- Submissions

Full Text
Advances in Complementary & Alternative medicine
Effects of the Combination of Beetroot and Pomegranate Extracts on Flow-Mediated Dilation and Fatigue in Adult Humans: A Randomized, Double-Blind, Placebo- Controlled, Dose-Ranging, Cross-Over Study
Yateesh Jayashankara1, Lincy Joshua2 and Jestin V Thomas2*
1BGS Global Institute of Medical Sciences, BGS Health and Education City, Karnataka, India
2Leads Clinical Research and Bio Services Pvt. Ltd., Karnataka, India
*Corresponding author:Jestin V Thomas, Leads Clinical Research and Bio Services Pvt. Ltd., Karnataka, India
Submission: January 10, 2025;Published: January 27, 2026

ISSN: 2637-7802 Volume 9 Issue 1
Abstract
Introduction: Dietary supplements rich in nitrates and ellagic acid such as beetroot and pomegranate
extracts respectively can enhance cardiorespiratory endurance and help in sustained performance,
efficient recovery, reduced fatigue, increased metabolic efficiency, preventing injury, and improved health.
In this study, a combination of hydro-alcoholic extracts of beetroot and pomegranate peel extract (BPE) is
investigated for its effects on flow-mediated dilation in the brachial artery and fatigue reduction.
Methods: This was a prospective, randomized (1:1:1=BPE 500mg: BPE 1000mg: Placebo), double-blind,
placebo-controlled, three-treatment, three-period, dose-ranging, crossover clinical interventional study
involving a population of 21 healthy individuals aged 20 to 35 years. The primary endpoint assessed
the mean Flow-Mediated Dilation (FMD); secondary outcomes comprised brachial artery diameter, blood
flow velocity, and Chalder Fatigue Scale (CFS). The efficacy endpoints included the change from baseline
in BPE500 and BPE1000 relative to placebo, measured by FMD of the brachial artery, brachial artery
diameter, and blood flow velocity at 1- and 2-hours post-dose on Day 1 and Day 7. CFS was evaluated at
baseline and on Day 7 of each treatment period to measure changes in fatigue.
Result: Twenty-one subjects completed three treatment periods. Both the doses of BPE showed significant
improvements (p<0.05) in mean FMD change compared to placebo as early as 1-hour post dose (BPE500-
increase by 36% on Day 1 and 40% on Day 7; BPE1000-increase by 39% on Day 1 and 44% on Day 7)
and lasting over 2-hour post dose (BPE500-increase by 44% on Day 1 and 43% on Day 7; BPE1000-
increase by 43% on Day 1 and 48% on Day 7). There were no significant differences between BPE500 and
BPE1000. Further, BPE1000 showed significant increase in blood flow velocity 2-hour post dose on Day 7.
In addition, both BPE500 and BPE1000 showed significant decrease in fatigue score on Day 7. No safety
issues were observed throughout the study.
Conclusion: Both BPE500 and BPE1000 showed significant efficacy in improving the flow mediated
dilation, and fatigue without causing any adverse effects. The product was found to be safe and well
tolerated. Further, long-term studies are required to establish the effects of BPE for energy and exercise
performance.
Keywords:Beetroot; Pomegranate; Flow Mediated Dilation (FMD); Physical endurance; Athlete; Fatigue
Abbreviations: AE: Adverse Event; BMI: Body Mass Index; BPE: Beetroot and Pomegranate Extract; BPE500: Beetroot and Pomegranate Extract 500mg; BPE1000: Beetroot and Pomegranate Extract 1000mg; CFQ/CFS: Chalder Fatigue Questionnaire/Chalder Fatigue Scale; CRP: C-Reactive Protein; CTRI: Clinical Trials Registry of India; FMD: Flow-Mediated Dilation; HPLC: High-Performance Liquid Chromatography; ICAM-1: Intercellular Adhesion Molecule-1; ICMR: Indian Council of Medical Research; NF-κB: Nuclear Factor Kappa-B; NO: Nitric Oxide; NO₃⁻: Nitrate; SAE: Serious Adverse Event; SD: Standard Deviation; SE: Standard Error; TNF-α: Tumor Necrosis Factor-Alpha; UPT: Urine Pregnancy Test; VCAM-1: Vascular Cell Adhesion Molecule-1; VO₂ max: Maximal Oxygen Uptake
Introduction
Cardiorespiratory endurance metabolism is a complex process that involves efficiently supplying oxygen to skeletal muscles and energy to physical activities over extended periods [1,2]. Though several systems within the body are involved, cardiovascular, and respiratory systems play vital roles specific to whole-body performance in gas exchange, oxygen transport, oxygen utilization, blood circulation, blood pressure regulation, energy production, heart function, etc., [1,3-6]. In sports, improved cardiorespiratory endurance is a performance deciding factor and helps in attaining sustained performance, efficient recovery, reduced fatigue, increased metabolic efficiency, preventing injury, and improved health [7,8]. Two principle metabolic processes involved in cardiorespiratory endurance are aerobic and anaerobic metabolisms. Aerobic metabolism extracts energy as Adenosine Triphosphate (ATP) from amino acids, carbohydrates, and fatty acids via aerobic capacity through oxygen supply while anaerobic metabolism uses glycolysis and fermentation to produce energy without the use of oxygen [9,10]. Aerobic metabolism produces more ATP than anaerobic but is slower in process.
Maximum Oxygen Uptake (VO2max), ventilatory thresholds, and energy efficiency or economy are limiting performance in endurance training and sports [8]. Nutritional supplementation and adjusting their intake, according to the exercise performed, improves athletic performance in healthy manner [11]. Beta vulgaris (Beetroot) extract is believed to enhance performance by increasing blood flow due to higher nitrate content. Besides, Beetroot is rich in antioxidants such as betalains, vitamin C, and micronutrients including potassium, sodium, magnesium, etc., [8,12,13]. Antioxidants properties of Beetroot protect the system from oxidative stress which might expedite recovery and subsequent performance [14]. Nitric oxide contributes to the maintenance of a healthy endothelial function, as it prevents longterm atherosclerotic disease progression and cardiovascular event rates because it exerts important vasodilatory, anti-inflammatory, antithrombotic, anti-proliferative, and anti-adhesive effects [15,16]. A validated non-invasive method to examine cardiovascular health effects of nutritional interventions that are known to affect NO bioavailability is brachial artery Flow-Mediated Vasodilation (FMD) [17]. Brachial artery diameter, and blood flow velocity are other important predictor of FMD [18].
Punica granatum (Pomegranate) extract is characterized for containing considerable amounts of biologically active phytochemicals including flavonoids (e.g. anthocyanins, catechins, quercetin, and rutin), other types of polyphenols, ellagitannins, and antioxidant vitamins. Many of these phytochemicals have been shown to possess antioxidant and anti-inflammatory properties plus additional biological activities such as inhibition of angiotensin converting enzyme. All these activities of the pomegranate are potentially beneficial for the treatment of hypertension and improvement of endothelial function [19,20]. Studies on nitrate supplementation have reported reduced hypertension, oxygen uptake, and blood lactate; enhanced exercise tolerance, exercise performance, and mitochondrial efficiency; and increased oxygenation and blood flow to skeletal muscle. Several exogenous food sources have been purported to naturally augment this pathway [21]. BPE contains homogenized beetroot extract standardized for nitrates and pomegranate peel extract standardized for ellagic acid in a proprietary ratio. Also, the polyphenol contents present in beetroot and pomegranate are believed to increase blood flow and vessel dilation [21]. The current study investigated the efficacy and safety of BPE on FMD and fatigue in healthy subjects.
Materials and Methods
This was a prospective, randomized (1:1:1), double-blind, placebo-controlled, three treatments, three period, dose-ranging, cross-over clinical interventional study. The total duration of the study was about 38 days including a screening period of 7 days. The study was initiated after obtaining written approval from an institutional ethics committee, BGS Global Institute of Medical Sciences Institutional Ethics Committee, Bangalore, India. The study was carried out as per the requirements of the Indian Council of Medical Research (ICMR) ethical guidelines, International Council for Harmonization (ICH) Guidance on Good Clinical Practice (E6R2) and the Declaration of Helsinki. The study was registered with the Clinical Trials Registry of India (CTRI/2023/10/058371).
Eligibility criteria
Inclusion criteria: Subjects who met all the following inclusion criteria were included in the study. Healthy and physically active male/female volunteers aged between 20 to 35 years; with BMI≥20 and ≤29.9kg/m2; systolic blood pressure ≤139 and diastolic blood pressure ≤89 mmHg; who were willing to abstain from alcohol, caffeine, and vigorous physical activity for 24 hours prior to every study visit; who agreed to have at least 8-hour sleep before the visit days and during the study period; who were willing to abstain from moringa, spinach or other ingredients known to increase vasodilation for 48 hours prior to every study visit; who were willing and able to understand and comply with the requirements of the study, consume the study investigational product as instructed, return for the required treatment period visits, comply with therapy prohibitions, and be able to complete the study
Exclusion criteria: Subjects who met any of the following criteria were excluded from the study. Subjects with hypersensitivity or history of allergy to the study product; with history of pulmonary disorders (asthma, COPD, pulmonary fibrosis, pneumonia, etc.); having FBG levels ≤125mg/dL; who were on/or had history of taking antihypertensives/diuretics; who were on performance enhancing supplements; smokers and tobacco users; with sleep disturbances and/or were taking sleep aid medication; with any chronic conditions which in the opinion of investigator may interfere with the study outcomes; who were pregnant, nursing, or planning a pregnancy within the study participation period; who had been treated with any investigational drug or investigational device within a period of 3 months prior to study entry.
Study procedures
After the informed consent process, demographic details such as date of birth, sex, ethnicity and race were obtained. Medical history including hypertension, diabetes mellitus, chronic liver, gastrointestinal, renal disorder, psychiatric disorders, surgeries, any other clinically significant medical and medication history, and details of prior and concomitant medications were obtained. During the screening visit, blood samples were collected for laboratory assessments, FBG and safety assessments (CBC, liver function tests [ALT, AST], and renal function tests [serum creatinine]). Urine Pregnancy Test (UPT) for females of childbearing potential was performed during Screening/Baseline Visit. At randomization visit, eligible subjects were randomly (double-blinded) assigned in 1:1:1 ratio to one of the three treatment groups i.e. BPE 500mg (BPE500), BPE 1000mg (BPE1000) or Placebo. BPE was a mixture of homogenized Beetroot and pomegranate peel extracts standardized to contain Not Less Than (NLT) 2.5% nitrates and NLT 10% ellagic acid (commercially known as BeepActiveTM), as measured by high-performance liquid chromatography (HPLC). Placebo contained microcrystalline cellulose.
Both the investigational product and placebo were manufactured by Samriddh Nutractive Private Limited, India. The subjects were instructed to consume two capsules every morning after breakfast, at the same time every day, for seven consecutive days. Subjects in BPE500 received one capsule of BPE500 and one capsule of placebo; subjects in BPE1000 received two capsules of BPE500 and subjects in placebo group received two capsules of placebo. The study investigational product was dispensed by designated personnel at the investigative site who did not participate in any other activity pertaining to subject’s safety and efficacy assessment that impacted the study outcome. There were three treatment periods and two washout periods of five days between the treatment periods. After finishing each washout period, the subjects crossed-over to next treatment as per their randomized treatment sequence.
Safety and efficacy parameters
Clinical assessments included all the measures required for the primary and secondary objectives of the study. FMD procedure was performed at baseline (pre-dose) and at 1 hour and 2 hours postdose on Day 1, Day 13 and Day 25 by Ultrasound (Samsung HS70A, South Korea). Baseline and post-deflation FMD, brachial artery diameter and blood flow velocity measures were quantitatively analysed using FMD Studio from Quipu (Quipu srl, Italy). Similarly, the procedure was repeated at 1 and 2 hours post dose on Day 7, Day 19 and Day 31. Chalder Fatigue Scale (CFQ) was administered at baseline and Day 7 of each treatment period to assess change in subjects mental and physical energy state. Safety assessments included monitoring of adverse events, physical examination, vital signs measurement, UPT (for females of childbearing potential), haematology and clinical chemistry tests.
Statistical analysis
A sample size of 21 subjects, was considered sufficient to detect a clinically important difference between groups with 80% power and a 5% level of significance. Statistical analyses were performed using R-software after all subjects had ended their participation in the study and the database was locked. Categorical variables were summarized using frequencies and percentages. Continuous variables were summarized using descriptive statistics (number of subjects with an observation [N], mean, SD, SE, median, minimum and maximum). The test for significance between the treatment groups was performed using independent t-test. The test for significance within treatment groups was performed using paired t-test. The criterion for a significant test by treatment was set at a p<0.05. Demographic and baseline characteristics such as age, height (cm), weight (kg), BMI (kg/m2) were summarized and tabulated by randomized treatment group and overall. The analysis was based on the randomized population.
Efficacy endpoint evaluation
Primary efficacy endpoint analysis: The primary efficacy
endpoint was the change from baseline for BPE500 and BPE1000
compared to placebo on FMD from brachial artery from baseline to
1-, and 2-hours post-dose on Day 1 and Day 7.
Secondary efficacy endpoint analysis: The secondary efficacy
endpoint was the change from baseline for BPE500 and BPE1000
compared to placebo on brachial artery diameter, and blood flow
velocity from baseline to 1-, and 2-hours post-dose on Day 1 and
Day 7 and Chalder fatigue scale scores on Day 7.
Safety analysis: The safety endpoints for the study were
clinically significant changes from baseline to the end of the
treatment in in laboratory parameters (AST, ALT, CBC, serum
creatinine), incidence of AE/SAEs reported, and vital sign
parameters. The analysis was performed on the safety population.
Result
Overall, 29 subjects were screened and 08 were screen failures in the study. A total of 21 subjects were randomized in the study and all 21 subjects completed the study (Figure 1). The study was conducted in 12 male and 09 female subjects (Table 1). All subjects in the study were of Indian origin. The mean (±SE) age of enrolled subjects was 29.62±0.83 years, body weight 67.58±2.00 kg, height 162.60±1.89 cm and BMI 25.18±0.65 kg/m2.
Table 1:Subject demographics.
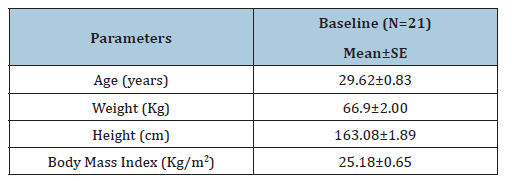
N-Number of subjects; SE-Standard Error.
Figure 1:Consolidated Standards of Reporting Trials (CONSORT) of BPE study.

Efficacy
Primary Efficacy Endpoint-FMD: Summary of mean FMD (%) change is presented in Figure 2a. BPE500 demonstrated a significant increase in mean FMD from baseline to Day 1 post-dose at 1 hour (BPE500=2.39±0.65 (36% increase), placebo=-0.08 ± 0.33, p<0.05) and post-dose at 2 hours (BPE500=2.88±1.05 (44% increase), placebo=0.01±0.34, p<0.05). This trend continued on Day 7, with measurements at 1 hour (BPE500=2.59±0.78 (40% increase), placebo=0.05±0.40, p<0.05) and 2 hours (BPE500=2.80±0.76 (43% increase), placebo=0.04±0.45, p<0.05), indicating a significant difference compared to the placebo. BPE1000 demonstrated a significant increase in mean FMD from baseline to 1-hour post-dose at Day 1 (BPE1000=2.58±0.62 (39% increase), placebo=-0.08±0.33, p<0.05) and 2 hours post-dose at Day 1 (BPE1000=2.84±0.54 (43% increase), placebo=0.01±0.34, p<0.05). This trend persisted at Day 7, with measurements at 1-hour post-dose (BPE1000=2.89±0.90 (44% increase), placebo=0.05±0.40, p<0.05) and 2 hours post-dose (BPE1000=3.14±0.98 (48% increase), placebo=0.04±0.45, p<0.05), demonstrating a significant difference compared to the placebo. Further, no significant difference was observed between BPE500 and BPE1000.
Figure 2:Graphical representation of
a) FMD mean change (%).
b) Brachial Artery Diameter (mm).
c) Blood Flow Velocity (cm/sec) and
d) Chalder Fatigue scores among BPE 500 mg (BPE500), BPE 1000mg (BPE1000), and placebo.

Secondary efficacy endpoint:
Brachial Artery Diameter: Summary of brachial artery diameter (mm) by visit and treatment for PP population is presented in Figure 2b. A significant difference in brachial artery diameter was noted between BPE500 and placebo at multiple time points: from pre-dose to Day 1 post-dose at 1 hour (BPE500=0.08±0.03, placebo=0.01±0.02, p<0.05), and post-dose at 2 hours (BPE500=0.09±0.04, placebo=-0.01±0.02, p<0.05). This trend persisted on Day 7, with measurements at 1-hour post-dose (BPE500=0.09±0.03, placebo=-0.02±0.07, p<0.05) and at 2 hours post-dose (BPE500=0.11±0.04, placebo=-0.01±0.05, p<0.05). However, this significant change was not attributed to BPE500 as it showed no significant difference at any of the post-dose timepoints compared to baseline. A significant difference in brachial artery diameter was noted between BPE1000 and placebo at multiple time points: from pre-dose to Day 1 post-dose at 1 hour (BPE1000=0.09±0.03, placebo=0.01±0.02, p<0.05) and post-dose at 2 hours (BPE1000=0.11±0.04, placebo=−0.01±0.02, p<0.05). This trend persisted on Day 7, with measurements at 1-hour postdose (BPE1000=0.11±0.4, placebo=−0.02±0.07, p<0.05) and at 2 hours post-dose (BPE1000=0.11±0.04, placebo=−0.01±0.05, p<0.05). However, this significant change was not assigned to BPE1000, as there was no significant difference at any of the postdose time periods compared to baseline. Further, no significant difference was observed between BPE500 and BPE1000.
Blood flow velocity: Summary of blood flow velocity (cm/sec) by visit and treatment for PP population is presented in Figure 2c. A significant difference in blood flow velocity was noted between BPE500 and placebo from pre-dose to Day 1 post-dose at 1 hour (BPE500=1.84±0.59, placebo=0.32±0.84, p<0.05) and at 2 hours post-dose (BPE500=2.65±0.59, placebo=0.62± 1.03, p<0.05). Additionally, significant differences were observed on Day 7 at 1-hour post-dose (BPE500=2.23±0.99, placebo=0.13±0.95, p<0.05) and at 2 hours post-dose (BPE500=2.88± 1.44, placebo=0.19±1.30, p<0.05). However, this significant change was not assigned to BPE1000, as there was no significant difference at any of the postdose time periods compared to baseline. BPE1000 demonstrated a significant increase in blood flow velocity from baseline to Day 7 post-dose at 2 hours (BPE1000=3.25±1.33, placebo=0.19±1.30, p<0.05) compared to the placebo. A significant difference in blood flow velocity was noted between BPE1000 and placebo from pre-dose to Day 1 post-dose at 1 hour (BPE1000=1.87±1.05, placebo=0.32±0.84, p<0.05) and post-dose at 2 hour (BPE1000=2.35±1.34, placebo=0.62±1.03, p<0.05). Additionally, significant differences were observed on Day 7 at 1-hour post-dose (BPE1000=2.69±1.22, placebo=0.13±0.95, p<0.05). However, this significant change was not attributed to BPE1000, as there was no significant difference at any of the post-dose time periods compared to baseline. Further, no significant difference was observed between BPE500 and BPE1000.
Fatigue: Summary of fatigue score by visit and treatment for PP population is presented in Figure 2d. BPE500 demonstrated a significant decrease in fatigue score from baseline to Day 7 (BPE500=−1.76±1.92, placebo=−0.19±0.87, p<0.05) as compared to placebo. Similarly, BPE1000 showed a significant decrease in fatigue score from baseline to Day 7 (BPE1000=−1.86±1.35, placebo=−0.19±0.87, p<0.05) in comparison to placebo. No significant difference was observed between BPE500 and BPE1000.
Safety
During the study, a total of 7 adverse events were reported by 4 subjects. One subject in each BPE500 and BPE1000 groups and 2 subjects in the placebo group experienced 2 (headache and fever), 1 (viral fever), and 4 (headache, fever and cold) adverse events, respectively. All the adverse events reported by the subjects were mild in severity and the causality of the adverse events was diagnosed by the investigator as not related to the investigational products. The outcomes of all the adverse events were noted as resolved before the end of the study. None of the subject reported any serious adverse events or was withdrawn from the study due to an adverse event. The liver function parameters, renal function parameters and hematological profile were within the normal range in the follow-up assessments. No clinically significant changes were observed in the vitals and physical examination during the study. The vital signs of the subjects were within the normal levels throughout the study. Overall, four subjects used at least one concomitant medication during the course of the study, of which one subject belonged to each BPE500 and BPE1000 groups and two subjects belonged to the placebo group. All four subjects used Acetaminophen and one subject from placebo group used Acetaminophen, Levocetirizine (5mg) combined with Montelukast (10mg).
Discussion
Beetroot and Pomegranate extract are known for enhancing exercise performance. NO3- and other metabolites in beetroot extract plays key role in physical endurance and exercise performance [22]. In addition to high nitrate concentration, high polyphenol contents in pomegranate juice or extract is believed to improve exercise performance [23]. This present study explored a proprietary ratio of Beetroot and Pomegranate homogenized extract mixture called as BPE (BPE) on FMD in healthy subjects. This was a prospective, double blind, randomized (1:1:1), placebo controlled, three-treatment, three-period, dose-ranging crossover study which explored safety and efficacy of the supplement. BPE contains homogenized extracts of Beetroot standardized for nitrates and Pomegranate standardized for ellagic acid in a proprietary ratio at the stage of extraction to get the final product BPE. The efficacy of BPE500 and BPE1000 on FMD was assessed using average FMD, brachial artery diameter, blood flow velocity and Chalder Fatigue Scale. FMD can describe any vasodilatation of an artery following an increase in luminal blood flow and internal-wall shear stress. It is widely believed to reflect endothelium-dependent and largely nitric oxide-mediated arterial function and has been used as a surrogate marker of vascular health [24,25].
Our results show that both the doses of BPE achieved significant improvements (p<0.05) in mean FMD change compared to placebo as early as 1-hour post dose (BPE500-increase by 36% on Day 1 and 40% on Day 7; BPE1000-increase by 39% on Day 1 and 44% on Day 7) and lasting over 2-hour post dose (BPE500 - increase by 44% on Day 1 and 43% on Day 7; BPE1000-increase by 43% on Day 1 and 48% on Day 7). Moreover, we have not observed any significant difference between BPE500 and BPE1000, suggesting optimal dose of BPE500. The significant change was observed as early as 1-hour post dose and observed lasting 2-hour post dose for both BPE500 and BPE1000 compared to placebo. Additionally, BPE1000 mg showed significant increase in blood flow velocity after 7 days of supplementation. The hydro-alcoholic extracts of Beetroot and Pomegranate Peel Extract (BPE) with relevance to the effects on flow-mediated dilation in the brachial artery and fatigue reduction may be associated with the activation of Sirtuin 1 [26]. The antiaging gene Sirtuin 1 is critical to vasodilation and mitochondrial function and biogenesis [27]. Sirtuin 1 activators present in BPE such as flavonoids (anthocyanins, catechins, quercetin, and rutin), and polyphenols are associated with the effects of BPE for energy and exercise performance [28].
Studies reveal the connection between nitrate supplementation and FMD which helps to understand improved endothelial mechanism and subsequent exercise tolerance. Inorganic nitrate present in beetroot is reduced to nitric oxide through various pathways and the nitric oxide generation [29]. Nitric oxide improves neurotransmission and blood flow, alterations in mitochondrial oxygen consumption, promotion of cognitive benefits, mood and cardiovascular function [29,30]. Beetroot juice showed acute improvement in exercise tolerance in adults and patients with peripheral arterial disease. Consumption of inorganic nitrate, either in the form of sodium nitrate or beetroot, has been shown to result in a significant increase in plasma nitrite levels and reduction of blood pressure in both younger (8 and 6mmHg; systolic and diastolic blood pressure; respectively) and older (5 and 3mmHg; systolic and diastolic blood pressure; respectively) adults. This positive effect on blood pressure has led to the suggestion that beetroot could potentially be used in medical settings as an alternative to traditional blood pressure lowering drugs [31-34]. Furthermore, increased nitric oxide bioavailability promotes vessel dilation, and increased blood flow [35]. Also, beetroot juice can improve cardiorespiratory endurance in athletes by increasing efficiency, which improves performance at various distances, increases time to exhaustion at submaximal intensities, and may improve the cardiorespiratory performance at anaerobic threshold intensities and VO2max [36].
Supplementary nitrates are reduced to nitrite through lingual anaerobic bacteria [29] and enters into systemic circulation where it further reduced to nitric oxide [37,38]. A study conducted on younger adults to examine the effects of acute supplementation of nitrate rich beetroot juice on cardiovascular responses, cognition, mood and perceptual responses revealed that beetroot significantly elevated plasma nitrite and nitrate concentration and reduced blood pressure; improved cognitive responses [39]. A randomized, double-blind placebo-controlled study was conducted to examine nitrate supplementation on microvascular and large-vessel endothelial function and blood pressure [40]. The study reported an increase in FMD by 1.5% following two weeks of beetroot consumption (p=0.04), whereas only a minimal (0.1%) change from the placebo group. In conclusion, this pilot study demonstrated that medium-term beetroot ingestion potentially improved blood pressure and large-vessel endothelial function in healthy older adults [40]. Another study evaluated the effect of beetroot juice consumption on macrovascular endothelial function (FMD) and muscle oxygen saturation parameters in pregnant women within a randomized, crossover, double-blind design in which 12 pregnant women consumed a single dose (140mL) of beetroot juice or placebo. The data demonstrate that a single dose of 140 mL of beetroot juice consumption improves macrovascular endothelial function, but not oxygen saturation parameters [41].
On the other hand, pomegranate is effective in physiological responses in individuals exhibiting physiological stress such as cardiovascular disease [42], oxidative stress [43], cellular inflammation [44], or joint or muscle damage [45]. Pomegranate enhance myocardial blood flow [46], antioxidant presence [42], reduces low-density lipoprotein cholesterol oxidation, blood pressure and carotid artery thickness [42]. Moreover, pomegranate attenuates oxidative stress by reducing free radical production, lipid peroxidation [47] and inhibits certain inflammation transcripts such as NF-κB, TNF-α and cyclo-oxygenase-2 (COX-2) [48,49]. A singlearm clinical study investigated thirteen hypertensive men aged 39-68 years on the acute effects of pomegranate juice consumption on blood pressure and markers of endothelial function showed. Comparison of pre- versus post-trial values revealed a significant reduction in both systolic blood pressure (7%) and diastolic blood pressure. However, changes in FMD (20%) as well as circulating levels of CRP, ICAM-1, VCAM-1, E-selectin, and IL-6 did not reach statistical significance. The study concluded that consumption of pomegranate juice could be considered in the context of both dietary and pharmacological interventions for hypertension [50]. Another randomized, double-blind, placebo-controlled, crossover study investigated the acute effects of pomegranate extract on blood flow, vessel diameter, and exercise performance including time to exhaustion in active individuals.
The study reported blood flow was significantly augmented with pomegranate extract thirty minutes post ingestion in comparison to placebo. Vessel diameter was significantly larger thirty minutes post exercise. Ingestion of the pomegranate extract was found to significantly augment time to exhaustion at 90% and 100% peak velocity. Acute ingestion of pomegranate thirty minutes prior to exercise may enhance vessel diameter, blood flow, and delay fatigue during exercise. Results of the current study indicated that pomegranate extract is ergogenic for intermittent running, eliciting beneficial effects on blood flow [21]. Our study results also showed significant decrease in fatigue score compared to placebo against both BPE500 and BPE1000. Overall, the analysis of our data revealed positive influence of BPE500 and BPE1000 among the subjects that could be helpful during exercise and may enhance vessel dilation, blood flow, and delay fatigue during exercise. Our safety analysis of BPE for both doses showed normal physical examination findings, vital signs and laboratory parameters throughout the study. None of the subject reported any SAE or was withdrawn from the study due to an AE or a SAE.
Conclusion
In the present study, the safety and efficacy of BPE500 and BPE1000 was studied in healthy subjects. BPE500 and BPE1000 showed significant efficacy in improving the flow mediated dilation, and fatigue without causing any adverse effects. The product was found to be safe and well tolerated. Further, long-term studies are required to establish the effects of BPE for energy and exercise performance.
Acknowledgment
We thank the participants of the study. All authors contributed to the study conception and design. Study product was supplied by Samriddh Nutractive Private Limited. Recruitment of volunteers, study procedure and overall study conduct was handled by Yateesh Jayashankara. Medical writing, editorial, and other assistance editorial assistance in the preparation of this article was provided by Leads Clinical Research and Bio Services Pvt. Ltd. All authors read and approved the final manuscript. The study was supported by Samriddh Nutractive Private Limited (Hyderabad, India).
Conflict of Interest
The authors declare no conflicts of interest.
References
- Mishchenko V, Suchanowski A (2010) Athletes’ endurance and fatigue characteristics related to adaptability of specific cardiorespiratory reactivity.
- Franklin BA, Eijsvogels TMH, Pandey A, Quindry J, Toth PP (2022) Physical activity, cardiorespiratory fitness, and cardiovascular health: A clinical practice statement of the ASPC Part I: Bioenergetics, contemporary physical activity recommendations, benefits, risks, extreme exercise regimens, potential maladaptations. American Journal of Preventive Cardiology 12: 100424.
- Opondo MA, Sarma S, Levine BD (2015) The cardiovascular physiology of sports and exercise. Clinics in Sports Medicine 34(3): 391-404.
- Mitchell JH, Saltin B (2003) The oxygen transport system and maximal oxygen uptake. Exercise physiology pp: 255-291.
- Jacob M, Chappell D, Becker BF (2016) Regulation of blood flow and volume exchange across the microcirculation. Critical Care 20(1): 319.
- Habler OP, Messmer KFW (1997) The physiology of oxygen transport. Transfusion Science 18(3): 425-435.
- Gao C, Gupta S, Adli T, Hou W, Coolsaet R, et al. (2021) The effects of dietary nitrate supplementation on endurance exercise performance and cardiorespiratory measures in healthy adults: A systematic review and meta-analysis. Journal of the International Society of Sports Nutrition 18(1): 55.
- Domínguez R, Cuenca E, Muñoz JLM, Fernández PG, Paya NS, et al. (2017) Effects of beetroot juice supplementation on cardiorespiratory endurance in athletes. A systematic review. Nutrients 9(1): 43.
- Pate R, Oria M, Pillsbury L (2012) Health-related fitness measures for youth: Cardiorespiratory endurance. Fitness Measures and Health Outcomes in Youth.
- Patel H, Alkhawam H, Madanieh R, Shah N, Kosmas CE, et al. (2017) Aerobic vs anaerobic exercise training effects on the cardiovascular system. World Journal of Cardiology 9(2): 134-138.
- Rodriguez NR, Marco NMD, Langley S (2009) American college of sports medicine position stand. Nutrition and athletic performance. Medicine and Science in Sports and Exercise 41(3): 709-731.
- Ormsbee M, Lox J, Arciero P (2013) Beetroot juice and exercise performance. Nutrition and Dietary Supplements 5: 27-35.
- Wylie LJ, Bailey SJ, Kelly J, Blackwell JR, Vanhatalo A, et al. (2016) Influence of beetroot juice supplementation on intermittent exercise performance. European Journal of Applied Physiology 116(2): 415-425.
- Kanner J, Harel S, Granit R (2001) Betalains a new class of dietary cationized antioxidants. Journal of Agricultural and Food Chemistry 49(11): 5178-5185.
- Raddino R, Caretta G, Teli M, Bonadei I, Robba D, et al. (2007) Nitric oxide and cardiovascular risk factors. Heart International 3(1): 18.
- Deanfield JE, Halcox JP, Rabelink TJ (2007) Endothelial function and dysfunction: Testing and clinical relevance. Circulation 115(10): 1285-1295.
- Thijssen DH, Bruno RM, Mil ACMV, Holder SM, Faita F, et al. (2019) Expert consensus and evidence-based recommendations for the assessment of flow-mediated dilation in humans. European Heart Journal 40(30): 2534-2547.
- Heiss C, Mateos AR, Bapir M, Skene SS, Sies H, et al. (2023) Flow-mediated dilation reference values for evaluation of endothelial function and cardiovascular health. Cardiovascular Research 119(1): 283-293.
- Stowe CB (2011) The effects of pomegranate juice consumption on blood pressure and cardiovascular health. Complementary Therapies in Clinical Practice 17(2): 113-115.
- Jurenka J (2008) Therapeutic applications of pomegranate (Punica granatum L.): A review. Alternative Medicine Review 13(2): 128-144.
- Trexler ET, Ryan AES, Melvin MN, Roelofs EJ, Wingfield HL (2014) Effects of pomegranate extract on blood flow and running time to exhaustion. Applied Physiology, Nutrition, and Metabolism 39(9): 1038-1042.
- Jędrejko M, Jędrejko K, Renaud VMG, Kała K, Muszyńska B (2024) Exploring the impact of alternative sources of dietary nitrate supplementation on exercise performance. International Journal of Molecular Sciences 25(7): 3650.
- Roelofs EJ, Ryan AES, Trexler ET, Hirsch KR, Mock MG (2017) Effects of pomegranate extract on blood flow and vessel diameter after high‐intensity exercise in young, healthy adults. European Journal of Sport Science 17(3): 317-325.
- Thijssen DHJ, Black MA, Pyke KE, Padilla J, Atkinson G, et al. (2011) Assessment of flow-mediated dilation in humans: A methodological and physiological guideline. American Journal of Physiology-Heart and Circulatory Physiology 300(1): H2-H12.
- Black MA, Cable NT, Thijssen DHJ, Green DJ (2008) Importance of measuring the time course of flow-mediated dilatation in humans. Hypertension 51(2): 203-210.
- Martins IJ (2016) Anti-aging genes improve appetite regulation and reverse cell senescence and apoptosis in global populations. Advances in Aging Research 5(1): 9-26.
- Martins IJ (2017) Single gene inactivation with implications to diabetes and multiple organ dysfunction syndrome. Journal of Clinical Epigenetics 3(3): 24.
- Martins IJ (2017) Nutrition therapy regulates caffeine metabolism with relevance to NAFLD and induction of type 3 diabetes. J Diabetes Metab Disord 4(19): 1-9.
- Kelly J, Fulford J, Vanhatalo A, Blackwell JR, French O, et al. (2013) Effects of short-term dietary nitrate supplementation on blood pressure, O2 uptake kinetics, and muscle and cognitive function in older adults. American Journal of Physiology-Regulatory Integrative and Comparative Physiology 304(2): R73-R83.
- Bentley DJ, Newell J, Bishop D (2007) Incremental exercise test design and analysis: Implications for performance diagnostics in endurance athletes. Sports Medicine 37(7): 575-586.
- Breese BC, McNarry MA, Marwood S, Blackwell JR, Bailey SJ, et al. (2013) Beetroot juice supplementation speeds O2 uptake kinetics and improves exercise tolerance during severe-intensity exercise initiated from an elevated metabolic rate. American Journal of Physiology-Regulatory Integrative and Comparative Physiology 305(12): R1441-R1450.
- Lansley KE, Winyard PG, Bailey SJ, Vanhatalo A, Wilkerson DP, et al. (2011) Acute dietary nitrate supplementation improves cycling time trial performance. Med Sci Sports Exerc 43(6): 1125-1131.
- Larsen FJ, Weitzberg E, Lundberg JO, Ekblom B (2007) Effects of dietary nitrate on oxygen cost during exercise. Acta Physiologica 191(1): 59-66.
- Kapil V, Khambata RS, Robertson A, Caulfield MJ, Ahluwalia A (2015) Dietary nitrate provides sustained blood pressure lowering in hypertensive patients: A randomized, phase 2, double-blind, placebo-controlled study. Hypertension 65(2): 320-327.
- Walker MA, Bailey TG, McIlvenna L, Allen JD, Green DJ, et al. (2019) Acute dietary nitrate supplementation improves flow mediated dilatation of the superficial femoral artery in healthy older males. Nutrients 11(5): 954.
- Arazi H, Eghbali E (2021) Possible effects of beetroot supplementation on physical performance through metabolic, neuroendocrine, and antioxidant mechanisms: A narrative review of the literature. Frontiers in Nutrition 8: 660150.
- Bailey SJ, Vanhatalo A, Winyard PG, Jones AM (2012) The nitrate‐nitrite‐nitric oxide pathway: Its role in human exercise physiology. European Journal of Sport Science 12(4): 309-320.
- Thompson KG, Turner L, Prichard J, Dodd F, Kennedy DO, et al. (2014) Influence of dietary nitrate supplementation on physiological and cognitive responses to incremental cycle exercise. Respiratory Physiology & Neurobiology 193: 11-20.
- Stanaway L, Markwick KR, Page R, Wong M, Jirangrat W, et al. (2019) Acute supplementation with nitrate-rich beetroot juice causes a greater increase in plasma nitrite and reduction in blood pressure of older compared to younger adults. Nutrients 11(7): 1683.
- Jones T, Dunn EL, Macdonald JH, Kubis HP, McMahon N, et al. (2019) The effects of beetroot juice on blood pressure, microvascular function and large-vessel endothelial function: a randomized, double-blind, placebo-controlled pilot study in healthy older adults. Nutrients 11(8): 1792.
- Souza MV, Oliveira GVD, Alvares TS (2018) A single dose of beetroot juice improves endothelial function but not tissue oxygenation in pregnant women: A randomised clinical trial. British Journal of Nutrition 120(9): 1006-1013.
- Aviram M, Rosenblat M, Gaitini D, Nitecki S, Hoffman A, et al. (2004) Pomegranate juice consumption for 3 years by patients with carotid artery stenosis reduces common carotid intima-media thickness, blood pressure and LDL oxidation. Clinical Nutrition 23(3): 423-433.
- Kaplan M, Hayek T, Raz A, Coleman R, Dornfeld L, et al. (2001) Pomegranate juice supplementation to atherosclerotic mice reduces macrophage lipid peroxidation, cellular cholesterol accumulation and development of atherosclerosis. The Journal of Nutrition 131(8): 2082-2089.
- Adams LS, Seeram NP, Aggarwal BB, Takada Y, Sand D, et al. (2006) Pomegranate juice, total pomegranate ellagitannins, and punicalagin suppress inflammatory cell signaling in colon cancer cells. Journal of Agricultural and Food Chemistry 54(3): 980-985.
- Shukla M, Gupta K, Rasheed Z, Khan KA, Haqqi TM (2008) Consumption of hydrolyzable tannins-rich pomegranate extract suppresses inflammation and joint damage in rheumatoid arthritis. Nutrition 24(7-8): 733-743.
- Sumner MD, Eller ME, Weidner G, Daubenmier JJ, Chew MH, et al. (2005) Effects of pomegranate juice consumption on myocardial perfusion in patients with coronary heart disease. The American Journal of Cardiology 96(6): 810-814.
- Kelawala NS, Ananthanarayan L (2004) Antioxidant activity of selected foodstuffs. International Journal of Food Sciences and Nutrition 55(6): 511-516.
- Afaq F, Malik A, Syed D, Maes D, Matsui MS, et al. (2005) Pomegranate fruit extract modulates UV‐B–mediated phosphorylation of mitogen‐activated protein kinases and activation of nuclear factor kappa b in normal human epidermal keratinocytes ¶. Photochemistry and Photobiology 81(1): 38-45.
- Huang THW, Yang Q, Harada M, Li GQ, Yamahara J, et al. (2005) Pomegranate flower extract diminishes cardiac fibrosis in Zucker diabetic fatty rats: Modulation of cardiac endothelin-1 and nuclear factor-kappaB pathways. Journal of Cardiovascular Pharmacology 46(6): 856-862.
- Asgary S, Keshvari M, Sahebkar A, Hashemi M, Kopaei MR (2013) Clinical investigation of the acute effects of pomegranate juice on blood pressure and endothelial function in hypertensive individuals. ARYA Atherosclerosis 9(6): 326-331.
© 2026 Jestin V Thomas. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






