- Submissions

Full Text
Research & Development in Material Science
Relevance of New Materials for Energy Storage
N K Kishore*
Department of Electrical engineering, Indian Institute of Technology Kharagpur, India
*Corresponding author: N K Kishore, Department of Electrical engineering, Indian Institute of Technology Kharagpur, Kharagpur 721 302, India
Submission: September 08, 2017; Published: October 05, 2017

ISSN : 2576-8840Volume1 Issue3
Editorial
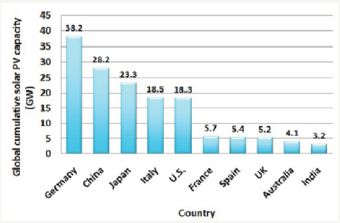
Today, perhaps electricity is the third most important commodity, next to only air and water. There is a continuous increase in demand for electricity. In fact the UN index for quality of life is per capita energy consumption. For a very long time electric power system is centrally controlled networks with generators located at the site of energy source and loads being large urban centers distant from power plants. Predominantly coal, oil, natural gas, and water have been energy sources from the beginning of electricity use. Among them, except water other three are fossil based fuels and are responsible for environmental pollution, necessitating locating power plants centrally and remotely. In view of dependence on the source and head of water, hydro generation is also centrally located. All this calls for transmission over long distances contributing to losses. Thus, there are low efficiencies in delivery, together with increased pollution and depletion of fossil fuels. This has brought developments in renewable energy sources based on solar and wind energy to the fore. Figures 1 & 2 show Global cumulative installed capacity of the wind and solar power in 2014 country wise, respectively [1,2].
Figure 1: Global cumulative installed capacity of wind in 2014 (country wise) [1].
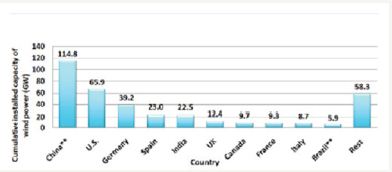
Figure 2: Global cumulative installed capacity of solar PV in 2014 (country wise) [2].
The renewable sources being clean, plants based on them are located close to loads giving rise to Distributed Energy Resources or also termed as a Dispersed generation (DG) into the fore. Of the various resources that are being employed, Solar and Wind are prominent.
Taking a look at the way electricity is used-now for over a long time it has become customary to be alternating commonly known as AC. With proliferation and development of consumer electronic devices use of electricity in direct or DC at low levels is becoming more common. Another perspective to look at is time of use. Although, electricity is required throughout the day-the use increases during nights. One of the resources that are being propelled is use of solar energy which is unavailable in nights. That amounts to having some way of storing excess energy during day time and use it at nights. From the very beginning of use of electricity, the storage of electrical energy is carried out through Cells connected together forming batteries. History of batteries may be traced as shown in Table 1 [3].
Table 1: Typical voltaic cell [3].
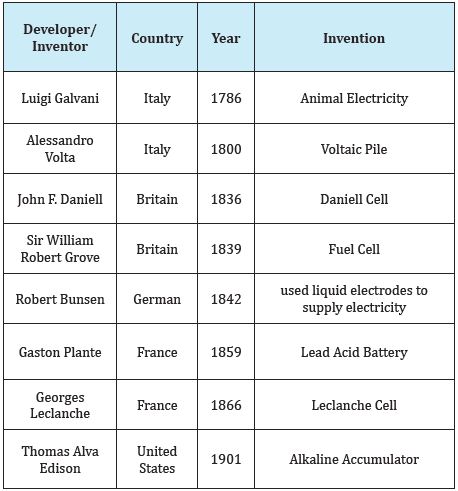
Figure 3: Step by step development in history of batteries [3].
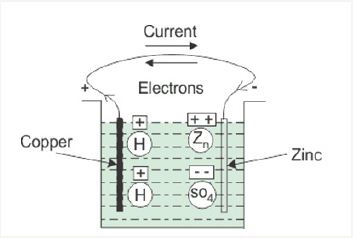
Although, it has been in use for a long time battery a form of Electro-Chemical energy storage is a fertile area of research. They vary in size from as small as a shirt button to that of a whole room forming a bank of battery. Figure 3 [3] shows the sketch of a typical voltaic cell. As may be noted it contains a jar having an electrolyte and two electrodes (made of copper and zinc) immersed in it connected through an external conductor and the electrolyte being sulfuric acid. Zinc dissolves in electrolyte leaving excess electrons on it (called Anode) forming negative electrode and copper attracts positive ions forming positive electrode (called Cathode). The electrons flow through external conductor and return back to positive electrode forming a closed electric circuit.
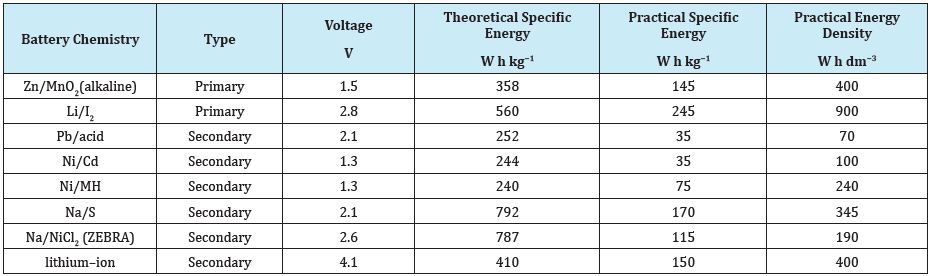
Thus, a battery has two electrodes and an electrolyte having ionic transport supporting electrons flow in the external circuit. So from a material stand point one has to look at Cathode Material, Anode Material and Electrolyte fundamentally. As increase in use and need of batteries comes to fore, requirements of having leak proof batteries and dry electrolytes and ability to be recharged by way of reversing REDOX reactions become important. This leads to two major category of batteries-Primary or Non-rechargeable or disposable and Secondary or Rechargeable. Another important issue is temperature rise in cells due to exothermic redox reactions leading to need for thermal management. Needless to stress, the requirements for improving energy densities necessary for adopting Electric Mobility towards pollution free environment. This has lead to progressive developments in newer materials and dry batteries. Table 2 lists various specifications of battery systems [4].
Table 2: Technical parameters of various battery systems [4].
Conclusiont
It may be said that the quest for newer electrode materials and electrolytes with high specific energy is ripe. It is believed this article motivates workers in the area to come forward to disseminate their findings towards a cleaner and more energy intensive environment for our progeny.
Acknowledgement
The author thanks Indian Institute of Technology Kharagpur for the encouragement and permission to publish this article.
References
- http://www.iea.org/topics/renewables/subtopics/wind/
- http://www.iea.org/topics/renewables/subtopics/solar/
- https://www.electrical4u.com/battery-history-and-working-principle-ofbatteries/
- Rosa Palacín M (2009) Recent advances in rechargeable battery materials: a chemist’s perspective. Chem Soc Rev 9: 10.1039/B820555H.
© 2017 N K Kishore. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






