- Submissions

Full Text
Research in Medical & Engineering Sciences
Anthelmintics and their Application in Veterinary Medicine
Enejoh OS and Suleiman MM*
Department of Pharmacology and Toxicology, Nigeria
*Corresponding author: Suleiman MM, Department of Pharmacology and Toxicology, Faculty of Veterinary Medicine, ABU, Zaria, Nigeria
Submission: August 31, 2017; Published: November 13, 2017

ISSN : 2576-8816Volume2 Issue3
Introduction
Parasitic diseases remain a major constraint to livestock productivity across all agro ecological zones and production systems in Africa, and gastrointestinal nematodes remain a major economic importance in domesticated livestock throughout the world [1] being the chief parasitizes responsible for disease-related production losses arising from stock mortality, severe weight loss and poor production, especially in small ruminants. The World Health Organization estimates that a staggering 2 billion people harbour parasitic worm infections. Parasitic worms also infect livestock and crops, affecting food production with a resultant economic impact. Also of importance is the infection of domestic pets. Indeed, the companion animal market is a major economic consideration for animal health companies undertaking drug discovery programmes. The parasitic helmets of animals are broadly classified into nematodes (round worms, whipworms, hookworms, pinworms, threadworms, and filarial worms), custodies (tapeworms), treaties (flukes, schistosomes), annelids (leaches) and acanthocephalans (thorny-headed worms). In the animal's body, the gastrointestinal tract is the abode of many helmets, but some live in tissues or their larvae migrates into tissues [2]. In ruminants, the most common cause of clinical helminthias is from infection with gastrointestinal nematodes of the order strongylida. The next most important parasite of sheep and cattle is the liver fluke. The major parasitic worm disease of horses is also generated by strongly round worms whose adult are blood suckers and whose larvae may cause colic, whereas their large scared nematode infections are rarely pathogenic. Fluke and tapeworms are rarely a cause for concern in equines. Pigs are infected by very large acaroids and by strangles but rarely suffer from pathogenic disease. Poultry and geese are susceptible to infection with an assortment of different helmets. Habitual parasites of domestic cats and dogs are intestinal tapeworms and scared nematodes. Dogs can suffer from pathogenic hookworm infections, particularly in warmer climates, where they may also be afflicted with heartworms [3].
Livestock producers have generally derived substantial benefits from the use of anthelmintics in controlling livestock parasitizes [4]. In the last 30 years, control of gastrointestinal nematode infections of ruminants has been achieved almost exclusively by use of pharmaceutically derived anthelmintics. Indeed, synthetic and semi-synthetically produced anthelmintics have for long been considered the only effective method of controlling helminthosis. However, in the extreme situations of subsistence farming where anthelmintics are either unavailable or unaffordable, massive mortalities of young stock are tragically commonplace in tropical Africa and Asia [5].
The use of medicinal plants for the prevention and treatment of gastro-intestinal parasitism has been used by livestock farmers and it has its origin in ethno veterinary medicine. For centuries, medicinal plants have been used to combat parasitism, and in many parts of the world are still used for this purpose (International Institute of Rural Reconstruction. For example, seeds of garlic, onion and mint have been used to treat animals that suffer from gastro-intestinal parasitism, whereas extracts of the tobacco plant have been used to treat the skin of livestock afflicted with external parasites [6]. Leaves, dried flowers and oil from Chenopodium ambrosioides, a shrub that originated from Central America and has been distributed around the world, have all been used as anthelmintics since the early 1900s [6]. Many plants have been screen for anthelmintic activities in recent times. Details will be discussed later.
By definition, anthelmintics are drugs that reduce parasite burdens in the animals to a tolerable level; they kill the parasites (vermicide), inhibit their growth or paralyse them (vermifuge). They also reduce the build-up of infective worm larva on the pasture, or eggs in the environment [2]. The "ideal" anthelmintic should have a broad spectrum of activity against mature and immature parasites (including hypo biotic larvae), be easy to administer to a large number of animals, have a wide margin of safety and be compatible with other compounds, not require long withholding periods because of residue(s), no unpleasant side effects and capable of economical integration and management system [3,7].
The era of modern anthelmintics started in the middle of the 20th century with the introduction of phenothiazine and piperazine, products that are considered to be the first generation of the broad spectrum drugs. The 2nd generation of truly broad spectrum anthelmintics were released in the 1960s and included the benzimidazoles, the probenzimidazoles, the imidazothiazoles and the tetra-hydro-pyrimidines. Following the early success of the introduction of the benzimidazoles, extensive research programmes were initiated during which successful structural modification resulted in the production of a series of benzimidazoles. Most recently, a 3rd generation of broad spectrum anthelmintics, the macrocyclic lactones, emerged in the early nineteen eighties. In addition, other compounds with a narrower spectrum have also been available on the market. These include substituted salicylanilides, phenols and organophosphates. Thus the pharmaceutical industry has, during the last 35 years, been able to produce a string of highly effective, broad and narrow spectrum anthelmintics, and veterinarians and livestock producers have used these extensively for parasite control either by drenching or injecting cattle, sheep and goats [2,7].
Classification of Anthelmintics
Anthelmintics are separated into classes on the basis of similar chemical structure and mode ofaction On the basis ofsimilar chemical structure, anthelmintics can be classified as: benzimidazoles, imidazolthiazoles, tetrahydropyrimidines, organophosphates, avermectins (macrolytic lactone), amino-acetonitrile derivatives (ADDs) [8]. Also, on the basis of the mode of action, anthelmintics can be classified as follow: Nicotin agonist, acetyl cholinesterase inhibitor, Gaba agonist, GluCl potentiator, calcium permeability increase, B-tubulin binding, proton ionophores, inhibitor of malate metabolism, inhibitor of phosphoglycerate kinase and mutase, and inhibitor of arachidonic acid [9]. Base on spectrum of action, anthelmintics can be classified as: broad spectrum (kill a wide variety of worms) or narrow spectrum (kill one or two varieties) [10].
Benzimidazoles
The benzimidazoles are the largest chemical family of anthelmintics used in domestic animals [2]. They are the first chemical class of modern anthelmintics developed. The first drug in this class is thiabendazole (TBZ) which was introduced in 1961 [8] and subsequently a number of further benzamidazoles were introduced as broad spectrum anthelmintics. They include albendazole, cabendazole, fenbendazole, flubendazole, mebendazole, oxfendazole, oxibendazole, parbendazole and triclabendazole. It is clear that their anthelmintic efficacy is due to their ability to compromise the cytoskeleton through a selective interaction with b-tubulin [11,12].
The benzimidazole compound can be grouped as follows:
a. Benzimidazole thiazolyls: Eg thiabendazole,cambendazole.
b. Benzimidazole methylcarbamates: which include: Parbendazole, mebendazole, Flubendazole, Oxibendazole, luxabendazole, albendazole, albendazole sulphoxide (ricobendazole), fenbendazole, oxfendazole.
c. Halogenated benzimidazole thiols: Example is Triclabendazole.
d. Pro-benzimidazoles: thiophanate, febantel and nitobimin.
Mechanisms of action
The benzimidazole and pro-benzimidazole bind to a specific building block called beta tubulin and prevent its incorporation into certain cellular structures called microbutbules, which are essential for energy metabolism. This binding causes disruption of the tubulin microtubule dynamic equilibrium [12]. It is important to note that microtubules are found in animals, plants and fungi cells. However, the rate constant for the dissociation of benzimidazone from parasite tubulin is much lower than the rate constant for the dissociation from mammalian tubulin. These differences in dissociation rate between benzimidazole and tubulin in host and parasites explain the selective toxicity of benzimidazole compound to parasites and its wide safety margin in the mammalian host.
Chemistry
Benzimidazole is a heterocyclic aromatic organic compound. In its structure, it is a bicyclic ring system in which benzene has been fused to the 4 and 5 positions of the heterocycle (Imidazole). It is an important pharmacophore and a privileged structure in medicinal chemistry. Most benzimidazole compounds are white crystalline powders, with fairly high melting point and which are insoluble or slightly soluble in water. Their aqueous solubility is markedly higher at low acidic pH values with the stomach/abomasums being the appropriate site for the dissolution of benzimidazole drugs particles after oral treatment [13].
The most prominent benzimidazole compound in nature is N-ribosyl-dimethylbenzimidazole, which serves as an axial ligand for cobalt in vitamin B12.1 [14]. The use of Benzimidazole dates many years back [15]. In 1990 various benzimidazole derivatives were synthesized with substitution of fluorine, propylene, tetrahydroquinoline and cyclised compound which resulted in compounds with increased stability, bioavailability and significant biological activity [16]. It was also showed that substitution on pyridine by electron donating group increases activity. In 1991 benzimidazole derivatives were synthesized by derivatization at N-H of benzimidazole by electron donating group and substitution with long chain of propyl, acetamido, thio, thiazole-amino, tetramethyl piperidine on pyridine resulting in good antiulcer activity [17].
Pharmacokinetics of benzimidazole
The drugs are generally administered orally. In the ruminants the rumen acts as a drug reservoir by slowing the digesta transit time throughout the abomasums, which results in improved systemic availability of the drugs compounds as a consequence of a greater dissolution of drug particles in the acid pH of the abomasums [18]. Benzimidazole anthelmintics are extensively metabolised in all mammalian species. The parent drug is usually short-lived and the metabolic products predominate in the plasma, all tissues, in the excreta of the host and in the parasites recovered from the treated animals [19]. The primary metabolites usually are products of oxidative and hydrolytic processes and are all more polar and water soluble than the parent drug. The metabolites are conjugated with glucoronide and/or sulphate to increase their polarities, which facilitates urinary and biliary excretion [14].
Once benzimidazole molecule has been absorbed from the gastrointestinal tract, it is rapidly distributed by the circulatory system throughout the entire body. During this process, the metabolic process necessary to facilitate its elimination commences [20]. The drugs transfer into the target parasites through transcuticular diffusion. This is usually the predominant pathway through which the drugs reach the nematodes. It is however worthy of note that the external surface of the nematode (curticle) and that of the cestodes and trematodes (tegument), influences the mechanism of entry of this drug. The mechanism of drugs entry to types of helminths seems to be dependent on the lipophilicity as a major phytochemical determinant of drug capability to reach therapeutic concentrations within the target parasites [21].
Anthelmintic spectrum of benzimidazole
The benzimidazoles are effective against a wide spectrum of nematodes. They are most effective against larval and adult round worms. Some such as thiabendazole, febantel, fenbendazole, oxfendazole and oxibendazole are ovicidal [2]. On a general note, the benzimidazole methylcarbamates are broad spectrum anthelmintics active against a wide varieties of gastrointestinal and lung nematodes, tapeworms, and trematodes [14]. The benzimidazole anthelmintics are virtually insoluble in water, which limits most of their formulations to suspension, paste, granule, tablets, blocks, powder and pellets for oral or intraruminal administration, or for administration in feed. Drench formulations are most frequently used in ruminant species, pastes are often preferred for horses, tablets for dogs and cats, and powder in feed administration for swine and poutry [14]. Some members of the group such as: parbendazole, cambendazole, oxfendazole, fenbendazole, and albendazole have been reported to be teratogenic at approximately four times their recommended doses; this limits their use in early stage of pregnancy, and sheep seem to be especially sensitive compared to other animals [22] (Tablel).
Table 1:The table below summarises the activities of the various members of the benzimidazole anthelmintics.
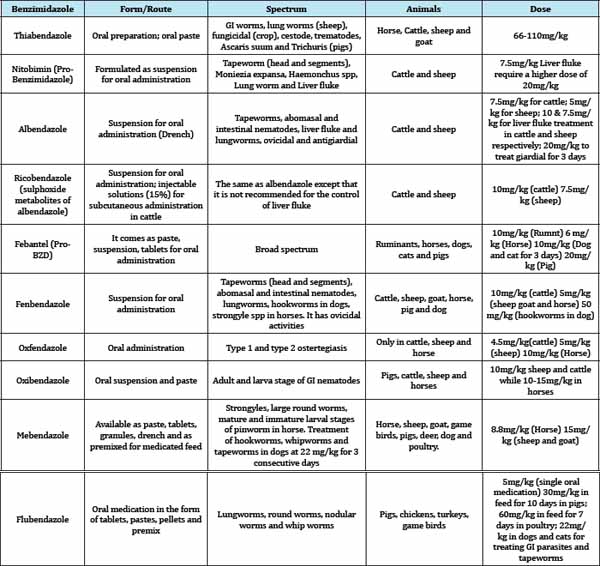
Imidazothiazoles
Tetramisole is the first imidazothiazole anthelmintics which was introduce into the veterinary market in 1967. However, the current and the most available imidazothiazole anthelmintic worldwide is the levamisole [13]. The other compound available is the butamisole which is a deravative of levamisole [2].
Chemistry
Tetramisole is a mixture of two optically active isomers, of which the laevorotatory (L) isomer, levamisole, was responsible for its efficacy against nematodes. The corresponding dextrarotatory (D) isomer (dexamisole) had little anthelmintic efficacy and its safety profile was no better than levamisole Brander et al. [3]. Thus, it was determined that the dosage could be reduced by one half using the L- isomer alone. Reducing the dosage in this way also appreciably increased the margin of safety, while leaving the anthelmintic potency unchanged [23].
Mode of action
Levamisole acts selectively as a cholinergic agonist [24,25] at the synaptic and extra synaptic nicotinic acetylcholine receptors on nematode muscle cells. This cause spastic paralysis of susceptible nematode by selectively gating acetylcholine receptor ion channels on nerve and muscle [26]. It is also been shown to inhibits the enzyme fumigate reductive but this is unlikely to be its mode of action in vivo [3].
Pharmacokinetics
Absorption rate and bioavailability of the drugs in this group depend on the route of administration. The drug is most rapidly absorbed following intramuscular or subcutaneous injection in cattle. The drug is widely distributed in the organism being more recovered in the tissues such as muscle, fat, kidney and particularly liver at two hour post oral and subcute administration [14]. The drug is rapidly and extensively metabolised in the liver through oxidation, hydrolysis and hydroxylation. It is rapidly eliminated from the body in urine and faeces within 24 hours and consequently has a short withdrawal period of 3 days for meat and 1 day for milk [3].
Spectrum of anthelmintic activity
Tetramisole, butamisole and levamisole are broad spectrum synthetic imidazothiazoles derivatives which are nicotinic like. They are effective against adult and larval nematodes of the gastrointestinal tract, heart, lungs, and kidney [2]. Levamisole is approved and marketed worldwide for use in cattle, sheep, swine, poultry, and dogs. It is however not generally used in horses because of its limited efficacy against many equine parasites. Levamisole is a nematodicidal compound effective against lung and GI tract nematodes but not effective against cestode and trematode parasites, and it is not ovicidal. One major advantage of levamisole is its formulation flexibility which allows for alternative routes of drug administration such as oral, parenteral and topical. More so, depending upon formulation, the drug is marketed as levamisole hydrochloride or phosphate salts (oral drench, feed premix, and injectable preparations) and levamisole base Ie pours on [14].
Levamisole have been shown to have immune modulatory effects, that is; it enhances immune responsiveness by restoring the number of T lymphocytes to normal when they are depleted. This characteristic of the drug has been used in man [27] and to limited animals [28] in several diseases.
Drug dosage and route of administration
Levamisole is administered as tablet, solution, oral drench, feed additive, subcutaneous injectable solution or topical pour on. The recommended dose is 8mg/kg in cattle, sheep, goats and pigs as single oral or subcutaneous dose or 10mg/kg for pour-on administration for cattle. Administration of levamisole in drinking water is used routinely in swine and poultry. However, injectable levamisole is preferred in cattle. The drug is approved for use in dogs and cats in some countries at the rate of 5 and 10mg/kg as tablets or oral solutions [14].
Toxicity
Mammalian toxicity with levamisole or especially with tetramisole is usually greater than for the benzimidazoles, although toxic signs are unusual unless the normal therapeutic dosage is exceeded. Levamisole toxicity in the host animal is largely an extension of its antiparasitic effect, i.e. cholinergic-type signs of salivation, muscle tremors, ataxia, urination, defecation, and collapse. In fatal levamisole poisoning, the immediate cause of death is asphyxia due to respiratory failure. Atropine sulfate can alleviate such signs. Levamisole may cause some inflammation at the site of SC injection, but usually this is transient [29]. The drug is neither embryo toxic nor teratogenic [30].
Tetrahydropyrimidines
Members of this group of anthelmintics are: pyrantel, morantel and oxantel. Pyrantel is the first compound within the tetrahydropyrimidine family introduced in 1966 as broad spectrum anthelmintic to treat gastrointestinal nematodes in sheep and thereafter, it was developed for use in cattle, swine, horses, dogs and cats. Later, pyrantel methyl esters called morantel; as well as the metaxyphenol analogue called oxantel were introduced as nematocidal compounds [23].
Chemistry
Pyrantel is prepared for use as pyrantel tartrate or pyrantel pamoate (embonate) and citrate salts. Pyrantel pamoate is insoluble in water and alcohol while the tartrate salt is more water soluble. Morantel is mainly formulated as tartrate salt. Aqueous solutions are subject to isomerization on exposure to light, with a resultant loss in potency; therefore, suspensions should be kept out of direct sunlight [14,23].
Mode of action
These drugs act selectively as agonist at synaptic and post synaptic nicotinic acetylcholine receptors on nematode muscle cells and produce contraction and spastic paralysis. Pyrantel and morantel are 100 times more potent than acetylcholine, although slower in initiating contraction [10,14].
Pharmacokinetics
Pyrantel tartrate is absorbed more readily than the pamoate salt. Pyrantel tartrate is well absorbed by pigs and dogs, less well by ruminants. Metabolism is rapid, and the metabolites are excreted rapidly in the urine (40% of the dose in dogs); some unchanged drug is excreted in the feces (principally in ruminants). Blood levels usually peak 4-6 hr after administration PO. The pamoate salt of pyrantel is poorly soluble in water as stated earlier; this offers the advantage of reduced absorption from the gut and allows the drug to reach and be effective against parasites in the lower end of the large intestine, which makes it useful in horses and dogs [7].
Drug formulation and administration
Pyrantel is used orally as a suspension, paste, drench, or tablets as well as by mixing it in feed. Regardless of the method of administration, a dose of 6.6mg pyrantel base/kg should be used. A powdered premix formulation containing 10.6% pyrantel tartrate is available for treating parasitic infection in swine via medicated feed. Overnight fasting is advised. Water should be available ad libitum during the fasting and treatment periods. In dogs pyrantel pamoate is also combined with febantel and praziquantel in a table form which is given to dog as a single dose. A sustained-release ruminal bolus for use in cattle, which releases the morantel over 90 days, has been developed. No withholding period (at least in some countries) is required for the biodegradable morantel bolus for cattle [14].
Spectrum of anthelmintic activity
It is effective against ascarids, large and small strongyles, and pinworms. Oxantel is combined with pyrantel in some anthelmintic preparations for dogs (and man) to increase activity against whipworms. Morantel in ruminants tends to be somewhat safer and more effective than pyrantel. Both pyrantel and morantel have a higher efficacy against adult gut worms and larval stages that dwell in the lumen or on the mucosal surface than against the stages found in the mucosa such as arrested Ostertagia larvae [8].
Organophosphate Compounds
The organophosphate anthelmintics were discovered in the 1950s, before the advent of the benzimidazoles and at that time, they show promise as potential broad spectrum parasicides, effective against both insects and nematodes; although their spectrum of activity against nematodes is fairly restricted [3]. Drugs in this group include: Haloxon, coumaphos, dichlorvos, crufomate and naphthalophos [2]. A number of organophosphates (OP) have been used as anthelmintics. Originally, they were used extensively as insecticides, then as ectoparasiticides. Dichlorvos is used as an anthelmintic in horses, pigs, dogs, and cats; trichlorfon in horses and dogs; and coumaphos, crufomate, haloxon, and naftalofos in ruminants. Haloxon is probably the safest organophosphate anthelmintics for use in ruminants [8].
Mode of action
Organophosphate inhibits many enzymes, especially acetyl cholinesterase, by phosphorylating their esterification site. This blocks cholinergic nerve transmission in the parasite, which results in spastic paralysis. The cholinesterase's of host and parasite and those of different species of parasites vary in their susceptibility to Organophosphates. Such variations are considered in development and production of Organophosphate. The susceptibility of cholinesterase enzymes to the Orgonophosphate, the rate at which the inhibition can be reversed, and the rates of inactivation of the various Organophosphates in the host animal largely determine the relative toxicity to different animals [10].
Spectrum of anthelmintic activity
Organophosphate tends to be labile to varying extents in alkaline media and may be partially hydrolyzed and inactivated in the alkaline region of the small intestine. For example, the oral dose rate for trichlorfon in cattle is 4.5 times the subcutaneous dose. In ruminants, organophosphates generally have satisfactory efficacy for nematode parasites of the abomasums (especially Haemonchus spp) and small intestine but lack satisfactory efficacy for parasites of the large intestine. Dichlorvos is particularly useful in pigs against all major parasites and was one of the first broad-spectrum anthelmintics to be used in this species. It is also effective against helminths and bots in horses. Trichlorfon (metrifonate) is used in horses because of its high degree of activity against bots, ascarids and oxyurids. It also has been used in ruminants and small animals. As with other organophosphate, trichlorfon tends to have a narrow margin of safety. Dichlorvos and trichlorfon are particularly effective against bots and ascarids and are effective against a broad spectrum of other GIT helminths also.
Metabolism
Organophosphate usually are rapidly oxidized and inactivated in the liver. Their margin of safety is generally less than that of the benzimidazoles, and strict attention to dosage is necessary. Because of its high volatility, dichlorvos is a particularly versatile organophosphate that can be incorporated as a plasticizer in vinyl resin pellets; it is released slowly from the inert pellets as they pass through the gi tract, which provides a therapeutic concentration along the tract. This controlled release governs the concentration available to the host as well as to the parasites and thereby increases the safety margin. When passed in the feces, the pellets still contain 45-50% of the original drug. Dichlorvos is rapidly absorbed and metabolized in the body.
Safety and toxicity
Certain precautions should be followed when using organophosphate. Generally, their toxicity is additive; thus, concurrent use of other cholinesterase-inhibiting drugs should be avoided. Atrorganophosphateine and 2-pam are used as antidotes to organophosphate toxicity. Organophosphate can be hazardous to man. Being lipid soluble, they are absorbed well through unbroken skin. They also have the prorganophosphateensity to interact with many other drugs. Organophosphate. Are relatively rapidly degraded in the body, and tissue residues are unlikely to pose a serious consumer hazard if specified withholding periods are followed. Perhaps the major risk arises from contamination of the environment through fecal excretion or accidental drug spillage [14,30].
Heterocyclic Compounds
The main drug in this group is the phenothiazine which was the first anthelmintic to demonstrate fairly wide range of activity against gastrointestinal nematodes in sheep, cattle, goats, horses and cattle since 1938. However, due to its toxicity, its use was limited in dogs, cats and humans. More so, there is development of some phenothiazine resistant strain since 1960s as well as competition from other broad-spectrum drugs. This has reduced the use of this compound as anthelmintics drug over the years [31].
Piperazine (Diethylenediamine)
Piperazine was discovered in 1900, and its anthelmintic moiety was discovered in 1954. The drug has good efficacy profiles against ascarid and nodular worm infections of all species of domestic animals, moderate for pinworm infections, and zero to variable to other veterinary helminths. Its use is limited in ruminants because ascarids are not a significant problem in this species. Its low cost and wide safety margin in domestic animals have been determinant for its extensive worldwide use in antiparasitic therapy [14]. Piperazine is available as hexahydrate and a variety of salts such as citrate, phosphate, tartrate or hydrochloride [3].
Mode of action
Piperazine acts by blocking transmission by hyperpolarizing nerve membranes at the neuromuscular junction leading to parasite immobilization by flaccid paralysis and consequent removal from predilection site and death. Recent studies [10,32] demonstrate that piperazine is a selective agonist of gamma-amino butyric acid (GABA) receptor resulting in opening of chloride channels and hyperpolarization of the membrane of the muscle cells of the nematode parasites. Mature worms are more susceptible to the action of piperazine than the younger stages. Immature adults and lumen dwelling larvae are sufficiently susceptible to be at least partially eliminated. Larval stages in host tissues, however, are relatively insusceptible. Because of subsequent larval development, repeated treatments are generally indicated within 2 weeks for carnivores and within four weeks for swine and horses [14].
Pharmacokinetics
The recommended dose in horses, swine and ruminants is 110mg/kg while that for dogs and cats is 45-60mg/kg. The drug is readily absorbed through the GIT and then extensively metabolized (60-70%). The remaining parent molecule is eliminated in the urine over 24 hours [33].
Diethylcarbamazine Citrate (Dec)
This drug is derived from piperazine and it is highly soluble in water, alcohol, and chloroform, but insoluble in organic solvents. The drug is used as preventive for heartworm disease in dog and previously used in the treatment of lungworm infection in ruminants. It is formulated as tablets or chewable that can be administered to dog every day especially during the mosquito vector season [14].
Organic Arsenicals
This class of antihelmintics are used principally in the treatment of canine heatworm caused by Dirofilaria immitis. They are the approved drugs used to treat the adult worm that lodge in the pulmonary artery and the right side of the heart. The drug include: Thiacetarsamide sodium and Melarsomine. They are not effective against microfilaria. Cautions need to be taken when administering the drugs since they are toxic at high doses [34].
Treatment of Hookworm
The drug commonly used for the treatment of hookworm (Ancylostoma caninum) is called Bephenium. It is a nicotine-like quaternary ammonium compound used mainly in dogs and cats. It is also effective against Toxocara and Trichuris to a lesser extent. It is poorly absorbed by the host and it has low toxicity. An analogue of bephenium which is strictly used in dog to control hookworm is Thenium closylate. It is 98% effective against adult and immature stages of Ancylostoma caninum but moderately effective against ascarids [3].
Anticestodal Drugs
Anticestodal drugs are used treat tapeworm infections. Tapeworm typically have a flat, segmented body and attaché to the host's intestine. Anticestodal drugs may be taeniafuges (which facilitate tapeworm expulsion from the host) or taeniacides (which cause death of the tapeworm in situ). Tapeworm infection in farm animals may be a minor problem and usually does not require treatment with specific anticestodal drug. Some of the broad spectrum benzimidazole compounds used to control nematode infections is also effective against tapeworms [14]. Treatment of tapeworms is often necessary in order to prevent disease due to larval stages in farm animals; to minimise meat inspection losses, and for aesthetic reasons in dogs and cats [3]. Some of the anticestodal drugs include.
Bunamidine
It is effective against the common tapeworm species and it is most effective when given after fasting. It is formulated as tablets (hydrochloride salt) or suspension (hydroxynaftoate salt) for oral administration in companion animals and ruminants, respectively. It acts by disrupting the tapeworm's tegument and reduces glucose intake, as a result, the subtegumental tissues are exposed and the worm destroyed by the host's digestive enzymes.
Niclosamide
This drug is highly effective against most tapeworms of dogs and cats though it has poor efficacy against Dapylidium spp and Echinococcus granulosus. It is also effective against common ruminant's tapeworms such as Moniezia expansa and Thysanosoma spp. In horse, it is used to treat against Anoplocephala spp. The drug acts by interfering with glucose absorption and oxidative phosphorylation which leads to the death of the parasites and their subsequent digestion within the gut. The drug is available as suspension for oral or intraruminal administration in ruminants and as tablets for oral treatment in companion animals [14].
Praziquantel
Praziquantel has high efficacy against cestods and schistosome infections of all species including humans [35]. It is also effective against most trematodes though its efficacy against Fasciola hepatical or hydatid cyst in humans is erratic. The drug is a synthetic isoquinoline-pyrazine derivative. The drug acts by inducing a rapid and sustained paralytic muscle contraction of the parasites and regimental disruption. This is followed by the exposure of the parasite antigens, binding, and penetration of host immune cells into the parasite [10]. Praziquantel also act by affecting the metabolic processes of the parasites. Metabolic changes include decrease in glucose uptake, glycogen storage, ATP content, and lactate release. All these effects are attributed either directly or indirectly to an alteration of intracellular calcium homeostasis [36]. The drug is available as tablets, paste and suspension for oral administration, and as solution for SC and IM injections [14].
The drug is absorbed orally, 80% bound to plasma protein, undergoes extensive first pass metabolism in the liver to many inactive products and it is distributed throughout the body and excreted in the urine [37]. The analogue of praziquantel is Epsiprantel which is highly effective against Echinococcos granulosus.
Antitrematodal Drugs
Disease (Fasciolosis) caused by trematodes (e.g. Fasciola hepatica) is the most common and economical important disease in domestical animals worldwide. Fasciolosis is an important zoonotic disease [38]. These leaf-shaped flatworms are generally characterised by the tissues they infect such as the liver, lung, intestine and blood [39]. The intermediate host is a mud snail called Lymnea truncatula. Some of the drugs discussed earlier are also effective against trematodes (flucks), but they lack wide therapeutic index. Examples include:
a. Benzimidazoles (albendazole, netobimin, triclabendazole).
b. Salicylanilides (brotianide, clioxanide, closantel, niclosamide, oxyclozanide, rafoxanide) and brominated salicylanilides (bromosalans).
c. Substituted phenols or nitrophenolic compounds (bithionol, disophenol, hexachlorophene, niclofolan, nitroxynil).
d. Aromatic amide or phenoxyalkanes (diamphenethide).
e. Bencenesulfonamides or sulphonamide (clorsulon).
f. Halogenate hydrocarbons (carbon tetrachloride, hexachloroethane, tetrachlorodifluoroethane, hexachloroparaxylene).
g. Bisphenolic compounds (hexachlorophene, bithionol).
Nitroxynil
It is effective against adult stages of Fasciola hepatica i.e. 8 weeks post infection and F. gigantica. It is not effective for the treatment of flukes younger than 6 weeks. It is also used to control Hemonchus contortus in sheep, Oesophagostomum species, Parafilaria bovicola and Bunostomum species in both sheep and cattle. Nitroxynil acts by producing a rapid spastic paralysis of the flukes [40,41] causes severe disruption of the tegument of Fasciola hepatica [39]. Nitroxynil is available as oral, intraruminal, subcutaneous, intramuscular preparations. The subcutaneous route has become the method of choice in practice.
Closantel
This drug in addition to treating flukes is effective against nematodes such as Hemonchus contortus, Oesophagostomum spp, Bunostomum species and Ostertagia species in both sheep and cattle. It is also effective against certain ectoparasites such as blood-sucking lice, ticks, mites, and certain grubs of rminants. It is used to treat nasal bot (Oestrus ovis) in sheep, adult stage of Ancylostoma caninum in dogs, Stroggylis vulgaris and Gasterophilus species in horse [14]. Closantel acts as uncouplers of the oxidative phosphorylatrion in the liver flukes. These results in metabolic changes such as increase in glucose uptake decrease in glycogen content, changes in respiratory intermediates and decrease in ATP synthesis [41].
Rafoxanide
Rafoxanide [42] was developed in 1969 and has been used extensively against fasciolosis and haemonchosis, bunostomosis in sheep and cattle as well as nasal but in sheep. The mode of action of rafoxanide is similar to that of closantel discussed above. The drug is available bolus or as a drenching suspension, which may also be used as intraruminal administration.
Oxyclozanide
This drug mode of action is similar to closantel. It is effective against flukes, Moniezia species (tapeworms) in sheep and cattle. It is available as oral drench (aqueous suspension) containing oxycloxanide only or in combination with levamisole hydrochloride or oxfendazole. It is also formulated as powder to be incorporated into feed.
Clorsulon
Clorsulon is highly effective against adult liver flukes in sheep and cattle. It is however more effective in cattle than in sheep and goats. The combination of clorsulon with ivermectin in a subcutaneous injectable formulation was designed for simultaneous treatment of Fasciola hepatica and nematode infections of cattle. Clorsulon is marketed as an oral drench for sheep and cattle and as an injectable formulation in which is combined with ivermectin for SC administration in cattle. The recommended dosage in cattle and sheep is 7mg/kg [43].
Benzimidazoles
These have already been discussed under drugs used against nematodes, but this is to stress that the benzimidazoles are broad- spectrum anthelmintics and are also effective against flukes. The chief flukicides among this group is the triclbendazole which holds and excellent efficacy against the adult and juvenile stages of Fasciola hepatica even down to one week old flukes [44]. Other drugs in this group used to treat flukes include: Albendazole, netobimin, and luxabendazole [3] (Table 2).
Table 2:The table below summarises the activities of the various members of the benzimidazole anthelmintics.
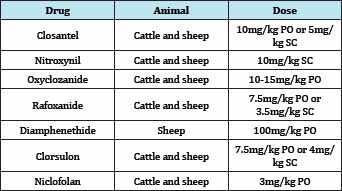
Macrocyclic Lactones (Endectocide)
The macrocyclic lactones (MLs) are now considered the most widely used broad-spectrum antiparasitic drugs in veterinary medicine. They possess unique features such as exceptional potency, high lipophilicity, and prolonged persistence of their potent broad-spectrum activity [14]. The avermectins and milbemycins are the macrolides produced through fermentation by soil dwelling actinomycetes called streptomyces. It is a unique combination which kills bothe endo and ectoparasites thus, giving it the name "edectocides" by which macrocyclic lactones are now regognised [45]. The avermectin's family includes a series of natural and semisynthetic molecules, such as abamectin, ivermectin, doramectin, eprinomectin, and selamectin. Abamectin is the naturally occurring avermectin approved for animal use and the starting material for the production of ivermectin. In fact, ivermectin, the first marketed endectocide molecule, is semisynthetic derivative of the avermectin family [46].
Doramectin was obtained by mutational biosynthesis in which the precursor called
Cyclohexane carboxylic acid was fed to a mutant strain of streptomyces avermitilis [47]. Eprinomectin is a
semisynthetic avermectin compound, developed for topical use in cattle [45]. Selamentin is a
semisynthetic monosaccaride oxime derivative of doramectin more recently introduced into the market
for parasite control in dogs and cats.
Meadectin, moxidectin, and milbemycin oxime belong to the milbemycin family. Moxidectin is a milbemycin compound produced by a combination of fermentation and chemical synthesis i. e. the chemical modification of nemadectin, the natural compound produced when streptomyces cyaneogriseus is grown under controlled culture conditions [48].
Various compounds from both families are used in livestock, companion animals, wildlife species, and humans. Ivermectin, doramectrin, and moxidectin are currently marketed as inject able, pour-on (cattle), and oral (sheep and goat) formulations. They are the most commonly used MLs worldwide to control endo- and ectoparasites in livestock's [45].
Mode of action
The macrolides act through selective toxic effects on insects, acarines, and nematodes. However, they do not possess efficacy against cestode and trematode parasites. The macrocyclic lactones induce reduction in motor activity and paralysis in both arthropods and nematodes. The parasitic effects are mediated through GABA and/or glutamate-gated chloride channels (GluCl), collectively known as ligand-gated chloride channels [49].
The newly proposed mechanism of action of the MLs is hyperpolarization, and flaccid paralysis of the invertebrate somatic muscles [50,51]. The endectocides cause paralysis and death of both arthropod and nematode parasites due to their paralytic effects on the pharyngeal pump which affects nutrient ingestion, and on the parasite somatic musculature limiting its ability to remain at the site of predilection in the host. In addition, MLs cause inhibitory effects on the female reproductive system and cause the reductions in parasite egg production [52].
Plants Used to Used as Anthelmintics
Hundreds of plants have been screened against helminths, and many of them have been shown to possess anthelmintic activities. Though, many of these plants have not been approved for global and commercial use, traditional livestock farmers are exploring these plants to their benefits. The plants used to treat helminthosis in man and in animals have been reviewed. Some of the plants include:
1. Minusop elengi stem bark.
2. Moghania vestita, roots and peels.
3. Melia azedarach; drupes.
4. Calotropis rocera, latex.
5. Punica granatum, root and stem bark.
6. Capparis deciduas, root bark.
7. Butea monosperma, seed.
8. Neolamarckia cadamba, stem bark.
9. Xylopia aethiopica, seed.
10. Gyandropsis gynandra, leaves and stem bark.
11. Evolvulus alsinoides, whole plant.
12. Carica papaya, seeds.
13. Semecarpus anacardium, nut.
14. Piper longum, essential oil.
15. Centratherum anthelminticum, seed.
16. Nigella sativa, essential oil.
17. Commiphora mukul, essential oil.
18. Trachyspermum ammi, seed.
19. Cucurbita maxima, seed.
20. Nicotiana tabacum, leaves.
21. Cannabis sativa, leaves
22. Ficus insipida, latex.
23. Trifolium repens, aerial shoot.
24. Cleome icosandra, leaves.
25. Strobilanthes discolour leaves.
26. Piper betle, essential oil.
27. Anacardium occidentalis, essential oil.
28. Callistemon viminalis.
29. Sapindus trifoliatus.
30. Momordica spp.
Other naturally occurring anthelmintics are: tobbacco, walnut, wormwood, clove, kalonji seeds, Garlic, Malefern, Pineapple, Diatomaceous earth, Soya and other legumes, Honey, water and vinegar are mixed with warm water act as Vermifuges. Most of the plants screened for anthelmintic activities reported are in vitro studies and very few in vivo studies. Worm samples often used are the Indian earth worm (Pheretima posthuma), Ascaridia galli, Safaris lubricious, Heligmoisodes polygyrus and Nippostrongyllus brasiliensis.
Conclusion
The use of anthelmintics still remained the effective method of treating and controlling helminthosis in man and animals. Appropriate use of these drugs for maximum efficacy is therefore advocated. Plants possessing anthelmintic activities should be further investigated so as to provide alternative drugs for the treatment of helminthosis in animals especially with this advent of anthelmintic drug resistance in livestock production.
References
- Prichard R (1994) Anthelmintic resistance. Vet Parasitol 54(1-3): 259268.
- Aliu YO (2006) Veterinary Pharmacology. (1st edn), Tamaza publishing company, Nigeria, pp: 467-482.
- Brande GC, Pugh DM, Bywater RJ, Jenkins WL (1991) Veterinary applied Pharmacology and Therapeutics. Education low price scheme, pp. 513549.
- Abdu PA, Jagun AG, Gefu JO, Mohammed AK, Alawa CBI, et al. (2000) A survey of ethno veterinary practices of agropastoralists in Nigeria. Ethnoveterinary practices, research and development, Proceedings of the International Workshop on Ethnoveterinary Practices, Kaduna, Nigeria pp. 14-18
- Meyers GH (1991) Parasite control importance in grazing beef cattle. Feedstuffs 63: 14.
- Guarrera MP (1999) Traditional antihelmintic, antiparasitic and repellant uses of plants in central Italy. J Ethnopharmacol 68(1-3): 183192.
- Aiello SE (2000) The Veterinary manual. (8th edn). Merck and Co, INC, White house station, NJ, USA.
- Schoenian S (2008) Small ruminant info sheet. University of Maryland extension, USA.
- Martin RJ (1997) Mode of action of Anthelmintic drugs. Vet J 154(1): 11-34.
- Sangster NC (2008) Anthelmintic resistance: past, present and future. Int J Parasitol 29: 115-124
- Borgers M, De Nollin S (1975) Ultrastructural changes in Ascaris suum intestine after mebendazole treatment in vivo. J Parasitol 6(1): 110-122.
- Lacey E (1990) Mode of action of benzimidazoles. Parasitology Today 6(4): 112-115.
- Riviere RE, Papich MG (2009) Veterinary Pharmacology and Therapeutics. (9th edn). Wiley-Black, pp. 1053-1145.
- Barker HA, Smyth RD, Weissbach H, Toohey JI, Ladd JN, et al. (1960) Isolation and properties of crystalline cobamide coenzymes containing Benzimidazole or 5,6- Dimethylbenzimidazole. J Biol Chem 235: 480488.
- Patil A, Ganguly S, Surana SA (2008) Systematic review of benzimidazole derivatives as an antiulcer agent. Rasayan J Chem 1(3): 447-460.
- Kubo K, Oda K, Kaneko T, Satoh H, Nohara A, et al. (1990) Synthesis of 2-(4- Fluoroalkoxy-2-pyridyl) methyl sulfinyl-1H-benzimidazoles as Antiulcer Agents. Chem Pharm Bull 38(10): 2853-2858.
- Ozkay Y, Tunali Y, Karaca H, Isikdag I (2010) Antimicrobial activity and a SAR study of some novel benzimidazole derivatives bearing hydrazones moiety. Eur J Med Chem 45(8): 3293-3298.
- Lanusse CE, Prichard RK (1993) Relationship between pharmacological properties and clinical efficacy of ruminant anthelmintics. Vet Parasito 49(2-4): 123-158.
- Alvarez L, Imperiale F, Sanchez S, Lanusse C (2000) Uptake of albendazole and albendazole sulphoxide by Haemonchus contortus and Fasciola hepatica in sheep. Vet Parasito 94(1-2): 75-89.
- Alvarez L, Sanchez S, Lanusse C (1999) In vivo and ex vivo uptake of albendazole and its sulphoxide metabolite by cestode parasites: relationship with their kinetic behavior in sheep. J Vet Pharmacol Ther 22(2): 77-86.
- Cross H, Renz A, Trees A (1998) In vivo uptake if ivermectin by adult male onchocerca ochengi. Ann Trop Med Parasitol 92(6): 711-720.
- McKellar Q, Harrison P, Galbraith E, Inglis H (1990) Pharmacokinetics of fenbendazole in dogs. J Vet Pharmacol Ther 13(4): 386-392.
- Barragry TB (1994) Veterinary drug therapy. Lea & Febiger Philadelphia, USA, pp. 83-115.
- Aceves J, Erlij D, Martinez-Maranon R (1970) The mechanism of the paralyzing action of tetramisole on Ascaris somatic muscle. Br J Pharmacol 38(3): 602-607.
- Aubry ML, Cowell P, Davey MJ, Shevde S (1970) Aspects of the pharmacology of a new anthelmintic: pyrantel. Br J Pharmacol 38(2): 332-344.
- Robertson SJ, Martin RJ (1993) Levamisole-activated single-channel currents from muscle of the nematode parasite Ascaris suum. Br J Pharmacol ogy 108(1): 70-178.
- Koller LD (1992) Chemical-induced immunomodulation. Journal of the American Veterinary Medical Association 181: 1102-1106.
- Desplenter L, McDonald LE (1983) Levamisole as an immunomodulator in the prevention of neonatal disease. In Veterinary Pharmacology and Toxicology, MTP Press Limited, USA, pp. 99-103.
- Reinemeyer C, Courtney C, Adams HR, Ames IA (2001) Antinematodal drugs. In Veterinary Pharmacology and Therapeutics, 8th (edn), Iowa State University Press, USA, pp. 947-979.
- Harder A (2000) Chemotherapeutic approaches t nematodes: current knowledge and outlook. Parasitology Research 88: 272-277.
- Jacobs DE (1987) Anthelmintics for dogs and cats. International Journal for Parasitology 17(2): 511-518.
- Tarello W (2001) Chronic fatigue syndrome in 15 dogs and cats with specific biochemical and microbiological anomalies. Comp Immunol Microbiol Infect Dis 24(3): 165-185.
- Thomas H, Gonnert R (1978) The efficacy of praziquantel against cestodes in cats, dogs and sheep. Research in Veterinary Science 24(1): 20-25.
- Day T, Bennett J, Pax R (1992) Praziquantel: The enigmatic antiparasitic. Parasitology Today 8(10): 342-344.
- Giorgi M, Meucci E, Vacarro E, Mengozzi G, Giusiani M, et al. (2003) Effects of liquid and freeze dried grape fruit juice on the pharmacokinetics of praziquantel and its metabolites 4-hydroxy praziquantel in beagle dogs. Pharmacological Research 47(1): 87-92.
- Mas-Coma S, Cotruvo JA, Dufour A, Rees G (2004) Human fascioliasis. In Waterborne zoonoses: Identification causes and control, World Health Organisation/IWA publishing, Switzerland, pp. 305-322.
- McKinstry B, Fairweather I, Brennan G, Forbes A (2003) Fasciola hepatica tegumental surface alterations following treatment in vivo in in vitro with nitroxynil (Trodax). Parasitology Research 91(3): 251-263.
- Fairwather I, Boray J (1999) Fasciollicides: Efficacy actions, resistance and its management. Vet J 158(2): 227-234.
- Alvinerie M, Floc'h R, Galtier P (1999) Plasma protein binding of nitroxynil in several species. J Vet Pharmacol Ther 14(2): 170-173.
- Swan G, Mulders M (1993) Pharmacokinetics of rafoxanide in suckling and weaned lambs following oral administration. J S Afr Vet Assoc 64(2): 67-70.
- Sundlof S, Whitlock T (1992) Clorsulon pharmacokinetics in sheep and goats following oral and intravenous administration. J Vet Pharmacol Ther 15(3): 282-291.
- Boray J, Crowfoot P, Strong M, Allison J, Schellenbaum M, et al. (1983) Treatment of immature and mature Fasciola hepatica infections in sheep with triclabendazole. Vet Rec 113(14): 315-317.
- Shoop W, Mrozik H, Fisher M (1995) Structure and activity of avermectins and milbemycins in animal health. Vet Parasitol 59(2): 139-156.
- Fisher M, Mruzik H, Campbell W (1989) Chemistry of Ivermectin, New York, USA, pp. 1-23.
- Bishop B, Bruce C, Evans N, Goudie A, Gration K, et al. (2000) Selamectin: a novel broad-spectrum endectocide for dogs and cats. Vet Parasitol 91(3-4): 163-176.
- Takiguchi Y, Mishima H, Okuda M, Terao M, Aoki A, et al. (1980) Milbemycins, a new family of macrolide antibiotics: fermentation, isolation and physico-chemical properties. J Antibiot (Tokyo) 33(10): 1120-1127.
- Forrester S, Prichard R, Dent J, Beach R (2003) Haemonchus contortus: HcGluCla expressed in Xenopa oocystes forms a gated ion channel that is activated by antiparasitic drug ivermectin. Molecular and Biochemical Parasitology 29: 115-121.
- Martin J, Robertson A, Wolstenholme A, Vercruysse J, Rew R, et al. (2002) Mode of action of macrocyclic lactones. In Macrocyclic lactones in Antiparasitic Therapy, Wallingford, Oxon, UK, pp. 125-140.
- Geary T (2005) Ivermectin 20 years on: maturation of a wonder drug. Trends Parasitol 21(11): 530-532.
- Fellowes R, Maule A, Martin R, Geary T, Thompson D, et al. (2009) Classical neurotransmission of ovijector of Ascaris suum: Localization and modulation activity. Parasitology 121(pt 3): 325-336.
- Goudie A, Evans N, Gration K, Bishop B, Gibson S, et al. (1993) Doramectin a potent novel endectocide. Vet Parasitol 49(1): 5-15.
- Sangster N, Gill J (1999) Pharmacology of anthelmintic resistance. Parasitology Today 15(4): 141-146.
© 2017 Enejoh OS, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






