mild reaction conditions, making furfural-based chemistry especially attractive for sustainable fuel production. Triglycerides, derived from vegetable oils and animal fats, follow more direct upgrading routes compared to carbohydrate-based biomass. Hydrolysis or transesterification of triglycerides produces fatty acids, fatty acid esters (biodiesel), and glycerol. Fatty acids and esters are efficiently converted into long-chain hydrocarbons via hydrotreating, decarboxylation, and ketonization reactions [11]. The glycerol can be further valorized into glycerol-carbonate, solketal, hydrocarbons, esters and ethers through carbonylation, acetalization, hydrogenation, and alkylation pathways using acid catalysts [12]. Recent advancesincluding biphasic reaction systems, continuous-flow reactors, and heterogeneous acid catalysts-have significantly improved process efficiency and sustainability. The choice of biomass feedstock plays a crucial role in the sustainable production of key platform molecules such as levulinic acid and furfural.
Importance of Heterogeneous Catalysis in Biomass Valorization
Heterogeneous catalysis plays a central role in the efficient conversion of biomass-derived platform molecules. Compared to homogeneous catalysts, heterogeneous systems offer advantages such as ease of separation, catalyst recyclability, reduced corrosion, and compatibility with continuous-flow processing. In biomass upgrading reactions, catalysts must operate in complex reaction environments that often include water, acids, and oxygenated compounds. These conditions place stringent demands on catalyst stability and resistance to deactivation. As a result, catalyst design strategies focus on optimizing metal dispersion, support properties, and metal-support interactions [13]. Nickel-based catalysts have attracted considerable attention as cost-effective alternatives to noble metals for biomass upgrading reactions. Nickel exhibits strong hydrogenation and hydrogenolysis activity and is significantly more abundant and affordable than precious metals such as Ru, Pd, and Pt. The catalytic performance of Ni-based systems is strongly influenced by metal particle size, dispersion, acidity and metalsupport interactions. Oxide supports such as Al2O3, ZrO2, TiO2, CeO2 and micro and mesoporous materials supports like zeolites, Al-MCM-41 and functionalized silica can modify the electronic properties of Ni, while carbon-based supports often enhance metal dispersion and resistance to sintering [13]. Bifunctional catalysts combining Ni metal sites with acidic supports are particularly effective for cascade reactions involving hydrogenation and dehydration steps. However, excessive acidity can promote side reactions and coke formation, necessitating careful control of acid site density and strength [14].
Catalytic Conversion of Levulinic Acid and Furfural into Fuel-Additives
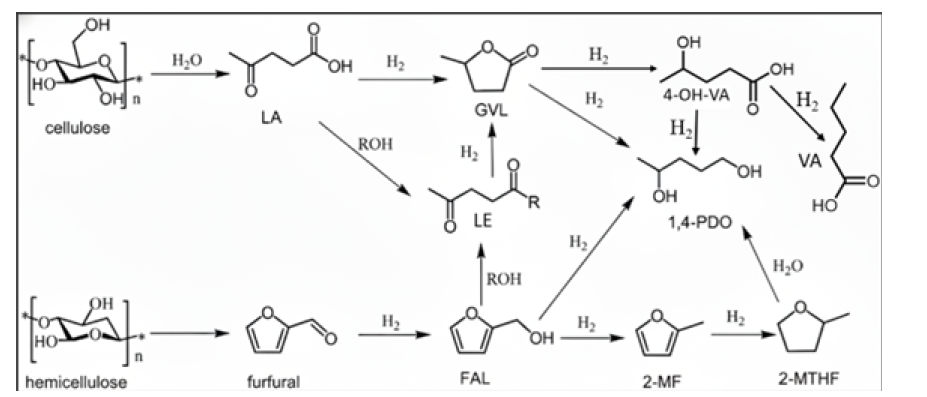
As shown in Figure 2, the hydrogenation of levulinic acid to γ-valerolactone is one of the most extensively studied biomasses upgrading reactions due to the versatility of GVL as a fuel additive and green solvent. The reaction typically proceeds via hydrogenation of the carbonyl group to form 4-hydroxypentanoic acid, followed by intramolecular esterification. Ni-based catalysts generally require higher reaction temperatures compared to noble metals but can achieve high selectivity under optimized conditions. The presence of formic acid, generated during levulinic acid production, can serve as an in-situ hydrogen source, further improving process integration [15]. Beyond GVL, levulinic acid can be upgraded to valeric acid and 1,4-pentanediol, which are attractive diesel-range fuel additives and fine chemicals respectively. These transformations typically involve ring opening of GVL followed by hydrogenation (Figure 2), [7]. Ni-based catalysts supported on acidic materials enable efficient conversion by providing both hydrogenation activity and acid-catalyzed ring-opening of GVL, particularly on Brønsted acidic sites. Control over reaction conditions is essential to suppress cracking and over-hydrogenation reactions [13].
Likewise, furfural is a highly versatile platform molecule that can be converted into a wide range of products through hydrogenation, hydrogenolysis, and rearrangement reactions (Figure 2). The selective hydrogenation of furfural to furfuryl alcohol is an important industrial process and reductive amination of furfural to furfurylamine used extensively as an intermediate synthesis of pharmaceuticals, agrochemicals and resins [10,16]. Nibased catalysts can selectively hydrogenate the carbonyl group of furfurals, while reaction severity and catalyst properties determine further hydrogenation to tetrahydrofurfuryl alcohol or ringopening products. Catalyst design strategies aim to balance activity and selectivity while minimizing deactivation [13,14].
Figure 2:Catalytic conversion routes of lignocellulosic carbohydrates to fuel-additive and value-added oxygenate compounds.
Applications as Fuel Additives and Fine Chemicals
World consumes roughly 103 million barrels of fossil-derived gasoline (petrol) and diesel per day as of 2024, which is equivalent of over 37 billion barrels annually. Therefore, cost of these oils were increased day-by-day, which can also affect on the environment. Air pollution from fossil fuel combustion is linked to millions of premature deaths annually and economic damages amounting to trillions of USD per year globally due to health costs and lost productivity. The combustion of diesel and petrol releases CO2 and other pollutants (such as NOx, CO, and particulate matter) that contribute to climate change and local air quality degradation, underscoring the need for cleaner alternatives. To reduce the carbon intensity and emissions profile of transport fuels while maintaining compatibility with existing infrastructure, biomassderived oxygenates and bio-blend stocks are increasingly used as additives in diesel and petrol.
Oxygenated bio-additives such as bioethanol, biomassderived compounds are widely blended with gasoline to increase the octane number and enhance combustion efficiency, leading to reduced emissions of unburned hydrocarbons and carbon monoxide. Bio-alcohols can also be blended with fuels to increase oxygen content and suppress particulate matter formation; however, their lower energy density and variable cetane number influence key fuel properties such as viscosity, ignition delay, and overall engine performance. Fatty-esters (biodiesel) represent the most established class of renewable diesel additives, offering improved lubricity, biodegradability, and general compatibility with compression-ignition engines, although higher blending ratios may result in increased NOx emissions, necessitating careful formulation and engine calibration.
Challenges and Future Outlook
Biomass valorization remains a promising route toward sustainable chemicals and fuels, although progress is hindered by the complexity of biomass feedstocks such as lignin and wastederived materials. Biomass-derived platform molecules offer an effective starting point for advancing the biorefinery concept, enabling fundamental insights into reaction mechanisms and transformation pathways that can be extended to more complex systems. However, aqueous-phase processing presents significant challenges, requiring efficient, water-tolerant, and reusable catalytic systems capable of operating in diluted, impurity-rich streams. Despite these challenges, recent advances in integrated catalytic processes highlight a bright outlook for biomass upscaling, and continued efforts to translate knowledge from model compounds to real renewable and waste feedstocks will be crucial for the widespread deployment of biorefineries.
Conclusion
Over the past recent years, research on biomass-derived platform molecules such as levulinic acid, furan derivatives, polyols, and fatty acids has expanded rapidly for the production of fuels and fuel additives. The main challenge in liquid fuel synthesis is to increase the CHx ratio while efficiently removing oxygen, preferably through C-C bond-forming condensation reactions rather than carbon-loss pathways. Although hydrodeoxygenation remains essential, reducing hydrogen demand and process complexity is critical. Consequently, the development of multifunctional solid catalysts capable of cascade reactions is a key objective. Lignocellulosic biomass represents an abundant, low-cost, and carbon-neutral feedstock, enabling the sustainable production of fuels and chemicals. While commercial implementation remains limited, because need to demonstrate clear understanding and strong potential of catalytic biomass upgrading toward sustainable fuels and fuel additives.
Acknowledgment
All the authors thank the Dr D Srinivasa Reddy Director CSIRIICT for constant help and support.
References
- Huber GW, Iborra S, Corma A (2006) Synthesis of transportation fuels from biomass: Chemistry, catalysts, and engineering. Chemical Reviews 106(9): 4044-4098.
- Corma A, Iborra S, Velty A (2007) Chemical routes for the transformation of biomass into chemicals. Chemical Reviews 107(6): 2411-2502.
- Alonso DM, Bond JQ, Dumesic JA (2010) Catalytic conversion of biomass to biofuels. Green Chemistry 12(9): 1493-1513.
- Zhou CH, Xia X, Lin CX, Tong DS, Beltramini J (2011) Catalytic conversion of lignocellulosic biomass to fine chemicals and fuels. Chemical Society Reviews 40(11): 5588-5617.
- Bozell JJ, Petersen GR (2010) Technology development for the production of biobased products from biorefinery carbohydrates-the US Department of Energy’s Top 10 revisited. Green Chemistry 12(4): 539-554.
- Horvat J, Klaić B, Metelko B, Šunjić V (1985) Mechanism of levulinic acid formation. Tetrahedron Letters 26(17): 2111-2114.
- Varimalla S, Modem V, Boggala S, Chatla A, Aytam HP, et al. (2026) Al-MCM41 supported Ni catalyst for the synthesis of valeric acid using aqueous levulinic acid: Synthesis, structure and activity studies. Journal of the Indian Chemical Society 103: 102340.
- Varimalla S, Boggala S, Manda K, Aytam HP, Akula V (2023) Rutile TiO2 a desirable support for Ni catalyst in the synthesis of γ-valerolactone: Influence of titania crystal phase on the product distribution. Applied Catalysis A: General 662: 119282.
- Varimalla S, Manda K, Boggala S, Nappuni RC, Inkollu S, et al. (2024) Effect of method of preparation of Ni and/or Cu supported on ZSM-5 catalysts for the aqueous phase hydrogenation of levulinic acid to γ-valerolactone. Catalysis Today 441: 114916.
- Mariscal R, Maireles Torres P, Ojeda M, Sádaba I, Granados ML (2016) Furfural: A renewable and versatile platform molecule for the synthesis of chemicals and fuels. Energy & Environmental Science 9(4): 1144-1189.
- Aalto P, Moilanen J, Jokinen J, Koivusalmi E, Myllyoja J, et al. (2007) Process for producing fuels and fuel components from biomass-derived feedstocks. WO Patent 2007068799.
- Matkala B, Boggala S, Basavaraju S, Akella VSS, Aytam HP (2024) Effect of Si/Zr ratio on the catalytic transformation of glycerol to solketal over sulfated Zr-MCM-41 catalysts. Energy & Fuels 38(21): 21134-21143.
- Sudarsanam P, Zhong R, Van den Bosch S, Coman SM, Parvulescu VI, et al. (2018) Functionalised heterogeneous catalysts for sustainable biomass valorisation. Chemical Society Reviews 47(22): 8349-8402.
- Babu S, Jojo L, James A, Melethil K, Thomas B (2023) Metal-based catalysis for biomass and renewables valorization-current status. Tetrahedron Green Chemistry 2: 100018.
- Peddakasu GB, Boggala S, Aytam HP, Manda K, Varimalla S, et al. (2025) Crude levulinic acid conversion to valeric acid over La-TiO₂ supported Ni catalysts: An environmentally friendly route to fuel additive synthesis. Catalysis Science & Technology 15(17): 4971-4980.
- Wang Y, Li M, Wang Z, Liu S, O’Young L (2025) Furfural production: A review on reaction mechanism and conventional production process. Industrial Crops and Products 230: 121103.
© 2026 Boggala Sasikumar. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






