- Submissions

Full Text
Research in Medical & Engineering Sciences
Effect of Oral Sub-Lethal Dose of Abamectin in the Rat: Serum ALT, AST, Urea, Creatinine and Histopathological Changes
Mohey Mekawy1, Ashur H Khali2 and Hussein A Kaoud3*
1Department of Forensic Medicine, Cairo University, Egypt
2Agency of Forensic Medicine, Egypt
3Department of Veterinary Hygiene and Environmental Pollution, Cairo University, Egypt
*Corresponding author: Hussein A Kaoud, Department of Veterinary Hygiene and Environmental Pollution, Faculty of Veterinary Medicine, Cairo University, Egypt
Submission: January 08, 2018; Published: January 30, 2018

ISSN: 2576-8816Volume3 Issue3
Abstract
In this study, the effect of ABA on serum ALT, AST, urea and creatinine were studied as well as histological changes in the liver and kidneys. Group 1 animals were given with abamectin at a dose of 30mg/kg B. Wt. (1/10 LD50), double oral doses \week for 15 days and one month. Group 2 animals were given with abamectin at a dose of 30mg/kg B. Wt. (1/10 LD50), double oral doses \week for 15 days and one month.
The results of the current study showed that in the administration of abamectin in 1/10 LD50, for 15 days and one-month (group 1 and 2) significantly increased plasma levels of ALT, AST, urea and creatinine in male rats treated, compared with control group. Changes in ALT and AST levels vary depending on exposure time, where an increase in enzyme activity was observed in animals of group 2 compared with that group 1.
The results also showed that the abamectin tended to cause a significant change in the liver and kidney rat. The permeability of the leukocyte pockets, congested blood vessels in the portal tract, destruction of some liver cells and vacuolation of liver cells. Significant necrosis of tubular cells, glomerular atrophy, and interstitial infiltration areas of round cells were found.
Keywords: Abamectin; Oral sub-lethal dose; ALT; AST; Urea; Creatinine; Histopathological changes
Introduction
Abamectin (ABA) is a powerful endo - and parasite agent with a wide range of activity in many animal species. It is a fermentation product produced by actinomycete Streptomycetesavermitilis [1]. Abamectin (ABA) is composed of about 80% of avermectin B1a and ≤ 20% avermectin B1b. ABA's mechanism of action is concerned with the gamma-aminobutyric acid (GABA) system and Cl channels. GABA receptors are performed for regulating the neural basal tone of the brain [2] and are in all nerve cells in the central nervous system (NES). Symptoms of toxicity of Abamectin in experimental animals are; pupil spasms, vomiting, convulsions and/or tremors and coma.
Abamectin, is an analog of Ivermectin, is made up of a mixture of avermectins. It is a macrocyclic lactone disaccharide, a member of the avermectins group which is extensively used as an anti- parasitic drug in agricultural and domestic animals. Abamectin has been extensively used to control insects for a wide range of agricultural products such as fruits, vegetables and ornamental crops [3]. They are also developed in commercial baits to control ants and cockroaches [4]. Abamectin acts as an insecticide by interfering with the nervous system of insects causing paralysis. It activates its function by stimulating the fused glutamate chloride channel in the invertebrate nervous system [5].
The Avermectin deactivate electrical activity in nerve and muscle preparations by increasing membrane behavior of conductance to chloride ions [6]. The action of abamectin involves G-aminobutyric receptors (GABA) in the peripheral nervous system. It appears that the channel glutamate gate (GWC), together with G-aminobutyric acid (GABA) spin-channel chloride (GAB) and the his-tamine-gated chloride channel (HisCl), is the target site of avermectin and ivermectin in insects and nematodes [7]. The aim of this research was to study the effect of ABA on serum ALT, AST, urea, and creatinine as well as histological changes in the liver and kidneys in rats.
Materials and Methods
Adult male albino rats (Rattus norvegicus), with body weights of 125-130g. The animals were housed in small groups (6 each), inside propylene cages. The temperature in the ex-perimental animal room was maintained at 24.5±1.5 °C with 12h dark: light cycle, and 72% humidity. The animals were provided with commercial pelleted rodent food and drinking water ad libitum. The animals could acclimatize to the laboratory conditions for one week prior to the start of the study. The experimental animals were divided into two groups of six adult males each. Animals of group-1 were administered with abamectin at a dose of 30mg/kg B. Wt. (1/10 LD50), two times a week, for a period of 15 days. Animals of group-2 were administered with abamectin at a dose of 30mg/kg B. Wt. (1/10, LD50), two times a week, for one month.
Animals
Adult male albino rats (Rattus norvegicus) of body weights of 125-130g, were used for the study. The animals were housed in groups (6 each), inside the stander cages. The temperature in the laboratory chamber was maintained at 24.5±1.5 °C with 12 hours dark: light cycle, and 75% humidity. The animals were supplied with commercial rodent food and drinking water ad libitum. Animals could adapt to laboratory conditions for one week before the start of the study. A gastric tube was used to administer the abamectin (5ml/kg B. Wt.) suspended in a car corn oil. Control groups were used to parallel study, only received oil corn twice a week for 15 and for one-month.
Blood Samples
Blood samples collected in a sterile tube then into a centrifuge tube. The samples allowed to coagulate at room temperature, then the serum was separated by centrifugation at 3000rpm for 20 minutes.
Methods
Determination of serum creatinine
The assay is based on creatinine reaction with sodium picrate [8] forming a red compound. The density of the colour formed was proportional to the creatinine concentration in the sample [9].
Determination of serum urea
Chromatography was used to detect serum urea with the Urea Assay Kit. The concentration of urea is determined by a combined enzyme assay, resulting in a chromatic (570nm) product, proportional to the present urea. The linear range for detecting this assay is between 1.0-5.0nmol.
Determination of Aspartate amino transferase (AST) and Alanine amino transferase (ALT) as liver marker
AST and ALT catalyse the amino group of glutamic acid to oxalacetic acid and pyruvic acid in reversible reaction. There is a substance known 2,4 dinitro-phenyl hydrazine reacts with pyruvate and oxaloacetate to form the corresponding 2.4- dinitrophenylhydrazine derivative of pyruvate 2.4- dinitrophenylhydrazine derivative of pyruvate in case the product is pyruvate and 2,4-dinitrophenylhydrazine derivative of oxaloacetate in case the product is oxaloacetate which can be measured spectrophotometrically at 546nm.
Histological Examination
The kidney and liver samples were excised, flushed with saline and then fixed in 10% buffered formalin PH 7.0 phosphate puffer. In brief, the fixed specimens were dehydrated, cleared in xylene and embedded in paraffin wax. Blocks were made, and 2-4μm-thick sections were cut using a sledge microtome. The tissue sections were deparaffinized, rehydrated and stained with haematoxylin and eosin (H&E). The stained slides were examined using bright field light microscopy to determine the histoarchitecture and the changes in kidney and liver.
Statistical Analysis
The analysis was carried out in triplicates for all determinations and results of the triplicates were expressed as mean±SE. The SPSS program (version 22 SPSS Inc., Chicago, IL, USA) [10] was used for the analysis of variance followed by the Minitab 17 for multiple comparisons of the means, P<0.05 between mean values were considered statistically significant.
Result
Table 1: Effects of abamectin on ALT, AST, urea and creatinine parameters in the blood plasma of male rats after repeated double oral doses \week for 15 days and one monthof abamectin.
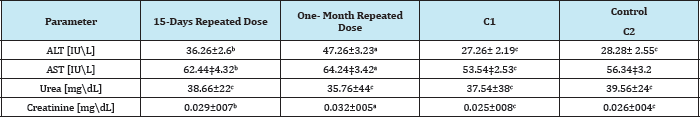
Values are means ±SEM. Means followed by the same letter(s) within each horizontal row are not significantly different at p < 0.05; ALT- alanine aminotransferase; AST- aspartate aminotransferase.
*Histopathological findings were normal except minimal congestion in two individuals of the control group (Table 1,2) ((Figure 1a & 1b).
Liver Histopathology
Light microscopy of the findings was revealed leucocytic sinusoidal permeation, congested blood vessels in the portal tract, degeneration of some hepatocytes and vacuolation of hepatocytes (Figure 2a).
Table 2: Histopathologic findings in liver and kidney.
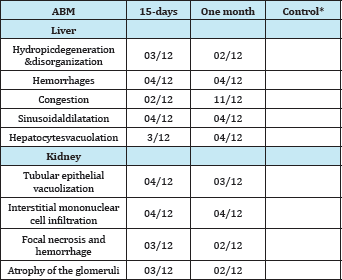
Figure 1a: Effects of abamectin on ALT, AST, urea and creatinine parameters in the blood plasma of male rats after repeated double oral doses \week for 15 days of abamectin.

Figure 1b: Effects of abamectin on ALT, AST, urea and creatinine parameters in the blood plasma of male rats after repeated double oral doses \week for one-month. of abamectin.

Kidney Histopathology
Figure 2a: Liver showing vacuolated hepatocytes (arrows) (H&E X 400)

Sections of the control rats' kidneys demonstrated normal renal histo-architecture of the kidney glomerular, and surrounding tubules (Figure 2b). Even the kidneys showed marked necrobiotic changes in abamectin-treated animals as compared to the normal histological examination of renal tissue in the control rats. A marked necrosis of tubular cells, atrophy of the glomeruli, and areas of interstitial infiltration of round cells were found (Figure 2c & 2d).
Figure 2b: Liver showing congested central vein (arrow head) and minute area of mononuclear cells infiltrations (arrow) (H&E X 400).

Discussion
Each drug was associated with liver toxicity almost because of the central role of the liver in the metabolism of the drug. Hepatic metabolism is, first and foremost, a mechanism to convert drugs and other compounds into products that are more easily excreted and usually have less drug activity than the carrier [11,12]. Metabolism may be higher activity and/or toxicity than the original drug.
In toxicological studies, a variety of biochemical parameters are measured to assess a wide range of physiological functions and metabolism that affect target organ identification and tissue injury assessment [13]. The combination of some common biochemical parameters provides better information than the recognition pattern, for example. enzymes such as ALT and AST, certain constituents (urea and creatinine) for glomerular function (Evans 1996) [14].
The results of the current study showed that in oral administration of abamectin, in 1/10 LD50, for 15 days and one- month (group 1 and 2) significantly increased (P< 0, 05) plasma levels of ALT, AST, urea and creatinine in male rats treated once a week, compared with control group.
Changes in ALT and AST levels varied depending on the time of exposure. An increase in enzyme activity was observed in group 2 compared to group 1. These results were consistent with the results obtained by [15]. ALT and AST levels activity was high as it is a dose-dependent pattern.
The activity of serum enzymes such as AST and ALT, representing the functional state of the liver [16]. Since some hepatic changes are irreversibly satisfactory [17], a higher AST may make the liver more susceptible to other pathological diseases [18,19]. Aspartate aminotransferase (AST) is an important indicator of liver damage in clinical studies. During liver injury, AST is secreted in the blood [20]. In deadly or damaged cells, these enzymes fall into the bloodstream [21].
The rise in liver enzyme activity may be due to liver weakness with a consequent reduction in the enzyme's biosynthesis and bio-change membrane permeability allowing enzyme leaks in the blood where the liver is vulnerable to direct exposure to toxic products. The liver plays a role in the detoxification of metabolic products and toxicants. In our study, increased AST and ALT levels could be due to liver toxicity that causing permeable changes and leakage of lysosomal enzymes promoting the release of enzymes [22,23]. Elevated ALT levels in this study suggest that hepatocellular tissue may be damaged by abamectin. Damage can be observed in pathogenic lesions in the liver of rats treated with abamectin. Abamectin was able to elevate levels of serum aspartate aminotransferase (AST) [24].
Effect on Kidney Function
Current results indicated that the oral administration of 1/10 LD50 ABM significantly (P< 0,05) increased the level of creatinine compared with control groups. Similar results were reported by [25,26]. Creatinine level is a useful indicator in the early deduction of renal toxicity induced by external compounds and agents. The current results have been explained that oral input of abamectin resulted in a marked increase in creatinine compared to controls. High serum creatinine concentration can be attributed to lower glomerular filtration in the kidneys and reflects a defect in kidney tubes.
Conclusion
Administration of abamectin in 1/10 LD50, for 15 days and one-month significantly increased plasma levels of ALT, AST, and creatinine in male rats. Changes in ALT and AST levels vary depending on exposure time. The results also showed that the abamectin tended to cause significant change to the liver and kidney of rats. The permeability of the leukocyte pockets, congested blood vessels in the portal tract, destruction of some liver cells and vacuolation of liver cells S and significant necrosis of tubular cells, glomerular atrophy, and interstitial infiltration areas of round cells.
References
- Burg RW, Miller BM, Baker EE, Birnbaum J, Currie JA, et al. (1979) The action of avermectin on identified central neurons from Helix andits interaction with acetylcholine and gamma-aminobutyric acid responses. Antimicrob Agents Chemother 15: 361-367.
- Turner MJ, Schaeffer M (2018) Mode of Action of Ivermectin. In: Campbell WC [Ed.], Ivermectin and Abamectin. pp. 73-88.
- Lankas G, Gordon LR (1989) toxicology. In: Campbell WC [Ed], Ivermectin and Abamectin. Springer-Verlag, New York, USA, pp. 363.
- Campbell WC (1989) Use of ivermectin in dogs and cats. Ivermectin and Abamectin. Springer, New York, USA, pp. 245-259.
- Cully DF, Vassilatis DK, Liu KK, Paress PS, Ploeg VDLH, et al. (1994) Cloning of an avermectin-sensitive glutamate-gated chloride channel from Caenorhabditis elegans. Nature (Lond) 371(6499): 707-711.
- Clark j, Scott M, Campos F, Bloomquist JR (1995) Resistance to avermectins: extent, mechanisms, and management implications. Annu Rev Entomol 40: 1-30.
- Cavera MCS, Walsh TK, Wolstenholme AJ (2007) Nematode ligand-gated chloride channels: an appraisal of their involvement in macro cyclic lactone resistance and prospects for developing molecular markers. Parasitology 134(pt 8): 1111-1121.
- Burtis A (1999) Textbook of clinical chemistry, in: (3ri edn), AACC.
- Young DS (2001) Effect of disease in clinical lab. In: (4th edn), Tests, AACC, Washington DC, USA.
- The SPSS program (version 22 SPSS Inc., Chicago, IL, USA).
- Poppers PJ (1980) Hepatic drug metabolism and anesthesia. Anaesthe- sist 29(2): 55-58.
- Tolman KG (1998) Hepatotoxicity of non-narcotic analgesics. Am J Med 105(1B): 13S-19S.
- Akhtar A, Deshmukh AA, Raut CG, Somkuwar AP, Bhagat SS, et al. (2012) Prallethrin induced serum biochemical changes in Wistar rats. Pestic Biochem Physiol 102(2): 160-168.
- Evans CO (1996) General introduction. p. 1-9. In: 'Animal Clinical Chemistry a Primer for Toxicologists201D; In: Evans GO, [ed.]. USA Taylor & Francis Inc., Frost Road, Suite 101, Bristol, 216 pp. Hsu DZ, Hsu CH, Huang BM, Liu MY 2001. Abamectin effects on aspartate aminotransferase and nitric oxide in rats. Toxicology 165(2-3): 189-193.
- Hsu DZ, Hsu CH, Huang BM, Liu MY (2001) Abamectin effects on aspartate aminotransferase and nitric oxide in rats. Toxicology 165(2-3): 189193.
- Cremer JE, Seville MP (1982) Comparative effects of two pyrethroids, deltamethrin and cismethrin on plasma catecholamines and on blood glucose and lactate. Toxicol Appl Pharmacol 66(1): 124-133.
- Helling TS, Wogahn BM, Olson SA, Evans LS, Reddy BR, et al. (1995) The effect of prostaglandin E1 on liver adenine nucleotides and cytoplasmic enzymes in a porcine model of normothermic hepatic ischemia. Hepatol- ogy 22(5): 1554-1559.
- Chamulitrat W, Spitzer JJ (1996) Nitric oxide and liver injury in alcohol-fed rats after lip polysaccharide administration. Alcohol Clin. Exp Res 20(6): 1065-1070.
- Nayak NC, Sathar SA, Mughal S, Duttagupta S, Mathur M, et al.(1996) The nature and significance of liver cell vacuolation following hepatocellular injury-an analysis based on observations on rats rendered tolerant to hepatotoxic damage. Virchows Archiv 428(6): 353-365.
- Kalender S, Ogutcu A, Uzunhisarcikli M, Acikgoz F, Durak D, et al. (2005) Diazinon-induced hepatotoxicity and protective effect of vitamin E on some biochemical indices and ultra structural changes. Toxicology 211(3): 197-206.
- Mansour SA, Mossa AH (2010) Oxidative damage, biochemical and histo-pathological alteration in rat exposed to chlorpyrifos and the role of zinc as antioxidant. Pest Biochem Physiol 96(1): 14-23.
- Choudhary N, Sharma M, Verma P, Joshi SC (2003) Hepato and nephrotoxicity in rat exposed to endosulfan. J Environ Biol 24(3): 305-308.
- Shrivastava AK, Raina R, Choudhary RK, Malik TK (1989) The acute toxicity and biochemial alterations in rats after single oral exposure to di- chlorvos. Pesticides 2(1): 35-40.
- Lowenstein M, Loupal G, Baumgartner W, Kutzer E (1996) Histology of the skin and determination of blood and serum parameters during the recovery phase of sarcoptic manage in cattle after avermectin (Ivomec) treatment. Appl Parasitol 37(2): 77-86.
- Eissa FI, Zidan NA (2010) Haematological, biochemical and his to pathological alterations induced by abamectin and Bacillus thuringiensis in male albino rats. J Basic Appl Sci 3(3): 2497-2505.
- Abd-Elhady HK, Abou-Elghar GE (2013) Abamectin induced bhiocemical and his to pathological changes in the albino rat, Rattus norvegicus. Plant Prot Res 53(3): 263-270.
© 2018 Mohey Mekawy, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






