- Submissions

Full Text
Annals of Chemical Science Research
Production of Briquettes from Sawdust Biomass and Coal Blend Using Molasses and Cornstarch as Binder via Response Surface Methodology (RSM)
Ifeoma Juliet Opara1*, Eno-obong Sunday Nicholas2 and Igbudu Sewuese Felicity1
1Department of Chemistry, Faculty of Physical Sciences, Federal University Wukari, Nigeria
2Department of Pure and Industrial Chemistry, Faculty of Physical Sciences, University of Nigeria, Nigeria
*Corresponding author:Ifeoma Juliet Opara, Department of Chemistry, Faculty of Physical Sciences, Federal University Wukari, Taraba State. P.M.B.1020 Wukari, Nigeria
Submission: December 28, 2025;Published: February 25, 2026

Volume5 Issue3February 25, 2026
Abstract
The decreasing availability of fuel wood has made it necessary that efforts towards the efficient utilization of agricultural wastes as low-cost energy sources. Some of these agricultural wastes include coconut shells, wood pulp, sawdust, cow dung and groundnut shells. In this study, briquettes were produced using coal and sawdust in different proportions as designed in RSM, utilizing molasses and cornstarch as binders. The briquettes produced were analyzed using FTIR, Proximate analysis, calorific value and physical tests such as ignition time, burning time. The results indicated that the briquettes produced using sawdust (25wt%) and coal blend (5wt %) with cornstarch (4wt%) as binder had a fixed carbon of 78wt% and a calorific value of 28.5MJ/kg, the briquette ignited at 40s and burned for 18min. While the briquettes produced using sawdust (25wt%) and coal blend (5wt %) with molasses (4wt%) as binder had a fixed carbon of 76.75% and a calorific value of 30.5MJ/kg, the briquette ignited at 26s and burned for 8min. This result suggests that cornstarch and molasses are very good binders for briquette production. The coal blend in sawdust biomass yielded a bio-coal briquette with shorter ignition time, higher combustion sustaining properties, decreased the generation of dust and soot, and emitted less SO2. The briquettes obtained in this study are efficient and environmentally friendly
Keywords:Biomass; Briquette; Coal; Corn starch; Sawdust; Molasses
Introduction
Processing of agricultural produce and clearing of farm land for farming are usually associated with agricultural waste. Direct burning of waste residues in a conventional manner is associated with very low thermal efficiency, fuel losses, and widespread air pollution [1,2]. Researchers have shown that many potential energies are present in these residues. Hence, there is a need to transform these wastes into forms that do not pose a threat to the environment. Biomass includes wood, forage, grasses and shrubs, animal waste, and waste arising from forest, agricultural, municipal and industrial activities, as well as aquatic biomass. Generally, biomass can be converted into energy either by a thermal or a biological process. The potential for the use of biomass as an energy source in Nigeria is very high [3]. This can be explained by the fact that about 80% of Nigerians are rural or semi-urban dwellers, and they depend solely on biomass for their energy sources. The global use of wood fuel for cooking and heating has devastating negative health effects, with 2 million deaths annually from pneumonia, cancer, and chronic lung diseases due to exposure to pollution from biomass combustion [4]. When firewood is burned inside the home, toxic fumes fill the lungs of children and threaten the health of the entire family. Thus, it has become necessary that an alternative source of wood fuel, which is not toxic and environmentally friendly, should be explored. These wastes provide energy when compressed into highdensity fuel briquettes. A briquette is a block of compressed coal, biomass, or charcoal dust that is used as fuel to start and maintain a fire [5]. Generally, the briquetting process has focused more on the production of smokeless solid fuels from coal and agricultural wastes. The common types of briquettes so far in use are coal briquettes, peat briquettes, charcoal briquettes, and biomass briquettes. Most recently, researchers have studied the effect of blending coal and biomass, such as enhancing the properties of coal briquettes using spear grass [6]. Coal was formed by the remains of vegetation that were buried underground for thousands of years under great pressure and temperature in the absence of air. Coal is a complex mixture of compounds composed mainly of carbon, hydrogen, and oxygen with small amounts of Sulphur, nitrogen, and phosphorus as impurities [7]. Bio-coal briquette is a type of solid fuel prepared by blending coal, biomass, binder and sulfur fixation agent. The high pressure involved in the process ensures that the coal particles and the fibrous biomass material interlace and adhere to each other as a result, do not separate from each other during combustion, transportation and storage. Adoption of briquette technology will not only create a safe way of utilizing the waste, but also improve the economy by converting waste into energy, and also contribute towards an eco-friendly environment.
Materials and Methods
Molasses
Sugarcane was ground, the juice was extracted by using a fine cloth, and was then heated at a low temperature till a syrupy texture was formed. It was then allowed to cool for few minutes and reheated at a low temperature till a dark syrup was formed.
Corn starch
Corn was soaked in water for some hours in order for the corn to soften and it was then ground using a grinding machine, sieved using a fine cloth, the filtrate was allowed to settle for some hours while the water settling on top was continuously disposed to get the cornstarch. It was then dried and sieved to attain a smooth and uniform particle size.
Coal
The coal from Onyeama mine was crushed and ground to fine powder. The coal powder obtained after grinding was sieved to obtain coal of particle size 1mm in diameter.
Sawdust
This was obtained from a saw mill in wukari, Taraba State. It was washed with distilled water and dried after which it was sieved to obtain fine particle size.
Bio-coal briquette formulation
Bio-coal briquettes were formulated using experimental runs from design expert as shown in Table 1 & 2. This entails using different percentages of sawdust, coal and binder. The quantity of the calcium hydroxide used was 5% of the whole briquette procedure, a quantity of starch ranging from 0-5g was used during the process, sawdust, coal and Ca (OH)2 was weighed out using a weighing balance. They were mixed thoroughly until a homogenous (uniform) mixture was obtained. A measured quantity of water was added to give a paste that can agglomerate. This was molded into block shaped briquette as shown in Figure 1.
Table 1:Experimental runs from design expert for production of briquettes using corn starch binder.
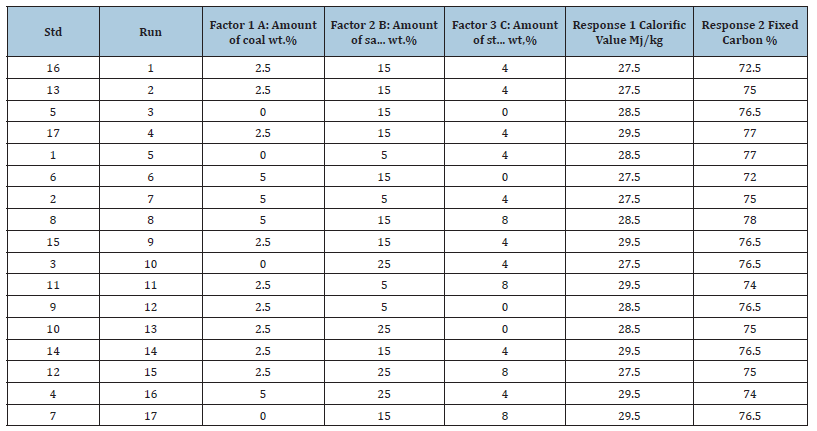
Table 2:Experimental runs from design expert for production of briquettes using corn starch binder.
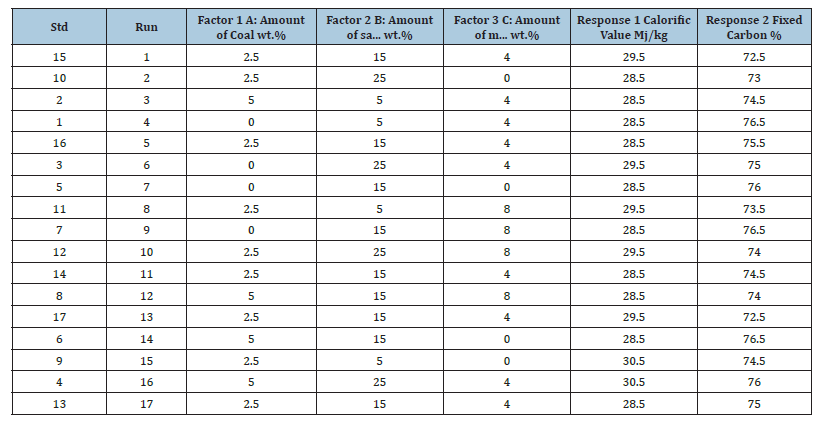
Figure 1:Photograph of sawdust and briquettes produced.

Proximate analysis of briquettes Determination of ash content
The ash content was determined by weighing 2g of the briquette samples into a crucible and placed in a furnace at a temperature of 250 °C for 3 hours. The ash content was calculated as Ash content (%) = weight of ash /initial weight of sample ×100
Determination of volatile matter
The volatile matter was determined by weighing 2g of the
briquette samples and placed in the furnace for 15min at 250 °C in a
partially closed crucible. The crucible and its content were retrieved
and placed in a desiccator to cool. The weight was recorded and the
volatile matter was calculated thus
VM = (W1-W2)/W1×100
Where W1 = original weight of sample, W2 = weight of sample
after cooling
Determination of moisture content
Moisture content was determined by weighing 2g of the briquettes samples and placed in an oven at a temperature of 105 °C for 4 hours. The initial and final weight of the samples were taken
Determination of fixed carbon
The fixed carbon of briquettes was determined using the
following formula
FC (%) = 100- (%VM + %AC + %MC)
Where VM = Volatile matter, AC = Ash content, MC = Moisture
content
Determination of calorific value
The calorific value of the briquettes was determined using oxygen bomb calorimeter. The calorific value (MJ/kg) of the samples under test was calculated from the temperature rise in the calorimeter vessel and the mean heat capacity of the system.
Physical characterization of briquettes Determination of ignition time
The ignition time is the time taken for a flame to raise the briquette to its ignition point. The briquette samples were ignited at the edge of their base with a lighter adjusted to give a steady light. The time taken for each briquette sample to catch fire was recorded as the ignition time of the sample. The test was repeated for each sample and the average time was taken.
Determination of burning time
This is the time taken for each briquette sample to burn
completely to ashes. Subtracting the time, it turned to ashes
completely from ignition time gives the burning time.
Burning rate = Ashing time – Ignition time
Results and Discussion
FTIR
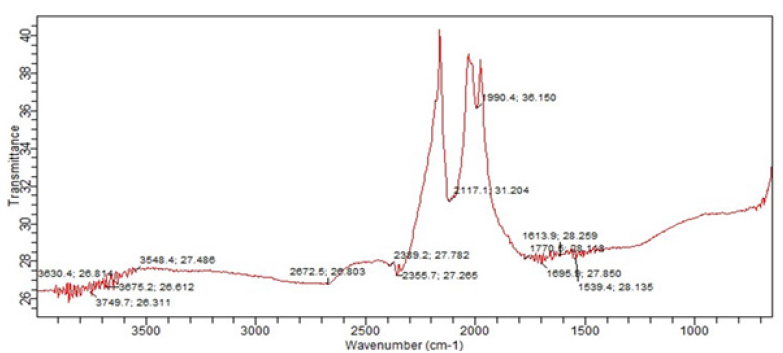
The FT-IR spectra of raw coal sample as shown in Figure 2 was carried out to determine the functional group of coal at various peaks and also to ascertain the presence of Sulphur for desulphurization. The peak at 3548.2 cm-1 could be due to O-H stretching vibration indicating the presence of carboxylic acid. The peaks at 2672.5cm-1 may be due to present of H-C=O group. The peak with a wave number of 2355.7cm-1 could be due to the presence of carbon dioxide (O=C=O). The peak at 2117.1cm-1 may indicate the present of an alkyne (-C≡C-) stretching vibration. The N-O group indicating presence of nitro compound may be due to the peak observed at 1539.4cm-1. The peak observed at 1613.9cm-1 is attributed to elemental sulphur. Due to the presence of sulphur in the raw coal as shown in the FT-IR spectrum, Ca (OH)2 was used to treat the coal as a desulfurizing agent in other to produce a smokeless briquette that is environmentally friendly.
Figure 2:FT-IR spectroscopy of coal sample.
Proximate analysis
The proximate analysis shown in Table 2 & 3 indicated that moisture content of briquettes produced using molasses and corn starch binders ranged from 8.5-13.5% and 8.5-13% respectively. The ash content ranged from 4-6% and 5-7% for briquettes produced using molasses and corn starch binders. The volatile matter is within the range of 7-9% and 6-8% for briquettes produced using molasses and corn starch binders. This implies that cornstarch binder briquettes and molasses binder briquettes are both good substitute of wood fuel since they do not contain high ash content which increased their calorific value. This result is similar to earlier report by Inegbedion [8]. The values obtained from the analysis were used to calculate fixed carbon. The calorific value of briquettes gives the indication of the intensity of the heating values. High calorific values are required in high quality briquettes so as to harness as much energy as possible from the briquettes. The interaction of sawdust, coal and the binders (molasses and corn starch) to yield higher fixed carbon and calorific values are shown in Figures 3-6. In this study, as the amount of interaction between coal increased to 5% and sawdust increased to 25%, the briquettes calorific value using cornstarch as binder increased from 27-29.5MJ/kg. On the other hand, at optimum level the briquettes calorific value using molasses as binder was found to be within the range of 28-30.5MJ/kg. Similar trend has been reported by Manyuchi et al. [9]. This shows that briquettes produced using molasses binder has higher calorific value due to high binder and sawdust concentration resulted in increased interlocking bonds with the coal fines resulting in a briquette that has a high heating value due to the additional strength provided by the combined effect of the high sawdust and binder concentration [10,11].
Table 3:Proximate analysis of briquettes produced using molasses and corn starch as binder.
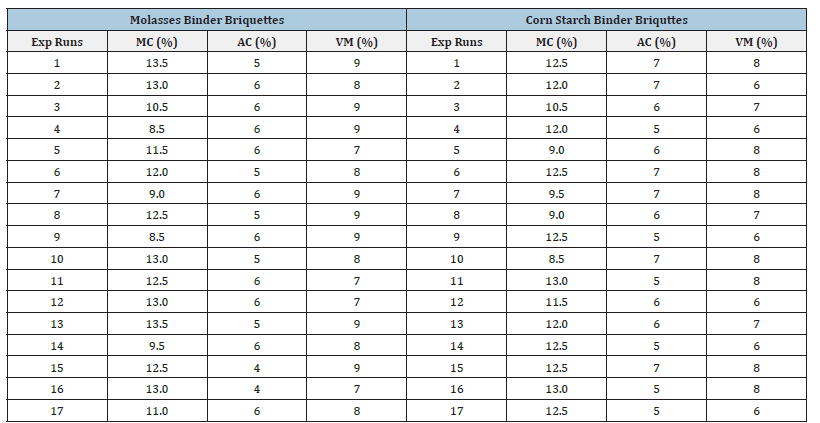
Figure 3:The 3D surface diagram showing the interaction between amount of coal, amount of sawdust and calorific value for corn starch binder briquettes.
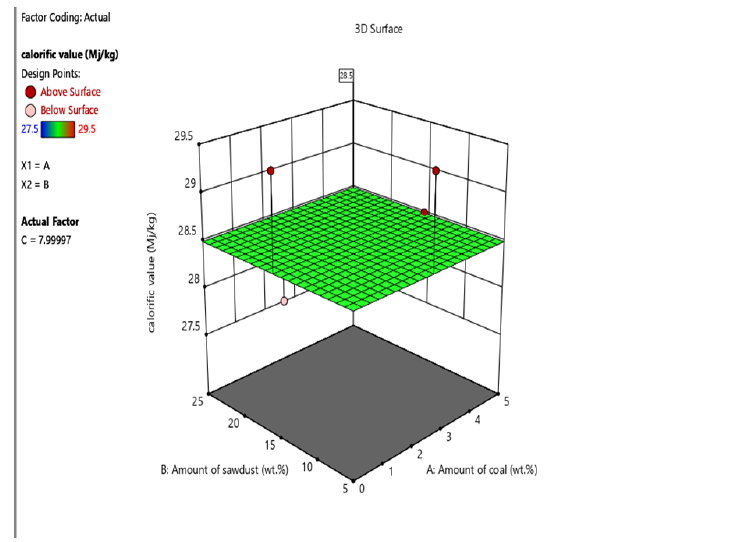
Figure 4:The 3D Surface Diagram showing the interaction between amount of coal, amount of sawdust and fixed carbon for briquettes produced using corn starch binder.
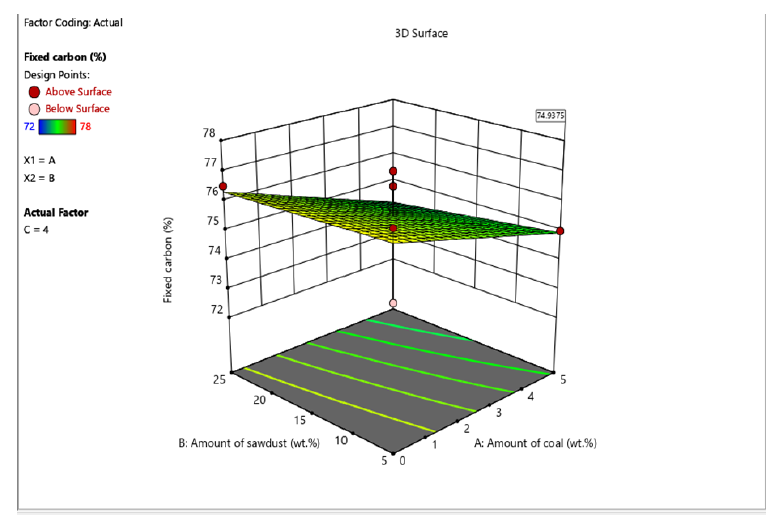
Figure 5:The 3D surface diagram showing the interaction between amount of coal, amount of sawdust and calorific value for briquettes produced using molasses binder.
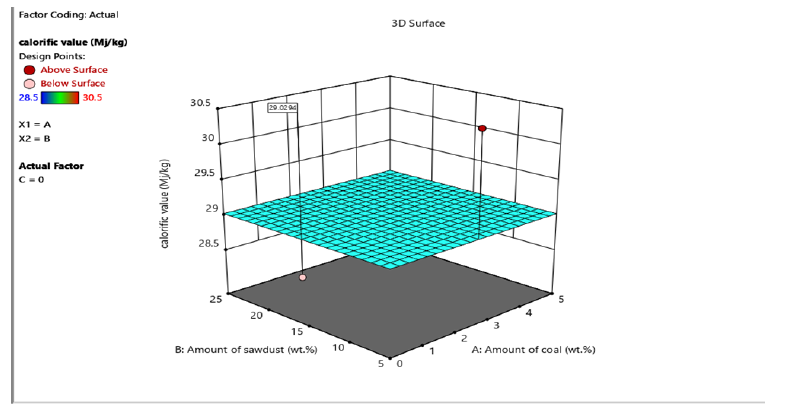
Figure 6:The 3D Surface Diagram showing the interaction between amount of coal, amount of sawdust and fixed carbon for briquettes produced using molasses binder.
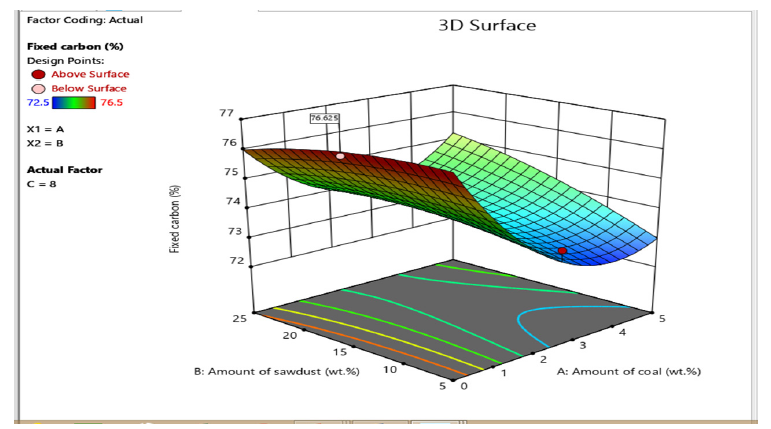
The higher the fixed carbon content, the higher the quality of the briquette [12]. At optimum level the briquettes produced using cornstarch as binder has higher fixed carbon ranging from 72- 78% and the briquettes produced using molasses as binder has a comparable fixed carbon ranging from 72-77%. The increase in the fixed carbon content can be attributed to the increased interlock of the coal-sawdust cornstarch particles as the briquetting process occurred [13].
Physical characterization of briquettes
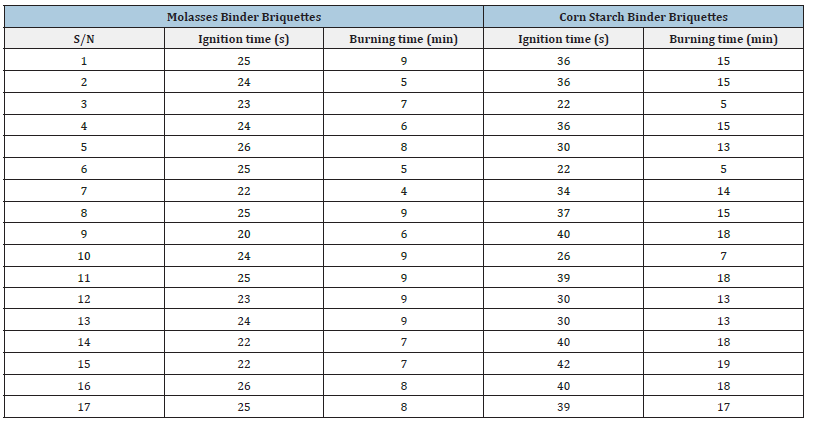
The ignition time of briquettes produced using cornstarch binder as shown in Table 4 was estimated at an average of 40s and that using molasses as binder was estimated at an average of 26s, this indicate that molasses binder briquettes ignites faster because its particles are not tightly held together due to weak interlocking of the concentration of the binder (molasses) to the coal-sawdust concentration and as a result of this factor the briquette burns fast, the burning time was estimated at an average of 8min compared with the cornstarch binder briquettes which its burning time was estimated at an average of 18min.
Table 4:Ignition and burning time values from briquettes produced using molasses binder.
Conclusion
Biomass from agricultural waste has potential for the production of briquette as alternative source of energy to wood fuel. Addition of coal to sawdust produced smokeless bio-briquette as energy source. The briquettes produced using sawdust, coal blend and corn starch or molasses as binder are of high calorific value and are smokeless when ignited. Briquettes produced with sawdust, coal blend and cornstarch as binder gave the calorific value of 29.5MJ/kg and fixed carbon of 74%, and ignites at 40s and burns for 18min. While Briquettes produced with sawdust, coal blend and molasses as binder gave the calorific value of 30.5MJ/kg and fixed carbon of 76% and ignites at 26s and burns for 8min. This shows that as the quantity of binder increases, there is an increase in the performance of the briquettes. Both briquettes produced using cornstarch and molasses as binder had comparable strength and can be used as substitute for wood fuel.
Conflict of Interest
The authors declare that they have no competing financial interests.
References
- Chinyere DC, Asoegwu SN, Nwandikom GI (2014) An evaluation of briquettes from sawdust and corn starch binder. The International Journal of Science & Technoledge 2(7): 149-157.
- Osarenwinda JO, Ihenyen OI (2012) The preliminary design and fabrication of manually operated briquetting machine. Journal of Applied Science and Environmental Management 16(2): 209-211.
- Juliet B, Vasilije M, Philip L (2016) Biomass resources and biofuels potential for the production of transportation fuels in Nigeria. Renewable and Sustainable Energy Reviews 63: 172-192.
- Singh RM (2013) Biobriquetting in Nepal-Scope and potentials: A review. Kathmandu University Journal of Science, Engineering and Technology 9(2): 104-120.
- Speight JG (2013) The chemistry and technology of coal, 3rd Edition. CRC Press, Boca Raton, FL, USA.
- Onuegbu TU, Ogbu IM, Ilochi NO, Okafor I, Obumselu OF, et al. (2010) Enhancing the efficiency of coal briquette in rural Nigeria using Pennisetum purpurem. Advances in Natural and Applied Sciences 4(3): 299-304.
- Meyers RA (2012) Coal structure. Elsevier (Academic Press). ISBN 978-0-12-493080-3.
- Inegbedion F (2022) Estimation of the moisture content, volatile matter, ash content, fixed carbon and calorific values of saw dust briquettes. MANAS Journal of Engineering 10(1): 17-20.
- Manyuchi MM, Mbohwa C, Muzenda E (2018) Value addition of coal fines and sawdust to briquettes using molasses as binder. South African Journal of Chemical Engineering 26: 70-73.
- Wilaipon P (2008) Density equation of bio coal briquettes and quantity of maize cob in Phitsanulok, Thailand. American Journal of Applied Sciences 5(12): 1808-1811.
- Sen R, Wiwatpanyaporn S, Annachhatre AP (2016) Influence of binders on physical properties of fuel briquettes produced from cassava rhizome waste. International Journal of Environment and Waste Management 17(2): 158-175.
- Taulbee D, Patil DP, Honaker RQ, Parekh BK (2009) Briquetting of coal fines and sawdust part I: Binder and briquetting-parameters evaluations. International Journal of Coal Preparation and Utilization 29(1): 1-22.
- Venter P, Naude N (2015) Evaluation of some optimum moisture and binder conditions for coal fines briquetting. The Journal of the Southern African Institute of Mining and Metallurgy 115(4): 329-333.
© 2026 Ifeoma Juliet Opara. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






