- Submissions

Full Text
Trends in Textile Engineering & Fashion Technology
Unique Electrochemical Properties of Ch900 Activated Carbon Cloth
Yury M Volfkovich*
AN Frumkin Institute of Physical Chemistry and Electrochemistry of Russian Academy of Sciences, Moscow, Russia
*Corresponding author:Yury M Volfkovich, AN Frumkin Institute of Physical Chemistry and Electrochemistry of Russian Academy of Sciences, Leninskii prospect 31, Moscow, 119071, Russia
Submission: December 01, 2022; Published: December 06, 2022

ISSN 2578-0271 Volume 7 Issue4
Opinion
In recent years, numerous studies have been carried out on the creation and improvement of electrochemical supercapacitors [1-3]. Carbon electrodes with a high specific surface area of ~500-2500m2/g is usually used as supercapacitor electrodes. For such electrodes, specific capacitance values from 50 to 300F/g have previously been obtained. Supercapacitors are used in electric vehicles, electric buses, cars (for starting) and in various electronic devices.
In this work, activated carbon cloth (ACC) CH900 manufactured by Kuraray Co. (Japan) was used as efficient supercapacitor electrodes [2-4]. Using the method of standard contact porosimetry, it was shown that this ACC has hydrophilic-hydrophobic properties. Its total specific surface area is 1520m2/g, and the hydrophilic specific surface area is 870m2/g [5]. Electrochemical studies of ACC CH900 were carried out in a wide range of potentials from -0.8 to 1.0V relative to the hydrogen reference electrode [2,6].
Figure 1:Capacitance-voltage cyclic curves for СН900-20 for 48% H2SO4 at potential rates: 1-0.5mV/s, 2-1mV/s, and 3, 4-2mV/s.
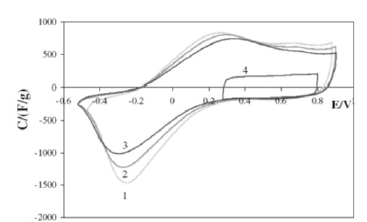
Figure 2 shows the dependence of the electrical resistance on the charging time of the tissue electrode measured by the impedance method. As you can see, as the charge increases, the resistance increases. This is explained by the change in time of the chemical composition of the electrode from carbon to C6H. Figure 3 shows the structural formula of this new chemical compound, which can be called carbon hydride or hydrogen carbide. The maximum capacitance obtained in [6] for the cloth electrode CH900 is 1110F/g, which is much higher than the values obtained earlier for carbon electrodes (maximum 300F/g).
Figure 2:Dependence of active resistance of the electrode on the charging time at Е= -400mV measured using the impedance technique in 40% sulfuric acid.
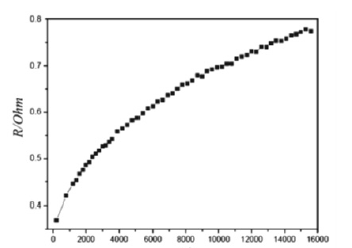
Figure 3:Structural formula of compound С6Н.

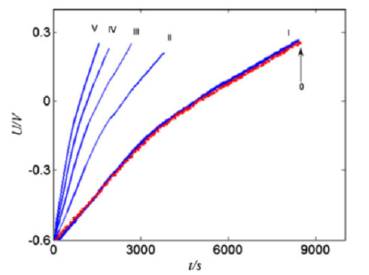
In [5], a mathematical model of the AСС electrode discharge was developed, which takes into account its specific surface area, ЕDL charging, hydrogen intercalation into carbon, solid-state diffusion of hydrogen in carbon, and diffusion of ions in an electrolyte solution. Figure 4 shows the model-calculated discharge curves for various currents, as well as the experimental curve for current density 2mA/ cm2. This figure shows that the model-calculated discharge curve almost coincides with the corresponding experimental curve. This indicates the correctness of the developed model of ACC electrode charging. Thus, in this work, an ultrahigh capacity of 1110F/g was obtained for the CH900 tissue electrode, which is much higher than the previously obtained values for carbon electrodes.
Figure 4:The discharge curves for various current densities. (0) The experimental curve 2mA/cm2 and the calculated curves (I) -2mA/cm2 , (II) -4mA/cm2, (III) -6mA/cm2, (IV) -8mA/cm2, (V) -10mA/cm2.
References
- Conway B (2013) Electrochemical supercapacitors: scientific fundamentals and technological applications. Springer Science & Business Media, Berlin, Germany, p. 636.
- Bagotsky VS, Skundin AM, Volfkovich YuM (2015) Electrochemical power sources: batteries, fuel cells, supercapacitors. John Wiely & Sons Inc, New York, USA, p. 372.
- Volfkovich YuM (2022) Supercapacitors: problems and development prospects. Russian Chemical Reviews 91(8): RCR5044.
- Volfkovich YuM (2022) Electrochemical properties of activated carbon cloths. Trends in Textile Engineering Fashion Technology 6(5).798-799.
- Volfkovich YuM, Filippov AN, Bagotsky VS (2014) Structural properties of porous materials and powders used in different fields of science and technology. Springer, London, p. 328.
- Volfkovich YuM, Bograchev DA, Mikhalin AA, Bagotsky VS (2014) Supercapacitor carbon electrodes with high capacitance. J Solid State Electrochem 18: 1351-1363.
© 2022 Yury M Volfkovich. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






