- Submissions

Full Text
Research in Medical & Engineering Sciences
Lean Enablers for Clinical Laboratories
Bohdan W Oppenheim1*, Michael H Kanter2, Onie Bueno3, Vincent L Dizon4, Louie M Farnacio5, Paulette L Medina6, Mike M Moradian7, Chiemi Tabata8 and Michael S Tiffert9
1PhD, Professor of Systems Engineering, Associate Director for Healthcare Systems Engineering, Loyola Marymount University, Los Angeles
2MD, CPPS, Regional Medical Director of Quality & Clinical Analysis, Southern California Permanente Medical Group (SCPMG), Executive Vice President, Chief Quality Officer, The Permanente Federation
3Director of Operations, Bacteriology and Molecular Microbiology, SCPMG, Regional Reference Laboratories, Chino Hills
4MBA MT(ASCP) DLM SBB. Director of Operations, Chemistry Services, Kaiser Permanente SCPMG Regional Reference Laboratories
5Director of Laboratory Services, Kaiser Permanente Southern California Regional Reference Laboratory
6Assistant Director of Operations, RRL Bacteriology, Kaiser Permanente SCPMG Regional Reference Lab
7PhD, Director of Operations, Molecular Genetic Pathology Regional Laboratory, Kaiser Permanente SCPMG Regional Reference Laboratories
8Assistant Director of Operations, RRL Automated Chemistry, Kaiser Permanente SCPMG, Regional Reference Laboratories
9MS, Section Manager, Cytogenetics, Regional Genetics Laboratory, Kaiser Permanente SCPMG, Regional Reference Laboratories
*Corresponding author: Dr. Bohdan W Oppenheim, Professor of Systems Engineering, Associate Director for Healthcare Systems Engineering, Loyola Marymount University, Los Angeles, USA
Submission: October 23, 2017; Published: November 27, 2017

ISSN : 2576-8816Volume2 Issue4
Abstract
A new intellectual product named Lean Enablers for Clinical Laboratories (LEfCL) is described. It is a collection of 136 best practices for clinical laboratory operations, containing collective wisdom on how to practice laboratory management, focusing on simultaneous improvements in test quality, productivity, laboratory capacity and stakeholder satisfaction, and reductions of test turnaround time and cost. The methodology follows an awarded method established in Systems Engineering and represents an extension of that engineering knowledge into clinical practice. The product has been developed by experienced directors from Kaiser Permanente regional clinical laboratory while pursuing a graduate Certificate in Lean Healthcare at Loyola Marymount University. LEfCL are organized into six well-known Lean Principles: Value, Value Stream Mapping, Flow, Pull, Perfection and Respect. The paper also identifies 46 Challenges that prevent success. Results of Lean improvements measured over the period of 2014-2016 are quoted for five laboratories. The LEfCL are not intended to become a mandatory practice. Instead, they should be used as a checklist of good practices. LEfCL are formulated as a comprehensive set of practices, intended for future training of new hires and as reference material. As such, they include both basic and advanced practices.
Keywords: Lean; Lean enablers; Clinical laboratories; Process improvement; Streamlining
Introduction
The healthcare industry in the US is the largest segment of the economy; exceeding three trillion dollars [1]. While the ACA seems to have started bending the overall healthcare cost curve downward [2], most of healthcare as well as individual patients are still experiencing high costs across the industry. The rapid changes following the introduction of the ACA and recent political turbulence tend to create stress within healthcare providing entities. Clinical laboratories are no exception and are feeling the challenges of higher number of patients, lower revenues, higher costs, quality and service challenges, and increasing regulatory oversights. This paper offers a methodology to help reverse this trend in clinical laboratory operations using Lean methodology.
Materials and Methods
Lean Thinking in Healthcare, or Lean Healthcare for short, is well established as the modern work paradigm and has been widely published, e.g. in [3,4]. It has been proven as the systematic approach to simultaneously reduce waste, cost and process throughput times, improve the capacity and quality, provide better services, and improve the work morale of the employees and other stakeholders. Lean routinely reduces the amount of waste in the system by 30-50-70% [5]. This paper extends the paradigm to clinical laboratories.
The tool that we have developed is called Lean Enablers. A Lean Enabler is a best practice minimizing waste, test turnaround time, cost and frustrations of providers and patients served, as well as maximizing test quality and work satisfaction, and is based on Lean Thinking. Each Enabler is typically one sentence long, stated in imperative form, eg, "Reduce batch sizes". Some Enablers are described in several short related sentences. Most Enablers listed in this paper are self-explanatory; a few which may not be are amended with brief explanatory paragraphs.
The general methodology used followed two large projects that involved six years of work by hundreds of experienced systems engineers and defense and commercial program managers from top global companies. The results have been published in [6], as well as in two books: [5,7]. In short, the methodology is based on capturing tacit knowledge and experience of experts, using intense brainstorming and comprehensive iterations and editing of the captured knowledge. Both publications received awards from the sponsoring organizations International Council on Systems Engineering (INCOSE) and Project Management Institute (PMI), as well as three awards from the Shingo Prize Institute in 2011, 2013 and 2017. While the scope of the present project developing Lean Enablers for Clinical Laboratories was significantly smaller than in the above-mentioned works (only 11 individuals working productively for about 10 weeks) fast progress was made possible by applying the former methods and lessons learned.
Setting
This study was undertaken in the Regional Laboratory of the Southern California Kaiser Permanente (KP) region. This laboratory is part of a complete laboratory system that provides services for 4.1 million members covering over 200 medical office buildings and fourteen hospitals. There are approximately 6000 physicians in the system. Patients receive essentially all their care within the KP system. The laboratory performs over 30 million tests per year and covers a broad range of routine and highly specialized clinical laboratory tests. Some routine tests and emergency room/hospital tests are performed in the local hospitals with non-stat routine and highly specialized tests being performed at the regional lab.
Definitions
Lean thinking1: Three concepts are fundamental to the understanding of Lean Thinking: value, waste, and the process of creating value without waste, which is captured in the so-called six Lean Principles. These concepts are summarized in this section in the general context of Lean clinical laboratories. Any reader who is new to the concepts of Lean Thinking would greatly benefit from reading the seminal books by [3,8]. Lean Thinking adopts several practices previously known by other names, such as Six Sigma, total quality management, concurrent engineering, and others. The criterion we use for adoption is simple: If a best practice promotes value, reduces waste, and can be captured into the Six Lean Principles, it is called Lean.
Value: The concepts of Value and customer are interlinked. Value has a meaning only for a specific customer or groups of customers. In clinical testing laboratories both the patient and the provider (physician or nurse) who ordered the test are the external customers. They need accurate test results as soon as possible in order to diagnose the patient and provide the needed treatment. Fast treatment often saves lives and is always conducive to better health. Another aspect of value is cost. Driven by the ACA, competition, and national imperative, clinical laboratories need to strive to keep lowering the costs of their testing as long as the test quality or throughput time are not jeopardized.
Waste: The ability to identify and eliminate waste is a critical skill for Lean practitioners. All work activities are classified into the following three categories [8]:
1. Value-added (VA) activities, which must satisfy the following three conditions:
a) Transform information or material, or reduce uncertainty (cannot be an unnecessary bureaucratic task that creates no value).
b) The cost of the value is reasonable to the customer (in more complex activities, such as clinical testing, the acceptance is implicit, that is, if the customer understood the details, he or she would approve of this cost.)
c) It is done right the first time. Rework resulting from former defects does not count.
2. Required (also called necessary) non-value-added (RNVA) activities, which do not meet the previous definition, but which cannot be eliminated because they are required by regulatory agencies, law, contract, company mandate, current technology, or other similar reason, eg the HIPAA - mandated activities.
3. Non-value-added (NVA) activities, which consume resources and create no value. They are pure waste, and should be eliminated immediately. Taiichi Ohno [9] classified waste into seven (later extended to eight [5] categories shown in the left column of Table 1. The right-hand side column lists examples of waste in clinical laboratories.
Table 1: Eight Types of Waste with Examples from Clinical Laboratories.

1This section has been adapted by the first author from chapter 3 of his book [5].
The Six Lean Principles
The process of creating value without waste is captured into six following Lean Principles. The name of a given Principle is listed in quotes, followed by the meaning and an explanation. The effectiveness of the Lean Principles has been demonstrated in a broad range of industries and work environments, including production; engineering and systems engineering; supply chain management; finance, banking and general administration; education; healthcare, and others. Oppenheim [5] contains a review of the literature which is not repeated here.
Principle 1, "Value": Capture the value defined by the customer stakeholders, who include the patient and the provider who ordered the test. The importance of capturing test value (understanding what needs to be done) with precision, clarity, and completeness cannot be overemphasized, to avoid unnecessary rework.
Principle 2, "Value Stream”: Map the value stream (plan the process) and eliminate waste. This is an important tool of Lean, which maps the current test process with all end-to-end linked tasks, control/decision nodes, and the interconnecting flows necessary to realize customer value, starting from specimen collection and ending in making the results available to the provider. During the mapping process, we identify and eliminate all NVA activities, minimize all RNVA activities, and enable the remaining activities to flow predictably and efficiently (the flow is described in Principle 3). Typically, we produce two maps, the Current State that identifies all wastes and frustrations, and Future State which is waste free and ready for implementation.
Principle 3, "Flow”: Flow the work through planned and streamlined value-adding steps of the Future State map, without stopping, batching, or idle time, unplanned rework, or backflow. Keep the samples moving through the required steps without waiting.
Principle 4, "Pull”: use the Just-in-time delivery of samples to the needed instrument or work station and to the final posting of the results for both the provider and patient to see. Samples should travel in as small batches as possible. Ideally, samples should be in constant motion from the point of collection until the posting of the results and archiving.
Principle 5, "Perfection”: Pursue perfection in all processes. Global competition is a brutal "race without a finish line," requiring continuous improvements of processes and products. Two features of Lean help prioritize processes for improvement: (a) making all imperfections in the workplace visible to all; and (b) prioritizing to eliminate the biggest impediments to flow and to reduce costs. Seeing problems as they appear in real time is conducive to making better decisions on corrective actions and better prioritization of improvements. When noticed early, imperfections tend to be easier and less expensive to fix; unnoticed early, they tend to grow to crisis proportions and require extensive actions to mitigate. Making imperfections visible is a motivator for applying continuous improvement in real time. The enterprise should create an effective infrastructure for capturing knowledge and lessons learned and for promoting continuous education to make each new process/step better than the last.
Principle 6, "Respect for People”: Respect the people in your program. A Lean enterprise is an organization that recognizes its people are the most important resource and is one that adopts highperformance work practices. In a Lean organization, people are encouraged to identify problems and imperfections honestly and openly in real time, brainstorm root causes and corrective actions without fear, and plan effective solutions together by consensus to prevent a problem from reoccurring. When issues arise, the system is blamed and not the messengers. People must not be afraid, and management must abandon the "blaming and shaming" habits and replace them with the spirit of teamwork and "we are in it together" Experienced and knowledgeable leaders lead, motivate and mentor, but also empower frontline employees to solve problems immediately. Such an environment requires a culture of teamwork, mutual respect and trust, open and honest communication, and synergistic and cooperating relationships of all stakeholders. As stated under Principle 5, a fundamental premise of Lean is to make all imperfections visible in order to eliminate them immediately, at the source, before they move downstream and grow to crisis proportions. Employees should be rewarded for bringing system (not people!) imperfections to the attention of the stakeholders. The imperfections should never be personalized; by definition, they are system problems. But in order to enable this behavior, the Respect for People Principle must be continually pursued. Lean employees and managers know that their real competitors are not their colleagues within the same enterprise, but rather an outside clinical facility.
Myths to Be Debunked About Lean
Even though Lean is a successful and well-established work organization paradigm in healthcare, eg (3), (4), several false myths about Lean make rounds in healthcare community. Therefore, we now debunk three worst myths:
1. In Lean 'people need roller skates' to work faster and harder
Most emphatically: Lean does not make people work faster or harder, and it does not ask employees to wear roller skates, but it expects people to work smarter [3]. Lean is not about moving faster between the work locations; instead Lean advises to reduce the amount of walking by re-architecting spaces, and then walk at a normal ergonomic speed. In Lean, nobody needs to work harder than the ergonomic rate determined by the education, training, and experience, but the employees are expected to identify and eliminate unnecessary waiting and defects and other wastes in the processes, to give them more time for adding-value work.
2. Lean cuts corners on quality
Lean never cuts corners on quality; quite the opposite is true: Lean improvements inherently improve work quality [8]. Lean advises to perform all tasks truly needed to deliver quality value; but also, to pay attention to waste and eliminate waste as much as possible.
3. Leans means "mean layoffs”
Properly implemented Lean is an enemy of layoffs. Lean uncovers productivity reserves which are utilized for good use: improvements of processes, training and education, increasing the laboratory capacity, and better service to stakeholders2.
Approach
In 2013 the first two authors of this paper (BWO and MHK) met to discuss the advantages of Lean in healthcare. Even though Kaiser Permanente (KP) as a system is highly ranked in the US healthcare [12], MHK of KP felt that there was opportunity to improve. The laboratory was chosen as an initial site to implement Lean because of its resemblance to other industries where lean has been successfully implemented. Loyola Marymount University (LMU) at the same time was developing a new master's program in healthcare systems engineering looking for real-world applications of Lean in healthcare.
As a result, a novel approach to Lean implementation was undertaken. A graduate three-course Certificate in Lean Healthcare was created at LMU, and 10 "students" were selected from the laboratory staff, all experienced managers and directors of their respective sections of the KP regional lab. Two courses, on Lean basics and advanced Lean, were followed by a hand-on project course in the laboratory under the supervision of BWO- a faculty in systems engineering. During the project students were required to design Lean improvements and develop a set of Lean Enablers for the clinical laboratories (LEfCL).
2The increasing number of patients enrolling into healthcare system due to the ACA and the significant shortage of medical professionals practically assures no layoffs for years to come. Also, people will never stop falling ill.
The result was 136 LEfCL described herein. Most of the Enablers are applicable across the laboratory industry and are cited in this paper. About 15 applicable only to specific situation of KP laboratories have been omitted from the present article.
Following the approach of[7], prior to formulating the Enablers, the team formulated 46 Challenges that clinical laboratories face today. Essentially, the methodology of capturing and developing the Challenges was identical to that of the Enablers: iterative collective brainstorming and editing. Most of the Challenges deal with operational problems. Some of the Challenges deal with cultural issues which are critically important for the operational problems to be addressed effectively. Lean cannot be implemented "mechanically", addressing only the tools and processes while ignoring human factors. Again, most of the Challenges are applicable across the laboratory industry and are cited in this paper. About 15 applicable only to specific situation of KP laboratories have been omitted from the present paper. Collectively the Lean Enablers attempt to answer the Challenges; however, the relationship is one-to-many and many-to-one rather than one-to-one. Most of the Challenges apply to all clinical laboratories across the industry, and these are listed without special headings. A subgroup of the Challenges applies to specific laboratory types, and these are listed under the types.
When creating the Challenges and Lean Enablers, the team made a significant effort to apply systems thinking ("the holistic big-picture thinking") for the entire laboratory system, including not only technical operations but also people and end-to-end throughput, including sample transport from collection to posting test result. Thus, the reader will see several Enablers that focus on the significance of process standardization, visual controls, facility layouts, transport, and cleaning and organization of the workplace. A one-time improvement is not a Lean process therefore the Enablers focus on perfecting the processes through continuous improvement. The Enablers promote allocation of teams to implement and measure the success of proposed solutions, as well as building in-process quality assurance from start as a preventive measure, and not relying on final inspection, which is applied too late to avoid errors. Critical emphasis is placed on workforce development, including practices such as cross training, creating multi-skilled workforce with a continuous improvement mindset, promoting participatory decision making and good leadership at all levels, as well as abandonment of the "blaming and shaming" culture3. LEfCL are formulated as a comprehensive set of practices, intended for future training of new hires and as reference material. As such, they include both basic and advanced practices.
Statistical Analysis
Ideally, one would wish to perform control tests measuring the effectiveness of each Lean Enabler individually, including measurements of the test turn-around-time (TAT), cost and defect reductions; capacity increase, and staff morale. The task of estimating these tangible benefits from the implementation of individual Enablers is not feasible in the busy production laboratories. Instead, we provide some aggregate benefits from the combined Lean improvements undertaken in each laboratory during the period from 2014 to 2016. Not all labs measured all the above benefits, so we quote only those measured or reliably estimated.
Results
In this section the authors use many Lean terms; these are explained in the Glossary. The Japanese words are shown in italic font.
Top challenges in managing clinical laboratories
Table 2 lists 46 challenges identified for clinical laboratories. The challenges describe typical problems that need to be eliminated. Likely, KP is not the only healthcare organization experiencing the challenges. KP has decided to publish the challenges in the spirit of improvement of professional practices across the industry. The Challenges listed herein deal only with broadly understood Lean aspects (waste and value and the flow of samples through the system) rather than the normal clinical, medical, chemical, biological, electronic or test instrument issues that are the subject of the CLS knowledge. The challenges are grouped into general types, those labeled with the first digit of 1, applicable to the entire enterprise; and those that apply to a specific laboratory, labeled with the first digits of 2 to 6. The latter are listed only for the laboratories that were represented in the project; but the reader should keep in mind that there are many other clinical laboratory types not covered here. The second digit is the sequential number of the given challenge in the group. The grammatical form of each challenge is a statement or observation agreed upon by a team.
Table 2: Challenges.
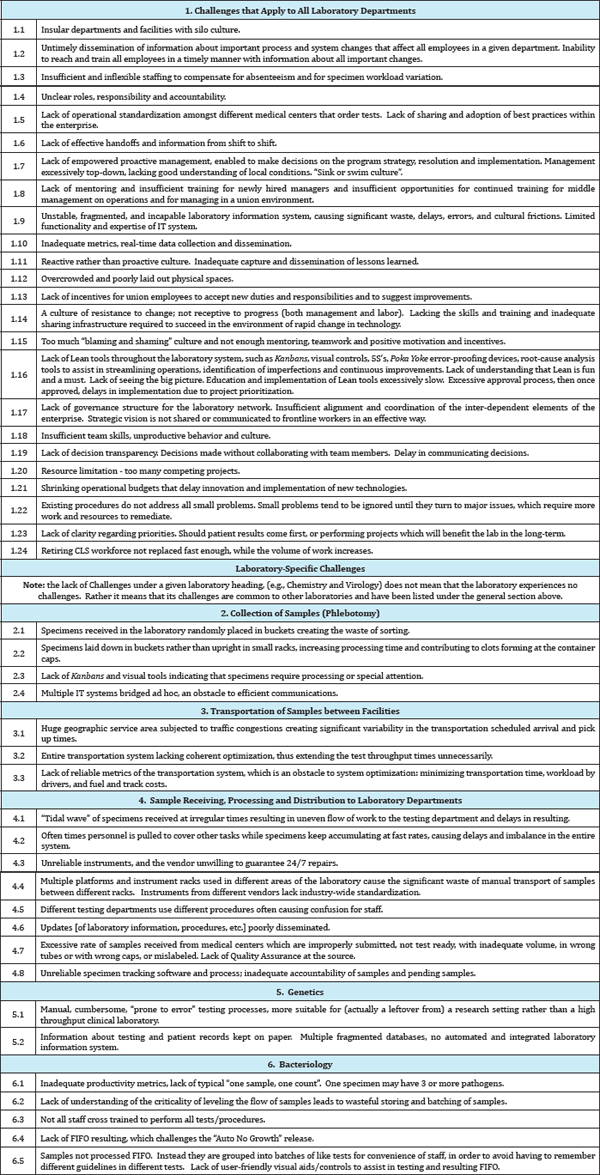
3KP is a union environment, with several unions represented in clinical laboratories.
Lean enablers
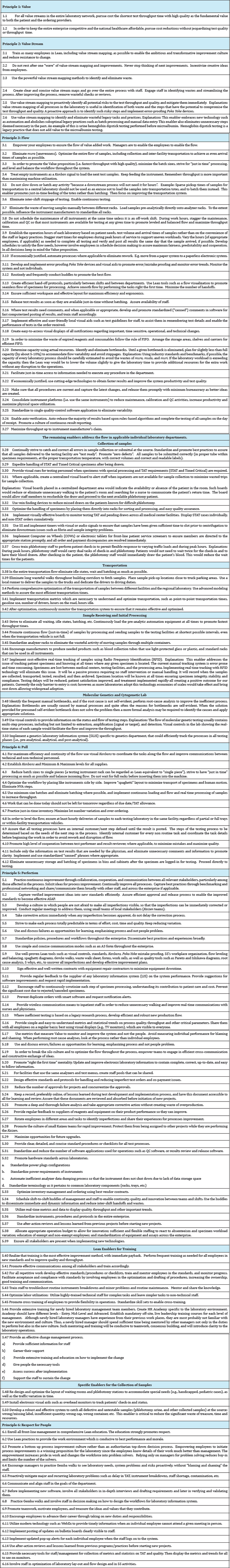
The 136 Lean Enablers are displayed in Table 3, organized by the Lean Principles (Value, Value Stream, Flow, Pull, Perfection, and Respect for People summarized above in the definitions section.) In most cases they are listed without additional subheadings. In several cases when a given Enabler applies to a specific laboratory department, it is listed under the department name. The Enabler numbers consist of two digits (x.y) where "x" is the Lean Principle number and "y" the consecutive Enabler number under that Principle. The readers will find the clear majority of the Lean Enablers to simply represent good sense. Our collective challenge is to translate this good sense into common sense (Table 3).
Table 3: Lean Enablers.
Numerical results of lean improvements
Lean improvements are described in the following testing laboratories: genetics, genetic Fluorescence in Situ Hybridization (FISH), automated chemistry, bacteriology, and bacteriology ova and parasite test laboratories. In each case, we first summarize the undertaken Lean improvements in a letter-numbered form, followed by a short table with available results. We close with Lean improvements in the Specimen Processing Department (SPD), which receives samples from inter-city transportation trucks, sorts the samples, prepares them for laboratory work, loads them into racks and loads the racks onto robots for automated transport to individual laboratory departments (all labs are in the same building but on different floors and corridors). Besides the five laboratories quoted here, several others attempted Lean improvements, but staff changes prevented the capture of improvement data.
Molecular Genetics Pathology Laboratory
The initial state in 2014 was a slow, highly manual process reminiscent more of a university research lab then a production clinical laboratory, involving manual entries and transcriptions between MS Office tools and different computers.
Lean improvements
a. Implementation of a new Genetics Laboratory Information System (GLIS). All processes now performed in one system from beginning to the end by one technologist, eliminating hand offs and the need for multi-system workflow. Hand offs were the main cause of delays and communication failures and eliminating them was a major improvement.
b. Elimination of the manual data transfers between different files and systems, which eliminated the need for multiple reviews to assure the transfers were performed correctly.
c. Elimination of manual transcription, which wiped out the transcription errors and eliminated the need for a daily review of the data entries. Prior to Lean, regular reviews were held for every step of testing to catch the mistakes, and were not fully successful in catching transcription errors.
d. Elimination of batching and inventory, tasks not needed, idle states, rework, waiting, human motion, and material transport; reduction of effort on tasks needed.
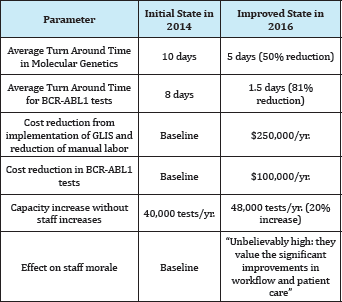
e. The reduction of TAT for the BCR-ABL1 tests from 8 to average of 1.5 days reduced the patient wait for chemotherapy and significantly improved the patient's care and experience.
The results are shown in Table 4.
Table 4: Results from Lean Improvements in Molecular Genetic Pathology, 2014-2016.
Molecular Genetics Fish Laboratory
Lean improvements
a. Implementation of the GLIS,
b. Batch size reduction: multiple FISH probes (up to 8) dropped on one slide which reduced both the number of slides to be washed and the amount of FISH probe needed (to 1μL from 4μL), causing a significant cost reduction.
c. The single piece flow initiated. Section managers receive cases as soon as they completed and cases are reviewed and sent for sign-out one case at a time replacing former batching.
d. Improved interfaces with medical centers allow for many of the samples to come already accessioned, which caused drastic reduction in wait times.
e. The ordering physician receives test result instantaneously after it is signed off electronically, replacing the former system in which several hours of delay occurred waiting for a technologist to first export cases into Laboratory Management System software and then separately to release them into Health Connect software before the physician would receive the result.
f. Elimination of rework: data entry into FileMaker database has been eliminated (which was a rework of data entry).
g. Better interfaces in the GLIS and the use of barcodes dramatically reduced transcription and misspelling errors of demographic and clinical information on forms. The physicians now place their own orders and enter the required information, thus eliminating the searches by laboratory staff for the information.
h. Due to the GLIS, elimination of paper folders for each FISH test, and of the need for copying of the genetic test requisition forms. The use of barcodes on slides eliminated handwritten lists.
i. Elimination of the manual flagging of STAT cases thanks to GLIS.
j. Electronic tracking and usage of the reagents before they expire.
k. Dramatic elimination of human walking with paper charts due to the electronics replacing paper.
l. Human walking reduction due to a placement of instruments much closer to the set-up area. m. Automation of the FISH washing eliminated variability in the final slide quality and ensured consistent test reproducibility
n. Sign offs of results performed in the evenings increased the number of test releases in a 24 hr period. Also, employees' shifts were changed to better address work flow and reduce overtime cost.
The results are shown in Table 5.
Automated Chemistry Laboratory
Lean Improvements:
a) Samples sent hourly to the lab to reduce batching
b) "Spaghetti" layout of lab spaces streamlined
c) Samples loaded directly onto instrument racks to eliminate manual transfer of samples between different racks (on subset of instruments only)
Table 5: Lean Improvements in FISH Laboratory, 2014-2016.
d) Originally, the daily maintenance, calibration and quality control was performed on all instruments during the morning shift, effectively idling all instruments, causing the buildup of large batches of samples, and extending the throughput times. This work was staggered so that only a third of the machines are idle per shift, and 2/3 are productive, to enable continuous testing and maximize throughput.
e) Batch size reduction effort in all applications and migration towards single piece flow (treating one rack as a "single piece").Distribution of received samples to instruments in real time.
f) Implementation of auto verification
g) Elimination of wasteful Hemastix testing
h) Elimination of manual uncapping of tubes with specimen
i) Limited progress towards standardization to a single tube type throughout the system.
j) Patient moving average provides a tool to continuously monitor stability and provides early detection and notification for analytical shifts not always detected by standard QC methods thereby giving only accurate patient results
k) Consolidation of tests and validation between different analyzers
The results are shown in Table 6.
Table 6: Lean Improvements in FISH Laboratory, 2014-2016.
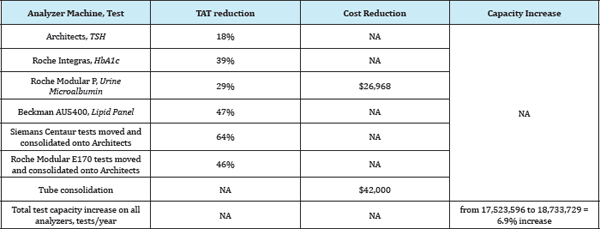
Bacteriology Laboratory
Lean improvements
a) Batching minimized in in-patient hospitals: Nurses instructed to send specimens through the tube system as soon as they are collected. If the tube system is down, a runner to pick up the samples from the hospital floor and deliver them for transportation to the lab.
b) Transport time reduction by building the new lab facility close to the medical centers served.
c) Batching of samples replaced with continuous workflow. As soon as the samples are received in the SPD, staff logs them for processing and delivers to the lab in 30 min intervals.
d) Elimination of waiting using laboratory automation system. Specimen identified with barcode is loaded into a sorter, moved through a conveyer belt to the processor, then into an automated incubator. Cultures can be read and resulted earlier because this system can detect clinically relevant growth quicker than with traditional incubators and manual reading.
e) Replacement of traditional biochemical method with mass spectrometer method to identify organisms. This reduced waiting for the identification from 24 hours to 32 minutes for 16 samples (about 2 min/sample).
f) Elimination of tasks not needed: Batches or transfer list eliminated since cultures are incubated in a "smart" incubator where each plate is assigned a unique location based on the barcode, allowing for random and rapid retrieval.
g) Elimination of double log in. The continuous workflow eliminated the need to acknowledge receipt into the ancillary department.
h) Implementation of automated image analysis software for detection and segregation of positive from negative samples using chromo genic agar. The software has a sensitivity of 100%, and a specificity of 90-96%. Plates can be segregated and resulted in groups of 40 instead of individually.
i) Elimination of transfer list reduced the need for paper.
j) Elimination of human motion: specimens are loaded onto a sorter, travel through a conveyor track, and are delivered and evenly distributed into the four specimen processors used. Once processed, the specimens are returned to the same location. One loading and unloading location replaced prior set up when each processor had to be manually loaded and unloaded.
k) Stocking of drawers by each work area with necessary supplies. One person spends one hour every other day to re-stock the drawers. Minimizes the need for CLS to get up each time they run out of supply.
The results are shown in Table 7.
Table 7: Lean Improvements in Bacteriology Laboratory, 2014-2016
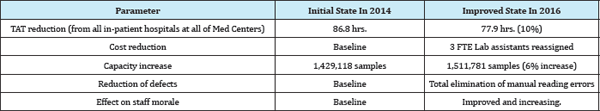
Bacteriology Ova and Parasite Testing Laboratory
Lean Improvements
a) Re-scheduling of all processing to be performed on the day shift by 1-2 lab assistants under CLS supervision, eliminating the waiting for CLS during afternoon and/or night shifts.
b) Elimination of defects due to the CLS staff now supporting Lab Assistants in real time about problem specimens.
c) Conversions to the "Total-Fix" vial from former "SAF" containers eliminated non-value adding tasks of adding formalin, and centrifuge/decant steps. The Total Fix vial being free of PVA, mercury, and formalin, is greener and safer to work with. The elimination of SAF vial and other stool collection containers from inventory simplified the ordering process for the supply staff. The elimination was gradual, stocking the new type vials as old ones were used.
d) Elimination of formalin, a flammable safety hazard and replacement of carcinogen and odor-emitting Xylene with a safer and less odorous xylene substitute has increased the morale of the staff.
e) Selection of a standardized commode stool collection container, and improved patient instructions for clarity.
f) Improved collection system combined with better instructions yield better specimen to examine.
g) Significant increase in capacity allowing for significant reduction of specimen sent to outside testing facility at large cost.
The results are shown in Table 8.
Table 8: Lean Improvements in Bacteriology Ova and Parasite Laboratory, 2014-2016.

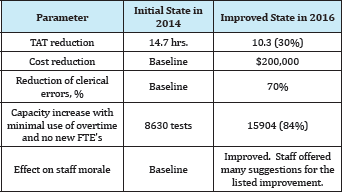
SPD
Lean improvements
a) Effort to minimize the number of untestable samples (bad or missing label, wrong container, inadequate volume of the sample, missing samples, etc.) sent from medical centers.
b) Reduction of batching of samples, more JIT processing
c) Sending samples to labs hourly rather than in large multi hour batches.
The results are shown in Table 9.
Table 9: Lean Improvements in SPD, 2014-2016.

Note: the increase in the numbers of specimen reflects the increase in the number of patients served, however, the increase was possible thanks to the Lean improvements. Also, note that many specimens are aliquoted into several others, so the number of tests is several times larger.
Conclusion and Discussion
The Lean Enablers for Clinical Laboratories (LEfCL) represent a collection of best productivity practices based on Lean Thinking that is minimizing waste and promoting value. The value is defined as the correct and accurate test with test results delivered to the ordering provider in the minimum time, at minimum cost, with maximum quality and improvement of staff morale. This paper presented 136 LEfCLs. The paper also presented the productivity and quality Challenges observed in clinical laboratories, which the LEfCLs remedy. Numerical results from five laboratories and from the Specimen Processing Department that serves all laboratories are quoted for the period of 2014 to 2016. Quoted are overall simultaneous improvements achieved from implementation of selected LEfCLs, including reduction in test turn-around-time, laboratory capacity increase, cost reduction, defect reduction, and staff morale improvements; however, not all these parameters were measured in all laboratories quoted. The two-year period coincided with a 14% increase in the number of tests performed due to the ACA-driven increased number of patients. Given the pressures to perform significantly more tests with minimal or no staff increases and even some staff reductions pushed the desired task of keeping accurate productivity metrics to a lower priority, so we can quote only aggregate results. Nevertheless, the Lean improvements shown vary from 6% to 81%. The undertaken improvements represent a small subset of the LEfCL. Therefore, the results quoted can be regarded as highly conservative estimates of the power of LEfCL. These results suggest that Lean can be a powerful paradigm for productivity, quality and staff morale improvements in clinical laboratories as it is in other healthcare operations, and that LEfCL represent a powerful improvement tool.
In order to achieve the goals set forth by the Lean Enablers one needs to have an efficient and timely process to introduce and implement the changes. In other words, a change process management is needed that will minimize resistance and create desirable results. The Lean Enablers proposed in the "Respect for People" section are intended to facilitate the change process.
The Lean Enablers should be particularly useful for planning new laboratories and training new hires. As Covey [13] stated one of the habits of highly effective people is to "begin with the end in mind." The Lean Enablers support this goal two-fold, by stressing the need for a clear understanding of the customer (provider who ordered the test and the patient) requirements and value perception, as well as proposing various effective laboratory management practices to efficiently fulfill these requirements. Lean Thinking can be ingrained in the laboratory "DNA" at the foundation level across all the people from the time they begin as team members. The benefits of this are that the people within the organization evolve to think in Lean terms and pursue Lean as a means by which the company delivers value to its customers. In organizations of this nature, Lean simply becomes the preferred way of working, and Lean Enablers become more of an automatic response by the people doing work for their customers on a daily basis, [7].
As stated earlier, this paper provides reference material. The Lean Enablers are not intended to serve as mandatory practices, but rather as both comprehensive training materials and as a checklist of best practices that can help with managing clinical laboratories more effectively. The Enablers have been collected and organized by highly experienced laboratory managers, from the highly ranked healthcare organization, therefore ought to represent a degree of wisdom to emulate by professionals engaged in clinical laboratory work.
One should not attempt to implement all Enablers at once; that would be a formidable task. Instead, the focus should be on those that will have the highest impact, and on the "low hanging fruit". Usually the highest priorities for improvements are: the biggest impediments to flow, impediments to test quality, and the sources of providers' complaints. Line staff should be encouraged to implement the Enablers which deal with the individual's work and do not need any significant coordination with others.
Oehmen [7] stated: "...a proactive way to utilize and implement the Lean Enablers is when an organization is operating without any major difficulties, but decides to find even better ways to provide greater value to their customers. Triggers can be the strategic planning of the value stream and then choosing to proactively improve some key processes that are operating well enough in the current state. Questions, such as 'what are our theoretical limits of performance?’ or 'how can we sustainably outcompete our competitors?' or 'what does true success for our customer really look like?’ are asked. Great levels of success are guaranteed when an organization attains world class business performance and sets the standard for everyone else."
The intent of the Lean Enablers must be clearly understood by those who will use this information. Most importantly, the laboratory leadership must understand the six Lean Principles. The maturity of an organization's Lean understanding will help determine the customizing and tailoring required for specific situations and the laboratory management leadership.
There are some limitations to this paper. We did not have a matched control group and did not completely analyze all aspects of the lab operation but merely measured what we could while operationalizing Lean. We cannot guarantee that our results would be generalizable to laboratories of different size or scope or those not part of an integrated delivery system. However, Lean has been applied across a wide range of industries and there is no reason to think that at least directionally our results would not generalize. Lastly, much of Lean implementation requires good leadership and the right culture.
To quote from [7]: "Much of the success of all Lean deployment truly rests with the quality of the Leadership of the organization. Leaders of the organization should define what their approach is, communicate it with great repetition, visibly participate with the Lean transformation activities, and provide reward and encouragement to those who are advancing the organization's Lean journey. Given this level of leadership support, these differing approaches become complementary and ultimately begin to achieve a Lean culture that is continuously improving itself through the implementation of Lean in the unending pursuit of perfection.”
Glossary (Japanese words are italicized)


Institution
Kaiser Permanente
References
- (2014) PCAST (President's Council of Advisors on Science and Technology), Report to the president better health care and lower costs: accelerating improvement through systems engineering. executive office of the president, USA.
- (2014) CBA (Congressional Budget Office), Updated estimates of the effects of the insurance coverage provisions of the affordable care act.
- Graban M (2011) Lean Hospitals: Improving Quality, Patient Safety, and Employee Engagement. (2nd edn), CRC Press, Boca Raton, Florida, USA.
- Toussaint J (2010) On the Mend: Revolutionizing Healthcare to Save Lives and Transform the Industry. Lean Enterprise Institute, Boston, USA.
- Oppenheim BW (2011) Lean for systems engineering with lean enablers for systems engineering, Wiley, Hoboken, New Jersey, USA, pp. 336.
- Oppenheim BW, Murman EM, Secor DA (2011) Lean Enablers for Systems Engineering. J Systems Engineering 14(1): 29-55.
- Oehmen J (2012) The Guide to Lean Enablers for Managing Engineering Programs. PMI-INCOSE-MIT LAI.
- Womack J, Jones D (2003) Lean Thinking: Banish Waste and Create Wealth in Your Corporation, (2nd edn), Simon & Schuster, New York, USA.
- Ohno T (2014) Workplace Management: Special (100th Birthday). McGraw-Hill, USA, Dec. 11, 201213.
- Oppenheim BW (2014) Lean Enablers for Clinical Laboratories, Kaiser Permanente Report, USA.
- Liker J (2004) The Toyota Way: 14 Management Principles from the World's Greatest Manufacturer, McGraw-Hill, New York, USA.
- Consumer Reports (2015).
- Covey SR (2002) The 7 Habits of Highly Effective People. Stephen covey. com Publication.
© 2017 Bohdan W Oppenheim, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






