- Submissions

Abstract
Progress in Petrochemical Science
Process Modelling and Simulation of Reactive Distillation for the Synthesis of High Purity Mono Ethylene Glycol
-
Open or CloseJofry Othman*, Norliza Abdul Rahman, Jarinah Mohd Ali and Siti Kartom Kamarudin
Chemical Engineering Programme, Faculty of Engineering and Built Environment, University Kebangsaan Malaysia, Malaysia
*Corresponding author:Jofry Othman, Chemical Engineering Programme, Faculty of Engineering and Built Environment, University Kebangsaan Malaysia, Malaysia
Submission: April 23, 2024; Published: May 17, 2024

ISSN 2637-8035Volume6 Issue3
Abstract
This study developed a suitable process modelling technique and simulation model for the non-catalytic synthesis of high purity Mono Ethylene Glycol (MEG) using the Reactive Distillation (RD) process. The feasibility of the RD process is demonstrated for performing the glycol reaction, dewatering and separation indistinguishable from the existing MEG production process with added advantages. Notably, a high purity MEG product of 99.8% is achieved within a single RD column configuration that avoids the need for the make-up of fresh demineralized water as in the current process. A working RD process model is generated that uses the established kinetics model parameters for MEG synthesis in combination with the equilibrium model and thermodynamic parameters in Aspen Plus® software. This included the power-law reaction variables for the main MEG synthesis along with the competing reactions that generate Diethylene Glycol (DEG), Triethylene Glycol (TEG) and Tetra Ethylene Glycol (TTEG) by-products. Hydrodynamics conditions for this process are also produced using a suitably packed column with the counter-current flow configuration of gas and liquid phases. The hydrodynamic variables including packing specification, maximum stage liquid holdup and maximum liquid superficial velocity, are generated to establish a maximum of 47.2% packing capacity and a low-pressure drop of 0.1kPa. Remarkably, compared with the conventional process RD promoted a 3.4% increase in MEG yield and a 53.2% reduction in energy usage. A lower water-to-ethylene oxide feed ratio of 12.9 is found to be suitable against the conventional ratio of 20 which reduces the separation loading while achieving a higher affinity for MEG selectivity of 91.4%. This subsequently generated a low energy usage through the utilization of reaction heat for separation with a net energy release of 0.24MW which lowers the operation cost.
Keywords:Reactive system; Mono ethylene glycol; Equilibrium model; Ethylene oxide
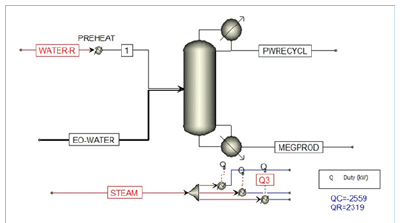
Figure 1:Reactive Distillation Column for MEG Production
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






