- Submissions

Full Text
Research in Medical & Engineering Sciences
The Laminous Subsumption-Epidermal Inclusion Cyst
Anubha Bajaj*
Deputy General Manager, LT Foods Ltd., LT Group Company, India
*Corresponding author: Anubha Bajaj, Deputy General Manager, LT Foods Ltd, LT Group Company (Daawat Basmati Rice), India
Submission: April 25, 2022Published: June 01, 2022

ISSN: 2576-8816Volume9 Issue5
Introduction
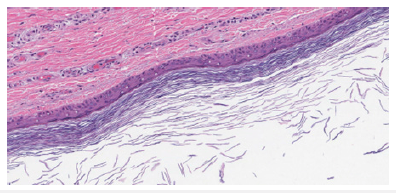
Preface Epidermal inclusion cyst is a frequently discerned, benign, noncontagious, cutaneous cyst incorporated with lamellar keratin. As the cyst wall is engendered from infundibular portion of hair follicle, the lesion may preponderantly be designated as an infundibular cyst. Sebaceous glands do not engender epidermal inclusion cyst and sebum does not configure as an intra-cavitary component. Thus, the terminology of “sebaceous cyst” is unwarranted and requires circumvention. Additionally designated as epidermoid cyst, epidermal cyst, follicular cyst- infundibular type, inclusion cyst or keratin cyst, the stable epidermal inclusion cyst may progress gradually. Normal cutaneous bacterial flora as Staphylococcus aureus or Staphylococcus epidermidis may infect an epidermal inclusion cyst. Disease Characteristics Generally, epidermal inclusion cyst occurs due to damage to pilo-sebaceous unit and is discerned within zones of inflamed or persistently irritated hair follicles. Lesions may concur with acne vulgaris. Neonatal epidermal inclusion cysts are designated as milia [1,2]. Epidermal inclusion cyst is frequently discerned upon the face, scalp, neck, trunk, dorsal region, or scrotum. No site of cyst emergence is exempt. Exceptionally, coalescing epidermal inclusion cysts may be exemplified upon trunk or extremities (Figure 1).
Figure 1Epidermal inclusion cyst depicting a layer of stratified squamous epithelium with a prominent granular cell layer and laminated keratin within the cavity.
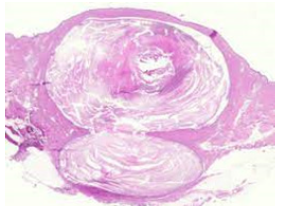
Majority of epidermal inclusion cysts emerge as a sporadic lesion. Although delineating a non genetic linkage, epidermal inclusion cyst may constitute a component of exceptional, hereditary syndromes such as Gardner’s syndrome, Favre-Racouchot syndrome with nodular elastosis, cysts and comedones along with Gorlin’s syndrome or basal cell nevus syndrome (Figure 2). Epidermal inclusion cysts depicted in Gardner’s syndrome are predominantly confined to posterior surface of pinna wherein lesions precede pubertal onset or commencement of colonic polyposis. A male predominance is observed with a male to female proportion of 2:1. Epidermal inclusion cyst is commonly discerned within second to fourth decade. Elderly subjects exhibiting chronic, actinic-induced cutaneous injury or infection with Human Papilloma Virus (HPV) demonstrate emergence of epidermal inclusion cyst (Figure 3). Epidermal inclusion cyst is categorized into primary cyst which arises directly from infundibulum of hair follicle. Impacted hair follicular orifice may configure the cyst. Frequently, epidermal inclusion cyst communicates with superimposed cutaneous surface although a miniature orifice or centric punctum may be discernible.
Figure 2Epidermal inclusion cyst delineating a stratified squamous epithelial lining and innumerable keratin flakes confined to the cyst cavity.

Figure 3Epidermal inclusion cyst demonstrating an encysted cavity coated with stratified squamous epithelium and lamellar keratin within the cavity.
Individuals with acne vulgaris delineate disrupted hair follicles and impaction of cutaneous pores with consequently enhanced proportion of epidermal inclusion cysts. Besides, the lesion may especially be associated with pre-existing comedones. •secondary epidermal inclusion cyst occurs subsequent to implantation of dermal follicular epithelium following trauma or in concurrence with comedones. Individuals subjected to cyclosporine therapy or treated with BRAF inhibitors such as imiquimod may display the occurrence of epidermal inclusion cyst. Epidermal inclusion cyst can resolve spontaneously. Besides, cyst reoccurrence may ensue in the absence of cogent therapy (Figure 4 & 5). Clinical Elucidation Epidermal inclusion cyst manifests as a discrete, consistent, or gradually progressive, mobile, cystic lesion commonly displaying a discernible, centric punctum. Typically, the comprehensively appearing cyst manifests as a subcutaneous nodule of magnitude extending to several centimeters. Epidermal inclusion cyst may enlarge, remain quiescent or undergo secondary inflammation.Fluctuant, infected epidermal inclusion cyst is apparently enlarged, distinctive and erythematous. The inflamed, painful epidermal inclusion cyst represents as a fluctuant subcutaneous nodule. Epidermal inclusion cyst is imbued with lamellar keratin with a “cheesy” appearance (Figure 6). As sebaceous glands do not engender epidermal inclusion cyst, the cyst cavity is devoid of sebum [3-5].
Figure 4Epidermal inclusion cyst exhibiting a layer of stratified squamous epithelium with a prominent granular cell layer and flaky keratin imbued within the cyst cavity.
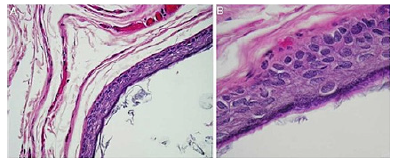
Figure 5Epidermal inclusion cyst exemplifying an encysted cavity coated with stratified squamous epithelium, encompassing fibro-connective tissue and lamellar intra-cavitary keratin.
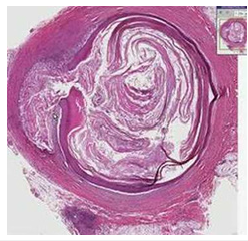
Figure 6Epidermal inclusion cyst enunciating an encysted lesion layered with attenuated stratified squamous epithelium and flaky keratin within the cavity.
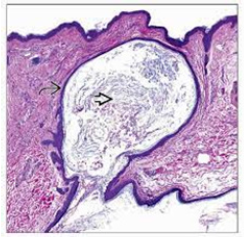
Generally asymptomatic, an inflamed, ruptured epidermal inclusion cyst demonstrates extrusion of cystic contents into encompassing cutaneous or subcutaneous tissue. Concurrent foci of active infection may or may not be discerned. Histological Elucidation Epidermal inclusion cyst can be confirmed with cogent tissue sampling. Upon gross examination, a subcutaneous yellowish nodule may be observed. Epidermal inclusion cyst is a unilocular cyst layered with stratified squamous epithelium delineating a prominent granular cell layer (Figure 7). The cystic cavity is imbued with acellular, lamellar keratin exhibiting a “basket weave” pattern. Epidermal inclusion cyst is accompanied by implantation of epidermal constituents within the dermis. The stratified squamous epithelium layer frequently exhibits a granular cell layer with numerous keratohyaline granules. Cyst rupture may induce granulomatous inflammation with the configuration of a keratin granuloma (Figure 8). Infected epidermal inclusion cyst may exemplify features such as discontinuity of cyst wall, an acute inflammatory cell infiltrate, invasion with neutrophils or an intense foreign body giant cell reaction. An estimated below< 1% of epidermal inclusion cysts are associated with malignant metamorphosis into basal cell carcinoma or squamous cell carcinoma [5-9].
Figure 7Epidermal inclusion cyst delineating a coating of stratified squamous epithelium, predominant granular cell layer and intra-cystic, lamellar keratin.
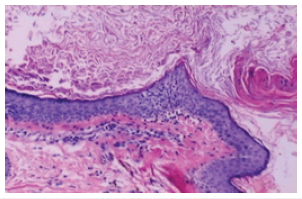
Figure 8Epidermal inclusion cyst demonstrating an encysted cavity coated with stratified squamous epithelium and lamellar keratin confined to the cyst cavity.
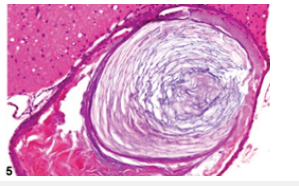
References
- Weir CB, St Hilaire NJ (2021) Epidermal inclusion cyst. StatPearls international, Treasure Island, Florida, USA.
- Zito PM, Scharf R (2021) Epidermoid cyst. StatPearls International, Treasure Island, Florida, USA.
- Sutedja EK, Tsaqilah L (2020) An unusual and rare case of generalized multiple epidermoid cysts with a giant epidermoid cyst. Int Med Case Rep J 29(13): 557-562.
- Tchernev G, Temelkova I (2019) Multiple epidermal cysts of the scalp: Dermatosurgical approach with favourable outcome. Open Access Maced J Med Sci 7(9):1509-1511.
- Paparella T, Fallat L (2018) A rare presentation of a giant epidermoid inclusion cyst mimicking malignancy. J Foot Ankle Surg 57(2): 421-426.
- Hoang VT, Trinh CT (2019) Overview of epidermoid cyst. Eur J Radiol 6: 291-301.
- Apollos JR, Ekatah GE (2017) Routine histological examination of epidermoid cysts; to send or not to send. Ann Med Surg (Lond) 13: 24-28.
- Boussemart L, Routier E (2013) Prospective study of cutaneous side-effects associated with the BRAF inhibitor vemurafenib: A study of 42 patients. Ann Oncol 24(6): 1691-1697.
- Pérez GJ, Scilletta A (2012) Giant earlobe epidermoid cyst. J Cutan Aesthet Surg 5(1): 38-39.
© 2022 Anubha Bajaj. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






