- Submissions

Full Text
Advances in Complementary & Alternative medicine
The Effects of Hyperbaric Oxygen Therapy on Sleep Quality: A Systematic Review and Meta- Analysis
Sakra Panthep Indra1*, Charnsiri Segsarnviriya2, Kittibhum Kawinchotpaisan3 and Thannawat Wattanaseth1
1Department of Anti-Aging and Regenerative Medicine, College of Integrative Medicine, Dhurakij Pundit University, Bangkok, Thailand
2Department of Otorhinolaryngology Head and Neck Surgery, Samitivej Thonburi Hospital, Bangkok, Thailand
3Department of Emergency Medicine, Navavej International Hospital, Bangkok, Thailand
*Corresponding author:Sakra Panthep Indra, Department of Anti-Aging and Regenerative Medicine, College of Integrative Medicine, Dhurakij Pundit University, Bangkok, Thailand
Submission: April 20, 2026;Published: April 27, 2026

ISSN: 2637-7802 Volume 9 Issue 2
Abstract
Background: Hyperbaric Oxygen Therapy (HBOT) has been increasingly investigated for its potential
role in modulating neurophysiological mechanisms associated with sleep regulation. This study aimed
to systematically evaluate the effects of HBOT on sleep quality through a systematic review and metaanalysis
of Randomized Controlled Trials (RCTs).
Methods: Electronic databases, including PubMed, ScienceDirect, Ovid, the Cochrane Library, and Google
Scholar, were searched to identify eligible studies comparing HBOT with control interventions and
assessing sleep quality.
Results: A total of four RCTs involving 243 participants were included. Pooled analysis using a randomeffects
model demonstrated no statistically significant difference in global PSQI scores between HBOT
and control groups (mean difference=-0.03; 95% CI, (-5.42, 5.36); p=0.99). Subgroup analysis based on
treatment pressure revealed no significant difference between <2 ATA and ≥2 ATA (p=0.15). However,
a trend toward greater improvement was observed in the <2 ATA subgroup (mean difference=-3.01;
95% CI, (-6.14, 0.11); p=0.06), whereas no significant effect was found in the ≥2 ATA subgroup (mean
difference=-0.23; 95% CI, (-2.30, 1.84); p=0.83). Sensitivity analysis excluding the study contributing
most to heterogeneity yielded consistent non-significant results (mean difference=-2.09; 95% CI, (-4.85,
0.67); p=0.14).
Conclusion: Current evidence does not support a statistically significant overall effect of HBOT on sleep
quality. Observed variability may be attributed to differences in clinical populations and treatment
protocols. Further large-scale, well-designed RCTs with standardized HBOT protocols are warranted to
clarify its therapeutic role in sleep modulation.
Keywords:Hyperbaric oxygen therapy; Sleep quality; PSQI
Abbreviations: ATA: Atmospheres Absolute; CI: Confidence Interval; HBOT: Hyperbaric Oxygen Therapy; I²: I-Squared Statistic; MD: Mean Difference; PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses; PSQI: Pittsburgh Sleep Quality Index; RCT: Randomized Controlled Trials
Introduction
Sleep is a fundamental biological process essential for maintaining neurocognitive function, emotional regulation, and systemic physiological homeostasis [1-3]. Impaired sleep quality has been associated with a wide range of adverse health outcomes, including cardiovascular disease, metabolic disorders, neurodegenerative conditions, and psychiatric illnesses [1-6]. Epidemiological data suggest that poor sleep quality affects a substantial proportion of the adult population worldwide [7,8]. Although conventional approaches such as pharmacotherapy, sleep hygiene strategies, and cognitive behavioral therapy for insomnia (CBT-I) are available, persistent sleep disturbances remain common, particularly among individuals with chronic inflammatory, neurological, or post-infectious conditions [4,9- 12]. Hyperbaric oxygen therapy (HBOT) has emerged as a potential therapeutic modality capable of influencing multiple patho physiological pathways implicated in sleep regulation [13- 16]. By delivering 100% oxygen under increased atmospheric pressure, HBOT enhances tissue oxygenation and has been shown to modulate neuroinflammation, improve mitochondrial function, promote cerebral perfusion, and regulate autonomic nervous system balance [13,15-19].
These mechanisms may indirectly contribute to improvements in sleep architecture, including reduced sleep latency, enhanced sleep efficiency, and improved subjective sleep quality. Previous clinical studies have reported beneficial effects of HBOT on sleeprelated out-comes in specific populations, such as patients with fibromyalgia, persistent post-concussion syndrome, and long COVID [20-23]. However, these findings remain fragmented and have not been systematically synthesized. To date, no comprehensive systematic review and meta-analysis has specifically evaluated the effect of HBOT on sleep quality using standardized outcome measures. Given the growing clinical interest in HBOT and its potential role in addressing treatment-resistant sleep disturbances, a rigorous synthesis of available evidence is warranted. Therefore, this study aimed to systematically review and quantitatively synthesize randomized controlled trials investigating the effects of HBOT on sleep quality, as assessed by the Pittsburgh Sleep Quality Index (PSQI) [24].
Materials and Methods
Protocol and registration
This systematic review and meta-analysis were conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guide lines [25], and it was registered in PROSPERO, CRD420251245503.
Search strategy and selection criteria: A comprehensive literature search was conducted independently by two reviewers (S.P. and C.S.) across five electronic data-bases, including PubMed, ScienceDirect, Ovid, The Cochrane Library, and Google Scholar, from inception through March 2025. The search strategy was developed around terms related to hyperbaric oxygen therapy and sleep. The complete PubMed search string was as follows: (“Hyperbaric oxygen therapy” OR “Hyperbaric oxygen treatment” OR “HBOT” OR “HBO”) AND (“Sleep”). The search strategy was adapted for each database as appropriate. All retrieved records were screened independently by two reviewers to assess eligibility. Titles and abstracts were reviewed first, followed by full-text assessment of potentially relevant studies. Disagreements regarding study inclusion or exclusion were resolved through discussion with a third reviewer until consensus was reached. In addition, reference lists of relevant articles were manually examined to identify any further eligible studies.
Inclusion and Exclusion Criteria: Studies were considered eligible if they were Randomized Controlled Trials (RCTs) comparing Hyperbaric Oxygen Therapy (HBOT) with a control or sham intervention in adult participants (aged≥20 years), and assessed sleep quality using a standardized outcome measure, including subjective and objective assessments, with sufficient data available for quantitative synthesis. Studies were excluded if they were non-randomized designs (including observational studies, case reports, case series, or re-views), did not include a control group, were published in languages other than English, or had insufficient data for analysis.
Outcomes: The primary outcome of this study was sleeping quality, assessed using both subjective and objective measures. Subjective sleep quality was evaluated using validated instruments such as the Pittsburgh Sleep Quality Index (PSQI), whereas objective parameters were assessed using methods such as actigraphy when available. For quantitative synthesis, the global PSQI score was used as the primary outcome because it was the most consistently reported measure across studies. Lower PSQI scores indicate better sleep quality. When available, changes in sleep outcomes before and after intervention, as well as between-group differences (HBOT vs. control), were extracted.
Data extraction: Data extraction was independently performed by two reviewers (S.P and C.S.) using a standardized data collection form. The following information was extracted from each included study: first author, year of publication, country, study population, sample size, participant characteristics, details of the HBOT intervention (including pressure, duration, and number of sessions), type of control group, and outcome measures related to sleep quality. Discrepancies between reviewers were resolved through discussion or consultation with a third reviewer to ensure accuracy and consistency of the extracted data.
Risk of bias assessment: The risk of bias of the included randomized controlled trials was assessed using the Cochrane Risk of Bias 2 (RoB 2) tool [26]. Two reviewers independently evaluated each study across five domains: bias arising from the randomization process, bias due to deviations from intended interventions, bias due to missing outcome data, bias in measurement of the outcome, and bias in selection of the reported result. Each domain was judged as low risk of bias, some concerns, or high risk of bias, leading to an overall risk of bias judgment for each study. Any disagreements between reviewers were resolved through discussion or consultation with a third reviewer to achieve consensus.
Data analysis: A meta-analysis was performed using a random effects model (DerSimonian and Laird method) to account for potential heterogeneity across studies [27,28]. Continuous outcomes were expressed as Mean Differences (MD) with 95% Confidence Intervals (CI) for PSQI scores between the HBOT and control groups. Statistical heterogeneity was assessed using the I² statistic, with values greater than 50% indicating substantial heterogeneity [27]. All statistical analyses were conducted using Review Manager (RevMan), version 5.4.1 (The Cochrane Collaboration, Copenhagen, Denmark).
Subgroup analysis: Subgroup analysis was conducted to explore potential sources of heterogeneity based on HBOT treatment pressure. Studies were stratified into two groups according to the applied pressure: <2.0 Atmospheres Absolute (ATA) and ≥2.0 ATA. The pooled effects were analyzed separately within each subgroup, and differences between subgroups were examined to determine whether treatment pressure influenced the effect of HBOT on sleep quality.
Results
Study selection
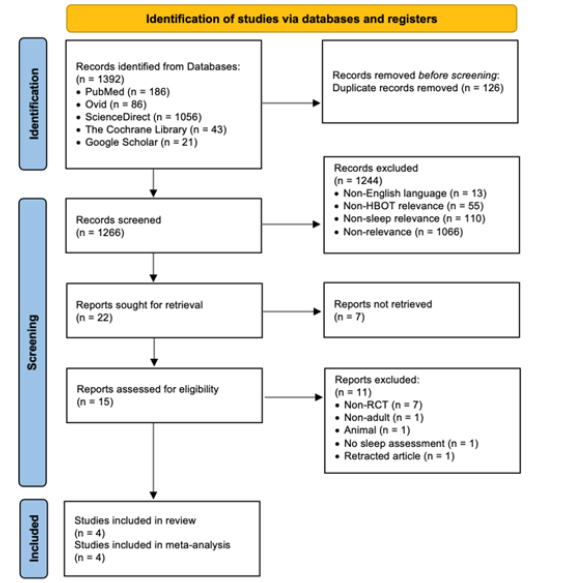
A total of 1,392 records were identified from five electronic databases, PubMed (n=186), Ovid (n=86), ScienceDirect (n=1,056), the Cochrane Library (n=43), and Google Scholar (n=21). After removing 126 duplicate records, 1,266 titles and abstracts were screened. Of these, 1,244 were excluded due to non-English language (n=13), lack of relevance to HBOT (n=55), absence of sleep-related outcomes (n=110), or general irrelevance (n=1,066). A total of 22 reports were sought for retrieval, of which 7 were not retrieved. Fifteen full-text articles were assessed for eligibility. Of these, 11 were excluded for the following reasons: non-randomized study design (n=7), non-adult population (n=1), animal study (n=1), absence of sleep assessment (n=1), and retracted article (n=1).
Ultimately, four randomized controlled trials [20-23] met the inclusion criteria and were included in both the qualitative synthesis and quantitative meta-analysis. The study selection process is summarized in the PRISMA 2020 flow diagram (Figure 1).
Figure 1:Prisma flow diagram of study selection.
Study characteristics
The main characteristics of the included studies are summarized in Table 1. A total of four randomized controlled trials [20-23], comprising 243 participants, were included in this meta-analysis. The study populations were heterogeneous and included patients with Mild Traumatic Brain Injury (mTBI), fibromyalgia, long COVID, and high-altitude-related insomnia. The sample sizes ranged from 20 to 80 participants, with mean ages varying from the mid-30s to approximately 50 years. HBOT protocols differed across studies in terms of pressure, treatment duration, and session frequency. Most studies applied HBOT at 2.0 Atmospheres Absolute (ATA) for 90minutes per session, five times per week for eight weeks, whereas one study used a lower pressure of 1.6 ATA administered once daily for 10 consecutive days. Control conditions also varied, including sham hyperbaric exposure and standard care without hyperbaric treatment.
Table 1:Characteristics of the included studies.
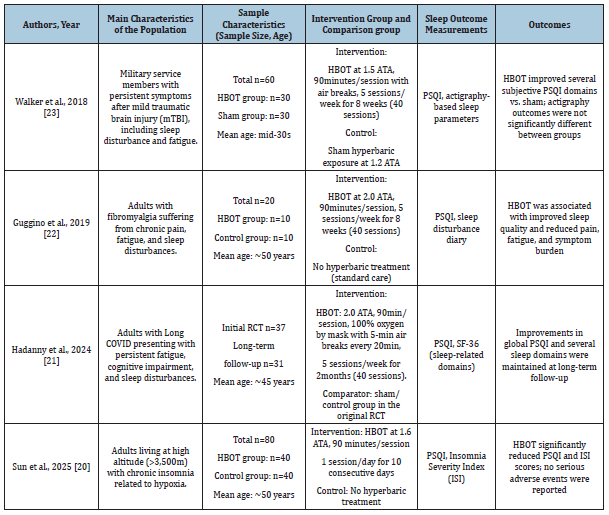
(HBOT: Hyperbaric Oxygen Therapy, ATA: Atmospheres Absolute, PSQI: Pittsburgh Sleep Quality Index, ISI: Insomnia Severity Index, SF-36: The Short Form-36 Health Survey, RCT: Randomized Controlled Trial, mTBI: Mild Traumatic Brain Injury, SD: Standard Deviation).
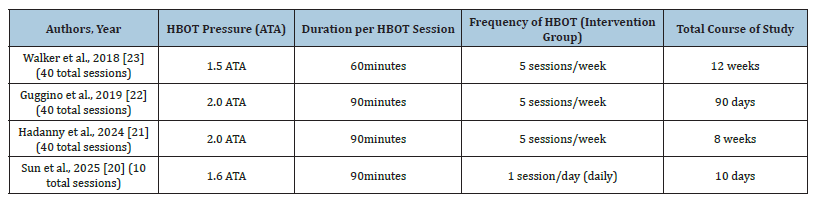
Sleep outcomes were assessed using validated instruments, primarily the Pittsburgh Sleep Quality Index (PSQI), with additional measures such as actigraphy, the Insomnia Severity Index (ISI), and the Short Form-36 (SF-36) in selected studies. Overall, although all studies evaluated the effect of HBOT on sleep quality, variations in patient populations, treatment protocols, and outcome measurements may have contributed to the observed heterogeneity in the pooled analysis. The HBOT protocols varied across the included studies in terms of pressure, session duration, and treatment frequency. Most studies applied HBOT at 2.0 ATA for 90 minutes per session, five sessions per week, with a total of approximately 40 sessions. One study used a lower pressure (1.5 ATA) with a shorter session duration, while another applied 1.6 ATA with daily sessions over a shorter treatment period (Table 2).
Table 2:Hyperbaric Oxygen Therapy (HBOT) treatment protocols across included studies.
Quality assessment of included studies
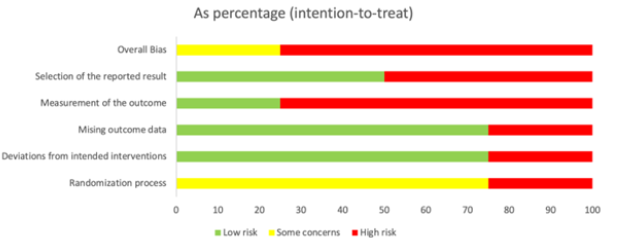
The risk of bias across the included studies is presented in Figure 2. Overall, the methodological quality varied among studies. One study was assessed as having low risk of bias, while the remaining studies were judged to have either some concerns or high risk of bias in at least one domain. Across individual domains, the risk of bias was most frequently observed in the measurement of outcomes (D4) and selection of the reported results (D5). Concerns related to the randomization process (D1) were also identified in several studies. In contrast, most studies demonstrated relatively low risk of bias in deviations from intended interventions (D2) and missing outcome data (D3). Figure 3 illustrates the proportion of studies classified as low risk, some concerns, and high risk across each domain. A higher proportion of high risk of bias was observed in outcome measurement and selective reporting domains, whereas deviations from intended interventions and missing outcome data were generally assessed as low risk. Overall, these findings indicate variability in study quality, which should be considered when interpreting the pooled results.
Figure 2:Quality assessment showing risk of bias in each included study.
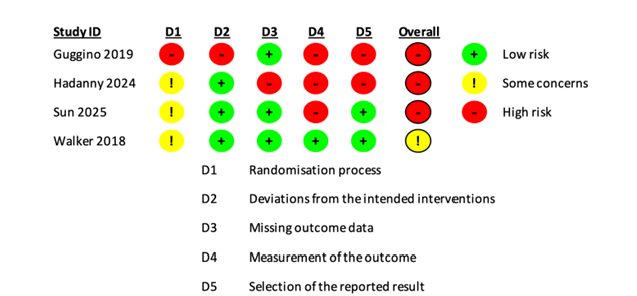
Figure 3:Quality assessment summary showing risk of bias of all included studies.
Meta-analysis
Overall effect of HBOT on sleep quality (PSQI): A randomeffects meta-analysis was conducted to evaluate the overall effect of Hyperbaric Oxygen Therapy (HBOT) on sleep quality, as measured by the Pittsburgh Sleep Quality Index (PSQI). A total of four randomized controlled trials [20-23] comprising 243 participants (HBOT: n=122; control: n=121) were included in the analysis. The pooled results showed no statistically significant difference between the HBOT and control groups (mean difference, -0.03; 95% CI, (-5.42, 5.36); p = 0.99) (Figure 4).
Figure 4:Forest plot showing the pooled effect of HBOT on sleep quality measured by PSQI.
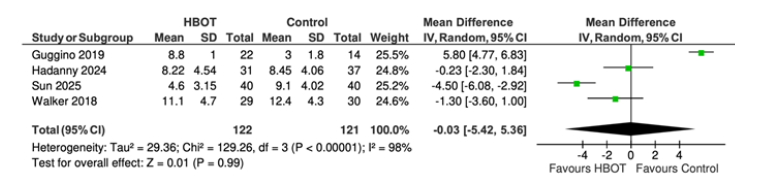
Heterogeneity assessment: Substantial heterogeneity was observed in the primary analysis (I²=98%) (Figure 4), indicating considerable variability across the included studies. Substantial heterogeneity was observed across the included studies. Overall, the pooled evidence did not support a statistically significant benefit of HBOT over control in improving PSQI scores. Potential sources of heterogeneity were explored through sensitivity and subgroup analyses. Visual inspection of the forest plot suggested variability in both the magnitude and direction of effect estimates among studies.
Sensitivity analysis: Given the substantial heterogeneity observed in the primary analysis, a sensitivity analysis was performed using a leave-one-out approach. The study by Guggino et al. (2019) was identified as a potential source of heterogeneity due to its discordant effect size and direction compared with the other studies and was therefore excluded from the re-analysis. After exclusion, three randomized controlled trials comprising 207 participants (HBOT: n=100; control: n=107) were included. The pooled analysis showed no statistically significant difference between groups (mean difference, -2.09; 95% CI, (-4.85, 0.67); p= 0.14) (Figure 5). Heterogeneity remained high (I²=83%).
Figure 5:Forest plot of the sensitivity subgroup meta-analysis of HBOT on sleep quality measured by PSQI, stratified by treatment pressure (<2 ATA vs. ≥2 ATA). The analysis was performed after exclusion of Guggino et al.
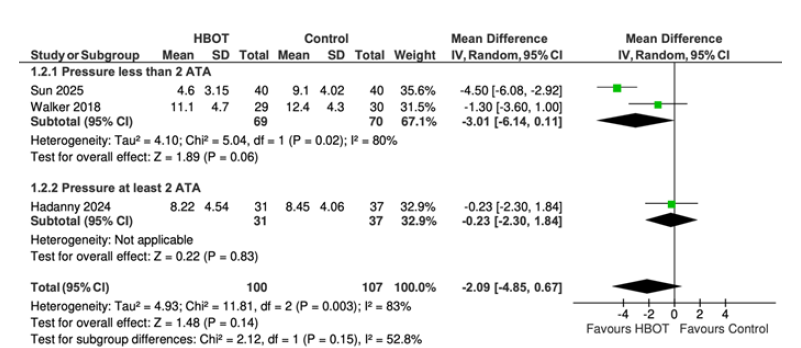
Subgroup analysis by HBOT pressure: Subgroup analysis was performed based on HBOT pressure (<2 ATA vs. ≥2 ATA) to explore potential sources of heterogeneity (Figure 5). In the <2 ATA subgroup, no statistically significant difference was observed, although a trend favoring HBOT was noted (mean difference, -3.01; 95% CI, (-6.14, –0.11); p=0.06). In the ≥2 ATA subgroup, no significant effect was found (mean difference, -0.23; 95% CI, (-6.14, 0.11); p=0.83).
Publication bias
Publication bias was assessed by visual inspection of the funnel plot. The distribution of studies appeared relatively symmetrical around the mean difference, suggesting no clear evidence of publication bias. Slight asymmetry was observed and may reflect between-study heterogeneity or differences in study characteristics rather than true publication bias. However, the small number of included studies (n=4) limits the reliability of this assessment, and formal statistical tests for publication bias, such as Egger’s regression, were not performed because fewer than 10 studies were included [27].
Discussion
This systematic review and meta-analysis evaluated the effect of Hyperbaric Oxygen Therapy (HBOT) on sleep quality measured by the Pittsburgh Sleep Quality Index (PSQI). The pooled analysis showed no statistically significant difference between HBOT and control groups (mean difference -0.03; 95% CI (-5.42, 5.36); p=0.99), indicating that current evidence did not demonstrate a statistically significant overall improvement in sleep quality with HBOT. A notable finding of this study is the substantial heterogeneity observed across the included trials. The high I² value (98%) suggests considerable variability in effect estimates, which may be attributed to differences in study populations, HBOT protocols (e.g., pressure, session frequency, and duration), and baseline sleep characteristics. This heterogeneity limits the interpretability of the pooled effect and highlights the need for more standardized study designs. Sensitivity analysis excluding one potentially influential study reduced heterogeneity (I²= 83%) and slightly shifted the pooled estimate (mean difference -2.09; 95% CI (-4.85, 0.67); p=0.14), although the effect remained statistically non-significant.
This finding suggests that individual studies may disproportionately influence the overall estimate but do not alter the overall conclusion. Subgroup analysis based on HBOT pressure (<2 ATA vs. ≥2 ATA) did not demonstrate a significant difference between sub-groups (I²=52.8%). Although a non-significant trend toward improved PSQI scores was observed in the <2 ATA subgroup (mean difference -3.01; 95% CI (-6.14, 0.11); p=0.06), no effect was observed in the ≥2 ATA subgroup (mean difference -0.23; 95% CI (-2.30,1.84); p=0.83). These findings suggest that treatment pressure may not be a major determinant of HBOT efficacy for sleep outcomes, although the limited number of studies restricts definitive conclusions. The absence of a significant effect contrasts with several theoretical and physiological mechanisms proposed for HBOT. Previous studies have suggested that HBOT may influence pathways relevant to sleep regulation through enhanced cerebral oxygenation, modulation of neuroinflammatory processes, reduction of oxidative stress, and improvement of mitochondrial function and tissue repair [2, 13-16,18,19,29]. At the molecular level, these effects may involve modulation of inflammatory mediators and cytokine-related signaling pathways implicated in sleep disturbance, including IL-6 and TNF-α, as well as oxidative stress signaling pathways that influence neuronal function, immune activation, and recovery processes [2,5,9,18,19].
HBOT has also been associated with mitochondrial biogenesis and metabolic regulation through pathways such as SIRT-1/PGC- 1α, which may support neuroplasticity and functional recovery in selected clinical conditions [18,30]. Such bio-logical effects may theoretically contribute to improved sleep quality, particularly in individuals with inflammation-related, neurological, or postinfectious conditions [5,9]. However, these mechanistic effects may not necessarily translate into measurable improvements in PSQI scores, especially in clinically heterogeneous populations, in studies with variable HBOT protocols, or in participants without clearly defined baseline sleep disorders [4]. Recent studies have further explored the role of HBOT in sleep-related conditions. A recent analysis reported that HBOT may improve sleep quality across diverse patient populations, supporting its potential role beyond disease-specific contexts [31]. In patients with chronic insomnia, particularly under hypoxic conditions such as high altitude, HBOT has been associated with significant improvements in PSQI and related sleep parameters [20].
Additionally, emerging evidence suggests that HBOT may influence sleep-disordered breathing through improved tissue oxygenation and modulation of respiratory physiology [32]. Ongoing research, including a recently published protocol for a systematic review and meta-analysis in post-stroke insomnia, further reflects the growing interest in this area and the need for more robust evidence [33]. These findings provide additional biological and clinical context for the potential effects of HBOT on sleep; however, variability in patient populations, underlying pathophysiology, and treatment protocols may contribute to the inconsistent results observed in the present me-ta-analysis. Regarding publication bias, visual inspection of the funnel plot suggested no clear asymmetry. However, given the small number of included studies (n=4), the reliability of this assessment remains limited [25,27]. Several limitations should be acknowledged. First, the small number of included studies reduces statistical power and limits the ability to detect small but clinically meaningful effects.
Second, substantial heterogeneity across studies complicates interpretation of the pooled estimates. Third, variations in HBOT protocols and participant characteristics may have contributed to inconsistent findings. Finally, reliance on PSQI as the primary outcome may not fully capture objective changes in sleep architecture, which are more appropriately assessed using standardized physiological measures such as polysomnography [24,34]. Despite these limitations, this study provides a structured synthesis of the current evidence on HBOT and sleep quality. The findings suggest that HBOT does not confer a significant benefit on PSQI outcomes under current clinical conditions. Future research should focus on well-designed randomized controlled trials with standardized HBOT protocols, stratification by baseline sleep status, and inclusion of objective sleep measures to better elucidate potential therapeutic effects.
Conclusion
This meta-analysis found no statistically significant effect of Hyperbaric Oxygen Therapy (HBOT) on sleep quality as measured by the Pittsburgh Sleep Quality Index (PSQI). The pooled evidence does not demonstrate a statistically significant overall improvement in subjective sleep outcomes with HBOT under current clinical conditions, although the findings should be interpreted cautiously because of substantial between-study heterogeneity. Substantial heterogeneity across studies and the limited number of available trials restrict the strength of these conclusions. Subgroup and sensitivity analyses did not identify a consistent modifying effect of treatment pressure or individual studies on the overall outcome. Further well-designed randomized controlled trials with standardized HBOT protocols and objective sleep assessments are needed to clarify the potential role of HBOT in sleep modulation.
Author Contributions
Conceptualization, S.P., C.S. and T.W.; methodology, S.P. and C.S.; validation, S.P. and C.S.; formal analysis, S.P. and K.K.; investigation, S.P.; resources, S.P. and C.S.; data curation, S.P.; writing original draft preparation, S.P.; writing review and editing, S.P., K.K. and C.S.; visualization, S.P.; supervision, C.S. and T.W.; project administration, S.P. All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data are available upon reasonable request to the corresponding author.
Acknowledgement
The authors gratefully acknowledge the support and assistance provided by Sarinrath Wongsirilukk, MD, during the entire process of this study..
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Medic G, Wille M, Hemels ME (2017) Short- and long-term health consequences of sleep disruption. Nat Sci Sleep 9: 151-161.
- Ahmadi F, Khalatbary AR (2021) A review on the neuroprotective effects of hyperbaric oxygen therapy. Med Gas Res 11(2): 72-82.
- Borbély AA, Daan S, Justice AW, Deboer T (2016) The two-process model of sleep regulation: A reappraisal. J Sleep Res 25(2): 131-143.
- Riemann D, Spiegelhalder K, Feige B, Voderholzer U, Berger M, et al. (2010) The hyperarousal model of insomnia: A review of the concept and its evidence. Sleep Med Rev 14(1): 19-31.
- Besedovsky L, Lange T, Born J (2012) Sleep and immune function. Pflugers Arch 463(1): 121-137.
- Mander BA, Winer JR, Walker MP (2017) Sleep and human aging. Neuron 94(1): 19-36.
- Leger D, Strambi LF, Han F, Poyares D, Uchiyama M, et al. (2024) Novel perspective of 'poor sleep' in public health: A narrative review. BMJ Public Health 2(2): e000952.
- Simonelli G, Marshall NS, Grillakis A, Miller CB, Hoyos CM, et al. (2018) Sleep health epidemiology in low and middle-income countries: A systematic review and meta-analysis of the prevalence of poor sleep quality and sleep duration. Sleep Health 4(3): 239-250.
- Rusch HL (2020) Links between stress, sleep, and inflammation: A translational perspective of resilience.
- Zhang Y, Ren R, Lei F, Zhou J, Zhang J, et al. (2019) Worldwide and regional prevalence rates of co-occurrence of insomnia and insomnia symptoms with obstructive sleep apnea: A systematic review and meta-analysis. Sleep Med Rev 45: 1-17.
- Edinger JD, Arnedt JT, Bertisch SM, Carney CE, Harrington JJ, et al. (2021) Behavioral and psychological treatments for chronic insomnia disorder in adults: An American Academy of Sleep Medicine clinical practice guideline. Journal of Clinical Sleep Medicine 17(2): 255-262.
- Riemann D, Espie CA, Altena E, Arnardottir ES, Baglioni C, et al. (2023) The European insomnia guideline: An update on the diagnosis and treatment of insomnia 2023. Journal of Sleep Research 32(6): e14035.
- Thom SR (2011) Hyperbaric oxygen: Its mechanisms and efficacy. Plast Reconstr Surg 127 Suppl 1(Suppl 1): 131S-141S.
- Efrati S, Jacob EB (2014) Reflections on the neurotherapeutic effects of hyperbaric oxygen. Expert Rev Neurother 14(3): 233-236.
- Ortega MA, Martinez OF, Montero CG, Peláez EC, Sáez MA, et al. (2021) A general overview on the hyperbaric oxygen therapy: Applications, mechanisms and translational opportunities. Medicina (Kaunas) 57(9): 864.
- Camporesi EM, Bosco G (2014) Mechanisms of action of hyperbaric oxygen therapy. Undersea Hyperb Med 41(3): 247-252.
- Fu Q, Duan R, Sun Y, Li Q (2022) Hyperbaric oxygen therapy for healthy aging: From mechanisms to therapeutics. Redox Biol 53: 102352.
- Schottlender N, Gottfried I, Ashery U (2021) Hyperbaric oxygen treatment: Effects on mitochondrial function and oxidative stress. Biomolecules 11(12): 1827.
- Wolde SDD, Hulskes RH, Weenink RP, Hollmann MW, Hulst RAV (2021) The effects of hyperbaric oxygenation on oxidative stress, inflammation and angiogenesis. Biomolecules 11(8): 1210.
- Sun B, He J, Chen H, Zeng W, Tian T, et al. (2025) Hyperbaric oxygen treatment for chronic insomnia at high altitude: A prospective, randomized, open-label, parallel-group trial. Travel Med Infect Dis 65: 102834.
- Hadanny A, Itskovich SZ, Catalogna M, Shina KE, Lang E, et al. (2024) Long term outcomes of hyperbaric oxygen therapy in post covid condition: Longitudinal follow-up of a randomized controlled trial. Sci Rep 14(1): 3604.
- Guggino G, Schinocca C, Pizzo ML, Liberto DD, Garbo D, et al. (2019) T helper 1 response is correlated with widespread pain, fatigue, sleeping disorders and the quality of life in patients with fibromyalgia and is modulated by hyperbaric oxygen therapy. Clinical and Experimental Rheumatology 37 Suppl 116(1): 81-89.
- Walker JM, Mulatya C, Hebert D, Wilson SH, Lindblad AS, et al. (2018) Sleep assessment in a randomized trial of hyperbaric oxygen in U.S. service members with post concussive mild traumatic brain injury compared to normal controls. Sleep Medicine 51: 66-79.
- Buysse DJ, Reynolds CF, Monk TH, Berman SR, Kupfer DJ (1989) The Pittsburgh sleep quality index: A new instrument for psychiatric practice and research. Psychiatry Res 28(2): 193-213.
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, et al. (2021) The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 372: n71.
- Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, et al. (2019) RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 366: l4898.
- (2019) In: Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, et al. (Eds.), Cochrane handbook for systematic reviews of interventions. The Cochrane Collaboration, London, UK.
- DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7(3): 177-188.
- Gottfried I, Schottlender N, Ashery U (2021) Hyperbaric oxygen treatment-from mechanisms to cognitive improvement. Biomolecules 11(10): 1520.
- Hsu HT, Yang YL, Chang WH, Fang WY, Huang SH, et al. (2022) Hyperbaric oxygen therapy improves parkinson's disease by promoting mitochondrial biogenesis via the SIRT-1/PGC-1α Biomolecules 12(5): 661.
- Barak KD, Shina KE, Lang E, Finci S, Elkarif V, et al. (2026) The effect of hyperbaric oxygen therapy on sleep quality across diverse patient populations. Frontiers in Neurology 17: 1690633.
- Quy SD, Hoc TV, Duy TN, Thao TTT, Van TN, et al. (2026) Hyperbaric oxygen therapy and its physio-mechanical effects on sleep breathing disorder: A systematic review. Pulm Ther 12(1): 39-55.
- Shi R, Meng W, Liu Z, Chang L, Lu R, et al. (2024) Hyperbaric oxygen therapy for poststroke insomnia: A systematic review and meta-analysis protocol. BMJ Open 14(3): e081642.
- Iber C, Israel SA, Chessonet AL, Quan SF (2007) The AASM manual for the scoring of sleep and associated events: Rules, terminology and technical specifications. American Academy of Sleep Medicine.
© 2026 Sakra Panthep Indra. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






