- Submissions

Full Text
Research & Development in Material Science
Effect of Substitution of Modified Pofa on Physico-Mechanical Properties of Porcelain
Abdullahi MG*, Takai ZI and Ibrahim MA
Department of Physics, Faculty of Science, Yusuf Maitama Sule University Kano, Nigeria
*Corresponding author: Abdullahi MG, Department of Physics, Faculty of Science, Yusuf Maitama Sule University Kano, Nigeria
Submission: September 29, 2022;Published: October 12, 2022

ISSN: 2576-8840 Volume 17 Issue 5
Abstract
To provide a sanitary environments and affordable materials that reduce the cost of living is one of the objectives of sustainable development goals. The 21st century needs civilization and developments in all aspects of life. The renewable sources such as recycling of by-product will help in sanitary and reducing cost of living as well as environmental sanitary. The present research is designed to utilize Palm Oil Fuel Ash (POFA) as an additive ingredient in the porcelain body and to identify the effect of substitution of this modified POFA on Porcelain. Also will determine which composition/element in the POFA contents that most increases the strength of porcelain when added to it. This involves the experimental work such as volume shrinkage, porosity, bending and compressive strengths and supported with a theoretical aspect. The chemical composition of Palm Oil Fuel Ash (POFA), shown that the silica content (SiO2) in POFA is much higher than in Ordinary Portland Cement (OPC). The presence of a higher amount of SiO2 would assist the pozzolanic reaction. However, the calcium oxide (CaO) is lower than in OPC, while the Loss on Ignition (LOI) value of POFA is higher than in Ordinary Portland Cement (OPC), at 3.10%. From the chemical analysis, it is revealed that POFA is a satisfactory pozzolanic material and may be used as a cement replacement.
Keywords:Physico-chemical properties; Ordinary Portland Cement (OPC); Palm Oil Fuel Ash (POFA); Loss on Ignition (LOI); Porcelain
Introduction
The solid waste from palm oil mill industry has been extensively increasing every year. Study shows that palm oil wastes were producing four million tons per year in Malaysia and generated near this amount in Nigeria as well. Therefore, to overcome this problem, the solution is to reuse this waste product and produced new composites that are beneficiary to human development [1].
Nigerian oil palm companies/industries are among the contributors that account for over half of the world’s total palm oil output and is expecting for continuous growing with the global increases in vegetable oil demand. Hence, it lead to a large contribution to the nation’s pollution problem of about 2.6 million tonnes of solid waste in the form of palm oil fuel ash (POFA) [2].
Based on the statistics Nigeria provided important percentages of about 12.16 and 14.02 tons per hectare of fresh bunch in 2010 and 2011 respectively. Whereas according to Malaysian Palm Oil Board (MPOB) is about 20.18 and 19.2 tons per hectare of fresh fruit bunch were produced in the year 2010 and 2011 as well. These make the total production of 90,566,927 tons and 90,070,272 tons of fresh fruit bunch in the year 2010 and 2011 [2]. According to estimation by Tay and Show, for every 100 tons of fresh fruit bunches processed, there were about 20 tons of nut shells, 7 tons of fibres and around 25 tons of empty bunches discharged from the mill industry [3].
Therefore, this waste product produced from the Palm Oil Industry caused criticism and complaint. However, due to increases in price of oil, it becomes costly and unreliable to continue with the diesel generators for power supply. For this concern, the empty fruit bunches of palm trees, shells and fibres were used extensively as a fuel to heat the steam for the electricity generation and during the palm oil extraction process [4]. After combustion in the steam boiler, there is around 5% of ash called Palm Oil Fuel Ash (POFA), being produced [5]. Therefore, as the production increases continuously, more ash will be produced which lead to potential environmental problem (Pollution) when it is disposed as a landfill material due to the limitation of it is utilization [6].
These abundant agriculture and industrial wastes are always discharge from the industries, these wastes also include POFA can be reutilized as potential cement replacement materials especially in the concrete construction. Based on this, effective consumption of POFA as a cement replacement will encourage the researchers to explore ideas for saving materials such as cement, and also to reduce the pollution in the country. According to Md. Rezaul Karim et al. [7], the use of POFA in concrete as cement replacement material is logical, worthy and attributable for the demand in concrete industry especially in the present situation for the reduction in the cost of construction material and minimization of waste disposal problem.
The recycling of these waste materials such as POFA and Ceramic Sludge (CS) is very useful, POFA as one of the pozzolanic materials that use for many applications such as cement replacement or even used as a renewable energy resource [8]. Pozzolanic materials are fine materials, which contain silica, alumina in higher amount and calcium oxide, iron oxide, magnesium oxide and alkalis in fewer amounts. Silicon oxide SiO2 is a major composition of POFA, when silica and alumina react in the presence of calcium oxide (CaO2) or calcium hydroxide Ca(OH2) they form cementitious materials which can be used as a cement replacement and can exhibit high strength concrete more than the normal cement. And the fineness the POFA particles the highest strength it developed.
Indigenously developed technology is also considered as an important index for exploring and promoting these unutilized resources, which are technological and economic potential of the nation’s industrialization processes. It has been established that, these abundant raw materials are available for the manufacture of porcelain as well as heated ceramic wares in most of the tropical countries such as Malaysia [9]. Porcelain is a ceramic material made by heating raw materials; generally consist of clay in the form of kaolin when heated in a kiln to temperatures between 1,200 and 1,400 degree of Celsius. The strength, toughness and translucence of porcelain usually arise from the formation of glass and the mineral mullite within the fired body at these high temperatures. Porcelain had been found to be veritable stoneware because of its very high density, industrial fast firing cycle, and tangible mechanical strength and wear resistance [9].
The mechanical properties of porcelain are improving when replacing quartz with other materials such as rice husk ash and silica fume, others are kyanite, bauxite, fly ash, sericitic pyrophyllite and silliminite sand [8]. The alumina also has significant influence on the mechanical properties of whiteware due to the formation of primary mullite [10].
According to Parasad et al. [11], show that rice husk ash with minor or negligible amount of carbon in cooperated in the whiteware composition, makes an improvement in the properties of porcelain. Hence, the flabby natures of rice husk ash (RHA) particles which unfavourably influenced the output of body preparation and possibly increase the loss caused by cracking and deformation.
Therefore, the present research is designed to utilize POFA as an additive ingredient in the porcelain body and to identify the effect of substitution of this modified POFA on Porcelain. Also to determine which composition/element in the POFA contents that most increases the strength of porcelain when added to it. This may involve the experimental work such as volume shrinkage, porosity, bending and compressive strengths and supported with a theoretical aspect.
Material and Method
Data collection
Figure 1:Graphical abstract for data collection.

The contents (waste product of palm oil nuts) used in this study was collected from the Palm Oil Mill industry assigned by the Principal researcher. This waste product from the mentioned industry was in the form of ash called Palm Oil Fuel Ash (POFA) and it used throughout the research. The equipment and apparatus used are Oven, Ball Mill Machine, Severs, Digital Balance BM Series H-8000 and Electric Furnace (Figure 1).
Experimental Procedures
Sample preparation
The POFA was placed in an oven and allowed it to dry at a temperature of 100 ºC to 110 ºC for at least 24 hours (1 day). When it was dried, the sample were removed and grinded in a ball mill machine to reduce the size of the particle to an appropriate size needed for the improving the reactivity. This milling time always took about an hour to an hour 30 minutes at 200 revolutions per second (rpm). After this grinding, the next step was sieving. The sample will sieves in a less than 150μm sever so that the particles greater than this value will be removed. The untreated POFA were heated separately to remove the excess carbon on it. This can only be done when as it heated to a temperature of about 600 ºC for 1.5 hours in an electric furnace. Furthermore, the Porcelain raw material powder was grounded separately in a ball mill, just like in the case of POFA. The powder was sieved and dried using sieve shaker and an oven. The treated POFA was incorporate into the body of porcelain powder from 2wt% to 10wt%. The composition was also mixed using a ball mill for 1 hour 30 minutes. The mixed powder was pressed into pellets and bars at a pressure of about 90 to 95MPa. We must ensure that all the pellets and bars was sintered at a temperature of about 1200 degree of Celsius for 2 hours soaking time, at a heating rate of 50 ℃per minute (Figure 2).
Figure 2:(a) Treated POFA furnace for 1.5h at 600 ºC (b) Prepared porcelain powder.

Sample analysis
The analysis involved an experimental procedure which was carried out in the laboratory. As this involved the sample preparation as mentioned above. Using this procedure the physical and mechanical properties of the pellets such as volume shrinkage, porosity, bending strength, and compressive strength were also determined. The chemical composition of the treated POFA was investigated using X-Ray Fluorescence (XRF) while the crystalline structure of the treated POFA was determined through XRD and the microstructural features were examined using FESEM.
Results and Discussion
X-ray fluorescence analysis
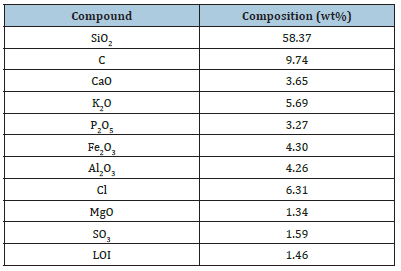
The chemical composition of treated POFA was analysed using X-ray fluorescence analysis (XRF), which was reported previously by Oyeleke et al. [12], Abdul et al. [13] and Weerachart et al. [14]. As can be seen in Table 1, there is present of different compounds with SiO2 as the major composition followed by C, Cl, CaO, K2O, Fe2O3 and Al2O3 with compositions 58.37, 9.74, 3.65, 5.69, 3.27, 4.30 and 4.26 respectively.
Table 1:Chemical composition of treated POFA..
XRD analysis
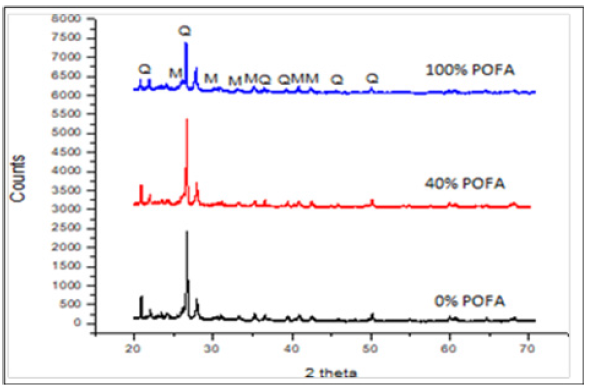
The X-ray diffraction analysis was used to study the structural properties of the composition materials and the result is presented in the Figure 3. It has been observed that the X-ray Diffraction (XRD) pattern of the samples containing three different of Palm Oil Fuel Ash (POFA), porcelain and mixture of POFA and Porcelain with sintering temperature of 1100 °C. This sintering temperature was specifically selected to ascertain the reason behind maximum compressive strength on this temperature. From the figure, it is clear that, the mineralogical phases of the samples are visibly seen. Figure 3 further revealed quartz as the major identified phases and also mullite can easily be seen from the XRD analysis. The peak of quartz decreases as the percentage of POFA increases, and the formation of mullite increases as the quartz decreased, as reported by [5] the mechanism behind the mullite growth was believed to be due to atmosphere induced changes to the composition in the oxidising atmosphere that facilitate mass transport and crystal growth. Therefore, in a pure porcelain the highest compressive strength was achieved. The presence of SiO2 and Al2O3 helped in mullitization of the crystal. The increment of the mullite is due to the melting of quartz grain which contributes to the formation of a homogeneous vitreous matrix [6]. According to Chaudhuri, mullite formation took place as a result of replacement of Al+3 in the porcelain structure owing to the role played by Fe+3 and Ti+4 that are refereed to foreign ions [7]. Since these foreign ions were found in the POFA, thus it helps in the formation of mullite as the POFA materials.
Figure 3:XRD pattern of three compositions of POFA, Porcelain and their combination sintering at temperature of 1100 ℃.
FESEM analysis
The morphological study of the samples has been investigated by using Scanning Electron Microscope (SEM) and the result is revealed in Figure 4, which indicated the present of POFA, Porcelain and the combined mixture, are shown as indicated in Figure 4a-4c, the mixture has the lowest porosity and hole around the surface due to the absence of POFA. In Figures 4b and low contain of it in Figure 4c, more porosity and hole around the surface due to the addition of POFA is pronounce 4c.
Figure 4:SEM micrographs of (a) POFA (b) Porcelain and (c) POFA/Porcelain.

Mechanical properties
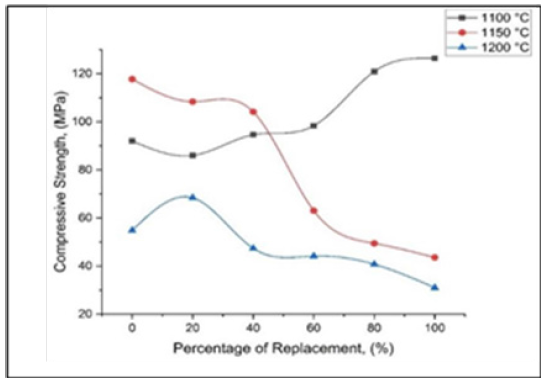
Researchers and industries considered compressive strength of porcelain to be the most important factor that determined the mechanical properties of porcelain. The result of compressive strength of this research is presented in Figure 5. It is indicated that, the compressive strength increases as the mixture annealed at 1100 °C, which is from 92.05MPa at 0% to 126.42MPa. The highest compressive strength for all the three sintering temperatures was obtained at 1100 °C which is 126.42MPa. However, the lowest values of compressive strength for all the three sintering temperatures were obtained at 1200 °C sintering temperature which is 31.03MPa at 100% of the replacement. At higher temperatures, these compounds would initiate pores which subsequently reduced the strength of the final product [8]. As a result of matrix reinforcement, the presence of strengthening effect was noticed, therefore, it is worthy to note that it is difficult to identify the matching of the coefficient of the dispersed particles with that of the glass matrix in porcelain body. Similarly, the reinforcement of the matrix structure in the porcelain body was realised due to the replacement of quartz with palm oil fuel ash (POFA), whereas, the subsequent dissolution of quartz was influenced by the increase in sintering temperature, hence leading to the overall formation of matrix structure. It is also pertinent to note that, SiO2 presence in meagre quantity facilitate the melting of other particles with the aid of feldspar [11,15]. Figure 2 shows the graph of compressive strength versus composition of Palm Oil Fuel Ash (POFA).
Figure 5:Graph of the compressive strength of porcelain against composition of Palm Oil Fuel Ash (POFA), Porcelain and the mixture of Porcelain/POFA at different sintering temperatures.
Conclusion
The benefit of POFA as can be seen in Figure 2 & 4 shows that visual look of the raw ash was considered as dark spongy and porous structure. Main components of the raw ash contain hard but light weightiness honeycombed scums, and asymmetrical shape and cellular textures. These explanations were in covenant with the findings of Oyeleke et al. [12], Abdul et al. [13] and Weerachart et al. [14].
Though, POFA beneficiation via sieving and all other processes revealed that the ash was having greyish form, when sieving it with the 150 test sieve. Furthermore, the matrix of the ground POFA retains very fine and powdery mid-air textured. The grey colour shows substantial volume of carbon insides been removed via the sieving exercise.
However, POFA as a complementary pozzolanic material in kaolin is very essential as well. The synopsis of SiO2, Al2O3 and Fe2O3 (Table 1, Chemical Composition of treated POFA) shows a vital role in the predictable effectiveness of mineral or natural admixtures. It also shows the Chemical and elemental composition of ground POFA, the chemical analysis tells more in agreement with the ASTM C618-01 (2001) description, the synopsis of SiO2, Al2O3 and Fe2O3 existent in the ground POFA satiates the requirements to be categorized as Class C supplementary cement material. Conversely, this expected that in a hydrated environment, the CaO in the Kaoin-POFA mixture may react with the supplementary cement material. In future, the predictable general effect of this reaction is that; the composite may chemically change into a natural cement structure of calcium silicates/aluminates in the suitable pH environment [16]. In addition, to that, the existing of magnesium oxide (MgO4, ≈17%), potassium magnesium silicate (K2MgSi3O8, ≈34% and magnesium aluminum oxide (MgAl2Fe1.8O4, ≈37%)) in the POFA also indicates its fitness as cementing agent in the POFA compound system [16]. It is predicted that the general effect of the minerals may extensively increase the reduction efficacy of the kaolin-POFA matrix, over the obstruction of harmful heavy metal ions existing in landfill leachate. Important amount of quartz (SiO2, ≈93% mineral noticed was in agreement with the findings of Foo & Hameed [17-35].
In conclusion the recycling of these waste materials (POFA) is very useful; POFA as one of the pozzolanic materials used as cement replacement or as an additive to porcelain is very vital for increasing its strength due to the mount of silica contents, alumina in higher amount and calcium oxide, iron oxide, magnesium oxide and alkalis in fewer amounts. Silicon oxide SiO2 is a major composition of POFA, when silica and alumina react in the presence of calcium oxide (CaO2) or calcium hydroxide Ca(OH2) they form cements’ materials which can be used as a cement replacement and can exhibit high strength concrete more than the normal cement. And the fineness the POFA particles the highest strength it will establish.
References
- Zarina Y, Mustafa Al-Bakri AM, Kamarudin Khairul Nizar H, Rafiza AR (2013) Review on the various ashes from palm oil waste as Geopolymer Material. Rev Adv Mater Sci 34: 37-43.
- Malaysian Palm Oil Board (MPOB) (2010) Economic and statistic.
- Tay JH, Show KY (1995) Use of ash derived from oil palm waste incineration as a cement replacement material. Resources, Conservation and Recycling 13(1): 27-36.
- Nurdeen M, Altwair MA, Johari M, Zeyad AM (2012) Pozzolanic characteristics of Palm Oil Waste Ash (POWA) and Treated Palm Oil Fuel Ash (TPOFA) Confernce: 2nd International Conference on Civil Engineering and Building Materials (CEBM, 2012) at Hong Kong.
- Sata V, Jaturapitakkul C, Kiattikomol K (2004) Utilization of palm oil fuel ash in high-strength concrete. Journal of Materials in Civil Engineering 16(6): 623-628.
- Safiuddin M, Salam MA, Jumaat MZ (2011) Utilization of palm oil fuel ash in concrete: A review. Journal of Civil Engineering and Management 17(2): 234-247.
- Rezaul Karim MFM, Zain M Jamil, Islam N (2011) Strength of concrete as influenced by palm oil fuel ash. Australian Journal of Basic and Applied Sciences 5(5): 990-997.
- Kumar Narendrabhai Patai V, Kumar N, Dilipbhai Shah (2015) Sustainability of porcelain and marble industrial waste powder to produce high performance concrete. American Journal of Civil Engineering and Architecture 3(3): 59-63.
- Fatiha N, Keat Z, Azlina TL, Subhash HKB, Abdul Rahman M (2005) Study of adsorbent prepared from Oil Palm Ash (OPA) for flue gas desulfurization. Septn and Purftn Tech 45: 50-60.
- Jomo U, Zaky M, Arafin Z (2014) Effect of palm oil fuel ash composition on the properties of morphology of porcelain-palm oil fuel ash composite. Journal of Teknologi (Science and Engineering) 70(5): 5-10.
- Parasad CS, Maiti KN, Venugopal R (2003) Effect of substitution of quartz by rice husk ash and silica fume on the properties of whiteware compositions. Ceramic International 29(8): 907-914.
- Oyeleke RB, Yusof Mb, Salim MR, Ahmad K (2011) Physico-chemical properties of palm oil fuel ash as composite sorbent in kaolin landfill liner system. International Journal of Renewable Energy Resources 1: 37-44.
- Abdul Awal ASM, Abubakar S (2011) Properties of concrete containing high volume palm oil fuel ash: A Short-term Investigation. Malaysian Journal of Civil Engineering 23(2): 54-66.
- Tangchirapat W, Jaturapitakkul C, Chindaprasirt P (2009) Use of palm oil fuel ash as a supplementary cementitious material for producing high-strength concrete. Construction and Building Materials 23(7): 2641-2646.
- Bergmann CP (2004) Traditional and glass powder porcelain: technical and microstructure analysis. J Eur Ceram Soc 24: 2383-2388.
- Chun Y, Yin W, Shabuddin WA, Ying PL (2008) Oil palm ash as partial replacement of cement for solidification/stabilization of nickel hydroxide sludge. J Haz Matls 150(2): 413-418.
- Araújo (2017) Polynomial regression with reduced over-fitting - The PALS technique. Measurement 124: 515-521.
- Deshmukh SD (2017) Regression: an efficient data analysis technique. Int J Innov Res Comput Commun Eng 5(3): 5901-5903.
- Eldagal, Elmukhtar O (2008) Study on the behaviour of high strength palm oil fuel ash (POFA) CONCRETE. Master Engineering Thesis (Civil and Structure), Universiti Teknologi Malaysia, Malaysia.
- Awal ASMA, Hussin MW (1997) The effectiveness of palm oil fuel ash in preventing expansion due to alkalis-silica reaction. Cement and Concrete Composite 19(4): 367-372.
- Jaturapitakkul J, Tangpagasit S, Songmue K, Kiattikomol K, (2011) Filler effect and pozzolanic reaction of ground palm oil fuel ash. Construction Building and Materials 25: 4287-4293.
- Jaturapitakkul C, Kiattikomol K, Tangchirapat W, Saeting T (2007) Evaluation of the sulfate resistance of concrete containing palm oil ash. Construction and Building Materials 21(7): 1399-1405.
- Koca C, Karakus N, Toplan N, Toplan HO (2012) Use of borosilicate glass waste as a fluxing agent in porcelain bodies. Ceram Process Res 13(6): 693-698.
- Galau, Ismail M (2010) Characterization of Palm Oil Fuel Ash (POFA) from different mill as cement replacement material. Faculty of Civil Engineering, Universiti Teknologi Malaysia 813100, UTM Skudai, Johor Bahru, Johor Malaysia, Malaysia.
- Mei FC, Kah PL, Hui JC, Ili Izyan SBR (2009) Removal of boron from ceramic industry waste water by absorption-flocculation mechanism using palm oil mill boiler (POMBA) bottom ash and polymer. Wat Resch 43(13): 3326-3334.
- Esposito L, Tucci A, Naldi D (2004) The reliability of polished porcelain stoneware tiles. J Eur Ceram Soc 25(9): 1487-1498.
- Jong LY, Teo DCL (2015) Concrete containing Palm Oil Fuel Ash (POFA) and Oil Palm Shell (OPS) subjected to elevated temperatures. UNIMAS e-Journal of Civil Engineering.
- Ismail M, Keok LS (2010) Fabrication of bricks from paper sludge and falm oil fuel ash. Concr Res Lett 1(2): 180-194.
- Megat Johari MA, Zeyad AM, Muhamad Bunnori N, Ariffin KS (2012) Engineering and transport properties of high-strength green concrete containing high volume of ultrafine palm oil fuel ash. Construction and Building Materials 30(3): 281-288.
- Brown OR, Yusof MBBM, Salim MRB, Ahmed K (2011) Compaction parameters of Kaolin clay modified with palm oil fuel ash as landfill liner. 2011 IEEE First Conference on Clean Energy and Technology (CET). IEEE, pp. 199-204.
- Chindaprasirt P, Jaturapitakkul C, Sinsiri T (2007) Effect of fly ash fineness on microstructure of blended cement paste. Constr Build Mater 21(7): 1532-1541.
- Chindaprasirt P, Homwuttiwong S, Jaturapitakkul C (2007) Strength and water permiability of concrete containing palm oil fuel ash and rice husk bark ash. Construction and Building Materials 21(7): 1492-1499.
- Garba SDI, Noh MZ, Siti Noraiza AR, Sharip N, Kutty NAA (2019) Effect of Iron (111) Oxide (Fe2O3) as an additive and substitution of quartz with POFA on physico-mechanical properties of porcelain. Int J Nanoelectron Mater 12(2): 175-184.
- Mukhopadhyay TK, Ghosh S, Ghosh J, Ghatak S, Maiti HS (2010) Effect of fly ash on the physico-chemical and mechanical properties of a porcelain composition. Ceram Int 36(3): 1055-1062.
- Sata V, Jaturapitakkul C, Rattanashotinunt C (2010) Compressive strength and heat evolution of concretes containing palm oil fuel ash. Journal of Material in Civil Engineering 22(10).
© 2022 Abdullahi MG. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






