- Submissions

Full Text
Techniques in Neurosurgery & Neurology
Deep Brain Stimulation in Parkinson’s Disease
Amit Kumar Ghosh1* and Sutirtha Hazra2
1Consultant Neurosurgeon, Institute of Neurosciences, India
2Registrar of Neurosurgery, Institute of Neurosciences, India Introduction
*Corresponding author: Amit Kumar Ghosh, Consultant Neurosurgeon, Institute of Neurosciences, Kolkata, India
Submission: July 23, 2019;Published: August 30, 2019

ISSN 2637-7748
Volume2 Issue5
Abstract
Deep brain stimulation (DBS) is an universally accepted therapy for medically refractory Parkinson’s disease (PD). Both globus pallidus internus (GPi) and subthalamic nucleus (STN) stimulation are effective in improving the symptoms of PD and reducing dyskinesias. STN DBS is the most commonly performed operation for PD as compared to GPi DBS. Ventral intermediate nucleus (Vim) DBS is used occasionally in selected cases as an alternative for tremor predominant PD patients.
Patient selection is very much essential in achieving good outcomes. Good response to levodopa challenge is most important predictor of favourable long-term outcomes. The DBS surgery is typically performed in an awake state and involves stereotactic frame application, CT/MRI imaging, anatomical targeting, electrophysiological confirmation and intra-operative clinical monitoring for efficacy and adverse effects after intra-operative stimulation and implantation of the DBS lead and pulse generator.
Anatomical targeting consists of direct visualization of the target and anterior and posterior commissure in MR images, reformatting with stereotactic atlases, CT-MRI fusion, getting the entry point and trajectory from the computer-based software and accordingly getting the frame co-ordinates.
Physiological verification is achieved by microelectrode recording followed by implantation of the DBS lead and intraoperative test stimulation to assess benefits and side effects. The various aspects of DBS surgery will be discussed.
Keywords: Deep brain stimulation (DBS); Parkinson’s disease(PD); Stereotaxy
Introduction
Parkinson’s disease is a neurodegenerative disease clinically manifested by tremor, rigidity, bradykinesia and postural instability. It is slowly progressive disease. It is the most common movement disorder in middle or later part of life with a prevalence of about 0.3% of the general population. Pathological findings in Parkinson’s disease are greatly diminished neuromelanin pigmented neurons in the substantia nigra with associated gliosis, and Lewy bodies present in many neurons. James Parkinson, in his original Essay (On 1817) about ‘The Shaking Palsy’, gave description of six patients in which he mentioned presence of tremor, festinating gait and flexed posture.
Nearly two centuries from Parkinson’s observations, and almost four decades after Cotzias’ dramatic demonstration of levodopa’s efficacy, the limitations and complications of levodopa treatment for Parkinson’s disease have become well documented.
Five years after initiation of therapy, a majority of patients develop medication related motor complications, namely levodopa induced dyskinesias (LID) and motor fluctuations. Deep brain stimulation (DBS) has been developed primarily to address these treatment related motor complications. Different motor and non-motor cortical areas project primarily to the striatum which has two major projections: the direct pathway to the globus pallidus pars interna (GPi) and the indirect projection to the GPi via the globus pallidus pars externa and the subthalamic nucleus (STN).
The GPi serves as the major output nucleus, which connects back to the cortex via the thalamus. Modulated by the substantia nigra pars compacta, the indirect pathway exerts surround inhibition and thus facilitates an excitatory drive to muscles responsible for a given movement and suppresses unwanted motor activity not relevant to the primary movement.
Thus, PD is thought to result from over-activation of the indirect pathway leading to an increased output from the GPi and a decrease in spontaneous movement. This model of the basal ganglia and its connections is, of course, an oversimplification of a complex network that, when disrupted, can result in a range of motor abnormalities. For example, a hyper-direct pathway, projecting directly from the cortex to the STN, and from there to the GPi has recently been added to this model [1-6].
Pathophysiology of PD<
The loss of dopaminergic neurons in the substantia nigra affects: the circuit described above and leads to the cardinal motor symptoms of PD. While the exact mechanism of this process is unknown, animal research as well as human recordings have provided functional and biochemical evidence that bradykinesia in PD results from excessive activity in the STN and the GPi. This leads to an exaggerated beta (10-30 Hz) synchronization within and between structures in the basal ganglia circuitry that could also contribute to rigidity and akinesia. The pathophysiology of rest tremor in PD is less clear and probably more complicated. This symptom most likely results from a dysfunction of both the striatopallidal- thalamocortical and the cerebellodentato-thalamocortical circuits, with hyperactivity and hypersynchronization between central oscillators.
Possible mechanism of action of DBS
DBS acts through delivering an electrical current in a specific target area of the brain. This current can be modulated through modification of voltage, frequency and duration of each electrical pulse delivered. The delivered energy creates an electrical field of variable size and shape according to the parameters used for stimulation. Although initially believed to stimulate the target, thus the name of the whole process, it seems that DBS actually excites the neuronal fibers, but inhibits the neural cells. In fact, GPi DBS decreases the GPi mean firing rate back to a normal range in animal models as well as PD patients, and high frequency DBS has a similar effect as dopamine replacement therapies, and promotes faster (about 70 Hz) nonhypersynchronous activity in the basal ganglia, correlated with clinical improvement. This might be achieved through stimulation of bypassing inhibitory pathways, synaptic inhibition, depolarizing blockade, synaptic depression, and simulation-induced disruption of pathological network activity. Overall, this leads to modifications of the firing rate and pattern of neurons in the basal ganglia, as well as local release of neurotransmitters such as glutamate and adenosine. In addition, it seems that DBS also increases blood flow and stimulates neurogenesis. Over the last few years, functional imaging, specifically functional magnetic resonance imaging (fMRI), positron emission tomography (PET) and single-photon emission computed tomography (SPECT), has been used in an attempt to clarify the mechanism of action of DBS. In fMRI, blood-oxygenlevel- dependent (BOLD) signals are acquired, and oxygenated blood marks areas of neural stimulation or inhibition. On the other hand, PET and SPECT allow for imaging of multiple activity markers, such as blood flow, glucose and oxygen metabolism. While fMRI is less powerful than nuclear medicine techniques, it provides a much better spatial and temporal resolution. Because of the suspected inhibitory DBS effects in electrophysiological studies, reduced STN blood flow or glucose metabolism would have been expected on functional imaging. However, the opposite has been found to be true in an overwhelming majority of imaging studies to date. In addition, BOLD activation in the area surrounding the electrode has been reported, despite the electrode imaging artifact preventing direct observation of the STN around the electrode. This discrepancy between apparent STN inhibition in single-cell studies and activation in imaging studies might be explained by a few hypotheses. First, electrophysiological recordings identify short neuronal modulation (in the order of milliseconds) while neuroimaging methods may reflect the summed activity changes over seconds to minutes. Second, non-neuronal contributions to the change in blood flow and/or glucose metabolism cannot be excluded, and could confound the results of neuroimaging. Finally, it is possible that PET and fMRI actually detect the increased activity in the axons, rather than in the cell bodies. Complicating matters further, some imaging studies after STN DBS have showed increased activity in the GPi while others reported decreased activity in that nucleus.
In summary, it is still unclear how exactly DBS affects the firing rate and pattern of neurons and how these
changes actually modify the symptoms of Parkinson’s disease. DBS is presently more of an empirically proven
treatment in search of physiological explanation.
Patient selection for DBS in PD
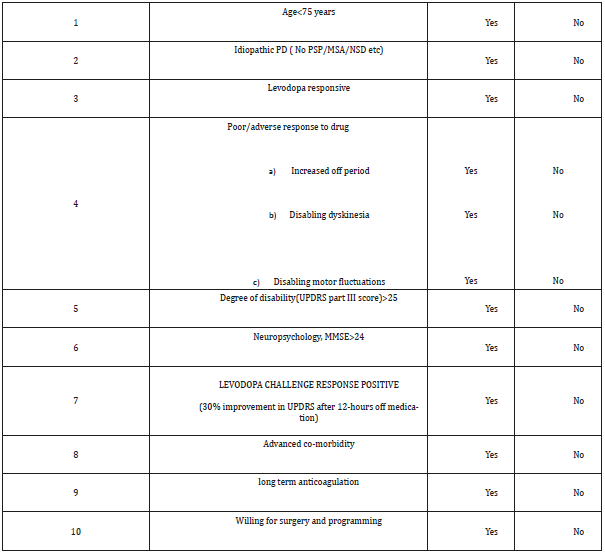
Patient selection is a critical first step as poorly chosen candidates may not have optimal benefits and have increased morbidity. Several factors must be considered before determining if a patient is an appropriate candidate for DBS surgery. A multidisciplinary approach involving the neurosurgeon, neurologist, and neuropsychologist is important to determine the appropriate surgical candidate. It is also important that the diagnosis of idiopathic PD be confirmed prior to proceeding with DBS surgery. Key to this assessment is evaluating the surgical candidate in both the on and off medication states with a corroborating levodopa challenge. Perhaps the best prognostic indicator of a patient’s suitability for DBS surgery is their response to levodopa. In general, a levodopa challenge following a 12-hour medication withdrawal should provide at least a 33% improvement in the motor section of the Unified Parkinson’s Disease Rating Scale (UPDRS).
Patient selection for DBS in PD
Patient selection is a critical first step as poorly chosen candidates may not have optimal benefits and have increased morbidity. Several factors must be considered before determining if a patient is an appropriate candidate for DBS surgery. A multidisciplinary approach involving the neurosurgeon, neurologist, and neuropsychologist is important to determine the appropriate surgical candidate. It is also important that the diagnosis of idiopathic PD be confirmed prior to proceeding with DBS surgery. Key to this assessment is evaluating the surgical candidate in both the on and off medication states with a corroborating levodopa challenge. Perhaps the best prognostic indicator of a patient’s suitability for DBS surgery is their response to levodopa.In general, a levodopa challenge following a 12-hour medication withdrawal should provide at least a 33% improvement in the motor section of the Unified Parkinson’s Disease Rating Scale (UPDRS).
Surgical technique
The basic components of DBS implantation surgery involve frame placement, anatomical targeting, physiological mapping, evaluation of macrostimulation thresholds for improvement in motor symptoms or induction of side effects, implantation of the DBS electrode and implantable pulse generator (IPG).
Head-frame placement
The CRW frame is the most commonly used followed by the Leksell frame. Placement of the frame is done under local anesthesia unless anxiety or uncontrollable movements necessitate the use of sedation or general anesthesia (Figure 1-5).
figure 1:Brief overview of the motor circuitry of the basal ganglia
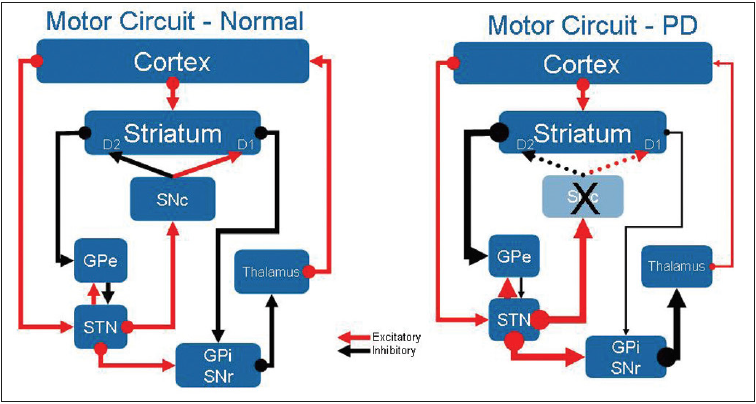
A: cortico-striato-pallido-thalamo-cortical (CSPTC) neural circuitry in normal state.
B: CSPTC in Parkinson’s disease (PD). SNc, substantia nigra pars compacta; GPe: Globus Pallidus Externus;
STN: Subthalamic Nucleus; GPi: Globus Pallidus Internus; SNr: Substantia Nigra reticulata
figure 2:Leksell stereotactic frame placed over the head of a patient showing the correct method for placement of the Leksell head-frame. The frame should be placed parallel to orbito-meatal line in order to approximate the AC-PC plane. It is attached to the patient’s head using four pins under local anesthesia.
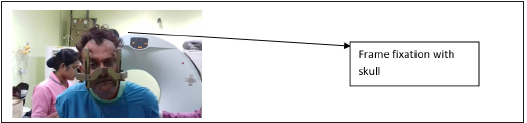
figure 3:The AC and the PC were marked and the centre of the AC-PC line determined.
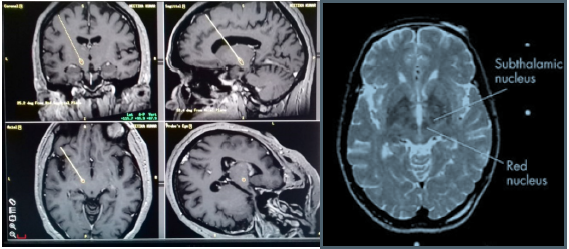
figure 4:
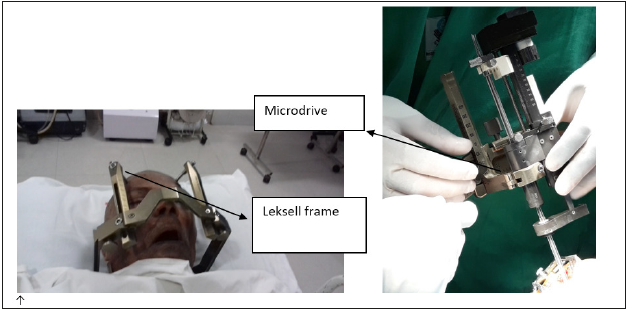
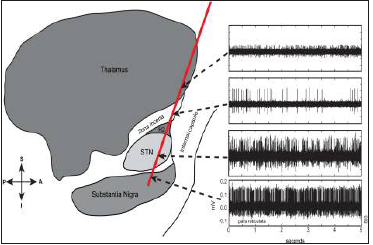
figure 5:Micro-electrode recording showing STN activity.
Imaging and anatomic targeting
Computerized Tomography (CT) scans and MRI are the two main imaging modalities used for targeting when performing DBS implantations. A thin cut stereotactic CT (2 mm slices with no gap and no gantry tilt) is obtained after frame placement and is then fused with the stereotactic MRI on a planning station (Stealth station). The advantage of fusing the CT with MRI is the ability to avoid image-distortions inherent to MR imaging adding to the stereotactic accuracy. To better define the STN, T2-weighted images (TR 2800, TE 90, flip angle 90˚, slice thickness 2.0 mm) were obtained.
The next step is planning the entry point and trajectory. The strategy here is to avoid surface and sub-cortical vessels. After trajectory planning, the patient is placed supine on the operating table and the frame attached to the table using an adaptor. Prophylactic antibiotics are given at least 30 min prior to incision. The head is prepped and draped in a sterile fashion. Under local anesthesia, a burr-hole is placed on the calculated entry point marked on the skull. The entry point is determined by the calculated arc and ring angles. Hemostasis is achieved with bone wax and bipolar cautery.
A Medronic Stim-Loc anchoring device (Medtronic, Minneapolis, MN) burr-hole base ring is then placed on the burrhole and secured with two screws which are used at the end of the procedure to anchor the DBS electrode.
The dura is then cauterized and opened exposing the underlying surface of the brain. The microdrive is then assembled and cannulae inserted 10mm above the target to avoid lenticulostriate vessels found deeper. Gel- foam and fibrin glue is applied on dural hole to minimize cerebrospinal fluid (CSF) loss and air entry into the skull. Subsequently, microelectrode recording and stimulation is undertaken (Table 1 &2).
Table 1:
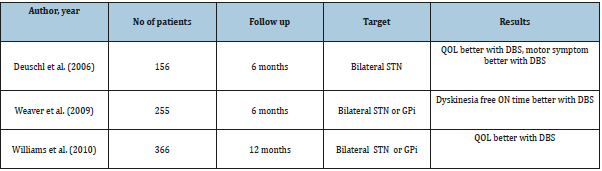
Table 2:In our institute, we follow a simple chart(below) for screening of patients for DBS in PD.
Microelectrode recording/Mapping
Microelectrode mapping is used to precisely define the target STN and its boundaries as well as nearby critical structures. We believe microelectrode mapping is crucial in order to give one the best chance for optimal placement of the DBS lead given anatomical inaccuracies due to image distortion and intraoperative brain shifts secondary to CSF loss, and pneumocephalus that can lead to inaccuracies in defining the initial target coordinates and shifts in the target itself once the skull is opened. Microelectrode mapping is performed using platinum-iridium glass coated microelectrodes dipped in platinum black with an impedance of around 0.3–0.5 Mo. These platinum-iridium microelectrodes are capable of recording single unit activity and can also be used for micro-stimulation up to 100 mAwithout significant breakdown in their recording qualities.
As the recording electrode was advanced, entry into the STN was identified by a sudden increase in the density of cellular discharge, with the characteristic irregular pattern of discharge-spikes of different sizes, occurring at random intervals. On coming out of the STN a quiet period (background noise) was seen followed by recording from the substantia nigra if the recording was continued far enough, described as high frequency (50–60 spikes/s) discharge pattern. 11 Characteristic STN recordings (visual and audio) were identified and the depth of the STN activity was noted. Identification of STN activity was only based on the visual identification. The centre of the point of best electrical activity was selected as the final target. The microelectrode was replaced with a permanent quadripolar macroelectrode (Medtronic electrode no. 3389) to target the centre of the STN electrical activity. The proximal part of this electrode consists of four nickel conductor wires insulated with a polytetrafluoroethylene jacket tubing. The distal part has four metallic noninsulated contacts of 1.5mm spaced at 0.5mm intervals. The diameter of the distal electrode is 1.27mm. Based on the clinical response any of the four contacts can be used for stimulation. Macrostimulation using the DBS electrode itself is then used to determine benefits and side effects. In most cases lateral skull x rays were obtained at this point with image intensifier carefully positioned to locate the target point in the centre of the Leksell-G frame rings.
Battery placement
Finally, under general anaesthesia, the wires were tunnelled and the stimulator (Kinetra; Medtronic Ltd) placed subcutaneously in the chest wall.
Programming parameters STN
DBS for PD: Initial programming is always refined by using intra-operative macrostimulation data and a mono-polar review to identify the thresholds of stimulation for improvement in parkinsonian motor signs as well as the thresholds for inducing side effects at the level of each contact. The four variables that are used in programming are choice of contacts (0, 1, 2 or 3 used either as the cathode or anode), frequency of stimulation (hertz), pulsewidth (ms) and amplitude (voltage).
Complication of DBS that may happen
Intraoperative-(%)
asymptomatic IVH (3.4)
symptomatic ICH (1.1)
asymptomatic ICH (0.5)
acute perilesional edema (0.3)
cortical/subcortical ischemic infarction (0.4)
vasovagal response (0.8)
hypotension (0.3)
confusion (0.4)
anxiety (0.7)
seizure (0.3)
arrhythmia (0.1)
aborted procedure (1.0)
Perioperative (2 weeks)
headache (4.2)
hemiparesis w/ or w/o decreased LOC (1.7)
confusion (1.0)
confusion/agitation (0.5)
respiratory distress (0.4)
seizure (0.4)
hallucinations (0.4)
somnolence (0.1)
fall (0.1)
long-term postop (>2 wks)
wound complications
wound infection–self limited (1.4)
wound infections
requiring system removal (1.0)
requiring lead removal only (0.1)
requiring IPG/extension removal only (0.4)
requiring debridement only (0.3)
skin erosion–device removal (0.3)
wound dehiscence–debridement only (0.3)
Hardware complications
lead fracture–lead revision (1.0)
lead malposition–lead revision (1.2)
lead migration–lead redirection w/o removal (0.5)
lead malfunction/high impedance–lead revision (0.3)
flipped IPG–revision (0.5)
malpositioned/uncomfortable IPG–revision (0.5)
IPG malfunction/high impedance–replace (0.1)
lead extension malfunction/high impedance–replace (0.1)
lead extension fracture–replace (0.4)
satisfaction-related complications
loss of system efficacy over time–lead revision (2.6)
decreased efficacy over time–no revision desired (1.4)
Postoperative management
In the immediate hours after surgery, it is important to keep arterial blood pressure in the normal range. In addition, the patient’s preoperative drug regimen should be restarted immediately after surgery to avoid problems with dopaminergic withdrawal. Patients should undergo postoperative CT scans and/or MRI scans to assess the electrode location and intracranial status. In addition, plain X-rays are obtained to assess the location and geometry of the leads and hardware. Parkinson’s medications may need to be adjusted depending on the patient’s status. Cognitive and behavioral changes may occur in the postoperative period, particularly in older patients. Patients can be discharged as early as 24 hours after surgery, depending on their neurological and cognitive status.
Conclusion
For the last 50 years, levodopa has been the cornerstone of PD management. However, a majority of patients develop motor fluctuations and/or LID about 5 years after the initiation of therapy. DBS of the STN or the GPi grant to patients with PD improved quality of life and decreased motor complications, and has been approved as such by the Food and Drug Administration in the US in 2002. We reviewed the experience and available literature on DBS for Parkinson’s disease over the last decade and arrive at the following understandings.
The success of DBS surgery depends on the accurate placement of the leads and meticulous programming of the stimulation. Therefore, it is best accomplished by an experienced team of neurosurgeon, neurologist, and support staff dedicated to the treatment. Reports of surgical complication rates and long-term side-effects of DBS are very variable, so benefits and potential adverse results should not be under- or over-emphasized.
While essentially equal in improving the motor symptoms of PD, STN and GPi might have their own benefits and risks, and the choice of the target should be individualized and adapted to the patient’s situation.
Knowledge to further improve DBS treatment for Parkinson’s disease, such as a more scientific and reliable protocol on programming, strategies to minimize cognitive and psychiatric complications, and the better long-term maintenance of the implanted device, are still lacking.
Data on the impact of DBS on non-motor symptoms affecting the quality of life of PD patients, such as pain, speech or gastrointestinal complaints, are still scarce. Further research in these areas will help make this useful treatment even more beneficial.
References
- Mehanna R, Lai (2013) Deep brain stimulation in Parkinson’s disease. Translational Neurodegeneration 2.
- Patel NK, P Plaha, Sullivan KO (2003) MRI directed bilateral stimulation of the subthalamic nucleus in patients with Parkinson's disease. J Neurol Neurosurg Psychiatry 74: 1631-1637.
- N A Hamid, R D Mitchell, P Mocroft (2005) Targeting the subthalamic nucleus for deep brain stimulation: technical approach and fusion of pre- and postoperative MR images to define accuracy of lead placement. J Neurol Neurosurg Psychiatry 76(3): 409-414.
- Deogaonkar M, Vitek JL (2008) Globus pallidus stimulation for parkinson’s disease. Textbook of stereotactic and functional neurosurgery, pp. 1577-1602.
- Andre M, Ali RR, Brian HK, Robert EG, Ashwini D, et al (2006) Deep brain stimulation for parkinson’s disease: surgical technique and perioperative management. Movement disorders 21(14): S247-S258.
- Albert JF, Simpson RK (2014) Risks of common complications in deep brain stimulation surgery: management and avoidance. J Neurosurg 120(1): 132-139.
© 2019 Behzad Saberi. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






