- Submissions

Full Text
Associative Journal of Health Sciences
Prevalence and Clinical Profile of Thyroid Disorders among Patients Attending Two Teaching Hospitals in Northern Sudan: A Hospital-Based Cross-Sectional Study
Amna Abdelraheem Omer Mohammed Ahmed1, Drocella Alshaib Fanous Hizgia1, Rahma Mohamed Elmubarak Abd Elgadir1, Saeed Abdullah Abdulrahman Abdullah Babgi1, Murtada Osman Eltahir1, Ghanem Mohammed Mahjaf2, Tibyan Abd Almajed Altaher3, Mosab Nouraldein Mohammed Hamad4*
1Department of Medicine, Faculty of Medicine, Elsheikh Abdallah Elbadri University, Sudan
2Department of Medical Microbiology, Faculty of Medical Laboratory Sciences, Shendi University, Shendi, Sudan
3Department of Clinical Chemistry, Faculty of Medical Laboratory Sciences, Shendi University, Shendi, Sudan
4Assistant professor, Microbiology department, Faculty of Medicine, Elsheikh Abdallah Elbadri University, Sudan
*Corresponding author:Mosab Nouraldein Mohammed Hamad, Assistant Professor, Microbiology Department, Faculty of Medicine, Elsheikh Abdallah Elbadri University, Sudan
Submission: January 14, 2026;Published: March 24, 2026

ISSN:2690-9707 Volume4 Issue3
Abstract
Background: Thyroid disorders constitute a significant global public health burden, disproportionately
affecting women and often linked to iodine deficiency. In Sudan, particularly in northern regions like
Atbara and Eldamer, updated local epidemiological data are scarce, hindering effective health planning
and interventions.
Objective: This study aimed to assess the prevalence, types, and clinical characteristics of thyroid disorders
among patients at Atbara and Eldamer Teaching Hospitals.
Methods: A descriptive cross-sectional study was conducted from January 2024 to August 2025. Using
convenience sampling, 103 adult patients with clinically and/or laboratory-confirmed thyroid diseases
were enrolled from medical and surgical departments. Data were collected via structured questionnaires
and medical record reviews, capturing demographics, clinical features, types of disorders, medication
use, surgical history, and thyroid function test results (TSH, T3, T4). Data were analyzed using SPSS version
28.
Results: The cohort comprised 103 participants, predominantly female (61.2%), with a median age of
35 years (IQR: 27-48). The most common disorders were goiter (25.2%), hypothyroidism (24.3%), and
hyperthyroidism (17.5%). Neck swelling (46.6%), tachycardia (28.2%), and general fatigue (21.4%) were
the leading symptoms. A high proportion of patients had a history of thyroid surgery (59.2%), and 30.1%
were on thyroid medication. Thyroid function tests revealed significant dysfunction: 15.5% had elevated
TSH, 8.7% had very low TSH, and 10.7% each had elevated T3 and T4 levels.
Conclusion: Thyroid disorders present a significant health burden in northern Sudan, particularly
among young to middle-aged women. The high prevalence of goiter, coupled with elevated surgical and
medication rates, underscores an urgent need for enhanced public awareness, early screening programs,
improved diagnostic capabilities, and strengthened iodine supplementation initiatives in the region.
Keywords:Thyroid disorders; Goiter; Hypothyroidism; Hyperthyroidism; Epidemiology; Sudan; Women’s health; Public health
Introduction
Thyroid disorders represent a significant global public health burden, affecting an estimated 200 million people worldwide and ranking among the most common endocrine diseases [1]. These conditions, which include hypothyroidism, hyperthyroidism, goiter, thyroiditis, and thyroid cancer, disrupt the synthesis and secretion of thyroid hormones-thyroxine (T4) and triiodothyronine (T3)-essential regulators of metabolism, cardiovascular function, neurodevelopment, and reproductive health [2,3]. The hypothalamic-pituitary-thyroid (HPT) axis maintains homeostasis through a delicate feedback mechanism; dysfunction at any level can lead to a spectrum of clinical disorders, from overt disease to subclinical presentations [4]. Globally, thyroid diseases exhibit a pronounced gender disparity, with women being 5-8 times more likely to be affected than men, particularly during reproductive and post-menopausal years [5,6]. The etiology is multifactorial, involving genetic predisposition, autoimmune processes, and environmental factors. However, iodine deficiency remains the single most significant preventable cause of thyroid dysfunction, particularly in low- and middle-income countries (LMICs) [7]. Iodine deficiency disorders (IDDs), such as endemic goiter and congenital hypothyroidism (cretinism), contribute to substantial morbidity, including impaired cognitive development in children and economic productivity losses in adults [8,9]. The World Health Organization (WHO) advocates universal salt iodization (USI) as a cost-effective strategy to eliminate IDDs, yet implementation and monitoring remain inconsistent in many regions [10,11]. In Africa, the burden of thyroid disease is exacerbated by nutritional deficiencies, limited healthcare access, and low public awareness [12,13]. Sub-Saharan Africa reports high prevalence rates of goiter, with studies in Ethiopia, Kenya, and Nigeria indicating goiter prevalence exceeding 30% in some iodine-deficient regions [14- 16]. Hypothyroidism and hyperthyroidism are also increasingly recognized, though data remain fragmented due to under-diagnosis and under-reporting [17]. In Sudan, thyroid disorders are a growing clinical and public health concern. Studies from the capital, Khartoum, suggest a high prevalence, with one report indicating that over 20% of patients attending endocrine clinics exhibit thyroid dysfunction [18]. Iodine deficiency is endemic in several regions, compounded by socioeconomic challenges, dietary habits, and variable iodized salt coverage [19]. However, epidemiological data are largely centralized, with a critical paucity of recent, population-based studies from northern states such as River Nile State. This gap not only hinders the development of targeted interventions and resource allocation but also makes it imperative to investigate the local burden to evaluate current strategies and guide future public health planning. Atbara and Eldamer Teaching Hospitals serve as primary referral centers for River Nile State, catering to a population of over 500,000. No contemporary study has systematically documented the prevalence, types, and clinical profiles of thyroid disorders in this catchment area. Addressing this knowledge gap is essential for informing local healthcare planning, guiding preventive strategies, and contributing to the national health database. Therefore, this hospital-based cross-sectional study aimed to assess the frequency, spectrum, and associated factors of thyroid disorders among patients attending Atbara and Eldamer Teaching Hospitals between January 2024 and August 2025. The findings are intended to provide evidence-based insights for policymakers, clinicians, and public health practitioners to enhance thyroid disease prevention, diagnosis, and management in northern Sudan.
Methods
Study design and setting
A hospital-based, descriptive cross-sectional study was conducted at Atbara Teaching Hospital (a 300-bed tertiary facility) and Eldamer Teaching Hospital (a 150-bed general hospital) in River Nile State, Northern Sudan.
Study period and population
The study was carried out from January 2024 to August 2025. The study population included all adult patients (≥18 years) diagnosed with thyroid disorders who attended the internal medicine and general surgery departments during the study period.
Sample size and sampling
A convenience sampling method was employed. A minimum sample size of 246 was calculated using the formula for crosssectional studies [20], with an estimated prevalence (p) of 20%, a 95% confidence level (Z=1.96), and a margin of error (d) of 5%. However, logistical and time constraints during the data collection period resulted in the enrolment of 103 eligible patients who met the inclusion criteria.
Inclusion and exclusion criteria
Inclusion criteria were: 1) age ≥18 years, 2) a confirmed clinical and/or laboratory diagnosis of a thyroid disorder (e.g., goiter, hypothyroidism, hyperthyroidism, thyroiditis, thyroid nodules, or cancer). Patients with incomplete medical records or those who declined to participate were excluded.
Data collection
Data were collected using a pre-tested, structured questionnaire
administered by trained research assistants, supplemented by a
thorough review of hospital records. The questionnaire covered:
• Section A: Sociodemographic data (age, sex, residence,
marital status).
• Section B: Clinical history (type of thyroid disorder, family
history, medication use, history of thyroid surgery).
• Section C: Clinical symptoms (e.g., neck swelling, fatigue,
weight changes, palpitations).
• Section D: Diagnostic data (results of Thyroid Function
Tests - TSH, T3, T4, and imaging where available).
Data analysis
Data were cleaned, coded, and entered into SPSS version 28 for analysis. Categorical variables were summarized using frequencies and percentages. Continuous variables, found to be non-normally distributed, were presented as medians and interquartile ranges (IQRs). Results were displayed in tables and figures.
Ethical considerations
Ethical approval was obtained from the Research and Ethics Committee of the Faculty of Medicine, El-Sheikh Abdallah Elbadri University. Written informed consent was obtained from all participants. Confidentiality and anonymity were maintained throughout the study.
Result
Sociodemographic characteristics
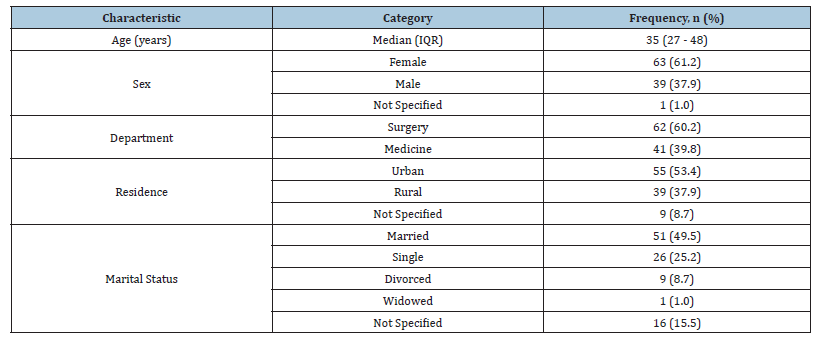
A total of 103 patients were included. The median age was 35 years (IQR: 27-48). The majority were female (63, 61.2%) and resided in urban areas (55, 53.4%). Most patients were recruited from the surgical department (62, 60.2%). Detailed characteristics are presented in Table 1.
Table 1:Sociodemographic characteristics of the study participants (N=103).
Clinical profile and types of thyroid disorders
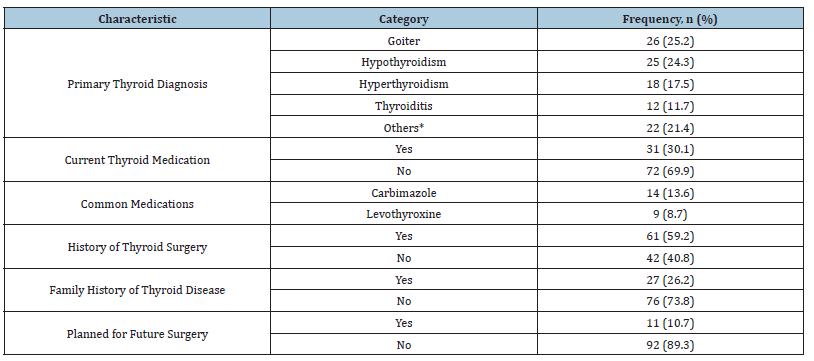
Goiter was the most common diagnosis (26, 25.2%), followed by hypothyroidism (25, 24.3%) and hyperthyroidism (18, 17.5%). A significant proportion of patients (61, 59.2%) had undergone previous thyroid surgery, primarily for cancer (6.8%) or hyperthyroidism (3.9%). Concurrently, 31 patients (30.1%) were on thyroid medication, with carbimazole (13.6%) and levothyroxine (8.7%) being the most common. A family history of thyroid disease was reported by 26.2% of participants. Details are shown in Table 2.
Table 2:Clinical characteristics and management of thyroid disorders (N=103).
Prevalence of Symptoms
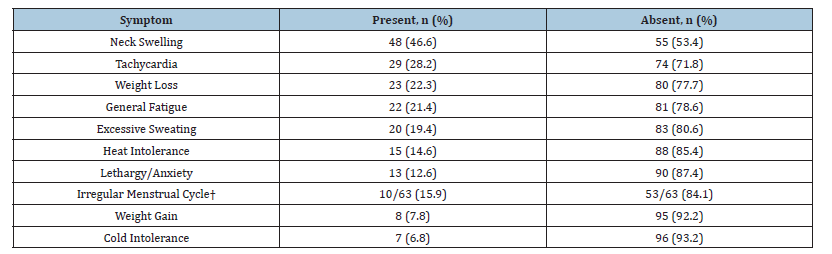
The most frequently reported symptom was neck swelling (48, 46.6%), followed by tachycardia (29, 28.2%), weight loss (23, 22.3%), and general fatigue (22, 21.4%). The distribution of symptoms is summarized in Table 3.
Table 3:Prevalence of self-reported symptoms among participants (N=103).
Thyroid Function Tests (TFTs)
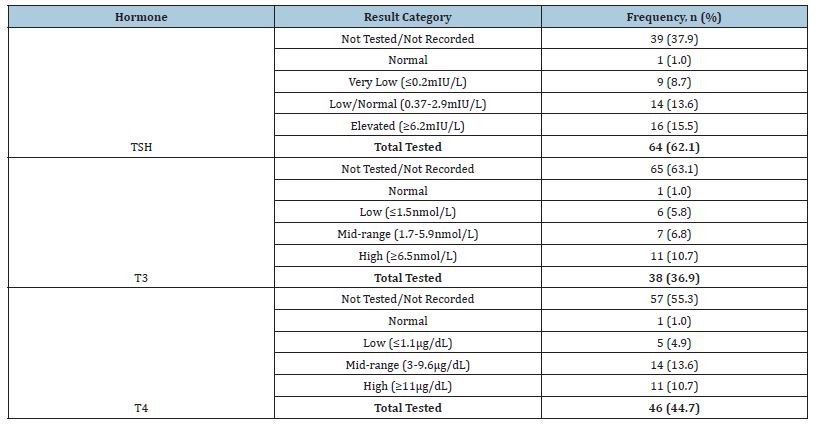
Thyroid function tests were performed on 65 patients (63.1%). Among those with available results, a wide spectrum of abnormalities was observed: 15.5% had elevated TSH (≥6.2mIU/L), suggesting hypothyroidism, while 8.7% had very low TSH (≤0.2mIU/L), indicative of hyperthyroidism. Elevated T3 and T4 levels were each found in 10.7% of tested patients. A significant number of patients (37.9% for TSH, 63.1% for T3, 55.3% for T4) had no TFT results recorded (“None”), highlighting a gap in routine diagnostic workup. Findings are consolidated in Table 4.
Table 4:Results of thyroid function tests (N=103).
Discussion
This study provides one of the first contemporary snapshots of the thyroid disease burden in Northern Sudan, revealing a high prevalence of goiter (25.2%), hypothyroidism (24.3%), and hyperthyroidism (17.5%) among patients attending two major teaching hospitals. The predominance of young to middleaged women (61.2%, median age 35 years) aligns with global epidemiological patterns, where autoimmune thyroid diseases such as Hashimoto’s thyroiditis and Graves’ disease peak during reproductive years [5,21]. This demographic trend underscores the need for gender-sensitive health interventions, particularly in regions where women’s healthcare access may be limited [22]. The high prevalence of goiter strongly suggests persistent iodine deficiency in River Nile State, consistent with findings from other iodine-deficient regions in Africa [14,23]. Goiter is a visible marker of longstanding iodine insufficiency and serves as an indicator of the effectiveness-or failure-of iodine supplementation programs [24]. Despite Sudan’s national salt iodization policy, our findings imply either inadequate implementation, low household coverage, or consumption of non-iodized salt in this region [25]. The coexistence of hypothyroidism (24.3%) further supports the link to iodine deficiency, as insufficient iodine impairs thyroid hormone synthesis, leading to compensatory TSH elevation and gland enlargement [26]. Notably, a substantial proportion of patients (59.2%) reported a history of thyroid surgery, primarily for cancer (6.8%) or hyperthyroidism (3.9%). This high surgical rate may reflect late presentation, limited access to long-term medical management (e.g., antithyroid drugs or radioiodine), or complications of advanced disease such as compressive symptoms or suspected malignancy [27]. In resource-limited settings, surgery often becomes the default intervention for large or symptomatic goiters, despite associated risks and long-term hormonal sequelae [28]. Concurrently, 30.1% of patients were on thyroid medication, indicating a significant chronic disease burden requiring ongoing pharmacologic management and follow-up.
Symptomatically, neck swelling (46.6%) was the most common complaint, reflecting the visible and palpable nature of goiter. Tachycardia (28.2%), weight loss (22.3%), and fatigue (21.4%) were also frequent, consistent with hypermetabolic and hypometabolic states seen in hyperthyroidism and hypothyroidism, respectively [29]. However, the high rate of non-specific symptoms highlights the diagnostic challenge in primary care settings, where thyroid disorders may be confused with anaemia, depression, or other chronic conditions [30]. A concerning finding was the incomplete biochemical evaluation: 37.9% of patients had no recorded TSH, and up to 63.1% lacked T3/T4 results. This gap underscores systemic limitations in diagnostic capacity, possibly due to reagent shortages, cost barriers, or lack of standardized protocols. Without accurate thyroid function testing, differentiation between subclinical and overt disease, as well as appropriate treatment titration, becomes challenging [31]. When compared with other studies, our prevalence figures for hypothyroidism and hyperthyroidism are higher than those reported in some African populations but consistent with data from iodine-deficient regions [17, 32]. For instance, a study in rural South India found lower hyperthyroidism rates (2.9%) but similar hypothyroidism prevalence [33], while research in Khartoum reported thyroid dysfunction in over 20% of endocrine patients [18]. Variations may be attributed to differences in study design, diagnostic criteria, population characteristics, and regional iodine status. In light of these findings and implications, the following actionable recommendations are proposed to mitigate the burden of thyroid disorders in Northern Sudan. This study’s strengths include its focus on an under-researched region, use of both clinical and laboratory criteria for diagnosis, and inclusion of surgical and medical patients, providing a broad clinical spectrum. However, several limitations must be acknowledged. The crosssectional design precludes causal inference. The hospital-based convenience sample, though informative, may not represent the general population, potentially overestimating severity. Recall bias may affect self-reported symptoms and family history. Importantly, we did not assess iodine status (e.g., urinary iodine concentration) or autoimmune markers (anti-TPO, anti-Tg antibodies), which are crucial for elucidating etiology [34,35]. The high proportion of missing laboratory data also limits the biochemical characterization of the cohort. The findings underscore an urgent need for a multipronged public health response. First, strengthening and monitoring universal salt iodization programs is critical to address the root cause of iodine deficiency [36]. Second, integrating thyroid screening into routine primary healthcare-especially for women of reproductive age-could facilitate early detection [37]. Third, improving diagnostic capacity through training, affordable testing, and standardized guidelines is essential for accurate management. Finally, community awareness campaigns about thyroid health, symptoms, and available treatments may reduce stigma and encourage timely care-seeking. Further studies should include population-based sampling, longitudinal design, and comprehensive etiological assessment (iodine status, autoimmune profiling, genetic factors). Research on the socioeconomic impact of thyroid disease and cost-effectiveness of interventions would also be valuable for policy planning.
Conclusion
This study demonstrates a significant burden of thyroid disorders among patients attending major teaching hospitals in Northern Sudan, with goiter, hypothyroidism, and hyperthyroidism being highly prevalent, especially in young and middle-aged women. The high rates of surgical history and ongoing medication use highlight the substantial clinical and healthcare system impact. The findings underscore an urgent need for comprehensive public health strategies, including reinforcing iodine supplementation, promoting early screening, and improving diagnostic and management capacities to reduce the burden of thyroid disease in this region.
Recommendations
A. Public Health Intervention: Reinforce and monitor
universal salt iodization programs across River Nile State to
combat iodine deficiency.
B. Early Detection: Implement targeted thyroid screening
programs in primary healthcare centers, focusing on high-risk
groups such as women of reproductive age.
C. Capacity Building: Ensure consistent availability and
utilization of thyroid function tests (TSH, T3, T4) and thyroid
ultrasound in hospital settings to improve diagnostic accuracy.
D. Clinical Guidelines: Develop and disseminate contextappropriate
clinical guidelines for the management of common
thyroid disorders.
E. Public Awareness: Launch community health education
campaigns to increase awareness about thyroid disorders, their
symptoms, and the importance of early medical consultation.
F. Further Research: Conduct large-scale, populationbased
studies incorporating measurements of iodine status,
autoimmune markers, and longitudinal follow-up to better
understand the etiology and natural history of thyroid diseases
in Sudan.
Declarations
Ethics approval and consent to participate
Approved by the Research and Ethics Committee of the Faculty of Medicine, El-Sheikh Abdallah Elbadri University. Informed consent was obtained from all participants.
Consent for publication
Not applicable.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors declare that they have no competing interests.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
- Garmendia Madariaga A, Santos Palacios S, Guillén-Grima F, Galofré JC (2014) The incidence and prevalence of thyroid dysfunction in Europe: a meta-analysis. J Clin Endocrinol Metab 99(3): 923-931.
- Brent GA (2012) Mechanisms of thyroid hormone action. J Clin Invest 122(9):3035-3043.
- Krude H, Biebermann H, Schnabel D, Ambrugger P, Grüters A (2000) Molecular pathogenesis of neonatal hypothyroidism. Horm Res 53(Suppl 1): 12-8.
- Persani L (2012) Clinical review: Central hypothyroidism: pathogenic, diagnostic, and therapeutic challenges. J Clin Endocrinol Metab 97(9): 3068-3078.
- Laurberg P, Jørgensen T, Perrild H, Ovesen L, Knudsen N, et al. (2006) The Danish investigation on iodine intake and thyroid disease, DanThyr: status and perspectives. Eur J Endocrinol 155(2): 219-228.
- Taylor PN, Albrecht D, Scholz A, Gutierrez-Buey G, Lazarus JH, et al. (2018) Global epidemiology of hyperthyroidism and hypothyroidism. Nat Rev Endocrinol 14(5): 301-316.
- Zimmermann MB, Boelaert K (2015) Iodine deficiency and thyroid disorders. Lancet Diabetes Endocrinol 3(4): 286-295.
- Hetzel BS (1989) The story of iodine deficiency: An international challenge in nutrition. Oxford University Press, UK.
- Bleichrodt N, Born MP (1994) A meta-analysis of research on iodine and its relationship to cognitive development. In: Stanbury JB (Ed.), The damaged brain of iodine deficiency. Cognizant Communication 5: 195-200.
- Andersson M, Karumbunathan V, Zimmermann MB (2012) Global iodine status in 2011 and trends over the past decade. J Nutr 142(4): 744-750.
- World Health Organization (2007) Assessment of iodine deficiency disorders and monitoring their elimination: A guide for programme managers. (3rd edn), WHO, Geneva, Switzerland.
- Zimmermann MB, Jooste PL, Pandav CS (2008) Iodine-deficiency disorders. Lancet 372(9645): 1251-1262.
- Assey VD, Peterson S, Kimboka S, Ngemera D, Mgoba C, et al. (2019) Tanzania national survey on iodine deficiency: Impact after twelve years of salt iodization. BMC Public Health 9: 319.
- Kassim IA, Moloney G, Busili A, Nur AY, Paron P, et al. (2014) Iodine intake in Somalia is excessive and associated with the source of household drinking water. J Nutr 144(3): 375-381.
- Gizak M, Gorstein J, Andersson M (2017) Epidemiology of iodine deficiency. In: Pearce EN, (Ed.), Iodine deficiency disorders and their elimination. Springer pp: 29-48.
- Salami BA, Oluwasola OA (2010) Prevalence of goiter among school children in a rural community in Southwestern Nigeria. Niger J Clin Pract 13(2): 226-229.
- Mbakwem A, Ojo O, Ozoh OB (2021) Thyroid disorders in Africa: A systematic review and meta-analysis. J Clin Transl Endocrinol 25: 100262.
- Eltom A, Eltom M, Idris M (2000) Iodine deficiency disorders in the Sudan: A review. East Mediterr Health J 6(5-6): 1057-1064.
- Elsayed NA, Ahmed AM, Ali R, et al. (2019) Iodine status among school children and pregnant women in Sudan. East Mediterr Health J 25(7): 464-471.
- Daniel WW, Cross CL (2013) Biostatistics: A Foundation for Analysis in the Health Sciences. 10th edn, Wiley.
- McLeod DS, Cooper DS (2012) The incidence and prevalence of thyroid autoimmunity. Endocrine 42(2): 252-265.
- Carrasco CA (2005) Thyroid hormone influences on brain development and neural function. Vitam Horm 71: 95-122.
- Engle-Stone R, Ndjebayi AO, Nankap M, Killilea DW, Brown KH (2014) Stunting prevalence, plasma zinc concentrations, and dietary zinc intakes in a nationally representative sample suggest a high risk of zinc deficiency among women and young children in Cameroon. J Nutr 144(3): 382-391.
- Delange F (1994) The disorders induced by iodine deficiency. Thyroid 4(1): 107-128.
- Sun D, Codling K, Chang S, Zhang S, Shen H, et al. (2017) Eliminating iodine deficiency in China: Achievements, challenges and global implications. Nutrients 9(4): 361.
- Lazarus JH (2005) Thyroid disorders associated with pregnancy: Etiology, diagnosis, and management. Treat Endocrinol 4(1): 31-41.
- Mekonnen A, Kifle M, Tesfaye K (2021) Thyroid surgery in a resource-limited setting: a 5-year retrospective review of indications, complications, and outcomes. World J Surg 45(5): 1412-1419.
- Sahli ZT, Najafian A, Schneider EB (2019) Association between thyroidectomy for benign disease and increased risk of depression and anxiety: a population-based study. JAMA Surg 154(6): 552-559.
- Chaker L, Bianco AC, Jonklaas J, Peeters P (2017) Hypothyroidism. Lancet 390(10101): 1550-1562.
- Canaris GJ, Manowitz NR, Mayor G (2000) The Colorado thyroid disease prevalence study. Arch Intern Med 160(4): 526-534.
- Biondi B, Cooper DS (2018) Subclinical hyperthyroidism. N Engl J Med 378(25): 2411-2419.
- Bekele K, Sileshi M, Getachew A (2020) Prevalence of thyroid dysfunction among adult patients in Addis Ababa, Ethiopia. Ethiop J Health Sci 30(2): 219-226.
- Yadav P, Kumar M, Singh A (2021) Prevalence of thyroid disorders in diabetic patients in rural South India. Int J Adv Med 8(1): 64-68.
- Hollowell JG, Staehling NW, Flanders WD, Hannon WH, Gunter EW, et al. (2002) Serum TSH, T (4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab 87(2): 489-499.
- McLachlan SM, Rapoport B (2004) Why measure thyroglobulin autoantibodies rather than thyroid peroxidase autoantibodies? Thyroid 14(7): 510-520.
- Zimmermann MB (2009) Iodine deficiency. Endocr Rev 30(4): 376-408.
- Vanderpump MPJ, Tunbridge WMG (2002) Epidemiology and prevention of clinical and subclinical hypothyroidism. Thyroid 12(10): 839-847.
© 2026 Mosab Nouraldein Mohammed Hamad. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






