- Submissions

Full Text
Research & Investigations in Sports Medicine
High-Level Athletic Sprinters: Effects of Tolerance Training on DNA Damage, Acute Phase Proteins and Creatine Kinase
Affonso HO1,2*, Kulnig V1 and Camilo CJ1
1Laboratory of Exercise, Brazil
2Pharmaceutical Sciences Graduate Program, Brazil
*Corresponding author: Helvio de Oliveira Affonso, Street Chafic Murad, Bento Ferreira, Vitória, Brazil
Submission: August 13, 2019;Published: August 29, 2019

ISSN: 2577-1914 Volume5 Issue2
Abstract
Several studies have suggest that tolerance and speed resistence training are highly beneficial for highlevel athletic sprinters in relation to maximal running performance. However ROS generation by high intensity exercise contribute to oxidant accumulation and fatigue depressing the contractile function of myofibers so strength levels decrease, strength and power imbalances ≥10% are greater risk of injuries. Thus how to safely periodize trainings to obtein biopositive adaptations without favoring the prevalence of injuries?
Aim: In the present study, we aimed to evaluate the acute muscle and DNA damage, as weel as, kinetics of biomarkers signalling inflammation induced of tolerance training. Athlete: One male elite sprinter (World Champion with team Brazil 4x100m in 2019 & Brazil’s second best performance ever) PB 10’’02. Body characteristics: 21 years of age, 80kg body mass, 182.0cm of body height, 6.7 % fat mass.
Methods: Three Blocks with five sprints, distance (10/20/30/40/50m) sum 450m; five minutes of rest between blocks and 30 seconds between sprints. The blood samples were collected from the fingertip for [La-] assessment before and 30 seconds after sprints using aportable analyser and after 24h of training, samples were collected from the antecubital vein in EDTA-containing Vacutainer glass tubes. All measurements were obtained via automatic biochemical analyzer. Parameters: C-Reactive protein ultrasensitive; C-Reactive protein quantitative; CK; DNA damage.
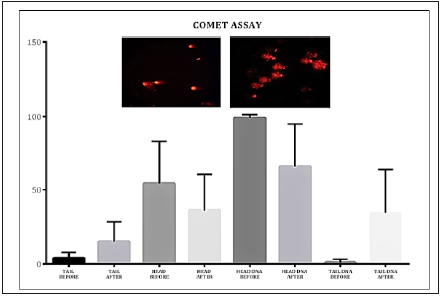
Results: The main results were together with the increase in DNA damage in after sprints compared to resting conditions high levels of CK (approximately 700%) and acute phase proteins (approximately 300/400%) and recovery optimal in approximately 96h.
Conclusion: This study suggests that optimal training or competition for maximum performance (strength, power and speed) should be combined with the best recovery before the next maximum stimulus. Still suggesting that program success may be favored by monitoring the best internal load levels (Biomarkers and comet assay).
Keywords: DNA damage; Acute phase proteins; Biomarkers; Signalling inflamation; Periodization; Injuries
Abbreviations: ROS: Reactive Oxygen Species; CK: Creatine Kinase; [La-]: Blood Lactate Concentrations; (mmol.l-1): Millimole/Liter; Min: Minute; us-CRP: C-Reactive Protein Ultra-Sensitive; CRP: C-Reactive Protein Quantitative; PB: Personal Best; m: Meters; DNA: Deoxyribo Nucleic acid
Introduction
Several studies confirm the benefits of regular physical exercise, it has also been shown that above certain intensity and duration, acute physical exercise may induce an increase in the production of reactive oxygen species (ROS) [1]. Anaerobic exercise (resistance training and sprinting) concomitant exposure to exercise mediated increased ROS, inflammation, and ischemia [2]. The high formation of ROS in amounts that exceed the capacity of the antioxidant defence system may result in DNA damage [3,4]. Interestingly, high levels of reactive oxygen species promote contractile dysfunction resulting in muscle weakness and fatigue [5]. Strength and power imbalances ≥10% are greater risk of injuries. Hamstring injury in elite sprinters was associated with weakness during eccentric action of the hamstrings and weakness duringconcentric action of the hip extensors. Studies recentes suggest that weakness of the hamstrings and possibly hip extensors is a cause of injury [6-8].
Lactate during trainings is considered to be the best biomarker for the assessment of intensity and performance bioenergetics in elite sprinters, particularly its anaerobic componente and is the end product of the anaerobic metabolism, diffusing from the muscle cell into the bloodstream when aerobic and other metabolic pathways are unable to keep up with the removal of pyruvate [9]. However tolerance and speed resistence training are highly beneficial for highlevel athletic sprinters in relation to maximal running performance, obviously very high intensity training to maximize performance and rank world. Solidly discussed in the scientific literature about that acute exercise causes apoptosis and inflammation [10,11], muscle metabolism parameters such CK are commonly increased after exercise and Elevated CK is commonly encountered in athletes [12]. CK levels should be monitored during and after exercise to evaluate recovery, that is, to determinewhether the levels return to basal, pre-exercise values, orwhether high values persist, which can be a signal of trauma, overtraining, or muscular pathology [13]. The purpose of this case report was show data regarding high intensity stimulus running in elite sprinter as well as the impacts and optimal recovery time through acute phase proteins, CK and DNA damage, drawing attention to the need to periodize training and competitions based on reliable biomarkers of the athlete’s total recovery, thus avoiding injuries related to imbalances and loss of strength and / or power, induced for ROS and DNA damage (Figures 1 & 2).
Figure 1:Kinetics of lactate, CK and acute phase proteins.
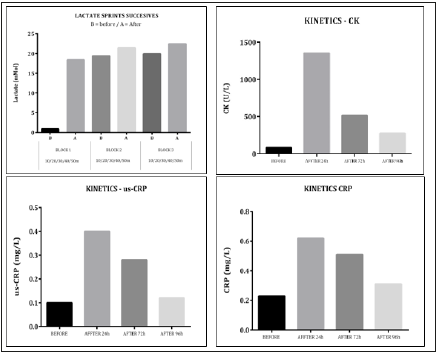
Figure 2:Acute damage DNA (before and after 3 bocks).
Case Presentation
This case report suggests high intensity training focusing on internal loads [La] in high levels all the time, combined with optimal external loads all the time to (maximal running performance). From the successive increase in sprint distance, thus causing considerable improvements in physiological capacity (tolerance), as well as increasing speed and speed resistance. However, we highlight the need for monitoring by biomarkers due to the significant increase damage to DNA, muscle and signaling of exercise-induced inflammation.
Methods
< Athlete
One male elite sprinter athletic (World Champion with time Brazil 4x100m in 2019 & Brazil’s second best performance ever) PB 10’’02, volunteered to participate in the current study and signed an informed consent form. Body characteristics were: 21 years of age, 80kg of body mass, 182.0cm of body height, 6.7% of fat mass. The body characteristics assessment was conducted before the beginning of the experimental protocol, with body mass and fat mass measured using the protocol sum of seven skinfolds [14]. All procedures were performed in accordance with the Declaration of Helsinki and approved by the university Vila Velha ethics committee n˚ 2.373.739/2017- CAAE 78770017.0.0000.5064.
Protocols
Sprints tests: Three (3) Blocks each with five (5) sprints successive, distance (10/20/30/40/50 m) total 450m; all sprints with maximal speed; five minutes of rest between blocks and 30 seconds between sprints (Tables 1-4).
Table 1:CK, us-CRP and CRP concentrations (basal, 24h, 72h and 96h after training).
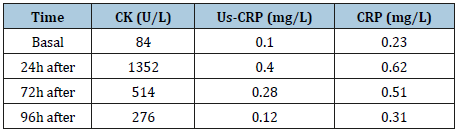
Table 2:Blood lactate (before) and after each block of sprints (10/20/30/40/50m).
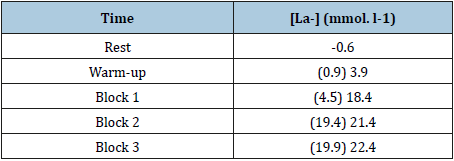
Table 3:Comet Assay (tail percentage and head percentage).
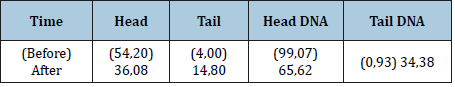
Table 4:Sprints and time.

Blood samples
The blood samples were collected from the fingertip for [La-] assessment before and 30 seconds after sprints test using aportable analyser (Lactate Plus®, Waltham, MA 02454-9141, EUA) and after 24h of training, samples were collected from the antecubital vein in EDTA-containing Vacutainer glass tubes (Becton, Dickinson and Co, Franklin Lakes-NJ), 9-10 a.m. All measurements were obtained via automatic biochemical analyzer (AU680, Olympus/Beckman Coulter, Munich-Germany) or hematological analyzer (Coulter LH750, Beckman Coulter, Brea, CA-USA). The parameters were: C-Reactive protein ultra-sensitive; C-Reactive protein quantitative; CK; DNA damage.
DNA damage
DNA damage were measured using the comet assay, with gel electrophoresis. Comet assay is a simple and sensitive method for quantitatively measuring DNA damage in individual cells [15]. Cells with increased DNA damage display increased DNA migration from the nucleus toward the anode. The migration is observed by fluorescence microscopy after staining with a fluorescent dye (ethidium bromide), and the intensity of the comet tail reflects the number of DNA damage measure with CASP LAB (comet assay software). The Alkaline Comet Assay is a well-validated technique for assessing DNA damage in individual cells [16].
Discussion
The present case study shows the importance of considering the increase inflamatory signaling and muscle damage during maximum sprints to periodize optimal training for elite sprinters, considering that ROS generation by high intensity exercise contribute to oxidant accumulation and fatigue depressing the contractile function of myofibers and results can lower strength levels, strength and power imbalances ≥10% are greater risk of injuries [17,18]. Several studies suggest that CK levels should be monitored during and after exercise to evaluate recovery, that is, to determine whether the levels return to basal, pre-exercise values, or whether high values persist, which can be a signal of trauma, overtraining, or muscular pathology. In summary, results of this case study suggest point to the conclusion that high intensity exercise (sprints and tolerance training) produces DNA damage and in he future research assessing these same variables, in addition to DNA damage and Supplementation on Antioxidant Status and exercises [19]. Finally, it seems to be in the balance between stimuli and recovery to maximize performance. Biomarkers can assist in decision making to define this optimal periodization.
Acknowledgement
We will like to acknowledge the Group Tommasi Laboratory of Vila Velha, Espírito Santo, Brazil, that allowed the execution of the exams beyond the athlete and staff that voluntarily participated in the study.
Conflict of Interest
There is no conflict of interest. Author have not received a specific grant for this research from any funding agency in the public commercial or not-for-profit sectors.
References
- Soares JP, Silva AM, Oliveira MM, Peixoto F, Gaivão I, et al. (2015) Effects of combined physical exercise training on DNA damage and repair capacity: role of oxidative stress changes. Age (Dordr) 37(3): 9799.
- Bloomer RJ, Goldfarb AH, Wideman L, McKenzie MJ, Consitt LA (2005) Effects of acute aerobic and anaerobic exercise on blood markers of oxidative stress. J Strength Cond Res 19(2): 276-285.
- Soares JP, Mota MP, Duarte JA, Collins A, Gaivão I (2013) Age-related increases in human lymphocyte DNA damage: Is there a role of aerobic fitness? Cell Biochem Funct 31(8): 743-748.
- Soares JP, Silva AI, Silva AM, Almeida V, Matos M, et al. (2015) Effects of physical exercise training in DNA damage and repair activity in humans with different genetic polymorphisms of hOGG1 (Ser-326Cys): Physical exercise effects in hOGG1 Ser326Cys. Cell Biochemunct 33(8): 519-524.
- Scott KP, Jackson MJ (2008) Exercise-induced oxidative stress: Cellular mechanisms and impact on muscle force production.Physiol Rev 88 (4): 1243-1276.
- Orchard J, Marsden J, Lord S, Garlick D (1997) Preseason hamstring muscle weakness associated with hamstring muscle injury in Australian footballers. Am J Sports Med 25(1): 81‐85.
- Croisier JL, Forthomme B, Namurois MH, Vanderthommen M, Crielaard JM (2002) Hamstring muscle strain recurrence and strength performance disorders. Am J Sports Med 30(2): 199‐203.
- Sugiura Y, Saito T, Sakuraba K, Sakuma K, Suzuki E (2008) Strength deficits identified with concentric action of the hip extensors and eccentric action of the hamstrings predispose to hamstring injury in elite sprinters. J Orthop Sports Phys Ther 38(8): 457‐464.
- Affonso HO, Silva AS, Fernandes RJ (2019) Can blood lactate concentrations rise significantly after very short duration swimming bouts? Ann Sports Med Res 6(1): 1139.
- Syu GD, Chen HI, Jen CJ (2011) Severe exercise and exercise training exert opposite effects on human neutrophil apoptosis via altering the redox status.
- Mitra S, Abraham E (2006) Participation of superoxide in neutrophil activation and cytokine production. Biochim Biophys Acta 1762(8): 732-741.
- Mougios V (2007) Reference intervals for serum creatine kinase in athletes. Br J Sports Med 41(10): 674-678.
- Becatti M, Mannucci A, Barygina V, Mascherini G, Emmi G, et al. (2017) Redox status alterations during the competitive season in élite soccer players: focus on peripheral leukocyte-derived ROS. Intern Emerg Med 12(6): 777-788.
- Jackson AS, Pollock ML (1978) Generalized equations for predicting body density of men. Br J Nutr 40(3): 497-504.
- Rodrigues BP, Campagnaro BP, Balarini CM, Pereira TM, Meyrelles SS, et al. (2013) Sildenafil ameliorates biomarkers of genotoxicity in an experimental model of spontaneous atherosclerosis. Lipids Health Dis 12: 128.
- Reid MB, (2016) Reactive oxygen species as agents of fatigue. Med Sci Sports Exerc 48(11): 2239-2246.
- Brancaccio P, Maffulli N, Limongelli FM, (2007) Creatine kinase monitoring in sport medicine. Br Med Bull 81-82: 209-230.
- Lippi G, Banfi G (2008) Distribution of creatine kinase in sedentary and physically active individuals. Am Heart J 155(6): e51.
- Ortiz-Franco M, Planells E, Quintero B, Acuña-Castroviejo D, Rusanova I, et al. ( 2017) Effect of melatonin supplementation on antioxidant status and DNA damage in high intensity trained athletes. Int J Sports Med 38(14): 1117-1125.
© 2019 Helvio de Oliveira. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






