- Submissions

Full Text
Research & Development in Material Science
Chitosan as Promising Materials for Biomedical Application: Review
Yadu Nath VK , Raghvendrakumar M1*, Aswathy V1, Parvathy P1, Sunija S1, Neelakandan MS1, Nitheesha Shaji1 and Vishnu KA2
1Department of Nanoscience and Nanotechnology, Mahatma Gandhi University, India
2School of Chemical Science, Mahatma Gandhi University, India
*Corresponding author: Raghvendrakumar M, Department of Nanoscience and Nanotechnology, Mahatma Gandhi University, India
Submission: October 10, 2017; Published: December 18, 2017

ISSN : 2576-8840Volume2 Issue4
Abstract
In the recent years, chitosan is considered as effective biopolymers for biomedical application. This is due to the fact that chitosan has a unique structure, various properties, and ubiquitous functionalities in character and provides the possibility for extension of this biopolymer in the in green chemistry, drug delivery, an anticancer agent, as well as wound healing etc. The inherent advantages offered by chitosan such as biocompatibility, genocompatibility, hemocompatibility, cytocompatibility, non-toxic, biologically suitable polymer properties make them attractive for biomedical application. The main aim of this review is to enlighten the importance of chitosan biopolymer for several biomedical technologies and engineering purposes.
Introduction
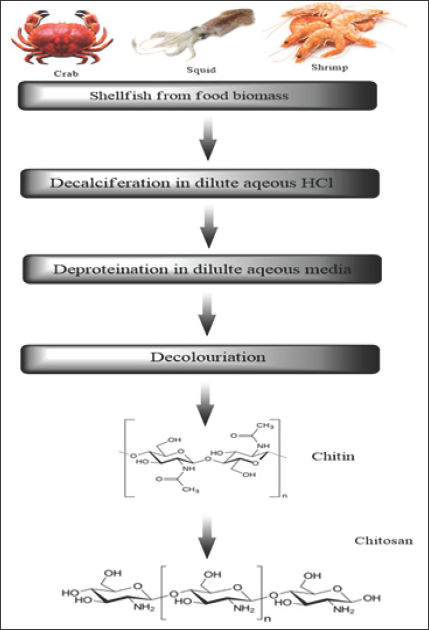
Figure 1: Commercial chitosan is derived from the shells of crustaceans.

Chitosan is cellulose like naturally derived polysaccharide (biopolymer), whereby the chitin shells (Figure 1) of crustaceans are treated with alkaline substances such as sodium hydroxide, which is also a biocompatible, biodegradable, non-toxic, antimicrobial and hydrating agent. The basic polysaccharide chitosan can be referred to as a chemically processed form of chitin. The cheapest source of chitosan is squid pen which is obtained as waste in the squid processing industry. Chitosan has a unique structure, various properties, ubiquitous functionalities and broad range of application in commercial as well as in bio medicinal field.They have contained the high percentage of (6.89%) nitrogen make them commercially valuable, and also a very good chelating agent. Chitosan is a bio polymer chain hitherto linked by two links namely P-(1-4) D-glucosamine and N-acetyl-D-glucosamine. The presence of these two links within the chain is random in nature (Figure 2).
Figure 2: Chemical structure of chitosan.
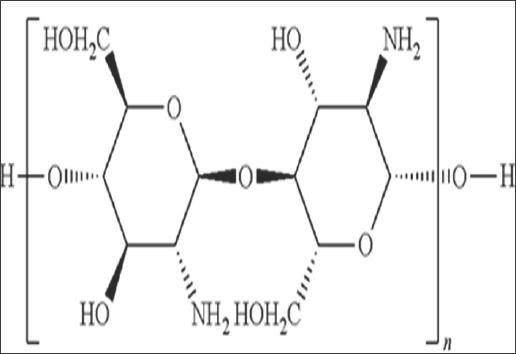
The chitosan is synthesized using its parent polymer Chitin by deacetylation method, due to its low solubility in aqueous and organic solvent chitin is not that much preferable to applications, while chitosan preferred in bio applications frequently. Chitosan is useful for multitudinous applications. The approximate molecular weight of chitosan found in the market lies in the range of 3800 to 20,000 Daltons. It is soluble in dilute acids such as acetic acid, formic acid etc. In the chitosan chain, nitrogen mostly occurs in the form of primary aliphatic amino group. The group undergoes amine based reactions like N-acetylation and Schiff's reaction. The derivatives of chitosan can be considered as substituted glucans and are obtained easily under mild conditions. Its chemical properties can be summed up as follows:
a. Linear aminopolysaccharide with large nitrogen content;
b. Rigid D-glucosamine structure exhibiting high hydrophilicity and crystallinity;
c. Weak base; deprotonated amino group is a powerful nucleophile (pKa 6.3);
d. Ability to form intermolecular hydrogen bonds leading to high viscosity;
e. It has large number of reactive functional groups for cross-linking and chemical activation;
f. Insoluble in water and organic solvents; soluble in dilute aqueous acidic solutions;
g. Forms salts with organic and inorganic acids;
h. Chelating and complex properties;
i. Ionic conductivity;
j. Polyelectrolytes (at acidic pH);
k. Cationic biopolymer with high charge density (one positive charge per glucosamine residue);
l. Flocculating agent and shows interaction with negatively charged molecules;
m. Adsorbing properties used for filtration and separation;
n. Thin film-forming ability; binding materials for isolation of bio molecules.
m. Adsorbing properties used for filtration and separation;
Naturally occurring polymers like Cellulose, Dextran, Pectin, Agar, Agarose have an acidic or neutral nature while chitosan is highly basic in nature. Emergent signs indicate that chitosan has the ability to decrease plasma and liver triacylglycerol (TG) in addition to the cumulative cholesterol (TC) levels, unveiling hypocholesterolemic as well as hypolipidemic effects [1]. Chitosan can also reduce the risk of cardiovascular diseases, potent fat-binding capacity, increase fecal neutral-steroid and excretion of bile acid, by in vitro studies. It has been reported that the Hypocholesterolemic effect of chitosan in humans' effectively decreases the plasma lipid levels also free from any side effects. Chitosan can easily bind with fat along with exhibiting cholesterol- and bile-salt-binding capacities. Different reaction conditions will yield various types of chitin with different physical and chemical properties. The molecular weight (Mw) and the Degree of Deacetylation (DD) and of a chitosan are the two key characteristics that prominently affect the chemical as well as physiological properties. The capacity of chitosan to bind with fat was pointedly more than that of cellulose, and it is also increased along with the increase in DD and Mw, whereas the cholesterol-binding ability was affected by the particle size [2].
The antimicrobial activity of chitosan along with its derivatives has been acknowledged and is deliberated to be one amongst the other vital functions, confirming the possible biological applications. Chitosan revealed antimicrobial properties with bacteria, fungi etc. Chitosan is also considered as the top amongst the other antimicrobial preservatives in the food industry. In addition to this, chitosan also inhibits the fungi growth and other microorganisms, specifically pathogens affecting plants [3]. It can persuade plants to synthesize enzymes (chitosanases) which can fight with invading microbes. It should be noted that chitosan shows comparatively more antimicrobial activity than chitin because of the positive charge of the amine group present in the chitosan. From the studies, it is discovered that chitooligosaccharides with higher degrees of polymerization (DP) show stronger reticence of fungi and bacteria than those by chitosan and chitooligo saccharides of lower DP [4]. At the same time, their inhibitory effects increase with the increase in the DD. Apart of this, partially hydrolyzed chitosan oligomers also exhibit antimicrobial activity against the common bacteria, mold, and yeasts that are found in food. Chitin, chitosan, along with their oligomers are recounted to exhibit activities elicitor in nature in numerous plants and have been broadly used as elicitors during the introduction of secondary products in the cell cultures of plants. During its attack by pathogens like fungi, viruses, and bacteria, higher plants tend to have a number of defensive reactions like the creation of phytoalexins, chitinase, and b-glucanase enzymes, inhibitors of proteinase, glycoproteins rich in hydroxyproline, active oxygen species, lignification. From most of the studies, it is seen clearly that, the chitosan along with its oligosaccharides show very strong antimicrobial activities and probably they can be used by humans safely [5].
The immunostimulating activity of chitosan and chitooligosaccharides has been noticed as they could produce non-specific host repellency when the animal body was infected with E [6]. Coli and viruses like Sendai. Studies also proved that chitooligosaccharides introverted the tumor growth by increasing the immune effects. Chitosan and chitosan derivatives also put on a display of an immunity-improving effect by enhancing the response towards antibody and anticancer activities. Moreover, chitosan is also recognized as a drug carrier which can enhance drug absorption as well as targeting and drug release. It also stabilizes the drug components. Chitosan can also act as a carrier of the gene to shield DNA and increase the period during which the genes can express themselves. Henceforth, chitosan has wide-ranging scenarios for applications as drug and gene carriers. For example, chitosan can also be used as a carrier of the drug to deliver the anticancer and antitumor agent. Also, conjugates of some kinds of agents capable of battling cancer with chitin and its derivatives acted as good anti-cancer agents [7].
Figure 3 shows the steps involved in the processing of chitin and chitosan, which are the useless products from crabbing and shrimp canning industry. During the processing mainly removal of proteins and the dissolution of high concentration of calcium carbonate in crab shells are the main part. Final chitosan is deacetylated in 40% sodium hydroxide for 1-3h at 120 0C to form 70% deacetylated chitosan. The chitin and chitosan processing includes crustacean shells, size reduction, protein separation, NaOH, washing demineralization (HCl), washing and dewatering, decoloration,chitin, deacetylation (NaOH), washing and dewatering, chitosan [8].
Figure 3: Processing of chitosan.
Biological Properties of Chitosan
Biodegradability of chitosan
It really is an essential to see the metabolic experience in our body biodegradation, when polymers utilized as drug delivery systems. When we look at the circumstance regarding the systemic absorption of hydrophilic polymers such as for example chitosan, they must have possessed the right molecular weight. If the employee polymer dimensions are bigger, then there is chance for the polymer to endure degradation. The biodegradation of chitosan could be the splitting of the polymer substance into smaller portions and also as monomers i.e. D-glucosamine, N-acetyl-glucosamine they will be catalyzed by enzymes or chemicals in vitro or perhaps in vivo. The biodegradation rate is a must for the usage chitosan in tissue engineering and drug-delivery systems. The amount of deacetylation (DD) and molecular weight (MW) will be the factors managing the biodegradation rates of chitosan. Chemical modifications to chitosan also influence the biodegradation rate. Biodegradation this is certainly chemical or enzymatic would offer fragments suited to renal clearance. In the case of chemical degradation, it refers to acid catalyzed degradation that is taking place when we look at the stomach [9]. Though it happens to be stated the oxidation-reduction depolymerisation and free radical degradation [10]. They are not likely to be an amazing source or degradation in vivo. Chitosan may be degraded by enzymes that hydrolyze glucosamine-glucosamine, glucosamine-N-acetyl- glucosamine and N-acetyl-glucosamine-N-acetyl-glucosamine linkages [11,12]. Chitosan is meant to be degraded in vertebrates mostly through lysozyme and also by bacterial enzymes when we look at the colon [13]. However, eight human chitinases (when we look at the glycoside hydrolase 18 family members) have now been recognized, for the reason that three of which may have revealed enzymatic action [14]. A variety of microorganisms synthesizes and/or degrades chitin, the biological substance of chitosan. Generally speaking, chitinases in microorganisms hydrolyze N-acetyl-β-1, 4-glucosaminide linkages randomly, this is certainly these are generally endochitinases. Chitinases are usually present in plants, and even though they do not have chitin structural elements. The family 19 chitinases are typically established to plants some have been establishing in bacteria [14,15]. This demonstrates simultaneously a safeguard mechanism in opposition to microbes and insects which have chitin structures and, in many cases, a symbiotic relationship [16]. So generally speaking we could state that simultaneously rate and degree of chitosan biodegradability in living organisms are influenced by the amount of deacetylation (DD) [17,18]. Increasing DD decreases the degradation rate. The degree of degradation relates to the rate, as most of the researchers are revealed over a finite lifetime. It really is potential that, presented appropriately some time recommended conditions; the chitosans would certainly degrade adequately for accompanying elimination. Chitinases separated from various organisms have actually extensively various properties. The suitable pH extends throughout the pH scale with chitinase separated from Saccharomyces cerevisiae with an optimum of pH 2.5 [19]. Chitinase separated from Oreochromisniloticus possessing an optimum of pH 9.0 [20]. The precise activity regarding the enzyme, preferably resistant to the same substrate, and it also differs mainly amidst different species. The three enzymatically active kinds of human chitinases known haven't been examined regarding the degradation of chitosan and/ or its types.
These sorts are;
a. di-N-acetyl chitobiose
b. Chitotriosidase,
c. The acidic mammalian chitinase (AMCase).
AM Case is recognized when you look at the gastrointestinal tract and lung [20] and has now demonstrated an ability to relax and play a crucial role in asthma [21-23]. This role happens to be presented to be mediated with the T helper cell type 2 reaction that prompts AMCase release from lung epithelial cells in an epidermal growth factor receptor (EGFR) dependent manner [14,15]. Neutralization of AM Case in a murine model created a reduced TH2 cell response and it surely will result in decreased inflammatory response [24]. Di-N-acetyl chitobiose was recognized in Tay Sachs and Sandhoff’s disease patient livers in 1974 [25] plus in rat liver in 1986. It absolutely was revealed to catabolise asparagine linked glycoproteins. Chitotriosidase is contained in excessive into the plasma of gaucher disease patients. It really is generated by activated macrophages. This enzyme has implications in certain diseases such as for example Gaucher disease and atherosclerosis. But in addition to protective mechanisms against fungi and parasites which may have chitinous structures.
Toxicity of chitosan
Chitosan is thoroughly noticed to be a non-toxic, biologically suitable polymer46. It really is approved for nutritional uses in Japan, Italy, and Finland [26-34] and has now been authorized because of the FDA to be used in injury dressings [35]. The modifications designed to chitosan will make it just about harmful and any residual reactants could be properly removed. It's very important to analyze in regards to the composition of chitosan along with a drug may possibly replace the pharmacokinetic and biodistribution profiles. By way of example, when it comes to chitosan/plasmid DNA nanoparticles, the in vivo kinetics and supply are mainly regulated because of the nanoparticle properties such as for example their shape and charge. Other sorts of, cellular uptake kinetics can be changed as a result of the charge interaction for e.g. in the case of DNA complexes. This controlling, or reduction, regarding the positive charges from the chitosan molecule, has possessions on its interaction along with cells in addition to microenvironment, and it also frequently trusted to reduced uptake and a minimize in toxicity. When it comes to a covalent drug conjugate, the polymer's physicochemical characteristics (hydrophilicity) and shape are altered (for example. micelle formation) with an arising influence on the distribution and cell uptake [36]. In addition, additionally, it is crucial to see the path of administration establishes the uptake, concentration, and contact some time cell kinds influenced. In a few reports, Schipper et al. [37] specified the consequence of chitosans with varying molecular weights and amount of deacetylation on Caco2 cells, HT29-H plus in situ rat jejunum. Through the toxicity, scientific studies unearthed that the amount of deacetylation and molecular weight are reliant. At high deacetylation the toxicity relates to the molecular weight in addition to concentration, at lower DD toxicity is less noticeable much less pertaining to the molecular weight. Nevertheless, almost all of the chitosan's examined failed to enhance dehydrogenase activity expressively when you look at the concentration range tested (1-500|ig/ml) on Caco-2 cells. The in situ rat jejunum research displayed no rise in LDH activity with some of the chitosans tested (50|ig/ml) [38,39]. The ciliary beat frequency of chicken embryo trachea was examined after use of trimethyl chitosan solutions and a marginal effect (comparable to physiological saline) was recognized [40]. For the style of drug delivery types predicated on chitosan, in vivo toxicity especially after long haul administration is a significant necessity In a somewhat long study (65 days), no negative influence on body weight was noticed if chitosan oligosaccharides were released [41]. Nevertheless, 50mg/kg intravenously applied chitosan triggered death, as a result of blood aggregation. A photo crosslinks chitosan, produced as a tissue glue similar to fibrin glue, azide-chitosan- lactose had not been toxic over a 1 month time period after administration of just one ml at 30mg/ml i.p., this is not photocrosslinked [42]. Nevertheless, no histological evaluation, enzyme or any other pathological measures were reported; not enough toxicity is induced by survival of >1 month.
Biocompatibility of chitosan
Biocompatibility could be the being compatible along with living tissue or an income system through not being toxic, injurious, or physiologically reactive rather than leading to immunological rejection. Facts associated with undesirable reactions among the biomaterials and living systems happens to be acquired from retrospective research in humans, and presently biomaterial-tissue interactions has been discovered by using bioassays in vitro plus in vivo. In previous research, biocompatibility evaluation mainly hinges on the area regarding the implant, and materials were classified as "very reactive" or "inert." This easy category has differed newly, because of the fact that a couple of materials can induce undesirable immune feedback, unwanted interactions with blood as well as other body fluids, and cause damage the genetic material in the chromosomal and DNA level. The good news is times almost all recently produced biomaterials suggested for usage in humans are confronted with in vitro assessments that are then followed closely by in vivo assessments. A number of clinical tests exploring the usage chitosan for medical function might not have been precisely authenticated and a few of those have examined merely very little biocompatibility studies (its operation or possibly in vitro cytotoxicity) without having the right control groups. By way of example, there clearly was issue that shrimp-allergic individuals may respond to glucosamine-including products since they may be made of the shells of crustaceans, shrimp, crab, as well as other shellfish [43].
Chitosan is actually polycationic in character at a pH<6 and simply interacts along with various negatively charged molecules, such as anionic polysaccharides, proteins, , essential fatty acids, bile acids, as well as others. It is essential to test the biocompatibility of chitosan derivatives prior to it really is found in the world of chitosan drug conjugate therapeutics since most of those are created to posses much longer plasma half-lives and afflicted by gradual degradation within your body. The detaching of monomers through the chitosan by degradation may possibly end up in serious complications, such as red cell lysis, immune protection system stimulation, tissue toxicity, and cell aggregation. Samples of biocompatible chitosan types revealed consist of modification with PEG, dextran, polyvinylpyrrolidone (PVP), arabinogalactan and poly-L-glutamic acid [44].
Chitosan derivatives are contained in a variety of kinds, such as solutions, films, porous structure scaffolds, pastes, sheets, hydrogels, fibers, tablets, nanoparticles, and microspheres. Chitosan films developed with acetic acid (chitosan-AA) and lactic acid (chitosan- LA) have now been tested with their potential as wound-healing materials when comparing to the commercial Omiderm [45].
Chitosan microspheres cross-linked with glutaraldehyde display sluggish biodegradability suited to the regulated delivery of several drugs; nevertheless, these synthetic cross-linking reagents are typical highly cytotoxic, that may impact the biocompatibility regarding the cross-linked biomaterials. So it will be significant to produce a cross-linking reagent this is certainly ideal for used in biomedical applications, or perhaps not cytotoxic, as well as that have to posses the proper execution a well balanced and biocompatible cross-linked item. As an example Geni pin is employed as a crosslinking reagent, with better biocompatibility and a more sluggish degradation rate compared to the chitosan microspheres crosslinked with glutaraldehyde [46].The biocompatibility reduces when we look at the order chitosan hydrochloride, chitosan glutamate, glycol chitosan, chitosan lactate, methylpyrrolidinone chitosan.
In vitro biocompatibility
The in vitro biocompatibility analysis of biomaterials is important since it is an important step for the growth of chitosan and its own derivatives for biomedical applications. A few experiments consist of biosafety evaluation, and biofunctionality analysis to determine the biocompatibility of chitosan and its own derivatives. In vitro screening centers around the evaluation of cytocompatibility and hemocompatibility, from then on in vitro genotoxicity ensure that your immune biocompatibility test can be executed. The tissue culture strategies can be applied for those studies and it is gaining importance and represents a great way to obtain all about the behaviour of cell when confronted with these biomaterials. "In vitro culture" is thought as an experimental system that synthetically reproduces environmentally friendly conditions essential to make sure the viability of cells or tissues harvested from an income organism, and it is widely utilized for the analysis of cell growth and differentiation. A suspension culture of chondrocytes was noticed to install, proliferate, and spread on porous chitosan microspheres, along with within these microcarriers, suggesting that chitosan is an appealing injectable tissue engineering scaffold [47].
Cytocompatibility
There are two evaluation stages requires testing the in vitro cytocompatibility of chitosan and its various derivatives. During the first stage, cytotoxicity tests are performed to screen the materials to be employed for external use or implantation. Cytotoxicity tests basically consist of morphological evaluations, which highlight and measure the cells that have died or have undergone regressive phenomena after exposed to the material in question [48-50].
In case there is the cytocompatibility examination appropriate choice of cell type is very important to make certain an appropriate and precise description of results and it is noticeably influenced by the conclusion use of the recently produced chitosan derivative product. When chitosan derivatives can be utilized as wound dressings, cultured cells, such as for example human keratinocytes or human dermal fibroblasts, may better fit the goal of cytocompatibility compared to the usage of cancerous cell lines. Compared to normal cell lines cancerous cell lines or transformed cell lines are far more resistant into the cytotoxic consequence of chitosan derivatives for that reason these cell lines may often undervalue the degree of cytotoxicity. For chitosan-derivative hydrogels, sheets, and pastes an indirect contact assay (agarose overlay assay) are suitable, for which cultured cells are covered with a permeable agar film, the test sample permitted to be added to the agar surface without getting in touch with membrane cells. It explained that extraction method also applicable for chitosan hydrogels, sheets, and pastes. In case there is chitosan hydrogels, sheets, and pastes direct-contact method could potentially cause the misunderstanding of cytotoxicity, using the fragments of sticky chitosan derivatives triggers challenges in MTT and MTS assays. The second stage involves cell cultures as a result tests. These tests ensure an even more exact assessment regarding the biocompatibility of these chitosan derivatives for the end use. Cell cultures are widely used to gauge the overall biological compatibility regarding the materials connected with toxic effects from the body, such as for example alterations regarding the metabolic pathways, alterations in organ functions, or modifications into the structure regarding the musculoskeletal system, that are responses to alterations occurring at the cellular level. Chitosan presents an increased in vitro cytocompatibility than does chitin. This property is apparently pertaining to the DD regarding the samples. As soon as the positive charge regarding the polymer improves, the interactions between chitosan in addition to cultured cells may also be improved as a result of the existence of free amino groups [48].
Hemocompatibility
Almost all of the biological feedbacks stimulated by materials are caused by the phenomena of surface initiation. The polycationic characteristics of chitosan prospect into the derivation of varied proteins and its own nonspecific binding to cell membranes could potentially cause thrombosis. This is why chitosan unsuitable for blood-contacting products. When chitosan and its own derivative come right into contact with blood, blood proteins are adsorbed on the surface leading to the attraction of platelets as well as other blood cells to cause platelet activation and thrombosis. The activation of platelets fairly lower for surface-sulfonated chitosan membrane in an aqueous alkaline medium that will be as a result of the phenomenon from the cationic NH3+ group [49].
The evaluation of hemolysis, red cell aggregation patterns, fibrinolysis, proteolysis, platelet activation, platelet adhesion, oxidative burst, and production of thrombin upon exposure are great for hemocompatibility research regarding the chitosan derivatives. If the chitosan derivatives are intravascularly utilized, their overall performance mainly hinges on their interaction with blood components. Experts are making an effort to create hemocompatible chitosan derivatives for a wide array of biomedical applications. N-acyl chitosans, like hexanoyl chitosan, have now been observed to obtain better hemocompatibility than N-acetyl chitosans [50]. The anticoagulant activity of chitosan is going to be pertaining to its paid down positive charge which will usually trigger interaction with negatively charged membranes of red blood cells.
Genocompatibility
Genotoxicity tests gauge the capability of a material resulting in mutations or gross chromosomal problems within affected cells. Coming from a variety of research it is available that chitosan has the capacity to mediate multiple biological activities, like the avoidance of genomic toxicity [51]. Chitosan oligosaccharide avoids mercury- stimulated chromosomal anomalies in mice, recommending that chitosan oligosaccharide is a nontoxic material which can be used as a suppressor of heavy metal-stimulated genotoxicity [52]. The conventional genotoxicity studies are time-taking in. Per currently growing technology named single-cell gel electrophoresis (SCGE) (comet assay) is a delicate biological tool utilized for Geno compatibility screening. For the assessment of Geno compatibility of a variety of chitosans as well as their derivatives comet assay happens to be achieving extensive recognition.
In vivo biocompatibility
The restriction of in vitro biocompatibility screening is the fact that cells hardly ever interact individually in vivo; examination built in vitro are simply just a straightforward section of complex in vivo mechanisms. The cultured cells try not to include any system to eradicate biological waste elements because culture environment is stagnant.
Which means material's toxicity can be over anticipated. There aren’t any standardized options for in vivo visualization of biocompatibility or inflammatory reactions to implanted biomaterials. Conventionally, biocompatibility is set via histological evaluation. Histology permits when it comes to a determination regarding the cell kinds and a wide range of cells nearby the implant, such as those from the immune protection system.
Alternatively, fluorescence imaging shows a collection of helpful strategies which were used by investigating tumor models, transplant rejection models, pulmonary inflammation and inflammation caused by arthritis [53].
Monocytes and neutrophils began the healing feedback if biomaterial is implanted. This feedback is followed closely by the propagation of fibroblasts and vascular endothelial cells. At the beginning of levels of wound healing, chitosan can speed the development of healthy granulating tissue. The in vivo biocompatibility of chitosan solutions that have glycerol- 2-phosphate (C-beta- GP) to be utilized when it comes to the parenteral administration of drugs was decided by injecting the C-beta-GP into the hind paw regarding the rate. C-beta-GP with a high DD yielded lower inflammatory reactions. Biocompatibility of thermosensitive chitosan-based hydrogels: an in vivo experimental approach to injectable biomaterials. Subcutaneous implantation of chitosan micro fibers into mice suggested that the implantation of rat muscle made of stem cells (rMDSCs) including chitosan micro fibers stimulated fewer host-tissue responses, together with reduced macrophage accumulation, then performed the injection of chitosan micro fibers itself, that might be as a result of the decreased immune reactivity regarding the transplanted rMDSCs [54]. This particular outcome implies that chitosan micro fibers may act as a biocompatible in vivo scaffold. In another type of analysis, the in vivo biocompatibility of a freshly formulated chitosan-agarose- gelatin (CAG) scaffold developed by using cryoablation technology was evaluated by subcutaneous implantation into Wistar mice.Neovascularization was noticed on the site regarding the interface, showing that CAG cryogel matrices not merely are biocompatible but in addition can trigger angiogenesis by developing new blood vessels [55].
Biomedical Application of Chitosan Nanoparticles
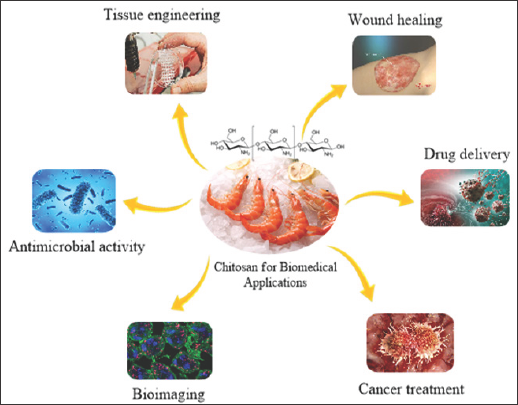
Chitosan nanoparticles have a large set of application in different filed of interests such as tissue engineering, bio imaging, cancer treatment, etc. shown in Figure 4. Some of the applications in medicinal filed was discussed below like bone tissue engineering, drug delivery etc. which explains the real role chitosan nanoparticles may reveal.
Figure 4: Illustrates the biomedical applications of chitosan NPs.
Chitosan in green chemistry application
Ionic liquids (ILs) happen to be attained so much more thought as green solvents due to the product regarding the advance of green chemistry. Additionally, it is a vital need for environmentally friendly protection [37]. The reception of ILs has appeared as a rebel which has been thrilled both the academia in addition to chemical companies. The terms and conditions room temperature ionic liquid (RTIL), nonaqueous ionic liquid, molten salt, liquid organic salt and fused salt have got all already been utilized to describe ILs (salts when you look at the molten state) [56]. The ILs have exceptional physicochemical characteristics which can be very helpful if traditional organic solvents are not really properly active or perhaps not appropriate [57].
In line with the mix of their inimitable characteristics in a way that ionic liquids possess a wide range of merits with their synthesis and extractions. These liquids have trivial vapor pressure this is certainly a liquid number of up to a lot more than 400K and densities higher than compared to water. The ionic liquids are miscible with substances inside an array of polarities. Additionally, demonstrates parallel dissolution in organic and inorganic substances. In several cases indicates that many processes could be amazing with traditional solvents due to their partial liquid range or their miscibility. Even greater probably could be the use of RTILs for chemical synthesis considering that the charged nature of these solvents may be subjective because of the synthesis itself. The main innovative and prominent feature of RTILs as solvents will there be probability to develop one with specific properties needed for a certain application, referred to as "designer solvents" [58].
Ionic liquids have now been related as a replacement solvents in various catalytic organic transformations [59,60]. These are generally successfully dissolved in biological macromolecules which can be linked by intermolecular hydrogen bonds, for example carbohydrates, cellulose, wool keratin and silk fibroin [61]. The Ionic liquid is also employed when it comes to dissolution of chitosan for waste water treatment, cosmetics, heavy metal and rock chelation, heterogeneous catalysts. Chitosan is made from an amino group and two hydroxyl groups when you look at the repeating hexosaminide residue. Through the chemical modification of every regarding the biopolymer, it's very important to make a well balanced homogeneous solution so that you can raise the efficacy of modification. Though, the solid inter and intramolecular hydrogen bonding amongst the chitin and chitosan chains reduces their solubility in a lot of organic solvents. So, dilute aqueous solutions of organic and mineral acids solutions are used when it comes to dissolution of chitosan. However, these solvents are corrosive and need an alkaline solution treatment procedure to get rid of the acid following the process. Moreover, in definite applications like transition metal sorbents and drug carriers [27] the polyelectrolyte solutions have limited application at the same time bioactive agents could be influenced because of the occurrence of acetic acid intake for dissolving chitosan. Consequently, a unique treating strategy for evolving potential applications among these bio- renewable resources is necessary [28].
Xie et al. [25] employed 1-butyl-3-methyl-imidazolium chloride solvent for chitin and chitosan. Then apply these as a substitute for amino-functionalized artificial polymers for catching and discharging CO2. When it comes to the creation of powerful CO2 recovery techniques from industrial waste gases is vital to simultaneously reutilization regarding the CO2 as a carbon resource in addition to environmental problems associated with greenhouse effects. Conventionally, the essential generally utilized process for CO2 recovery is a chemically reversible CO2 fixation with amines at room temperature to make ammonium carbamates. Through heating, the CO2 will be released through the ammonium carbamates [8]. Amino-functionalized synthetic polymers and ionic liquids happen to be also founded to fixate CO2 from the basis among this concept [29].
Chitosan is a normal polyamine and as a consequence, it could be the right replacement for amino-functionalized polymer when it comes to CO2 fixation. Although the ionic liquids will not totally interrupt the crystalline domains of chitosan and just a partial dissolution regarding the option would be obtained, it deceptively will not distress their use. The chitin/IL and chitosan/IL are in place when it comes to reversible fixation of skin tightening and. This green friendly system has the possibility not merely of recovering CO2 from the industrial exhaust but it additionally for CO2 sensing [30]. An original polymer/RTIL composite material predicated on chitosan and 1-butyl-3-methyl-imidazolium tetrafluoroborate (BMIM.BF4) were well-known by Lu et al. [22] The composite system could willingly make use of as an immobilization matrix to entrap proteins and enzymes. Hemoglobin (Hb) was chosen as a version protein for the research the composite system. The bioactive composite film-modified glassy carbon (GC) electrodes were produced by direct electron transfer amongst the protein in addition to GC electrode. The movie electrode revealed melodramatically enhanced bio electrocatalytic action to oxygen and trichloroacetic acid alongside it offers effective stability in solution.
Two well-described quasi-reversible redox peaks for hemoglobin was detected in the composite film-modified GC electrode. Thermogravimetric analysis (TGA) indicates that the chitosan-BMIM.BF4-Hb composite has finest thermal stability compared to that of chitosan-Hb itself.These unique composite materials may possibly deliver a fantastic electrochemical sensing platform for redox proteins and enzymes. Thus have possible applications in direct electrochemistry, biosensors, and biocatalysis by direct electron transfer.
A delicate glucose biosensor, making use of an electrodepositing chitosan-ionic liquid-glucose oxidase biocomposite onto nano-gold electrode, was generated by Zeng et al. [11]. A nano-gold electrode is produced by electrochemically depositing gold nanoparticles onto a flat gold electrode surface monitored by immersing into a bath containing benzoquinone (BQ), chitosan, glucose oxidase (GOD) and IL for electrodeposition of enzymatic electrode. This biosensor shows a wild amperometric response (<5s) to glucose with a good up-to-date sensitivity, this is certainly 2.8 times a lot better than a biosensor synthesized by electrodepositing chitosan- IL-GOD biocomposite on a flat gold electrode. The enlightening limit for glucose was 20-fold better set alongside the biosensor designed from the flat gold electrode. The biosensor offers higher reproducibility, long-time period storage space stability, and acceptable anti-interference capability [31]. Chitosan/cellulose composites, hybrids composite, as biodegradable bio sorbents, was produced on such basis as an ionic liquids; no agent that is crosslinking adsorption for heavy metal and rock ions by Lu et al. [22] The chitosan/cellulose that is freeze-dried was displayed to put on higher adsorption capability as well as enhanced stability. The interaction regarding the two components in addition to materials that are resulting in the ability to Ni(II) was verified by IR and XPS. In inclusion,some other heavy metal and rock ions, were efficiently adsorbed using the appropriate capacity Cu(II) > Zn(II) > Cr(VI) > Ni(II) >Pb(II) [12]. An method that is inventive the building of an auspicious glucose sensor, GOx/C60-Fc-CS-IL, that achievements the interactive advantages of fullerene (C60), ferrocene (Fc), chitosan and ionic liquid (IL) for glucose oxidase (GOx). The electrocatalytic action of C60 and Fc unusually upgraded the electron relays which activate the oxidation regarding the glucose and enhanced the reaction that is electrochemical the chitosan-IL network offering an accepting micro environment which conserved the bioactivity of GOx. The biosensor exhibited a rather sensitivity that is high low detection boundaries, quick response time, substantial calibration range and exceptional long-term stability as much as 30 weeks [32]. Both in cross-linking attitudes and in dosage forms while chitosan is relatively nontoxic, biocompatible material, attention must be taken to safeguard that it is pure and free from contaminations, such as protein, metal or other contaminants that might potentially cause various harmful things. Afterward each chemical reaction, understanding must certainly be filled to systematically eradicate the un-reacted reagents, particularity those who are cytotoxic, in order to prevent results that are mystifying.
Drug delivery
Conveyance of the medicinal drug to a specific place in the body was a major complication in the remedy of various diseases [54]. Associating already designed medicinal therapeutics to drug delivery entity easily evolves in terms of performance that is efficiency, safeness of the patient, and fewer side effects. Drug delivery entities that are built by Polymers can boost the pharmacokinetics of medicine, advances the therapeutic index, lower their side effects and accordingly escalate the resourcefulness of the entire system [55] . Considering a process in drug delivery mechanism, first of all, the drug conveyed to the site where treatment is required, the drug acts on the present issues and accumulates in the area, so the next time drug only needs to contain lower dose amount of therapeutics [56] . Chitosan NPs have developed into a colossal significance in polymeric therapeutic conveyance entity for the advancement of recent pharmacological and medicinal release to the targeted site including good bio distribution, elevated specificity, and sensitivity. Chitosan is one of the most popular natural polymers have wide application in the discipline of drug delivery entities due to its cationic functionality and the aqueous medium solubility of the material. It also has some upper hand due to its non-toxicity and degradability in nature with no harmful effect. As a therapeutic delivery entity, chitosan NPs have drawing attention towards their vast relevance in, such asstoring protein therapeutics, genetic and adverse tumor chemical therapeutics by means different path ways often intake such as oral, nasal and intravenous. The affected site specific delivery of this chitosan therapeutic conveyance entity was exceptionally greater due to the positive charge of nanoparticles, which give them an advantage on drawing attention to the negatively charged cell membranes. The hydrobhobic nature of chitosan influences the efficient encapsulation of hydrophilic therapeutics into the chitosan Nps.
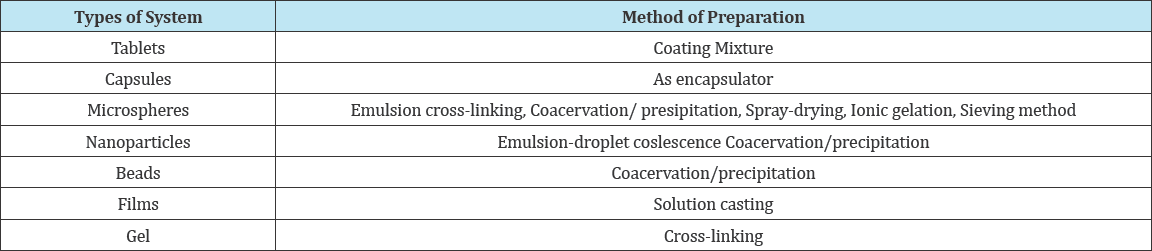
Due to the better bio adhesive functionality and capability to control the discharge of the active constituents, it was utilized in topical conveyance system. Chitosan shows cationic nature due to its primary amino groups which define many of the characteristics, like disciplined drug discharge, in situ gelation transfection, and permeation enlargement and efflux pump inhibitory properties used in therapeutic conveyance system. In gene delivery, chitosan NPs can inter-react with negatively charged DNA and transform into a polyelectrolyte complex. Nuclease degradation was ineffective when DNA was in this complexes leading to better transfection efficiency. Storing considerable amounts of anti-cancer therapeutic using chitosan NPs is crucial for outstanding results in cancer treatment. Absorption and bioavailability of drug encapsulated into chitosan NPs can be improved, so they can be used to deliver gene drugs, protein drugs and can protect them effectively from enzyme degradation in-vivo. Blood capillary was capable enough to administrate chitosan intravenously, the bio distribution of NPs can vary confide in the size, surface charge, and hydrophobicity of the administrated particles. Chitosan acts as an inactive ingredient added to tablet or capsule in order to control the release of the drug .The elimination of chitosan after the delivery was easy through renal clearance, however, this applies for chitosan with suitable molecular weight. Enzyme degradation is required for very large molecular weight chitosan. Previously numerous investigations has been made known the biomedical applications of chitosan NPs. Lu et al. [22], synthesized copolymer of poly (L-lysine) with chitosan and studied its efficiency in relation to plasmid DNA adherence capability as well as gene transfection effect in HEK 293T cell with pristine chitosan polymer. Synthesised chitosan- montmorillonite NPs stored with isoniazid therapeutic and investigated the influence of particle size on the release characteristics. They found that swelling and release of isoniazid from the NPs were found to increase with a decrease in the PH of the medium and the size of the NPs. They also found that mucoadhesive property of NPS was superior in gastric pH and expanded with lowering in particle size. Agnihotri, et al. [26] expressed the chemical modification of chitosan or its derivatives and the application of its micro and NPs in drug delivery Table 1 shows the different chitosan based drug delivery.
Table 1: Chitosan based drug delivery systems prepared by different methods [34].
Antimicrobial activity
Even with the enormous advancement in antimicrobial of the antimicrobial systems and suboptimal pharmacological progression, Distinct contagious inflammation still left which is challenging us with no cure, because of a cluster of reasons such as the evolution and advancement of resistant clones, the insufficiency of the antimicrobial systems and suboptimal pharmacological characteristics of the ongoing antimicrobial structures, which sometimes are difficult to reach active concentrations inside bacterial strains or in some body sites. Chitosan is a promising biocompatible and biodegradable natural polymer shows unique antibacterial activity Zheng et al. [62].
The antimicrobial activity is due to the fact that chitosan is a cationic polyelectrolyte polymer. Low-molecular-weight chitosan can go through microbial cellular areas, bind with DNA and limit DNA interpretation and mRNA functions, while high-molecular- weight chitosan can merge to the negatively charged components on the microbial cellular areas. It forms an impermeable layer around the cell, changes cell permeability and blocks transport into the cell., Microbes are capable of hooking to the exterior of chitosan NPs drastically in the small instance of just 30 minutes; thus chitosan NPs displays antimicrobial activity [63].
The degree of chitosan polymerization and type of chitosan affects the antimicrobial activity. Establish that chitosan NPs demonstrates decent antibacterial activity contradicts to a numerous amount of gram negative and gram positive bacteria [51], patterned the antimicrobial activity of copper loaded NPs and chitosan NPs [42]. They discovered that chitosan NPs displays superior antimicrobial activity than copper loaded NPs. Chitosan NPs express advanced interaction with gram negative bacteria due to the hydrophilicity and negative charge on the cell surface than those of gram-positive bacteria, which resulted in stronger antibacterial activity against them.
It was also stated that the quantity of chitosan binding to the bacterial cell wall was reliant on the surrounding pH value, molecular weight and degree of acetylation of chitosan. Low environmental pH increases the positive charge in the chitosan polymer, which drawing towards to the bacterial cell wall more often. Antifungal activity of chitosan varies with the fungus due to the effects of molecular weight and degree of acetylation of chitosan. Chitosan exhibit antifungal activity against several phytopathogenicfungi like Penicillium sp. in citrus fruit, Botrytis cinerea in cucumber plants, Phytophthorainfestans, Alternariasolani and Fusariumoxysporum in tomatoes etc. A permeable chitosan film formed on the crop surface which interfered with the fungal growth and activated several defense processes like chitinase accumulation, proteinase inhibitor synthesis, callus synthesis and lignification, is the suggested mechanism involved in the activity.
Chitosan as an anticancer agent
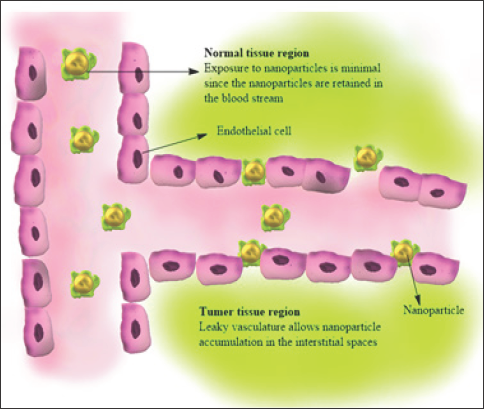
Although there is considerable medical progress in medical research, cancer is still one of the high ranking causes of death in the world. Conventional therapy has several disadvantages. A major problem is an inability to administrate therapeutic agents selectively to the target sites without adverse effects on healthy tissues [60]. Successful cancer management depends on accurate diagnostics along with specific treatment protocols. Nanoparticles are the colloidal carrier with size on the nanoscale. They are particularly attractive for cancer treatment due to small size, varied composition, surface functionalization, and stability. Recent studies have shown that chitosan and its derivatives exhibited antitumor activity in both in vitro and in vivo models. Chitosan nanoparticles can act on tumor cells directly to interface with cell metabolism, inhibit cell growth, or induce cell apoptosis. Tokoro and co workers found that the antitumor effect of chitosan derivatives is due to the increase in secretion of interleukin-1 and 2 which caused maturation and infiltration of cytolytic T-lymphocytes. It also has an antitumor role through improving the body's immune function. Long circulating macromolecules (polymer drug conjugates) and nanosized particulates (such as micelles and liposomes) accumulate passively at the tumor due to the Enhanced permeability effect (EPR effect).Some studies revealed that chitosan involved in the direct killing of tumor cells by inducing apoptosis as shown in Figure 5 the nanoparticle will accumulates on the leaky vasculature.
Figure 5: Antitumor activity of chitosan NPs.
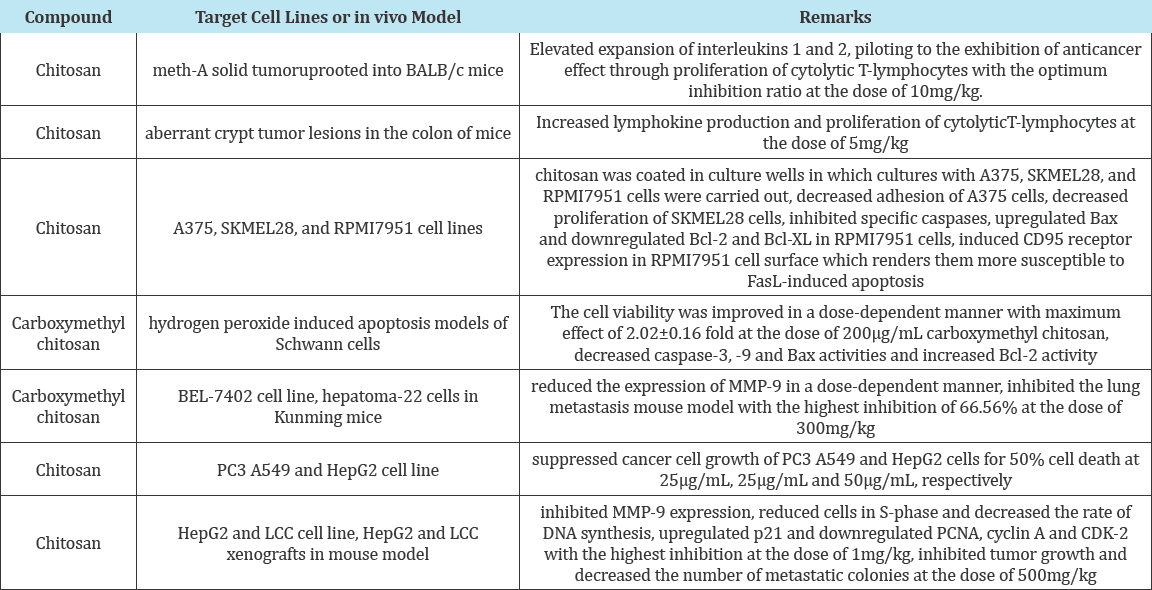
Chitosan oligosaccharide- arachidic acid conjugate has been successfully prepared and used for the development of selfassembled NPs for doxorubicin, Table 2 shows the rest of chitosan derivatives as antitumouragents . The biodegradable polymer- drug conjugate of doxorubicin conjugated with a stearic acid grafted chitosan nano-sized oligosaccharide showed high efficiency for cellular uptake and found efficient on the suppression of tumor growth [61].
Table 2: A summary of antitumor activities of chitosan and its derivatives [75].
Chitosanfor tissue engineering
In bone tissue engineering: Bone tissue engineering is a research field which develops new 3D scaffolds with highly interconnected porous structures. These scaffolds should match the characteristics of tissues that we have replaced. These tissue engineering scaffolds should contain some properties like, biocompatibility, osteoinductive, osteoconductive and mechanically well-suited in order to restore bone which has been lost or damaged. Santo et al. [24], synthesized hybrid scaffolds based on poly (D, L- lactic acid) incorporated with chitosan chondroitin sulfate NPs with drug release capabilities due to the presence of chitosan /chondroitin sulfate NPs, these scaffolds show increased swelling properties and adequate mechanical properties for cell adhesion and support for bone tissue engineering. Hybrid scaffolds based on calcium phosphate and chitosan have been used as bone graft substitutes because of their in-situ setting ability and bioactivity. Meng et al. [25] synthesized bioactive cement based on CPC (chitosan- polyglactinfiber scaffold) containing chitosan nanospheres as an injectable material for the bone regeneration [64]. They found that these scaffolds increase the formation of more new bones and degradation of the bioactive cement in the bone defects. Mansur and costa studied the physical, mechanical, biological properties of hybrid scaffolds containing poly (vinyl alcohol), chitosan and bioactive glass. The results show that these hybrid scaffolds can use for bio tissue engineering due to their mechanical, morphological and cell viability properties.
In cartilage tissue engineering: In recent years, different chitosan- based biomaterials have been developed as scaffolds for cartilage tissue engineering because of its biocompatibility, high porous structure, non- cytotoxicity, flexibility and elasticity. Kim et al. [65], stabilized transforming growth factor (TGF-^1) signalling in the chitosan hydrogel for use in cartilage regeneration. They found that TGF-^1 linked chitosan promotes cartilage regeneration in a rat partial- thickness chondral defect model was confirmed. Silva et al. [66] synthesised chitosan and chondroitin sulphate 3D nanostructures to support the attachment and proliferation of bovine chondrocytes. The results show that cells were attached, proliferated and metabolically active over the entire 3D chitosan and chondroitin sulfate nanostructure scaffolds.
Anti-inflammatory effects and repair of arthritic tissue: Chitosan can neutralize the gastric acids due to the alkaline nature of the free amino group present in it. It forms a shielding membrane in the stomach, so chitosan can be used to cure acid indigestion and peptic ulcer. The anti-inflammatory mechanism of chitosan is because of the acid hydrolysis of chitosan to glucosamine hydrochloride or its sulfate, phosphate and other salt preparation by salt conversion. These monosaccharides found in connective tissues and cartilages are the structural units of the proteoglycans. These tissues can be restored and regenerated by absorbing these monosaccharides when they are damaged or inflamed [67].Therefore, these monosaccharides are an active treatment for the prevention and curing of rheumatoid arthritis, as well as bone hyperplasia. They are also used as antiarthritic drugs in clinical practice. Moreover, contrasting with some usual anti-arthritic steroidal anti-inflammatory drugs or analgesic and anti-inflammatory drugs, these monosaccharides have no harmful side effects or deadliness and can be taken for a long period. The experiments pointed that taking glucosamine for two weeks can eliminate arthritic pain and improve movement in patients suffering from severe arthritis.
Chitosan for wound healing: Wound healing is a pathway of biologically regulated reactions allied to the general phenomenon of growth and tissue regeneration. It progresses through a series of stages in which different cellular and matrix components act together to re-establish the damaged and replacement of lost tissues [68]. Many studies have been reported about the beneficial effects of chitin and chitosan for wound healing using in-vitro, animal and clinical studies. The effects of chitin and its derivatives on the proliferation of fibroblast and on the synthesis of cytokinesis in vitro. They found that cell proliferation is accelerated indirectly by chitin and its derivatives when these materials are used invivo. The effect of chitin and chitosan NPs with different degree of deacetylation (DDA) but the similar molecular weight on the proliferation of skin fibroblast and keratinocytes in-vitro. Their results reveal that chitosan with relatively high DDA (89%) strongly stimulated fibroblast proliferation while samples with lower DNA showed less activity.
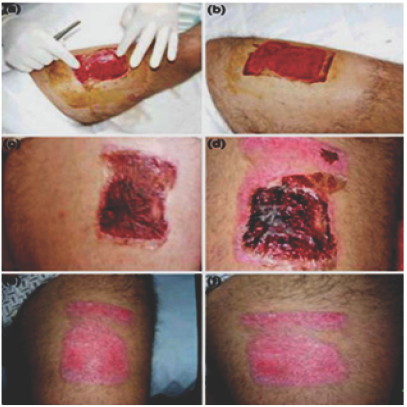
The composite films made of chitosan acetate exhibits properties leading to wound healing. Some medical reports regarding the wound healing are given below. The report seems positive for the effective healing of wounds caused by burning, infection etc. The Figure 6a shows the film set up previous compressive hemostasis, Figure 6b shows the film after application. The Figure 6c shows the third-day evolution. An absence of liquid collections and the presence of clots in the film are observed. On Figure 6d it is possible to see the sixth-day evolution, there is partial epithelialization and some partial film falls off is observed. The Figure 6e shows a great degree of epithelialization. Figure 6f the injury after 14th-day evolution [49].
Figure 6: Films application on a tattoo removal.
6a: The film setup
6b: The film after the application
6c: The third day evolution
6d: The sixth day evolution6(e,f): Fourteenth day of evolution [69].
Chitosan NPs in gene delivery and bioimaging
The cellular delivery of nucleic acids is the foundation of gene therapy and it has potential to treat many currently incurable diseases. Chitosan NPs shows many features of an ideal gene delivery system. Chitosan-DNA NPs formulation readily done by coacervation between positively charged amino groups on the chitosan and negatively charged phosphate groups on the DNA resulting increases in stability and enhancing gene transfer properties. Application of nano-chitosan in bioimaging is gaining rapid attention due to its biocompatible properties. Nie, et al. [69] studied the effects of chitosan NPs on bioimaging. They have checked the effect of quantum dot for bio detection and bio imaging. They also incorporated the QDs into the chitosan NPs and checked the efficiency of both media on bio imaging. They found that the fluorescence of quantum yield of QDs in chitosan is found to be 11.8% higher than that of free QDs.
Currently, polymer-inorganic hybrid nano spheres have attracted growing attention because of the synergic homes arising from each the compound nanospheres and inorganic nanomaterials, significantly those that square measure biocompatible and capable of emitting light area unit considerably useful in statement nanosphere journey in cell and frame, detection illness websites and reportage medical helpful resource reaction. Compared with natural fluorescent dyes, the inorganic light-emitters similar to quantum dots (QDs) showcase extra sturdy fluorescence intensity, larger photostability, a broader excitation wavelength vary, and a narrower spectrum [4] so, thorough analysis has focused on the utility of QDs as imaging dealers for multi-coloured imaging cells, tissues, and animals [4-7] but, the scientific space of experience and clinical applications of QDs region unit restricted by suggests that of their unhealthy biocompatibility and instability in various physiological environments. Several ways of vicinity unit being advanced to beat these boundaries. The foremost wealthy approach has been to coat QDs with biocompatible soluble polymers, on the thanks to growth QDs mixture stability in resolution and organic surroundings, and improves their bio compatibility.
Molecular imaging permits the noninvasive assessment of biological and organic chemistry processes in living subjects. Such technologies thus have the potential to reinforce our understanding of unwellness and drug activity throughout pre symptomatic and clinical drug development. Molecular imaging permits a repetitive and noninvasive study of similar living subject exploitation identical or different biological imaging assays at completely different time points, therefore harnessing the applied math power of longitudinal studies, and reducing the rate of animals needed and value. Chitosan could be a hydrophilic and non-antigenic biopolymer and contains a low toxicity toward class cells. Hence, it's nice potential as a biomaterial owing to its glorious biocompatibility. Conjugated to further materials, chitosan composites end in a replacement category of biomaterials that possess mechanical, physicochemical and practical properties that have potential to be used in advanced medical imaging applications. Chitosan [poly (1, 4-p-D-glucopyranosamine)], an abundant natural biopolymer, is made by the deacetylation of polyose obtained from the shells of crustaceans. It's a polycationic chemical compound that has one group and 2 hydroxyl radical teams within the continuance hexosamine residue. Chitosan has nice potential as a biomaterial owing to its bio-compatible properties. It hydrophilic, nonantigenic and contains a low toxicity toward class cells. Additionally, chitosan is thought to facilitate drug delivery across cellular barriers and transiently open the tight junctions between animal tissue cells. polyose and chitosan area unit aminoglucopyrans composed of N-acetylglucosamine (GlcNAc) and glucosamine (GlcN) residues. These polysaccharides are renewable resources which are currently also being explored intensively for their applications in pharmaceutical, cosmetics, biomedical, biotechnological, agricultural and food industries [68,70].
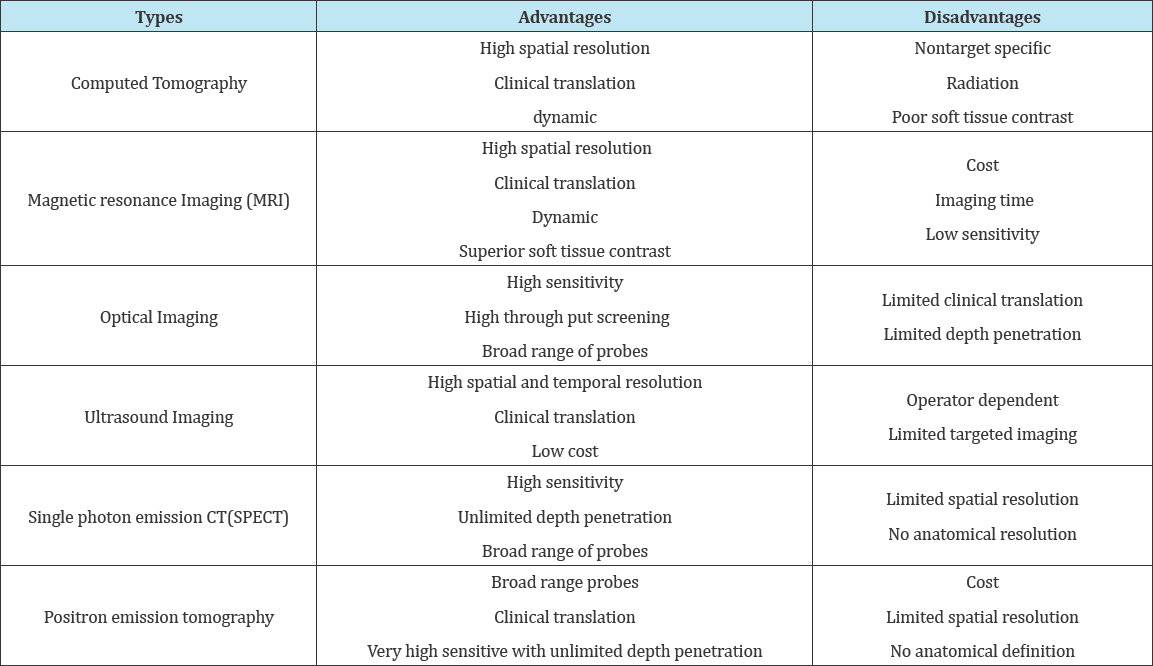
The goal of molecular imaging is to check biological processes noninvasively. Molecular imaging plays a vital role in an effort the challenges of the characterization of biological processes at the cellular level in living objects. Most of the diagnostic techniques that area unit applied for routine clinical use have a counterpart within the experimental analysis setting. Hence, it's attainable to style pre symptomatic experiments that not solely facilitate to outline the clinical protocol, however, can even give a mechanistic understanding of the discovered biological response. Molecular imaging holds the promise of the noninvasive assessment of biological and organic chemistry processes in living subjects. Since the beginning of X-ray technology for medical imaging, several noninvasive methodologies are unreal and with success used for applications starting from clinical identification to analysis in cellular biology and drug discovery. Such technologies so have the potential to reinforce our understanding of malady and drug activity throughout pre symptomatic and clinical drug development. The advantage of molecular imaging techniques over a lot of standard readouts (e.g. immunohistochemistry) is that they will be performed within the intact organism with spare abstraction and temporal resolution for learning biological processes in vivo. Moreover, molecular imaging permits a repetitive and noninvasive study of equivalent living subject exploitation identical or various biological imaging assays at completely different time points, so harnessing the applied math power of longitudinal studies, and reducing the rate of animals needed and price. Imaging techniques have potential impact in the day-after-day activities of the medicine mortal. Every imaging modality has bound benefits Moreover as limitations, and also the alternative of associate degree imaging modality, or a combination of techniques, is decided by the particular biological queries being asked. Every modality has its own characteristics in terms of sensitivity and determination, complexity, time of knowledge acquisition and money price. Most of the techniques exhibit helpful overlap with one another. However, there's no single modality that matches all applications [70] (Table 3).
Table 3: Different types of molecular imaging.
Molecular imaging techniques normally want a distinction agent to report on the organic process of interest. These imaging agents' square measure ordinarily classified in 2 main classes, i.e. endogenous and exogenous agents. Endogenous agents are either naturally gift within the tissue of interest, or they’re made in place following genetic manipulation. Exogenous distinction agents square measure injected, in most cases intravenously, and aren’t intrinsic to biological tissues. Endogenous agents the foremost ordinarily utilized newsperson systems square measure the fluorescent proteins, similar to inexperienced fluorescent macromolecule (GFP) (and its mutants) and also the luciferin/ luciferase light system, each of that square measure introduced into cells of interest via molecular biological techniques. These systems will either be constitutively expressed or their expression is created to depend upon the presence or absence of alternative factors. GFP proteins need no any substances or substrates to figure. Though fluorescent proteins provide wonderful and really numerous opportunities for extracting molecular and cellular level info in tiny animals, they presently haven't any role in clinical applications . On the opposite hand in luminescence imaging, a substrate (typically luciferin) is run to associate in a nursing animal that has been designed to provide the luciferase protein specified once the substrate and protein meet, the animal pigment changes. This results in the emission of light, so sanctionative detection of sites of luciferase expression. Bioluminescence is accustomed overcome a number ofthe problems encountered with endogenous fluorophores and has promising applications in vivo tracking of injected cells. However, current drawbacks embody point uncertainty of sunshine emitting cells thanks to nonhomogeneous scattering, light-weight penetration problems and a lucid want for a stable expression of the optical reporter. Like mixture particles used for drug delivery, imageable nanoparticles are conjugated with ligands to focus on them to a molecular marker of interest, permitting indirect marker detection by totally different imaging techniques. What is more, pH- and temperature-sensitive nanoparticles is accustomed visualize regional variations in these parameters with trendy imaging techniques. Self-assembling nanoparticles are usually composed of copolymers with each hydrophobic and hydrophilic segment. The core of the self-assembled nanoparticles provides an efficient loading compartment for several hydrophobic and hydrophilic drugs, fluorescent probes, and distinction agents, whereas the shell improves the suspension stability of nanoparticles in solution [68].
Chitosan is an exemplary biopolymer in biological applications due to its biocompatible properties. it's a natural polycationic chemical compound and composed of D-glucosamine and N-acetyl- D-glucosamine joined by b-(1.4)-glycosidic bonds, and so has one free radical and 2 free radical teams within the repetition hex- osaminide residue. This cluster may be changed with hydrophobic segments to enhance the self-assembling capabilities by increasing unit hydrophobic interactions between segments. Conjugated to further materials, chitosan composites end in a replacement category of biomaterials that possess mechanical, chemistry and useful properties that cannot be achieved either by native chitosan or the incorporated material alone. The incorporation of laborious and brittle phosphate or hydroxyapatite into chitosan yields a bioresorbable composite with favourable mechanical properties for bone and gristle tissue engineering. Moreover, incorporation of imaging agents like Fe3O4 for imaging into the self-assembled nanoparticles may enhance hepatocyte-targeted imaging and also the particle may serve as MR molecular imaging agent. Numerous inorganic materials, as well as metals, may be incorporated within the chitosan composite preparations and their combined characteristics have tried using for many biomedical applications. Chitosan polyion advanced composites may be prepared by interactions of chitosan with natural and artificial polyanion molecules. Making ready fluorescent chitosan quantum dot composites permits the mixture of targeted drug and sequence delivery with optical imaging. Polyacrylic acid (Carbopol), AN anionic artificial chemical compound having mucoadhesive properties, is extensively used with chitosan to make polymer composites, which have longer circulation times in vivo, leading to higher bioavailability of incorporated therapeutic agents. The latter composites also can be created to contain distinction agents for imaging functions. The power of chemical element-loaded chitosan bubbles to market the exchange of oxygen within the presence people. Oxygen delivery is increased by sonication and each frequency and time period people affected the exchange dynamics [69].
Lee et al. [71] have developed novel self-assembling nanoparticles composed of amphipathic soluble chitosan-linoleic acid (WSC- LA) conjugates for encapsulation of SPIOs as a distinction agent to focus on hepatocytes. The WSC-LA conjugates self-assembled into core-shell structures in solution. The SPIO clusters fashioned within the hydrophobic core had an awfully high imaging relaxivity. The T2-weighted mister pictures of the liver showed a symptom drop of sixty-nine.2, 78.2, and 62.9% at thirty min, 1 h, and a couple of h when injection of the nanoparticles, severally. These SPIO-loaded WSC-LA nanoparticles may be used as a distinction agent to assist within the diagnosing of viscus diseases [72].
Saha et al. [72] discussed the success ready biocompatible chitosan nano particles encapsulating each CdSe-ZnS QDs and Gd- DTPA. These multifunctional nano beads may be used as fluorescent biomarkers and additionally as distinction agents for imaging. The careful characterization was done by the authors incontestible that their chitosan-based nanobeadsare spherical, concerning fifty nm in size and possess a far improved quantum yield still as favorablerelaxivity of the Gd-DTPA. The 3MPA-modified soluble CdSe/ZnS-3MPA QDs exhibit AN intense, slim spectrum with a peak at 565 nm. Similarly, nanobeads encapsulating each QDs ANdGd- DTPA possess an equally intense and slim spectrum, with its peak position at 567nm. These multifunctional chitosan nanobeadsare promising for future medical specialty applications. Hwang et al. loaded HGC nanoparticles with the antineoplastic drug docetaxel (DTX) employing a qualitative analysis technique, and also the ensuing docetaxel-loaded HGC (DTX-HGC) nanoparticles had a mean diameter of 350 nm in solution. The authors showed that these nanoparticles were well spread and stable for two weeks in vitro beneath physiological conditions (pH 7.4 and 37 °C) with a sustained drug unharness profile. The DTX-HGC nanoparticles were moderately stable within the presence of excess bovine albumen that steered that these nanoparticles may also be stable within the blood stream. The authors additionally evaluated the time- dependent excretion profile, in vivo biodistribution, the circulation time, and tumor targeting ability of DTX-HGC nanoparticles by employing a non-invasive live animal imaging technology. Beneath best conditions for cancer medical care, the DTX-HGC nanoparticles showed higher anticancer effectuality like reduced tumor volume and exaggerated survival rate in A549 carcinoma cells-bearing mice and powerfully reduced antineoplastic drug toxicity compared thereto of free DTX [147]. Ex vivo labeling of cells with nanoparticles for ensuant in vivo detection is of huge interest to varied medical specialty applications. To check these cells, Ge Y et al. [69]. ready novel fluorescent/magnetic nanoparticles coated with changed chitosan possessing a magnetic iron compound core and a covalently hooked up dye . So as to localize the magnetic nanoparticles within the cells, the authors performed microscopy and microscopy. The practicability and potency in labeling cancer cells (SMMC-7721) was additionally evaluated, showing a high level of incorporation, as incontestible by flow cytometry and resonance imaging. These new magneto-fluorescent nano agents so have the potential for advanced bio imaging studies [70].
Gd-DTPA could be a widely used imaging distinction agent. Since its incorporation in nanoparticles, its potential for in vivo molecular imaging applications has exaggerated enormously. in an exceedingly similar fashion, chitosan-based Gd-nanoparticles are ready by incorporating Gd-DTPA victimization the emulsion-droplet union technique. Their unharness properties ANd their ability for long retention of Gd-DTPA within the tumor indicated that these Gd nanoparticles could be helpful as an i.t. inject able device for metal neutron-capture medical care (Gd-NCT). The extent of Gd loading was totally different for various forms of chitosans utilized in the preparation. The very best Gd load was achieved with a hundred deacetylated chitosan in V-J Day Gd-DTPA solution, and also the particle size was 452 nm, whereas chitosan with lower deacetylation level made abundant larger particles with weakened Gd-DTPA content. Similarly, CMS-Gd-DTPA ready at Gd-DTPA: chitosan ratios of one.5:1 and 1.7:1 had fairly regular, spherical shapes but, all CMS-Gd-DTPA ready at a magnitude relation of 15:1 Gd-DTPA: chitosan were nonspherical, rough-faced, folded and/or aggregate. Method improvement expedited production of gadolinium-loaded chitosan nanoparticles with an especially high Gd-DTPA content (45.3%) and an acceptable size for injection (452nm). Adolinium- loaded chitosan nanoparticles displayed prolonged retention in tumor tissue when in vivo intratumoral injection. Kumar et al. [73] additionally represented the chemistry and preparations of Holmium-166 and Samarium-153 chitosan complexes that are primarily suited for pharmaceutical applications [74].
References
- Nasti A, Zaki NM, Leonardis PD, Ungphaiboon S, Sansongsak P, et al. (2009) Chitosan/TPP and chtiosan/TPP-hyaluronic acid nanoparticles: systematic optimization of the preparative process and preliminary biological evaluation. Pharm Res 26(8): 1918-1930.
- Thanou M, Verhoef JC, Junginger HE (2001) Chitosan and its derivatives as intestinal absorption enhancers. Adv Drug Deliv Rev 50 Suppl 1: S91-S101.
- Agnihotri SA, Aminabhavi TM (2004) Controlled release of clozapine through chitosan microparticles prepared by a novel method. J Control Release 96(2): 245-259.
- Kim IY, Seo SJ, Moon HS, Yoo MK, Park IY, et al. (2008) Chitosan and its derivatives for tissue engineering applications. Biotechnol Adv 26(1): 1-21.
- Stavroula G, Nanaki, Ioannis A, Koutsidi, Ioanna K, et al. (2012) Miscibility study of chitosan/2-hydroxyethyl starch blends and evaluation of their effectiveness as drug sustained release hydrogels. Carbohyd Polym 87(2): 1286-1294.
- Tiyaboonchai W (2003) Chitosan nanoparticles: A promising system for drug delivery. Naresuan University J 11(3): 51-66.
- Bamrungsap S, Zhao Z, Chen T, Wang L, Li C, et al. (2012) Nanotechnology in therapeutics: a focus on nanoparticles as a drug delivery system. Nanomedicine 7(8): 1253-1271.
- Zargar V, Asghari M, Dashti A (2015) Review on chitin and chitosan polymers: structure, chemistry, solubility, derivatives, and applications. Chem Bio Eng Rev 2(3): 204-226.
- Rodrigues S, Dionisio M, Lopez CR, Grenha A (2012) Biocompatibility of chitosan carriers with application in drug delivery. J Funct Biomat 3(3): 615-641.
- Patel MP, Patel RR, Patel JK (2010) Chitosan mediated targeted drug delivery system: A review. J Pharm Pharm Sci 13(4): 536-557.
- Wang JJ, Zeng ZW, Xiao RZ, Xie T, Zhou GL, et al. (2011) Recent advances of chitosan nanoparticles as drug carriers. Int J Nanomed 6: 765-774.
- Zheng LY, Zhu JF (2003) Study on antimicrobial activity of chitosan with different molecular weights. Carbohydr Polym 54(4): 527-530.
- Termsarasab U, Cho HJ, Kim DH, Chong S, Chung SJ, et al. (2013) Chitosan oligosaccharide-arachidic acid-based nanoparticles for anti-cancer drug delivery. Int J Pharm 441(1-2): 373-380.
- Boateng JS, Matthews KH, Stevens HNE, Eccleston GM (2008) Wound healing dressings and drug delivery systems: A Review. J Pharm Sci 97(8): 2892-2923.
- Malmo J, Varum KM, Strand SP (2011) Effect of chitosan chain architecture on gene delivery: comparison of self-branched and linear chitosans. Biomacromolecules 12(3): 721-729.
- Su Y, Hu Y, Du Y, Huang X, He J, et al. (2015) Redox-responsive polymer- drug conjugates based on doxorubicin and chitosan oligosaccharide-g- stearic acid for cancer therapy. Mol Pharm 12(4): 1193-1202.
- Tripathy S, Das S, Chakraborty SP, Sahu SK, Pramanik P, et al. (2012) Synthesis, characterization of chitosan-tripolyphosphate conjugated chloroquine nanoparticle and its in vivo anti-malarial efficacy against rodent parasite: A dose and duration dependent approach. Int J Pharm 434(1-2): 292-305.
- Peniche H, Peniche C (2011) Chitosan nanoparticles: a contribution to nanomedicine. Polymer Int 60(6): 883-889.
- Shi SF, Jia JF, Guo XK, Zhao YP, Chen DS, et al. (2012) Biocompatibility of chitosan-coated iron oxide nanoparticles with osteoblast cells. Int J Nanomedicine 7: 5593-5602.
- Cao J, Zhou NJ (2005) Progress in antitumor studies of chitosan. Chin J Biochem Pharm 26(2): 127.
- Qi L, Xu Z (2006) In vivo antitumor activity of chitosan nanoparticles. Bioorg Med ChemLett 16(16): 4243-4245.
- Yu H, Chen X, Lu T, Sun J, Tian H, et al. (2007) Poly(L-lysine)-graftchitosan copolymers: synthesis, characterization, and gene transfection effect. Biomacromolecules 8(5): 1425-1435.
- Duncan R (2003) The dawning era of polymer therapeutics. Nat Rev Drug Discov 2(5): 347-360.
- Santo VE, Duarte ARC, Gomes ME, Mano JF, Reis RL (2010) Hybrid 3D structure of poly (D,L- lactic acid) loaded with chitosan/chondroitin sulfate nanoparticles to be used as carriers for bio macromolecules in tissue engineering. J supercrit fluids 54(2010): 320-327.
- Meng D, Dong L, Wen Y, Xie Q (2015) Effects of adding restorable chitosan microspheres to calcium phosphate cements for bone regeneration. Mater Sci Eng C Mater Biol Appl 47: 266-272.
- Agnihotri AS, Mallikarjuna NN, Aminabhavi TM (2004) Recent advances on chitosan-based micro- and nanoparticles in drug delivery. Journal of Controlled Release 100(1): 5-28.
- Rinki K, Dutta PK, Hunt AJ, Macquarrie DJ, Clark JH (2011) Chitosan aerogels exhibiting high surface area for biomedical application: preparation, characterization, and antibacterial study. Int J Poly Mat & Poly Biomat 60(12): 988-999.
- Hsu SC, Don TM, Chiu WY (2002) Free radical degradation of chitosan with potassium persulfate. Polym Degrad Stab 75(1): 73-83.
- Zoldners J, Kiseleva T, Kaiminsh I (2005) Influence of ascorbic acid on the stability of chitosan solutions. Carbohydr Polym 60 (2): 215-218.
- (2009) KEGG Pathway, Amino sugar and nucleotide sugar metabolism, Kanehisa Laboratories, Japan.
- Kean T, Thanou M (2011) Chitin and chitosan-sources, production and medical applications. In: Williams PA, Arshady R (Eds.), Natural Products Desk Reference, CRC Press, India.
- (2009) Polymers, their sources, chemistry and applications, Kentus Books, London, pp. 327-361.
- Funkhouser JD, Aronson NN (2007) Chitinase family gh18: evolutionary insights from the genomic history of a diverse protein family. BMC Evol Biol 7: 96.
- Huet J, Rucktooa P, Clantin B, Azarkan M, Looze Y, at al. (2008) X-ray structure of papaya chitinase reveals the substrate binding mode of glycosyl hydrolase family 19 chitinases. Biochemistry 47: 8283-8291.
- Collinge DB, Kragh KM, Mikkelsen JD, Nielsen KK, Rasmussen U, et al. (1993) Plant chitinases. Plant J 3(1): 31-40.
- Kasprzewska A (2003) Plant chitinases-regulation and function. Cell Mol Biol Lett 8(3): 809-824.
- Schipper G, Varum KM, Artursson P (1996) Chitosans as absorption enhancers for poorly absorbable drugs 1: influence of molecular weight and degree of acetylation on drug transport across human intestinal epithelial (caco-2) cells. Pharm Res 13(11): 1686-1692.
- Yang YM, Hu W, Wang XD, Gu XS (2007) The controlling biodegradation of chitosan fibers by n-acetylation in vitro and in vivo. J Mater Sci Mater Med 18(11): 2117-2121.
- Xu J, McCarthy SP, Gross RA, Kaplan DL (1996) Chitosan film acylation and effects on biodegradability. Macromolecules 29(10): 3436-3440.
- Correa JU, Elango N, Polacheck I, Cabib E (1982) Endochitinase, a mannan-associated enzyme from Saccharomyces cerevisiae. J Biol Che 257: 1392-1397.
- Molinari LM, Pedroso RB, ScoarisDde O, Nakamura TU, Nakamura CV, et al. (2007) Identification and partial characterisation of a chitinase from Nile tilapia, Oreochromis niloticus. Comp Biochem Physiol B Biochem Mol Biol 146: 81-87.
- Boot RG, Blommaart EF, Swart E, Vander Vlugt GK, Bijl N, et al. (2001) Identification of a novel acidic mammalian chitinase distinct from chitotriosidase. J Biol Chem 276(9): 6770-6778.
- Hartl D, He CH, Koller B, Da Silva CA, Homer R, et al. (2008) Acidic mammalian chitinase is secreted via an adam17/epidermal growth factor receptor-dependent pathway and stimulates chemokine production by pulmonary epithelial cells. J Biol Chem 283(48): 3347233482.
- Zhu Z, Zheng T, Homer RJ, Kim YK, Chen NY, et al. (2004) Acidic mammalian chitinase in asthmatic Th2 inflammation and IL-13 pathway activation. Science 304(5677): 1678-1682.
- Stirling JL (1974) Nn'-diacetylchitobiase activity in tay-sachs disease and sandhoff's disease. Biochem J 141(2): 597-599.
- Kuranda MJ, Aronson NN (1986) A di-n-acetylchitobiase activity is involved in the lysosomal catabolism of asparagine-linked glycoproteins in rat liver. J Biol Chem 261: 5803-5809.
- Hollak CE, Weely SV, Van Oers MH, Aerts JM (1994) Marked elevation of plasma chitotriosidase activity. a novel hallmark of gaucher disease. J Clin Invest 93(3): 1288-1292.
- Renkema GH, Boot RG, Muijsers AO, Donker-Koopman WE, Aerts JM (1995) Purification and characterization of human chitotriosidase, a novel member of the chitinase family of proteins. J Biol Chem 270(5): 2198-2202.
- Malaguarnera L (2006) Chitotriosidase: the yin and yang. Cell Mol Life Sci 63(24): 3018-3029.
- Thanou M, Verhoef JC, Junginger HE (2001) Oral drug absorption enhancement by chitosan and its derivatives. Adv Drug Delivery Rev 52(2): 117-126.
- Illum L (1998) Chitosan and its use as a pharmaceutical excipient. Pharm Res 15(9): 1326-1331.
- Wedmore JG, McManus, Pusateri AE, Holcomb JB (2006) A special report on the chitosan-based hemostatic dressing: experience in current combat operations. J Trauma 60(3): 655-658.
- Carreño-Gómez B, Duncan R (1997) Evaluation of the biological properties of soluble chitosan and chitosan microspheres. Int J Pharm 148(2): 231-240.
- Sgouras D (1990) The evaluation of biocompatibility of soluble polymers and assessment of their potential as specific drug delivery systems, in Pharmacy, Keele, University of Keele, UK.
- Wan KW, Malgesini B, Verpilio I, Ferruti P, Griffiths PC, et al. (2004) Poly (amidoamine) salt form: effect on ph-dependent membrane activity and polymer conformation in solution. Biomacromolecules 5: 1102-1109.
- Schipper NG, Varum KM, Stenberg P, Ocklind G, Lennernas H, et al. (1999) Chitosans as absorption enhancers of poorly absorbable drugs. 3: influence of mucus on absorption enhancement. Eur J Pharm Sci 8(4): 335-343.
- Thanou M, Verhoef JC, Romeijn SG, Nagelkerke JF, Merkus FW, et al. (1999) Effects of n-trimethyl chitosan chloride, a novel absorption enhancer, on caco-2 intestinal epithelia and the ciliary beat frequency of chicken embryo trachea. Int J Pharm 185(1): 73-82.
- Cárdenas G, Anaya P, von Plessing C, Rojas C, Sepúlveda J (2008) Chitosan composite films. Biomedical applications. J Mater Sci Mater Med 19(6): 2397-2405.
- Landegren U (1984) Measurement of cell numbers bymeans of the endogenous enzyme hexosaminidase: applications to detection of lymphokinesand cell surface antigens. J Immunol Methods 67(2): 379388.
- Mao S, Shuai X, Unger F, Wittmar M, Xie X, et al. (2005) Synthesis, characterization andcytotoxicity of poly(ethylene glycol)-grafttrimethyl chitosan block copolymers. Biomaterials 26(32): 6343-6356.
- Ruoslahti E, Bhatia SN, Sailor MJ (2010) Targeting of drugs and nanoparticles to tumors. J Cell Biol 188(6): 759-768.
- Zheng, Lian-Ying, Zhu JF (2003) Study on antimicrobial activity of chitosan with different molecular weights. Carbohydrate polymers 54(4): 527-530.
- Prashanth, Harish KV, Tharanathan RN (2007) Chitin/chitosan: modifications and their unlimited application potential-an overview Trends in food science & technology 18(3): 117-131.
- Li Z, Hassna RR, Hauch KD, Xiao D, Zhang M (2005) Chitosan-alginate hybrid scaffolds for bone tissue engineering. Biomaterials 26(18): 39193928.
- Kim KJ, Li B, Winer J, Armanini M, Gillett N, et al. (1993) Inhibition of vascular endothelial growth factor-induced angiogenesis suppresses tumour growth in vivo. Nature 362(6423): 841-844.
- Silva JM, Georgi N, Costa R, Sher P, Reis RL, et al. (2013) Nanostructured 3D constructs based on chitosan and chondroitin sulphate multilayers for cartilage tissue engineering. PLoS One 8(2): e55451.
- Boateng JS, Matthews KH, Stevens HN, Eccleston GM (2008) Wound healing dressings and drug delivery systems: a review. Journal of pharmaceutical sciences 97(8): 2892-2923.
- Xu ZP, Stevenson GS, Lu CQ, Lu GQ, Bartlett FP, et al. (2006) Stable suspension of layered double hydroxide nanoparticles in aqueous solution. J Am Chem Soc 128(1): 36-37.
- Ge Y, Zhang Y, He S, Nie F, Teng G, et al. (2009) Fluorescence modified chitosan-coated magnetic nanoparticles for high-efficient cellular imaging. Nanoscale Res Lett 4(4): 287-295.
- Sahu S, Behera B, Maiti TK, Mohapatra S (2012) Simple one-step synthesis of highly luminescent carbon dots from orange juice: application as excellent bio-imaging agents. Chemical Communications 48(70): 8835-8837.
- Wanlop W, Praneet O, Tanasait N, Ruktanonchai U, Lee RJ, et al. (2008) Evaluation of chitosan salts as non-viral gene vectors in CHO-K1 cells. Int J Pharm 348(1-2): 161-168.
- Tokumitsu H, Ichikawa H, Saha TK, Fukumori Y, Block LH (2000) Design and preparation of gadolinium-loaded chitosan particles for cancer neutron capture therapy. STP Pharm Sci 10(1): 39-49.
- Kumar MN, Muzzarelli RA, Muzzarelli C, Sashiwa H, Domb AJ (2004) Chitosan chemistry and pharmaceutical perspectives. Chem Rev 104(12): 6017-6084.
- Brenner, David J, Hall EJ (2007) Computed tomography-an increasing source of radiation exposure. N Engl J Med 357(22): 2277-2284.
© 2017 Yadu Nath VK, et al. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






