- Submissions

Full Text
Advances in Complementary & Alternative medicine
Globally Used Herbal Supplements with Blood-Thinning Effects and their Interactions with Warfarin and Aspirin in Stroke Care: A Narrative Review
Leday AJ1,4, Iyengar D1,3 and Weerasinghe P1,2*
1UT HEALTH McGovern Medical School, The University of Texas Health Sciences Center at Houston (UTHealth) Houston TX, USA
2Department of Pathology and Laboratory Medicine, The University of Texas Health Sciences Center at Houston (UTHealth) Houston TX, USA
3Department of Family Medicine, The University of Texas Health Sciences Center at Houston (UTHealth) Houston TX, USA
4Medstar Georgetown University Hospital, 3800 Reservoir Rd NW, Washington DC, USA
*Corresponding author:Priya Weerasinghe, Department of Pathology and Laboratory Medicine, McGovern Medical School, The University of Texas Health Science Center at Houston (UTHealth), Houston, TX 77030, USA
Submission: April 07, 2026;Published: April 24, 2026

ISSN: 2637-7802 Volume 9 Issue 2
Abstract
Herbal and dietary supplement use is increasingly common, making awareness of herb-drug interactions an important component of patient counseling and medication safety. In stroke care, anticoagulant and antiplatelet agents such as warfarin and aspirin are widely used for prevention and treatment, yet many commonly consumed herbs and supplements may alter hemostasis and increase bleeding risk. This narrative review summarizes the literature on five commonly used herbs with potential blood-thinning properties: garlic, turmeric, cinnamon, cayenne pepper, and ginger. For each, the origin, pharmacologic effects, mechanisms of action, and reported interactions with warfarin and aspirin were examined. The reviewed literature suggests that these herbs may affect coagulation and platelet function through differing mechanisms, resulting in variable levels of clinical risk when used concurrently with standard antithrombotic therapies. Some may potentiate warfarin activity and increase bleeding complications, whereas others may act through alternative pathways with less direct influence on INR but continued relevance to hemorrhagic risk. The findings of this review support the need for clinicians to routinely inquire about herbal and supplement use in patients taking blood-thinning medications. Incorporating herbal counseling into routine stroke care may improve medication safety and reduce preventable adverse events. Additional research is needed to clarify the magnitude and clinical significance of these interactions and to better inform evidence-based counseling practices.
Keywords:Herbal supplements; Herb-drug interactions; Stroke care; Warfarin; Aspirin; Turmeric; Garlic; Ginger; Cinnamon; Cayenne pepper bleeding risk; Hemostasis; Platelet function; Anticoagulants; Antiplatelet therapy
Abbreviations: INR: International Normalized Ratio; PTT: Partial Thromboplastin Time; PT: Prothrombin Time; vWF: Von Willebrand Factor; GPIb: Glycoprotein Ib Receptor; GPIIb/IIIa: Glycoprotein IIb/IIIa Receptor; 12-HETE: 12-Hydroxyeicosatetraenoic Acid; DOACs: Direct Oral Anticoagulants; CIM: Complementary and Integrative Medicine; aPTT: Activated Partial Thromboplastin Time
Introduction
Stroke, or cerebrovascular disease, is the “third major cause of morbidity and mortality in many developed countries” [1]. It is broadly classified into two major subtypes: ischemic stroke and hemorrhagic stroke. Both are strongly influenced by the function, or dysfunction, of the body’s hemostatic and coagulation pathways. In ischemic stroke, excessive coagulation can promote thrombus formation within cerebral blood vessels, obstructing blood flow and depriving brain tissue of oxygen. In contrast, hemorrhagic stroke occurs when damaged blood vessels rupture and bleed into the brain parenchyma. In some cases, ischemic stroke may progress to hemorrhagic transformation, a complication in which damaged or weakened cerebral vessels begin to bleed following the initial ischemic event. This risk may be further increased when blood-thinning medications are not carefully optimized. Because neuronal injury begins within minutes of stroke onset, hospitals often implement rapid intervention metrics such as “door-to- CT” and “door-to-needle” times to facilitate timely diagnosis and treatment. Loss of blood supply and subsequent brain tissue death can lead to substantial impairment in memory, movement, sensation, and other essential neurologic functions. For this reason, stroke care depends heavily on the careful balance between therapeutic benefit and the potential risks of bleeding, toxicity, and drug-drug interactions.
A substantial body of literature has examined interactions between anticoagulant or antiplatelet agents and conventional medications. However, far less attention has been given to the contribution of commonly consumed herbs and spices that may also possess blood-thinning properties. This gap is clinically relevant, particularly in patients taking agents such as warfarin and aspirin for stroke prevention or secondary prevention, where even modest changes in coagulation or platelet activity may alter bleeding risk. Food plays an important role not only in American culture, but across societies worldwide [2]. Beyond its nutritional value, food is closely tied to socioeconomic status, religion, celebration, tradition, and personal identity. Cultural beliefs and regional practices strongly influence dietary patterns, including the routine use of specific herbs and spices. The concept of “luxury foods,” for example, has been described according to refinement in qualitative characteristics, quantity, and variability [3], illustrating how the meaning and value of food differ across social, economic, and political contexts. Even in institutional settings such as prison systems, food can function as an expression of identity, community, customs, and values [4].
Herbs and spices reflect this same cultural significance while also occupying an important place in traditional and homebased health practices [5]. In many parts of the world, their use is further shaped by longstanding healing traditions, including Traditional Chinese Medicine, Ayurvedic medicine, Latin American folk medicine and other Indigenous and regional medical practices. Many commonly used spices contain anti-inflammatory, antiproliferative, and antiangiogenic properties, making them attractive as natural remedies, especially in settings where access to prescription medications may be limited or where traditional healing practices remain prominent [6]. Although the use of such products may be convenient, affordable, and culturally meaningful, their biologic activity may also create unintended risks when they are taken concurrently with pharmaceutical agents that affect coagulation or platelet function. Given the widespread global use of herbal products and the narrow therapeutic balance required in stroke prevention and management, greater attention to herb-drug interactions is warranted. More careful counseling regarding herbal and supplement use may help reduce preventable complications and bleeding risk in patients receiving blood-thinning medications. This review therefore examines commonly used herbs with bloodthinning effects and their potential interactions with warfarin and aspirin, with particular attention to their relevance in stroke care and global health (Figure 1).
Figure 1:Various herbs with potential blood-thinning properties: Garlic (1), ginger (2), cinnamon (3), cayenne pepper (4), and turmeric (5).

Ischemic and hemorrhagic stroke
Stroke can be broadly divided into two major categories: ischemic stroke and hemorrhagic stroke. Ischemic stroke occurs when a blood vessel becomes obstructed, resulting in reduced blood flow and oxygen delivery to the brain. In contrast, hemorrhagic stroke occurs when a cerebral vessel ruptures and bleeds into the brain, likewise impairing oxygen delivery and normal neurologic function [7]. Although these two stroke types are distinct, a combination of both may occur in the serious complication known as hemorrhagic transformation of ischemic stroke. This complication may arise as a consequence of the initial ischemic injury itself or as a result of reperfusion injury following treatment of ischemic stroke [8,9]. Because both stroke types are closely tied to disturbances in vascular integrity and hemostatic balance, treatment strategies must be carefully tailored. In ischemic stroke, the primary goal is restoration and preservation of cerebral perfusion, whereas in hemorrhagic stroke, treatment is directed toward limiting ongoing bleeding and reducing further vascular damage. Antithrombotic medications commonly used in stroke prevention and management include anticoagulants such as heparin and warfarin and antiplatelet agents such as aspirin and indomethacin. These drugs exert their effects through interference with the coagulation cascade and platelet aggregation pathways, respectively.
Hemostasis
Platelets play an essential role in maintaining vascular integrity and supporting the delivery of nutrients and oxygen to vital organs [10]. Primary hemostasis is the process by which platelets aggregate at sites of endothelial injury to form an initial platelet plug. Damage to the endothelial lining triggers vasoconstriction and the release of endothelin. Exposure of the subendothelial matrix promotes release of Von Willebrand factor (vWF) from Weibel-Palade bodies. vWF binds to the exposed site of injury and facilitates platelet adhesion through platelet Glycoprotein Ib (GPIb) receptors. This interaction activates platelets through a conformational change, promoting degranulation and further release of vWF and other mediators that recruit additional platelets in a positive feedback loop. Platelets also bind to one another through induced expression of Glycoprotein IIb/IIIa (GPIIb/IIIa) receptors. With the additional involvement of thromboxane A2 and cyclooxygenase activity, platelet aggregation proceeds and an initial platelet plug is formed. However, this plug is temporary and unstable.
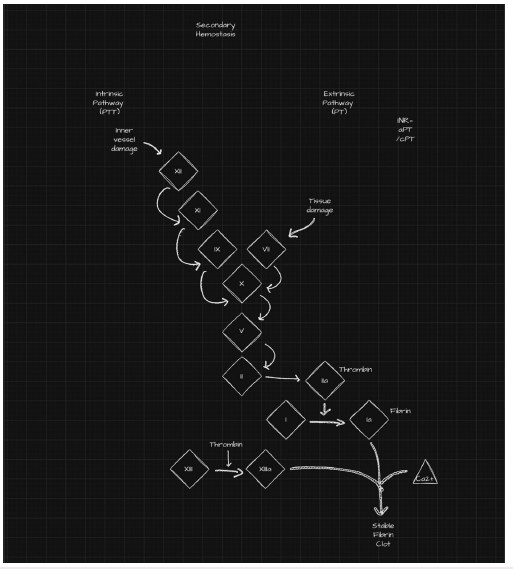
Long-term stabilization requires secondary hemostasis through activation of the coagulation cascade (Figure 2). The coagulation cascade stabilizes the initial platelet plug through the generation of fibrin, thereby reducing the risk of continued hemorrhage. This process is classically divided into the intrinsic and extrinsic pathways, which converge into a common pathway [10]. The intrinsic pathway is activated by endothelial damage and exposure of subendothelial components, whereas the extrinsic pathway is activated by tissue injury and the release of tissue factor. As the cascade progresses, clot formation is reinforced through amplification mechanisms. First, activation of sequential clotting factors increases the local concentration of active intermediates. Second, thrombin generation amplifies clot formation through positive feedback on multiple upstream clotting factors. These coordinated reactions ultimately result in formation of a stable fibrin clot (Figure 3). Clinically, the time required for clot formation through these pathways is used to assess hemostatic function. The intrinsic pathway is primarily evaluated by Partial Thromboplastin Time (PTT), whereas the extrinsic pathway is assessed by Prothrombin Time (PT). The International Normalized Ratio (INR) standardizes PT values by comparing an individual’s PT with that of a healthy control sample. INR is commonly used to monitor anticoagulant therapy, particularly warfarin, in order to optimize treatment while minimizing adverse effects. Coagulation is essential for prevention of hemorrhage, but both excessive and insufficient coagulation can contribute to stroke pathology.
Figure 2:Primary hemostasis: platelet aggregation and formation of an unstable platelet plug.
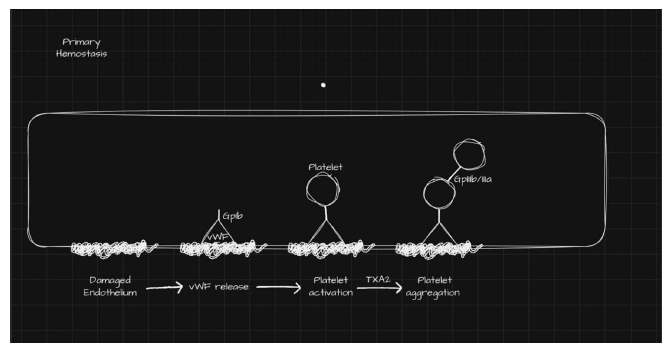
Figure 3:Secondary hemostasis (the coagulation cascade): Formation of a stable fibrin clot.
Hypercoagulability, or excessive activation of the coagulation cascade, may promote inappropriate thrombus formation and increase the risk of ischemic stroke. In contrast, hypocoagulability, characterized by impaired clot formation or an inability to control bleeding, may contribute to hemorrhagic stroke or hemorrhagic transformation of ischemic stroke. Causes of hypocoagulability may include thrombocytopenia, reduced levels of coagulation factors, or overly aggressive reperfusion strategies. Thus, precise regulation of hemostatic pathways is critical to maintaining vascular balance. One important regulatory molecule in this system is thrombin, which participates in both procoagulant and anticoagulant feedback mechanisms. Through its central role in coagulation, thrombin promotes clot formation, but it is also involved in pathways that limit excessive clotting and facilitate fibrinolysis. Thrombin contributes to activation of mechanisms that lead to plasmin generation, allowing fibrin breakdown, and it also interacts with endogenous anticoagulant systems, including antithrombin, to restrain further coagulation [11]. Many current medical therapies target thrombin directly or indirectly to modulate hemostasis in patients at risk for stroke.
Medications affecting hemostasis
Medications that act on clotting pathways must be carefully managed in order to avoid excessive bleeding, hypercoagulability, or other complications that may contribute to stroke. Warfarin, a coumarin derivative commonly used in stroke prevention and treatment, exerts its anticoagulant effect by inhibiting vitamin K-dependent activation of clotting factors II, VII, IX, and X [12,13]. Because warfarin has a narrow therapeutic index, it must be dosed carefully and monitored closely using INR measurements to reduce the risk of under-anticoagulation or bleeding complications. Aspirin, or acetylsalicylic acid, acts by irreversibly inhibiting cyclooxygenase, thereby reducing thromboxane A2 synthesis and impairing platelet aggregation [14].
Because cyclooxygenase and thromboxane A2 are central to platelet activation during primary hemostasis, inhibition of this pathway reduces platelet clot formation and contributes to aspirin’s well-known blood-thinning effect. Aspirin monotherapy has been associated with an increased risk of hemorrhagic stroke, and this risk may increase further when aspirin is combined with other blood-thinning agents. Unlike warfarin, aspirin therapy is not routinely monitored with a standardized laboratory measure in most clinical settings. For this reason, potential interactions with other substances become especially important to recognize during patient counseling and treatment planning. Vitamins, particularly vitamin K, as well as supplements and over-the-counter medications, may influence both primary and secondary hemostasis and may also alter the metabolism or activity of anticoagulant and antiplatelet agents [15]. Failure to consider interactions between blood-thinning medications and highly unregulated, commonly consumed herbal products may therefore result in serious and preventable adverse consequences.
Garlic
Garlic is one of the oldest known horticultural crops and is believed to have originated in Middle Asia, where it was used for a wide range of purposes, including treatment of depression and rheumatism [16]. Across history, garlic has held both medicinal and cultural importance. In ancient Egypt, it was reportedly used to provide strength and sustenance to laborers while also reducing hunger [17]. Garlic bulbs have also been identified in archaeological discoveries from ancient pyramids, further reflecting its historical value. Over time, garlic continued to be used for both religious and medicinal purposes. It was even referred to as “Russian Penicillin” because of its use by Russian physicians in the treatment of respiratory tract diseases. Today, garlic remains widely used both as a culinary ingredient and as a supplement believed to promote general health and prevent illness. As with any biologically active substance, however, safe use requires understanding both its therapeutic properties and its adverse effects. One of the bestknown active derivatives of garlic is ajoene, a sulfur-containing compound that irreversibly inhibits platelet aggregation [18]. This antiplatelet effect is mechanistically similar to the action of aspirin and is particularly relevant in patients at risk for stroke.
The concentration of ajoene appears to correlate with the potency of garlic’s blood-thinning effect, with the highest levels found in oil macerates. Accordingly, supplements containing garlic oil preparations may have greater antiplatelet activity than other formulations. Clinical observations further suggest that garlic supplementation may alter hemostasis in meaningful ways. Case reports have described surgical complications related to poor hemostasis in patients without underlying coagulopathy or use of prescription blood thinners, who were later found to be self selfmedicating with garlic supplements. These findings suggest that garlic may exert clinically relevant antithrombotic effects and raise concern for increased bleeding risk when used concurrently with warfarin or aspirin, given its aspirin-like effect on platelet aggregation. At the same time, not all data have demonstrated significant interaction. In a randomized controlled study of 48 patients with deep vein thrombosis, cerebrovascular accident, thrombosis, valvular heart disease, atrial fibrillation, or prosthetic valves who were receiving warfarin therapy, participants were assigned to receive either aged garlic extract or placebo [19]. All patients were adults and had not used garlic remedies within the previous three months.
The study found no significant difference in bleeding risk between the experimental and control groups and concluded that aged garlic extract did not significantly increase hemorrhagic risk. However, this finding should be interpreted with caution, as aged garlic extract represents a distinct preparation that is less commonly used and contains reduced levels of sulfur compounds. Because sulfur-containing constituents are thought to mediate much of garlic’s biologic activity, the generalizability of these findings to typical dietary or supplemental garlic intake may be limited. Preparation method also appears to be important. The Health Effects of Garlic reports that allicin, another sulfur-containing active substance, is generated when raw garlic is crushed and the enzyme alliinase interacts with its precursor compound [20]. However, alliinase is deactivated by heat, and cooked garlic may therefore be less therapeutically active than raw garlic. Together, these findings suggest that the form and preparation of garlic are important considerations when counseling stroke and cardiovascular patients regarding garlic use during antiplatelet or anticoagulant therapy.
Cinnamon
Cinnamon has been widely used for thousands of years, with records of its use dating back to at least 2800 BC [21]. Historically, it served a variety of cultural, medicinal, and ceremonial functions, including use in Egyptian embalming practices, anointing traditions described in the Bible, and medicinal applications among the Romans. Cinnamon was also a highly valued trade commodity, influencing major voyages such as those of Christopher Columbus and Vasco da Gama and shaping global trade routes, including those involving South India and Sri Lanka. Because of its perceived value and broad therapeutic potential, cinnamon became associated with wealth, power, and medicinal significance. Today, it remains widely used worldwide in traditional dishes, herbal medicine, and the fragrance industry. Four principal types of cinnamon are commonly used: Mexican, Indonesian, Vietnamese, and Cassia cinnamon. These varieties differ in color, flavor, and, importantly, coumarin content. Coumarin is a hepatotoxic and potentially carcinogenic compound from which warfarin was originally derived. Cinnamon also contains several active constituents, including cinnamaldehyde, cinnamate, and cinnamic acid, in addition to a variety of essential oils.
These compounds have been associated with antihypertensive and vasodilatory effects in animal and human studies. Initially, these vascular effects were thought to occur mainly through nitric oxide production; however, other mechanisms have also been proposed. For example, 2-methoxycinnamaldehyde isolated from Cassia cinnamon has been shown to reduce expression of vascular cell adhesion molecule-1 in TNF-α-activated endothelial cells [22]. In C. philippinensis, a less commonly used species, cinnamophilin has been reported to block thromboxane synthase and thromboxane A2 receptors in smooth muscle while also reducing calcium influx and release, thereby contributing to vasodilation. Other constituents, including quercetin and flavones, have also been reported to influence fibrinogenesis and clot degradation [23]. Although the precise mechanism responsible for cinnamon’s blood-thinning effect remains incompletely defined, available evidence supports its capacity to influence vascular and hemostatic function. The clinical implications of cinnamon use are particularly important in patients receiving anticoagulant therapy. Because warfarin is derived from coumarin and cinnamon contains variable amounts of coumarin depending on the species, concomitant use may increase bleeding risk [24].
Cassia cinnamon, one of the most commonly consumed forms, contains approximately 5mg of coumarin per teaspoon. In patients already taking warfarin, additional coumarin exposure from cinnamon may further impair coagulation and increase the risk of hemorrhage. At the same time, some findings suggest possible therapeutic benefit when cinnamon is used with antiplatelet therapy. In a randomized controlled study of 122 patients with transient ischemic attack or mild stroke, cinnamon was evaluated as an adjunct to aspirin for prevention of stroke recurrence [25]. Patients receiving aspirin plus Chinese cinnamon had a 3.2% 90-day recurrence rate, whereas those receiving aspirin alone had a 15% recurrence rate. These findings suggest that cinnamon may have a potentially useful role in stroke care under carefully controlled circumstances. However, such use would require close attention to cinnamon type, concomitant medications, and adverse effect risk. As with garlic, preparation also appears relevant, as heat has been shown to deactivate some of cinnamon’s active compounds [26]. This should be considered when counseling patients, since culinary preparation may alter the concentration and activity of its biologically active constituents.
Turmeric
Turmeric, a plant in the ginger family, has a history spanning approximately 4000 years and is deeply rooted in Indian culture, where it continues to be widely produced and consumed for culinary, cosmetic, and medicinal purposes [27]. Traditionally, it has been used in the treatment of conditions such as rheumatoid arthritis, conjunctivitis, and dyspepsia. Within Ayurvedic practice, turmeric has also been valued for its proposed benefits in improving circulation, supporting digestion, increasing energy, and providing antiparasitic and antiseptic effects. Beyond traditional medicine, turmeric is used widely in food products such as cheese, butter, cake icing, and orange juice, to which it imparts a characteristic yellow color. Because of its broad cultural significance and extensive use, turmeric intake has been reported to reach as high as 1000mg/day in some Asian populations. In the United States, billions of dollars are spent annually on turmeric and other herbal therapies, underscoring the importance of evaluating its safety and interaction potential. Curcumin, along with related curcuminoids, is the principal active component of turmeric and is responsible for both its yellow color and many of its proposed medicinal properties.
Investigation of curcumin has been complicated by its poor oral bioavailability and chemical instability [28]. Nevertheless, in vitro studies have shown that curcumin may inhibit platelet aggregation through several mechanisms. One proposed pathway involves reduced thromboxane formation, leading to increased 12-lipoxygenase activity and subsequent production of 12-hydroxyeicosatetraenoic acid (12-HETE), a molecule that inhibits platelet aggregation and, at higher concentrations, may further suppress thromboxane formation. Additional proposed mechanisms include inhibition of cyclooxygenase pathways and calcium signaling. These observed effects on coagulation and thrombosis may have relevance in stroke and other thrombotic conditions [29]. It has also been reported that daily turmeric consumption may help maintain an anticoagulant state by significantly prolonging activated partial thromboplastin time and activated prothrombin time [30]. Data on turmeric’s interaction with blood-thinning medications are mixed. In a study using Wistar rats, combinations of curcumin with warfarin or clopidogrel were compared with warfarin or clopidogrel alone [31].
Although the combinations did not significantly alter prothrombin time or platelet aggregation rate, they did affect the pharmacokinetics of both drugs, increasing exposure to warfarin and clopidogrel by 1.6-fold and 1.8-fold, respectively. One proposed mechanism is inhibition of cytochrome P450 isoenzymes and P-glycoprotein, which may reduce drug metabolism and prolong the presence of active compounds in the bloodstream [32]. This is particularly relevant for warfarin because of its narrow therapeutic index. Even modest changes in warfarin metabolism can significantly increase bleeding risk. The interaction with clopidogrel may be more complex. Because clopidogrel requires CYP-mediated activation, inhibition of this pathway may reduce its conversion to its active form and potentially decrease therapeutic efficacy. In such cases, turmeric use may paradoxically contribute to inadequate antiplatelet effect and increase the risk of thrombosis if not accounted for during treatment planning.
Ginger
Ginger has been used for at least 2500 years, with early records tracing its use to China [33]. In addition to its culinary role, it has historically been used to address a broad range of conditions, including snakebites, nausea, poor digestion, hair loss, cardiovascular disease, hypercholesterolemia, and even as a mosquito repellent. Its perceived value led to the establishment of ginger plantations in Jamaica by the Spanish in the 1600s. Today, ginger remains widely used for its antimicrobial, antiinflammatory, antitumor, and antioxidant properties [34]. At the same time, concerns persist regarding its safety when combined with anticoagulant or antiplatelet medications such as warfarin, aspirin, and clopidogrel. One case report described a 70-year-old woman receiving long-term warfarin therapy for a cerebrovascular accident whose INR increased from 2.7 to 8.0 after initiation of a “Ginger Rescue” supplement [35]. The patient reported no other recent dietary changes. One week after discontinuing the supplement and holding three doses of warfarin, her INR declined to 2.6.
When warfarin was restarted at the original dose of 7.5mg daily, the INR remained within therapeutic range.
This observation suggests a possible clinically significant interaction and highlights the need to counsel patients receiving anticoagulants about ginger-containing supplements. The principal active components of ginger are believed to include terpene and phenolic compounds, the concentrations of which may vary depending on growing conditions, heat exposure, and storage duration [36]. In vitro studies have shown that ginger may decrease angiotensin-converting enzyme activity and arginase, leading to increased nitric oxide levels and vasodilation. In hypertensive rats, ginger treatment was associated with a reduction in blood pressure. Ginger extract has also been reported to suppress nitric oxide synthase and cyclooxygenase.
A cross-sectional study involving 4628 participants supported these vascular findings, reporting a lower probability of hypertension among individuals who consumed ginger daily [37]. Despite these hemodynamic effects, direct evidence regarding ginger’s anticoagulant interaction profile remains inconsistent. One rat study found that ginger did not significantly affect the anticoagulant activity of warfarin and likewise did not significantly alter platelet aggregation [38]. Additional studies have similarly failed to identify a clear interaction between ginger and warfarin. However, the case reports described above remain concerning. In another report, a 76-year-old woman developed an INR as high as 10 with associated epistaxis after using ginger products [39]. Her INR returned to normal after discontinuation of ginger. Taken together, these conflicting findings suggest that evidence remains insufficient to define the safety of ginger use in patients taking blood thinners such as warfarin [40]. One possible explanation for the inconsistency across studies is a dose-dependent effect, as well as differences in formulation and preparation.
Cayenne pepper
Cayenne pepper, also known as red pepper or capsicum, has been used since at least 5500BC in Mexico, including as a food preservation agent [41]. In addition to its culinary use, cayenne pepper has long been recognized for medicinal properties that include antimicrobial, anti-inflammatory, analgesic, antihypertensive, and thermoregulatory effects, and has also historically been regarded as an aphrodisiac. Among Native American communities, capsaicin-containing peppers were used for pain relief for centuries. Today, capsaicin continues to be used worldwide and is even incorporated into prescribed treatments for conditions such as neuropathy and osteoarthritis. Its analgesic effect is believed to arise in part from depletion of substance P, an important mediator of pain signaling between the peripheral and central nervous systems [42]. Despite these benefits, cayenne pepper is often described as contraindicated in individuals using aspirin, blood-thinning medications, or other herbs with anticoagulant properties. Capsicum species comprise more than 31 varieties [43]. Among the most commonly used are C. annuum, C. baccatum, C. chinense, C. frutescens, and C. pubescens. C. annuum has been reported to contain compounds with anticoagulant and antiplatelet properties.
Some studies suggest that capsaicin may prevent clot formation relevant to stroke. Hogaboam et al. [42] found that capsaicin disrupted platelet aggregation in the presence of thrombin, even after aggregation had already begun, and that this effect was reversible after removal of capsaicin [44]. In addition to these hemostatic effects, capsaicin has also demonstrated potential neuroprotective properties. One study identified TRPV1-dependent downregulation of NMDA receptors in cortical neurons [45], while another reported delayed stroke onset and increased survival time with long-term capsaicin exposure through a related TRPV1- mediated mechanism [46]. Despite these potentially beneficial findings, few studies have directly assessed the interaction between capsaicin and anticoagulant or antiplatelet medications. Some reports suggest that capsaicin may be more effective than commonly used agents such as aspirin and indomethacin in certain experimental settings [47], but the absence of robust clinical data makes it difficult to define its true safety profile in patients receiving blood-thinning therapy. For now, concurrent use is often discouraged as a precaution, and further research is needed to clarify the risks and benefits of cayenne pepper in this context.
Discussion
Stroke care remains a major clinical priority, particularly as stroke continues to rank among the leading causes of morbidity and mortality in developed countries. Prevention and treatment of ischemic stroke rely heavily on careful regulation of hemostasis through anticoagulant and antiplatelet therapies such as warfarin and aspirin. Because these medications act on tightly balanced coagulation and platelet pathways, factors that alter hemostasis may significantly affect clinical outcomes. Failure to recognize interactions between prescribed blood-thinning medications and commonly used herbal products may therefore place patients at increased risk of bleeding complications, including hemorrhagic stroke and hemorrhagic transformation. The herbs reviewed in this paper, including garlic, cinnamon, turmeric, ginger, and cayenne pepper, have each been reported to possess blood-thinning, antiplatelet, anticoagulant, or otherwise hemodynamically relevant effects. However, the extent and mechanism of these effects vary considerably across herbs, and the available evidence is often limited by differences in formulation, preparation, dosage, and study design. While not all of these herbs can be conclusively linked to direct effects on primary or secondary hemostasis, the literature suggests sufficient potential for clinically meaningful interactions to justify greater caution in patients receiving warfarin, aspirin, and other antithrombotic therapies.
An additional consideration is the potential for interactions between these herbal agents and Direct Oral Anticoagulants (DOACs), including dabigatran, apixaban, rivaroxaban, and edoxaban. Compared with warfarin, the evidence base for herb DOAC interactions remains more limited, and current guidance suggests that many proposed interactions are supported primarily by mechanistic reasoning, expert opinion, and case reports rather than robust clinical trials. Nevertheless, caution is warranted, as garlic and ginger may contribute antiplatelet or anticoagulant effects, curcumin may influence P-glycoprotein-mediated drug handling, and cinnamon, particularly Cassia cinnamon, has been implicated together with ginger in a reported fatal bleeding event in a patient receiving dabigatran. In contrast, evidence for cayenne pepper remains sparse and largely theoretical. As the use of DOACs continues to expand in stroke prevention and management, further research is needed to clarify the magnitude and clinical significance of bleeding risk when these agents are used concurrently with commonly consumed herbal products. Beyond these clinically established concerns, future studies may also benefit from examining exploratory mechanistic biomarkers such as SIRT1, which has been implicated in pathways related to oxidative stress, inflammation, mitochondrial dysfunction, and ischemic injury [48].
At the same time, because SIRT1 remains an emerging rather than established clinical biomarker in stroke, its assessment would be most informative when considered alongside standard laboratory markers, particularly platelet count, PT, INR, and aPTT, as well as appropriate liver and renal function tests. Incorporating both conventional clinical measures and selected mechanistic biomarkers may provide a more comprehensive evaluation of coagulation status, bleeding risk, and potential systemic effects associated with concurrent use of herbal products and antithrombotic therapies in stroke patients. These findings also carry important clinical, educational, and professional implications. More thorough counseling regarding the possible benefits, risks, and interactions of herbal products may help reduce preventable adverse events and support safer individualized treatment planning. As herbal supplement use continues to expand across culturally diverse populations, physicians and other healthcare professionals must be prepared not only to recognize potential herb-drug interactions, but also to discuss them effectively within the broader context of patient-centered care.
Conclusion
Herbal products commonly used in everyday diets and traditional health practices, including garlic, cinnamon, turmeric, ginger, and cayenne pepper, may exert clinically relevant effects on hemostasis. When used concurrently with antithrombotic therapies such as warfarin, aspirin, and direct oral anticoagulants, these agents have the potential to alter coagulation and platelet function, thereby increasing the risk of bleeding complications. Although the strength of evidence varies and is often limited by heterogeneity in study design and reliance on case reports or mechanistic data, the cumulative literature suggests that these interactions are sufficiently plausible to warrant clinical attention. Given the widespread and often underreported use of herbal supplements, routine assessment of herbal product use should be incorporated into standard medication histories, particularly in patients receiving anticoagulant or antiplatelet therapy for stroke prevention and management. Increased awareness, patient counseling, and careful clinical judgment are essential to minimizing preventable adverse events. Future research should prioritize well-designed clinical studies to better define the magnitude, mechanisms, and clinical significance of herb–drug interactions involving both traditional anticoagulants and newer direct oral anticoagulants. Strengthening the evidence base in this area will be critical to informing clinical guidelines and improving patient safety. Overall, integrating awareness of herbal medicine into clinical practice, medical education, and ongoing professional development represents an important step toward safer, more comprehensive, and culturally responsive care in stroke prevention and treatment.
Acknowledgement
The authors thank Dr. Maximilian Buja, Professor in the Department of Pathology and Laboratory Medicine, for his review of this article, and the Department of Pathology and Laboratory Medicine for its support of this project.
Disclaimer
The views expressed in this manuscript are those of the author(s) and do not necessarily reflect the official policy or position of the Defense Health Agency, the Department of War, nor any agencies under the U.S. Government.
References
- Unnithan AKA, Das JM (2025) Hemorrhagic stroke overview. StatPearls Publishers, Florida, USA.
- Monaco GL, Bonetto E (2019) Social representations and culture in food studies. Food Research International 115: 474-479.
- Veen MVD (2003) When is food a luxury? World Archaeology 34(3): 405-427.
- Simanovic T, Gosev M (2019) Is food more than a means of survival? An overview of the Balkan prison systems. Appetite 143: 104405.
- Tapsell LC, Hemphill I, Cobiac L, Patch CS, Sullivan DR, et al. (2006) Health benefits of herbs and spices: The past, the present, the future. The Medical Journal of Australia 185(S4): S1-S24.
- Kim DC, Ku SK, Bae JS (2012) Anticoagulant activities of curcumin and its derivative. BMB Reports 45(4): 221-226.
- Kovács KB, Bencs V, Hudák L, Oláh L, Csiba L (2023) Hemorrhagic transformation of ischemic strokes. International Journal of Molecular Sciences 24(18): 14067.
- Hong JM, Kim DS, Kim M (2021) Hemorrhagic transformation after ischemic stroke: Mechanisms and management. Frontiers in Neurology 12: 703258.
- Saad J, Asuka E, Schoenberger L (2023) Physiology, platelet activation. StatPearls Publishers, Florida, USA.
- Chaudhry R, Usama SM, Babiker HM (2025) Physiology, coagulation pathways. StatPearls Publishers, Florida, USA.
- Seto AH (2018) 3-interventional pharmacology. The Interventional Cardiac catheterization handbook. (4th edn), Elsevier Publishers, Amsterdam, Netherlands, pp: 88-106.
- Li JH, Voora D (2018) Chapter 16-cardiovascular pharmacogenetics. Genomic and precision medicine. (3rd edn), Elsevier Publishers, Amsterdam, Netherlands, pp: 291-307.
- Arif H, Aggarwal S (2023) Salicylic acid (Aspirin). StatPearls Publishers, Florida, USA.
- Cordier W, Steenkamp V (2011) Herbal remedies affecting coagulation: A review. Pharmaceutical Biology 50(4): 443-452.
- Philipp S (2020) The origins and distribution of garlic: How many garlics are there?
- Petrovska BB, Cekovska S (2010) Extracts from the history and medical properties of garlic. Pharmacognosy Reviews 4(7): 106-110.
- Lawn A, Sains P (2012) Not just a vampire repellent: The adverse effects of garlic supplements in surgery. Annals of the Royal College of Surgeons of England 94(6): 451.
- Macan H, Uykimpang R, Alconcel M, Takasu J, Razon R, et al. (2006) Aged garlic extract may be safe for patients on warfarin therapy. The Journal of Nutrition 136(3 Suppl): 793S-795S.
- Tattelman E (2005) Health effects of garlic. American Family Physician 72(1): 103-106.
- Kawatra P, Rajagopalan R (2015) Cinnamon: Mystic powers of a minute ingredient. Pharmacognosy Research 7(Suppl 1): S1-S6.
- Rao PV, Gan SH (2014) Cinnamon: A multifaceted medicinal plant. Evidence-Based Complementary and Alternative Medicine 2014: 642942.
- Ahmadi E, Shahri MM (2019) The antioxidant and anticoagulation effects of coumarin and quercetin from cinnamon methanolic extract, and the assessment of cinnamon powder effect on plasma parameters in diabetes, and the disinfectant activity in diabetic patients. Herbal Medicine Journal 4(3): 103-110.
- Maadarani O, Bitar Z, Mohsen M (2019) Adding herbal products to direct-acting oral anticoagulants can be fatal. European Journal of Case Reports in Internal Medicine 6(8): 001190.
- Zhang L, Li Z, Wu Y, Fan Y, He Z, et al. (2022) Cinnamon and aspirin for mild ischemic stroke or transient ischemic attack: A pilot trial. Clinical Therapeutics 44(4): 482-490.
- Santos RR, Andrade M, Madella D, Martinazzo AP, Moura LDAG, et al. (2017) Revisiting an ancient spice with medicinal purposes: Cinnamon. Trends in Food Science & Technology (62): 154-169.
- Prasad S, Aggarwal BB (2011) Turmeric, the golden spice: From traditional medicine to modern medicine. Herbal Medicine: Biomolecular and Clinical Aspects. (2nd edn), Chapter 13.
- NIH (2020) Turmeric. National Center for Complementary and Integrative Health.
- Keihanian F, Saeidinia A, Bagheri RK, Johnston TP, Sahebkar A (2018) Curcumin, hemostasis, thrombosis, and coagulation. Journal of Cellular Physiology 233(6): 4497-4511.
- Liu AC, Zhao LX, Lou HX (2013) Curcumin alters the pharmacokinetics of warfarin and clopidogrel in Wistar rats but has no effect on anticoagulation or antiplatelet aggregation. Planta Medica 79(11): 971-977.
- Bahramsoltani R, Rahimi R, Farzaei MH (2017) Pharmacokinetic interactions of curcuminoids with conventional drugs: A review. Journal of Ethnopharmacology 209: 1-12.
- Moghaddasi MS, Kashani HH (2012) Ginger (Zingiber officinale): A review. Journal of Medicinal Plants Research 6(26): 4255-4258.
- Modi M, Modi K (2024) Ginger Root. National Library of Medicine National Center for Biotechnology Information.
- Rubin D, Patel V, Dietrich E (2019) Effects of oral ginger supplementation on the INR. Case Reports in Medicine 2019: 8784029.
- Mao QQ, Xu XY, Cao SY, Gan RY, Corke H, et al. (2019) Bioactive compounds and bioactivities of ginger (Zingiber officinale Roscoe). Foods 8(6): 185.
- Wang Y, Yu H, Zhang X, Feng Q, Guo X, et al. (2017) Evaluation of daily ginger consumption for the prevention of chronic diseases in adults: A cross-sectional study. Nutrition 36: 79-84.
- Jiang X, Williams KM, Liauw WS, Ammit AJ, Roufogalis BD, et al. (2005) Effect of ginkgo and ginger on the pharmacokinetics and pharmacodynamics of warfarin in healthy subjects. British Journal of Clinical Pharmacology 59(4): 452-432.
- Vaes LP, Chyka PA (2000) Interactions of warfarin with garlic, ginger, ginkgo, or ginseng: Nature of the evidence. The Annals of Pharmacotherapy 34(12): 1478-1482.
- Krüth P, Brosi E, Fux R, Mörike K, Gleiter CH (2004) Ginger-associated overanticoagulation by phenprocoumon. Annals of Pharmacotherapy 38(2): 257-260.
- Bortolotti M (2013) Red pepper: From the kitchen to the pharmacy. Journal of Gastrointestinal and Liver Diseases 22(3): 253-256.
- Pawar SS, Bharude NV, Sonone SS, Deshmukh RS, Raut AK, et al. (2011) Chillies as food, spice and medicine: A perspective. International Journal of Pharmacy and Biological Sciences 1(3): 311-318.
- Batiha GE, Alqahtani A, Ojo OA, Shaheen HM, Wasef L, et al. (2020) Biological properties, bioactive constituents, and pharmacokinetics of some Capsicum spp. and Capsaicinoids. International journal of molecular sciences 21(15): 5179.
- Hogaboam CM, Wallace JL (1991) Inhibition of platelet aggregation by capsaicin. An effect unrelated to actions on sensory afferent neurons. European Journal of Pharmacology 202(1): 129-131.
- Pasierski M, Szulczyk B (2022) Beneficial effects of capsaicin in disorders of the central nervous system. Molecules 27(8): 2484.
- Xu X, Wang P, Zhao Z, Cao T, He H, et al. (2011) Activation of transient receptor potential vanilloid 1 by dietary capsaicin delays the onset of stroke in stroke-prone spontaneously hypertensive rats. Stroke 42(11): 3245-3251.
- Wang JP, Hsu MF, Hsu TP, Teng CM (1985) Antihemostatic and antithrombotic effects of capsaicin in comparison with aspirin and indomethacin. Thrombosis Research 37(6): 669-679.
- Martins IJ (2018) Indian spices and biotherapeutics in health and chronic disease. Health 10(4): 374-380.
- Martins IJ (2018) Sirtuin 1, a diagnostic protein marker and its relevance to chronic disease and therapeutic drug interventions. EC Pharmacol Toxicol 6(4): 209-215.
- Tang H, Wen J, Qin T, Chen Y, Huang J, et al. (2023) New insights into Sirt1: Potential therapeutic targets for the treatment of cerebral ischemic stroke. Front Cell Neurosci 17: 1228761.
© 2026 Weerasinghe P. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






