- Submissions

Full Text
Techniques in Neurosurgery & Neurology
Myxofibrosarcoma of Base of Skull in Paediatric Age Group: A Case Report
Zafar Ahmed Sheikh*
Mahatma Gandhi Memorial Medical College, India
*Corresponding author:Zafar Ahmed Sheikh, Mahatma Gandhi Memorial Medical College, G1 Kasturi Apts, 12 Shankar Nagar, Saket Extension, Indore-452018, Madhya Pradesh, India
Submission: March 08, 2018;Published: August 31, 2018

ISSN 2637-7748
Volume2 Issue1
Abstract
Myxofibrosarcoma is a common sarcoma affecting extremities of elderly age group. It rarely affects the younger age group and the head neck reg ion. It exhibits an infiltrative growth pattern and has a high local recurrence rate. We report a 14 year old patient with a myxofibrosarcoma of base of skull. The patient’s age and clinical presentation favored a bed side diagnosis of a dermoid cyst or langerhans cell histiocytosis, radiology was suggestive of an intraosseous hemangioma and surgical pathology favored a low grade Osteosarcoma. However, tissue immunohistochemistry clinched the diagnosis of a Myxofibrosarcoma. He underwent a near total excision with adjuvant radiotherapy and remains recurrence free at one and a half year follow up. Tissue immuno chemistry played a pivotal role in the management of our patient. We have attempted to bring forward the pertinent neurosurgical issues related to the management of this common soft tissue sarcoma which is rarely encountered by Neurosurgeons.
Introduction
Soft tissue sarcomas are a heterogeneous and a diverse group of neoplasm of mesodermal origin. Myxofibrosarcoma is one of the common histologic types of Soft Tissue Sarcomas. Formerly myxofibrosarcoma was considered as the myxoid variant of Malignant Fibrous Histiocytoma. The 2002 WHO classification of tumors included them as a distinct entity after Angervval & kindblom [1] recognized them to be so and coined the term Myxofibrosarcoma. To date less than 20 patients have been reported to have head neck Myxofibrosarcoma and ours is the first case of a skull base myxofibrosarcoma in the pediatric age group.
Case History
figure 1:Preoperative lateral and posterior profile view depicting the left occipital swelling.
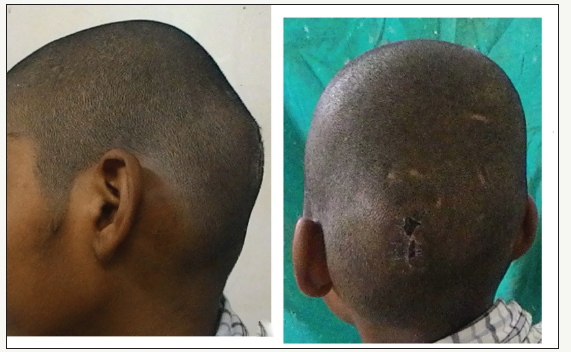
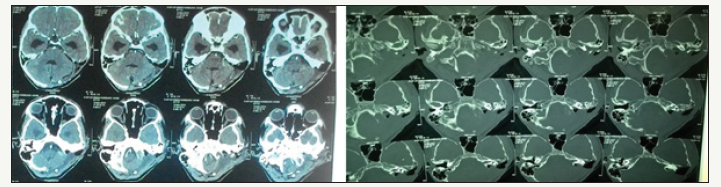
figure 2:Contrast enhanced Computed tomogram of head with axial, coronal and saggital cuts revealing the intensly enhancing occipital mass.

figure 3:Postoperative front profile depicting the left hypoglossal paresis. And the post resection defect.
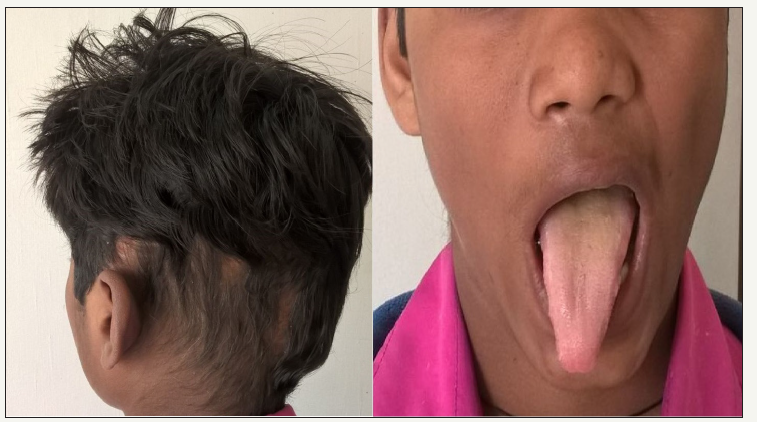
A 14-year-old boy presented with a oneyear history of a painless, progressive swelling over the left occipital region (Figure 1). He denied constitutional symptoms and was neurologically intact on examination. Contrast enhanced computed tomogram (Figure 2) head revealed a large, hyper vascular, expansile lytic mass of the left occipital bone. It was well corticated with occasional bony internal septations. It was heterogeneously hyper intense and showed intense post contrast enhancement. The basal cisterns were effaced with significant mass effect on brainstem and cerebellum. An angiogram revealed retention of contrast on delayed phase images suggesting a possibility of intraosseous hemangioma of occipital bone. Intraoperatively, the lesion was pearly white and fleshy in consistency. Outer and inner bone tables were paper thin. The bone tables were breached but the dura and scalp was uninvolved. He underwent a near total excision, with the nubbins infiltrating jugular foramen being left behind. Postoperatively, the patient was clinically stable, with a transient lower cranial nerve paresis which recovered gradually (Figure 3).
Pathological evaluation of the surgical specimen revealed a low grade Osteosarcoma. Immunohistochemistry clinched the diagnosis of a myxofibrosarcoma (Figure 4). He received adjuvant radiotherapy in view of the residual disease (Figure 5) and the IHC findings. At 30 months follow up he remains recurrence free.
figure 4:Top row from left ALK1 poitive, BCL2 positive, CD 31 & CD 34 positive in richly vascular stroma, Desmin negative , EMA negative , GFAP negative, Ki 67 15%, S 100 negative, SMA positive and VIMENTIN strong positive Bottom Row H& E preparations showing Spindly cells with abundant pink matrix (osteoid). The nuclei are irregular pleomorphic and hyperchromatic. Occasional mitosis. Vacularity seen. (ALK)1 Activin receptor-like kinase BCL1 - “b cell lymphoma #1” CD 31 cluster of differentiation 31 CD 34 cluster of differentiation 31 EMA Epithelial Membrane Antigen GFAP Glial Fibrillary acid protein Ki 67 Proliferation index SMA Smooth Muscle Actin H& E Hematoxylin and eosin stain
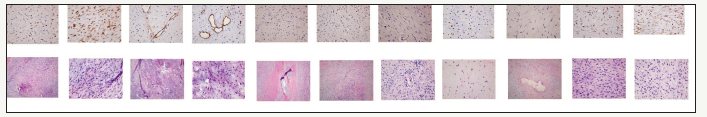
figure 5:Postoperative operative contrast enhanced computed tomogram of head showing the residual lesion.
Discussion
Myxofibrosarcoma is the most common sarcoma in the elderly age group. Its peak incidence is in the seventh decade with Slight male Preponderance. It involves the extremities in over 75% cases, trunk, retroperitoneum and mediastinum being the less favored sites. It exhibits a slow and painless progression [2]. Based on the plane of origin Mentzel et al classified myxofibrosarcoma into two subgroups: superficial (dermal/subcutaneous) and deep (intramuscular/sub fascial) [3]. The superficial group of tumors exhibit a longitudinal pattern of extension and infiltration, whereas the intramuscular/sub fascial tumors form a single discrete mass.
These tumors have been subdivided into three or four grades based on the degree of cellularity, nuclear pleomorphism and mitotic activity [3,4]. Mutter et al. [5] have reported a local recurrence rate of 14.3% for Myxofibrosarcoma [5]. Other groups have reported a high Local recurrence rate of upto 61% [1,3,6]. An infiltrative growth tendency with ability to extend along facial and vascular plane may be implicated in the high local recurrence rates found in these tumors [3-5,7].
The tendency of myxofibrosarcoma to be of progressively higher grade in recurrences by becoming gradually more cellular, more pleomorphic, more mitotically active, and more necrotic seems to be a distinctive feature and is in contrast to most other sarcomas [3].
Imaging
On Magnetic resonance imaging Myxofibrosarcoma exhibit abnormal signal infiltration along fascial planes. Presence of a tail sign is common and associated with an adverse local recurrence free survival [8]. Ahmadi et al. [9] have proposed that a fairly uniform dural enhancement in vicinity of a neoplasm on Contrast MRI is most likely reactive but focal breach in continuity of enhancing adjacent dura implies infiltration and warrants dural excision, where possible [9,10].
Treatment
A wide margin surgical resection aimed at achieving negative margins is the main modality of treatment of Myxofibrosarcomas. Radiation is an effective adjuvant treatment in these patients and should be considered in the local management of this disease [5]. The need for follow-up imaging to assess tumor recurrence or progression of residual disease makes selection of materials used in closure of the cranial defect after cranial surgery a key element of operative planning. Poly ethyl ethyl ketone and titanium implants produce minimal metallic artifact on MRI and CT, making it an excellent option for cranial reconstruction when autologous bone is unavailable or not suitable.
Treatment Data to guide the optimal management strategy for Myxofibrosarcoma of the calvaria and skull base is sparse Myxofibrosarcoma of skull present a challenging management conundrum owing to the combination of exceedingly rare occurrence, varied pathologic features, infiltrative tendency, high local recurrence rate and stage progression in recurrences. Currently ensuring a tumor free margin, an expert pathological review, adjuvant radiotherapy and a close surveillance form the corner stone of treatment plan. The future of management lies in understanding the molecular basis of the tumors to develop and direct therapy at them.
References
- Angervall L, Kindblom LG, Merck C (1977) Myxofibrosarcoma. A study of 30 cases. Acta Pathol Microbiol Scand A 85(2): 127-140.
- Fischer CDM, Unni KK, Mertens F (2002) Pathology and Genetics of Tumours of soft tissue and Bone. In: Fischer CDM, Unni KK, Mertens F (Eds.), Lyon: IARC Press, France
- Mentzel T, Calonje E, Wadden C, Camplejohn RS, Beham A, et al. (1996) Myxofibrosarcoma. Clinicopathologic analysis of 75 cases with emphasis on the low-grade variant. Am J Surg Pathol 20(4): 391-405.
- Huang HY, Lal P, Qin J, Brennan MF, Antonescu CR (2004) Low-grade myxofibrosarcoma: A clinicopathologic analysis of 49 cases treated at a single institution with simultaneous assessment of the efficacy of 3-tier and 4-tier grading systems. Hum Pathol 35(5): 612-621.
- Robert W Mutter, Samuel Singer, Zhigang Zhang, Murray F Brennan, Kaled M Alektiar (2012) The Enigma of Myxofibrosarcoma of the Extremity. Cancer 118(2): 518-527.
- Gronchi A, Lo Vullo S, Colombo C, Collini P, Stacchiotti S, et al. (2010) Extremity soft tissue sarcoma in a series of patients treated at a single institution: local control directly impacts survival. Ann Surg 251(3): 506-511.
- Kaya M, Wada T, Nagoya S, Sasaki M, Matsumura T, et al. (2008) MRI and histological evaluation of the infiltrative growth pattern of myxofibrosarcoma. Skeletal Radiol 37(12): 1085-1090.
- Yoo HJ, Hong SH, Kang Y, Choi JY, Moon KC, et al. (2014) MR imaging of myxofibrosarcoma and undifferentiated sarcoma with emphasis on tail sign; diagnostic and prognostic value. Musculoskeletal European Radiology 24(8): 1749-1757
- Ahmadi J, Hinton DR, Segall HD, Couldwell WT (1994) Surgical implications of magnetic resonance-enhanced dura. Neurosurgery 35(3): 370-377
- Mutter RW, Singer S, Zhang Z, Brennan MF, Alektiar KM (2012) The Enigma of Myxofibrosarcoma of the Extremity. Cancer 118(2): 518-527.
© 2018 Zafar Ahmed Sheikh. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






