- Submissions

Full Text
Research in Pediatrics & Neonatology
An Easy and Practical Tool to Detect the Neurodevelopmental Status in the First Two Years of Life
Magda Di Renzo1*, Elena Vanadia1, Paolo Pace2, Teresa Mazzone3, Lidia Racinaro1, Monica Rea1 and Alberto Villani4
1Institute of Ortofonologia (IdO), Rome, Italy
2Child Neuropsychiatry ASP, Trapani, Italy
3Pediatric Primary Care, ASL, Rome, Italy
4University of Rome-Tor Vergata and Hospital Bambino Gesù, Rome, Italy
*Corresponding author: Magda Di Renzo, Institute of Ortofonologia (IdO), Rome, Italy
Submission: September 04, 2023; Published: September 25, 2023

ISSN: 2577-9200 Volume7 Issue5
Abstract
The main stages of neurodevelopment in the pediatric age are well defined and described in the main books of Pediatrics. In recent years, a significant increase in neurodevelopmental delays and disorders has been described in the pediatric literature. It is also well known that intercepting these situations early allows an early intervention, in many cases fundamental, thanks to an early diagnosis and to obtain the best desired results. In Italy all children have the right to free pediatric assistance thanks to the National Health System (SSN). Periodic check-ups (health reports) are foreseen and regulated from birth to 14 years of age. In this study we verified the usefulness of using specific questionnaires to evaluate neurodevelopment particularly in the first 2 years of life during the periodic check-ups. The possibility for pediatricians to have a tool that is easy to compile and based on the well-known stages of neurodevelopment at the different ages in which health assessments are usually carried out, allows situations of delay or defect to be highlighted, identifying them and allowing those who test positive in screening to be referred to specialist centers. Early identification through a questionnaire in which the main stages of neuro-development are listed for different ages allows pediatricians to select the population that really needs a specialist evaluation, avoiding overloading specialist centers. The results of this study on 1993 subjects aged between 1 and 24 months are preliminary and encourage the use of this rapid and cost-free tool (questionnaire) in the early identification of situations worthy of further diagnostic investigation.
Keywords:Neurodevelopmental screening; Psychomotor development; Neurodevelopment delay; Screening form; Pediatrician
Introduction
The neurodevelopmental screening and monitoring form 0-2 years presented in this research was constructed following some basic assumptions. Chief among these is the importance of early detection of developmental disorders in order to establish therapy and/or family support programs as soon as possible, so that children’s vulnerabilities or conditions of altered environmental stimulation can be recognized and addressed in a broader perspective of treatment, which also becomes a prevention for the onset of emotional-behavioral disorders, secondary developmental delays and conditions of distress. In fact, it is known that the first two years of life represent one of the periods if not the main one-in which brain displays the greatest plasticity; this is valid both in adaptive and maladaptive terms [1], whereas in this second case a condition of hindered functional recovery or the development of an undesired symptom is defined. Always in relation to the early detection of disorders, the Form contains some questions relating to the so-called alarm bells for neurodevelopmental disorders, such as language, movement and autism spectrum disorders but also neurosensory disorders and /or Infantile Cerebral Palsy (ICP) [2-5]. Neurodevelopmental disorders, previously defined as developmental disorders, which typically onset in childhood are very frequent because they affect about 15-20% of the population between childhood and adolescence at different level of severity. Their determinism and evolution are influenced by social, environmental and biological factors [6,7]. However, a high percentage of them has been calculated that is not identified during follow-up, even specialist ones, in particular for those forms that we could define as “subthreshold” and in particular in the first years of life [8].
Another important assumption relates to the concept of development trajectories. Referring again to the brain plasticity typical of the first years of life and considering that initially the child’s development proceeds in an integrated and synactive way [9], it is clear that some limitations or dysfunctions in the early processes of maturation and regulation can interfere with subsequent physiological acquisitions also in other domains of development, but also that by intervening early on the dysfunctions it is possible to positively modify the development trajectories. Furthermore, propose a tool that is not specific for the identification of a single disorder, but which focuses attention on the main areas of development ranging from physio-logical regulation (sleep, nutrition, etc.) to motor development, from the modulation of behavioral states to attention and intersubjectivity indices, from language (first gestural and then verbal) to sensory integration is a concrete and easy way to encourage punctual observation and monitoring of children over time, from a global and developmental perspective. This tool allows you to highlight the delay with respect to the expected behaviors for the specific age. As already mentioned, it is of fundamental importance to give parents and caregivers the opportunity to better understand and respond to children’s needs by directing care and treatment processes according to a correct timing, which also means optimizing re-sources, acting for health promotion and disease prevention, taking into account the biopsycho- social model which places the interaction of biological, psychological and social factors at the center of health and disease [10] . The main objective of this work, then, is to define, on the basis of 1993 forms com-piled by pediatricians, the domains in which developmental delays or alterations are most frequently observed in the various age groups in early childhood.
The theoretical basis of the screening form
During the first years of life, emotionally nourishing relationships lay the foundation for lifelong health and well-being. Research and clinical experience demonstrate that first relationships significantly influences brain development, social emotional and cognitive skills, that is the future individual trajectory [11] . For this reason, dealing with the psychophysical development in early childhood means having care of those parameters of physical growth, both regulatory and relational, that have a strong impact on neurological, adaptive and psychological development of the child in later stages. The division of the questionnaire into 5 age groups, was chosen on the basis of the timing with which in Italy pediatric health balances are expected in the first two years. The Italian National Health system (SSN) guarantees free health care from 0 to 14 years. Pediatricians must carry out periodic check foreseen in the following ages: 1 month, 2-3, 4-6, 7-9, 10-12, 15-18 months, 2-3 years. This ensures that all patients adhered to the study because the questionnaires were administered and completed during the regular check-ups scheduled during the health assessments [11] . An added value of the questionnaire is to be replicable and usable in the different health balances, precisely because it is specific for each age and this allows both to monitor the changes with respect to any criticalities that emerged from the previous balance, the onset of symptoms or signs that must alert the pediatrician to the child’s neuro-psycho-behavioral development. On the international scene, there are several standardized scales and checklists used in pediatric practice that investigate specific domains of development and allow the early detection of some pathological frameworks. For example the Dubowitz [12] Examination to evaluate both physical and neurological characteristics and estimates the baby’s gestational age within 1-2 weeks of the true gestational age [12]; the Prechtl’s [13] assessment of general movements for the functional assessment of the young nervous system [13], the M-CHAT and M-CHAT-R/F for early identification of autism spectrum disorders [14,15].
However, the scales described so far have the limit to evaluate some specific functions, and not to put together observations regarding the overall profile of the child. There are also standardized tools to define the level of development and harmony/disharmony of individual skill profiles, including the Griffiths [16] Scales of child development 3rd edition [16] and the Bayley [17] Scales of Infant and Toddler Development 3rd edition [17], most often used in mental health services. Bayley [17] and Griffiths [16] Scales, while evaluating the child in its overall development profile, provide for an individual administration, which lasts at least an hour; therefore, unlike the questionnaire that we present, are not suitable for use during pediatric monitoring visits. Finally, some checklists are available, including rating scale for caregivers, which detect any deviations of the physiological trajectory of a child’s development, for example the Milestones checklist [18]. However, these types of checklists are more generic, as they are designed to be used by nonprofessional people (especially parents) and therefore they are not designed for medical/psychological supervision. The questionnaire that we presented is based on theoretical constructs and scientific literature related to the different domains of development in the first two years of life. For all domains there are similar range of age in which children acquire and show new abilities, usually divided in 5 or 6 stages from 0 to 24 months [11]. Neurological development: The brains’ many functions do not develop at the same time. Although basic sensation and perception systems are fully developed at about kindergarten age, systems such as memory, decision making and emotion continue to develop well into childhood [19]. The foundations of many of these abilities are constructed during the early years. Infants and toddlers use their sense and motor abilities to manipulate their surroundings and learn about the environment. They understand a cause-and-effect relationship and as the frontal lobe matures and memory develops, children can imagine what may happen without physically causing an effect. This is the emergence of thought and allows for the planning of actions.
Sensory development
Development of the senses begins in early fetal life, initially with structures and then in-utero stimulation initiates perception. After birth, environment accelerates each sensory organ to nearly complete maturity several months after birth [20]. The first twelve months of a baby’s life are full of sensory advancements. Sight, smell, hearing, touch and taste all develop faster during the first 12 months of life than during any time thereafter. For example, in the first three months the baby can see limited colors and begins to make eye contact, he can fix and follow a slow horizontal arc and eventually will follow past the midline. At about 12 months, depth perception and distance judgements improve, it’s easy grabbing of objects. There is an increase in smell preference and reaction. Baby can recognize and react to songs and sounds; he is now able to grasp handheld toys like rattles and tethers and enjoys a greater variety of taste [21]. Motor development: At birth the child’s motility is dominated by a motor activity of a reflex type, so he is unable to make voluntary movements and for this reason he reacts with tears to a posture set by the adult which he perceives as unsafe and cannot change. In the first two years of life, psychomotor development is considered within stages of development according to age groups, each of these stages collects functional skills that mature in the various stages of the child’s development [22]. For example, in the 0-3 months phase the flexor tone predominates; in the 3-6 month phase, in a sitting position he holds his head straight, the spine is straight except at the lumbar level (kyphosis); between 6 and 9 months he maintains the sitting position and is able to lean on his arms; between 9 and 12 months it crawls; in the 12- 18 month stage he begins to walk on his own; between 18 and 24 months autonomous walking is more rapid (greater balance and stability, no longer brings hands forward for defense) [23].
Language development: there is more than one theory related to linguistic development, from Skinner who believed that children learn language through operant conditioning [24], to the Chomsky’s one based on innate abilities to learn language (“language Acquisition Device”) [25]. Bruner went beyond the previous positions by theorizing the LASS (Language Acquisition Support System) as a system that allows the LAD (language Acquisition Device) to be made operational thanks to the interactions of the child with the caregiver [26]. Jean Piaget’s theory of language development suggests that children use both assimilation and accommodation to learn language [27] and Vygotsky’s theory of language development focused on social learning and the zone of proximal development [28]. Camaioni studied communication in its preverbal aspects, emphasizing the importance of gestures as photosymbiotic forms [29]. With respect to linguistic acquisitions, in the typical development the child in the first 6 months laughs, screams, cries, blows and responds with vocalizations; between 6 and 12 months he gradually passes from bubbling to single words, to which he is attributing a precise meaning starting to use them voluntarily. Between 12 and 18 months, he recognizes his name and immediately turns away if he hears his name. He uses single words and understands simple commands. Within 2 years the vocabulary expands exponentially and from the word phrase passes to the association of two or more words [29]. Psychological development: For Piaget [27] young infants process experiences and events by balancing assimilation and accommodation. Assimilation consists of elaborating new information’s and fitting it into previously understood mental schemas. Accommodation means adapting and revising a previously understood mental schema according to the new information’s. Piaget divided child development into four stages. The first stage, Sensorimotor (ages 0 to 2 years of age), is the time when children master causality and object permanence. Between 0 and 2 months infants actively seek stimuli and respond more vigorously to changing ones; between 3 and 6 months children engage in a purposeful sensory exploration of their bodies, and this builds the concepts of cause and effect and self-understanding. Between 7 and 12 months object permanence emerges, as the toddler looks for objects. Between 13 and 18 months toys can be explored, made to work and novel play skills emerge. Gestures and sounds can be imitated. Egocentric pretend play emerges too. Between 19 and 24 months new problem solving strategies emerge even without rehearsal. Thoughts and imagination arise, and there is the ability to plan actions. Object permanence is wholly established, and objects can be searched for by anticipating where they may be [30].
Socio-emotional and intersubjective development: Social emotional development covers two important concepts of development including the development of self or temperament and relationship to others or attachment. Temperament is an innate component that directs the child’s approach to the world and his interaction with the environment; it includes activity level, distractibility, intensity of emotions, regularity, sensory threshold, tendency to approach versus withdrawing, adaptability, persistence, mood quality [30]. In healthy children, social-emotional stages develop on an expected trajectory, and monitoring these steps is a fundamental part of preventative health supervision visits. For ex-ample, in the first 2 to 3 months infant learns to regulate physiologically and needs easy routines. Around 4 months of age turn taking conversation (vocalizations) begin. The infant learns to manipulate his environment. Between 6 to 12 months, infants establish attachment relationships with a responsive caregiver. Around 8 months of age, joint attention skills develop. An infant will look in the same direction as the caregiver and follow his gaze and gradually becomes able to share experiences. Between 12 to 18 months, the infant learns to explore his environment with support from a caregiver. Around 12 months of age, the child takes part in interactive play (peekaboo) and around 18 months of age, the child brings the object to show or give it to the caregiver. Between 18 and 30 months, autonomy, that is individuation, emerges so that child’s temperament manifests itself more [31,32]. Trevarthen stressed that the meanings of language and cognition are co-created thanks to the exchange of the child with the caregiver on the basis of an innate matrix that makes him ready to interact from the first moments of life [33,34].
Regulatory development
In the first two years of life, the regulatory processes to which greater attention must be paid are physiological regulation (neonatal vagal tone and sleep-wake cyclicity), emotion regulation (response to stress at 3, 6, and 12 months) and attention regulation by focused attention and delayed response (during the 2nd year). Ruth Feldman has defined in her work that regulatory functions showed stability across time, measure and levels, and also the coherence of the regulation construct and with neurobiological models on self and consciousness [35]. Also [36] purposed a theoretical model to understand and assess the individual infant focus on the dynamic, continuous interplay of various subsystems within the organism: The autonomic system, the motor system, the state organizational system, the attention al interactive system, and the self-regulatory system. The organism forges ahead negotiating emerging developmental agenda while simultaneously seeking to attain a new level of modulated, functional competence [36]. Therefore, starting from the theoretical-scientific knowledge on development and from the already existing standardized tools, we have selected those items that the pediatrician can easily evaluate within the health assessments or ask the parents and which, correlated to each other on the basis of the presence/absence and their evolution, can allow early detection not only of major neurological or psychological disorders (e.g. infantile cerebral palsy, neurosensory deficits, autism spectrum disorders), but also minor cases or vulnerability of neurobehavioral development. We have also considered some of the conditions that most frequently interfere with physiological psychomotor acquisitions, for example positional plagiocephaly and gastro-esophageal reflux [37]. In fact, we think that for a pediatrician, being able to have a tool that also contemplates the correlations between the different developmental systems, can represent a valid aid to clinical practice in order to be able to give the right advice to parents and to be able to direct any investigations and referrals to other specialists.
Method and Materials
The screening form
The screening Form is divided into 5 Modules of 15 or 20 items, specific for 5 age groups: 0-3 months, 4-6 months, 7-12 months, 13-18 months and 19-24 months. Each Module is calibrated for the specific age group considered and it can be used independently of the others. Responses to items are organized on a 3-point Likert scale, where 0 indicates the absence of problematic behaviors, 1 indicates the occasional (sometimes) presence of specific behaviors and 2 indicates the frequent presence of specific behaviors. The sum of the scores assigned to each item provides a total score which will be compared with the cut-off (indicated at the footer) on the basis of which implement the suggested operational choice (typical= nothing to report; low risk=retest between 3 months; high risk= retest in 1 month). Typical refers to a child who is able to carry out what is foreseen for his/her age; Low risk refers to a child who shows difficulties in carrying out what is foreseen for his/her age; High risk refers to the children who are not able to respect the behaviors and skills expected for their specific age on the basis of the consolidated milestones of neurodevelopment. Furthermore, in each Module there are “critical” items, highlighted in bold: if even in only one of these items the child receives a score of “2”, an in depth study would be appropriate. The administration time of the questionnaire is about 5/10 minutes. The Screening Form is shown in the Appendix 1.
Procedure
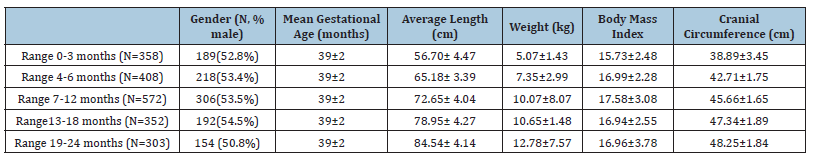
The Screening Form was sent to 25 Pediatricians from 4 Italian Regions, Liguria (16%), Abruzzo (24%), Lazio (55%) and Campania (5%), and was completed by them during the periodic routine check-up of 1993 children. The children (N= 1993) were aged between 1 and 24 months, subdivided into age groups: 0-3 months (N=358); 4-6 months (N=408); 7-12 months (N=572); 13- 18 months (N=352) and 19-24 months (N=303). Half of them were male (N=1059; 53%). The data were collected between January 2019 and June 2021. 19.2% of the data (N=382) were collected in the post-lockdown period (i.e. after May 2020). Characteristics of are shown in Table 1. Pediatricians filled out the Screening Form for typical developmental children; there was no known risk factor for a neurodevelopmental disorder. The pregnancies were full-term and normal.
Table 1:Characteristic of sample, divided by age group.
Inclusion criteria
Children with the following characteristics were not included in the research: (a) neurological disorders or focal neurologic signs; (b) severe sensory deficit (blindness and deafness); (c) history of severe birth injuries such as asphyxia, head trauma or epilepsy; (d) preterm children (<36 weeks of pregnancy); (d) chronic comorbidities and genetic syndromes. Pediatricians gave feedback to families on screening outcome.
Data analysis
Inferential tests on the normal distribution of scores were conducted, on the basis of which the raw scores corresponding to the 5th percentile were calculated. In particular, the total raw scores were transformed into z points and then into T points, whereby scores between -1.5 and +1.5 standard deviations from the mean (i.e. T points ≤60) were considered Typical; scores between 1.5 and 2 standard deviations from the mean (i.e, T points between 61 and 69) were considered Low Risk; scores greater than 2 standard deviations from the mean (i.e, T points ≥70) were considered High Risk. Descriptive analyzes of the items were also conducted to identify those most frequently representing a difficulty in the child.
Result
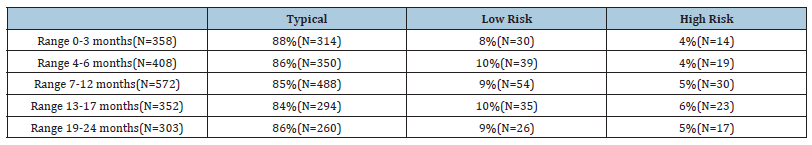
A preliminary analysis was conducted on the total raw scores obtained in each age group from the total sample, to establish the cut-off points beyond which the scores are indicative of a probable neurodevelopmental difficulty, for each individual age group. Scores falling between the 25th and 100th percentile were defined as Typical, scores between the 6th and 24th percentile were defined as Low Risk, scores lower than or equal to the 5th percentile were defined as High Risk. Analyzes of variance were also conducted to verify the effect of the variable “gender” on the total scores. The cut off scores emerged are shown in Table 2. Table 3 shows the numbers and percentages of children in the sample falling into the three different risk probability ranges, divided by age group.
Table 2:Clinical cut-off by age groups.
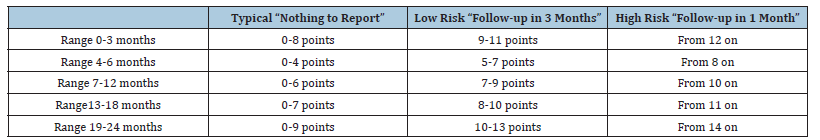
Table 3:Numbers and percentages of children in the sample falling into the three different risk probability ranges, divided by age group.
Range 0-3 months
Specifically, in the children of this age group (N=358), emerged that 88% were within the Typical range (25th-75th percentile), 8% of the children fell within the Low Risk range and 4% of children were in the High Risk range. The items in which children with High Risk most often had the score “2”, that is the score which indicates the frequent presence of problematic behaviors, were: the child does NOT adapt to being held (64%); the child does NOT show attention to surroundings sounds (78%); the child is not very attracted by colored objects (86%); the child does NOT imitate mouth movements (93%) and the child had difficulty with breastfeeding attachment in the first 3 months of life (93%). In this age group, no significant differences emerged for the “gender” variable (F1,357 = 0.08; p =.92).
Range 4-6 months
In children of this age group (N= 408), emerged that 86% were within the Typical range, 10% of children were in the Low Risk group and 4% of children fell in the High Risk category. The items in which children with High Risk most often had the score “2” were: the child is NOT easy to dress/undress (37%) and the child’s sleep is irregular (difficulty falling asleep, restless sleep, frequent awakenings) (41 %). In this age group, no significant differences emerged for the variable “gender” (F1,407= 1.90; p =.17).
Range 7-12 months
In children of this age group (N= 572), emerged that 85% were within the Typical category, 9% of children were in the Low Risk group and 5% of the children were in the High Risk group. The items in which children with High Risk most often had the score “2” were: the child is NOT autonomous in his movements (40%), when the child gets hurt a lot, he cries little (33%), the child sleeps too much compared to other children ( 33%), the child does NOT repeat sounds and/or first words (97%), while sitting or on all fours the child does NOT turn to pick up an object (33%), the child does NOT imitate the other’s gestures (70%), the child’s sleep is irregular (43%) and the child is NOT able to stand upright with support (37%). In this age group, no significant differences emerged for the variable “gender” (F1,571= 0.06; p =.80).
Range 13-18 months
In children of this age group (N=352), emerged that 84% were within the Typical range, 10% of children were in the Low Risk group and 6% of the children in the High Risk group. The items in which children with High Risk most often had the score “2” were: the child tends to smell objects or people (30%), the child overreacts to denial (35%), the child does NOT produce at least 5 words with meaning (83%), the child’s sleep is irregular (43%), when he hurts a lot, he cries little (30%) and the child does NOT flip through the pages of a book (39%). In this age group, no significant differences emerged for the variable “gender” (F1,351= 2.03; p =.16).
Range 19-24 months
In children of this age group (N=303), emerged that 86% were in the Typical range, 9% of children were in the Low Risk group and 5% of the children in the High Risk group. The items in which children with High Risk most often have the score “2” were: the child tends to bring objects to the mouth and/or lick them (53%), the child does NOT make sentences of 2 or more words (59%), the child has eating problems (35%), the child does NOT complete a task (65%), the child does not blow /suck (41%), does NOT produce at least 20 meaningful words (88%) and the child overreacts to denial (53%). In this age group, no significant differences emerged for the variable “gender” (F1, 302= 2.11; p =15).
Discussion
In the 0-3 months range, the main alarm bells revealed by the screening form concern the child’s ability, while awake, to pay attention to the world around her/him and to interact with it, such as, for example, the difficulty to adapt when held in the arms, to show attention to sounds and colors around her/him, to imitate movements of the parent’s mouth and to breastfeeding attachment. These aspects risk having an impact on the development of self-regulation necessary for subsequent learning [38-40]. In fact, in children with typical development, in this first trimester of life, we observe the eye gaze as well as the ability to pay attention to objects, to look and respond to a familiar face, until the appearance of the social smile [38-40]. Looking at the adult’s mouth and imitating some facial movements represent the precursors of preverbal communicative intentionality [41]. It is interesting to note how the items in which the “High Risk” children received high scores seem to indicate a vulnerability between sensory-perceptive-motor development and early relational skills, aspects which are very important to analyze within health checkup and screening, in order to identify early vulnerability of the child or the family system [42,43]. For example, the coexistence of “difficulty to breastfeeding attachment” and “difficulty to imitate mouth movements” may underlie a sensory-motor immaturity that could have repercussions on the relationship sphere or, otherwise, a social-affective vulnerability that limits the specialization of motor functions (in this case oral) [35].
In the 4-6 month range, emerged alarm bells mainly related to difficulties in sensory and physiological regulation, for example the child “does not like to be dressed or undressed” and “has difficulty in sleep regulation”. In accordance with these findings [44-46], the co-presence of other alarm bells may or not define the difference between a physiological transition phase (when sleep is the only indicator and a spontaneous resolution is possible) and a disorder that affects multiple subsystems, whereas specialist assessments should be carried out and parents should be supported in understanding child’s characteristics and environmental factors that could represent “risk” or “protective” elements. In the 7-12 month range, alarm bells emerged concerning difficulties mainly related to motor development (“the child is NOT autonomous in his movements”; “while sitting or on all fours he does not turn around to pick up an object”; “the child is not able to stand upright with support”) and communicative-linguistic (“the child does not repeat sounds and/or first words”; “the child does not imitate the other’s gestures”), but sleep regulation difficulties also remain. At this age the child should begin to gain his first forms of independence, so starting to move away (moving away from his parents or trying to reach objects) stimulates the need to communicate more [27]. Difficulties in these areas seem to be related to maturation variables, in an age range that foresees significant changes and acquisitions. But it should be emphasized that motor, linguistic and sleep regulation difficulties also represent early warning signals for autism spectrum disorders [47,48]. In our opinion, it is therefore essential to pay attention to the quantity and quality of the indicators, in order to early identify children at developmental risk and to establish early and appropriate intervention and/or monitoring programs.
In the 13-18 months range, alarm bells emerged concerning difficulties related to sensory and emotion regulation (“the child tends to smell objects or people”; “the child overreacts to denial”; “when he hurts a lot, he cries little”) but also remain those related to linguistic and communicative development (“the child does not produce at least 5 words with meaning”; “the child does not flip through the pages of a book”) and the regularity of sleep. Compatibly with developmental processes, at this age emotion regulation is particularly “critical”, that is the individual ability to regulate own emotions, both positive and negative, attenuating them, intensifying them or simply maintaining them [49]. The processes through which emotions can be regulated can be of intrinsic type, when the subject acts by regulating himself (regulation in self), or of extrinsic type (regulation in other), when someone acts to regulate the emotions of the other [50,51]. Emotion regulation is initially mediated by the caregiver, later in the course of development it appears as a more autonomous and conscious modality [52-54]. This, more than other areas of development, can easily be affected positively or negatively by environmental conditions, including dysfunctional family dynamics, parental psychopathologies, adverse, stressful or traumatic events. It is also based on the temperament of each child which, representing a constitutional characteristic, is the least modifiable in the personality structure but, if recognized, allows the environment to modulate the stimuli and favor a positive organization of the character [55,56]. Furthermore, emotion regulation correlates, in terms of developmental trajectories, with the risk of developing disorders of the emotional sphere, behavior and conduct. Therefore, also in this case, the difficulties observed represent early risk indicators for detecting conditions at risk, in particular behavioral and psychopathological.
Finally, in the 19-24 months range, the screening form revealed that the difficulties concern various developmental domains: Sensory and emotion reactivity (“the child tends to bring objects to mouth and/or lick them”; “the child overreacts to denial”), communication (“the child does not produce at least 20 meaningful words”; “the child does not make sentences of 2 or more words”; “the child does not blow/ suck”), nutrition (“the child has eating problems”) and attention (“the child does not complete a task”). At this age the child should imitate adults, discover language and recognize the world of emotions, through play and social interaction [29]. Usually, even when he still speaks little, he should be able to communicate through gestures, point to ask, to show and to share. This is also an age in which the expectations of the environment are greater and the comparison with peers highlights the differences [57,58]. However, while on neurodevelopmental disorders, autism, there is always greater attention to early detection, the delays or atypia in areas such as language and emotional regulation are still too often underestimated, depriving children of the possibility of intervening in the most critical developmental window in which, therefore, the system is more receptive. We believe that the results here described highlight that there are areas and domains of development in which, based on the age group and therefore on the developmental stage, children may more frequently present conditions of vulnerability; on the other hand, it is often precisely in those areas that the first alarm bells for major developmental disorders arise.
The goal is to look at development has a whole, rather than going in search of a single pathognomonic indicator of a disorder, so as not overlook conditions which although apparently less serious could make the child more fragile even in the future and undermine the serenity of family and social relationships. For example, while it is known that the absence of babbling and the subsequent delay in language acquisition, as well as asymmetries or other alterations in motor development could both represent the expression of a deficit in the specific developmental area, as well as be the first manifestations of a global disorder, on the other hand we are still witnessing many delays in diagnosis and consequently in therapy. Still too often, in fact, clinicians wait until the age of 3 year before carrying out a specialist evaluation for language delay, perhaps also because not everyone knows the importance that the first processes of regulation, sensory and motor integration can have in determinism of future neuropsychological and socialaffective disorders, such as in the more well-known dyspraxia, but also in sensory processing disorders [59] and in those of emotion, attention and behavior regulation. There is another very important factor in clinical practice with early childhood, which is gradually entering the protocols of outpatient and hospital contexts; that is, the impact of somatic dysfunctions (the most frequent is plagiocephaly) on the adaptation of the child [60]. Many babies develop plagiocephaly, so the incidence in the general population should be monitored. Recent research finds a significant increase and defines plagiocephaly as a risk factor of developmental delays [60-63]. These authors suggest clinicians to monitor infants with plagiocephaly and to prompt referral to early intervention services, who may identify infants with longer term developmental needs [63].
Conclusion
The present research illustrates a large observational study of data collection and the prevalence with which, in the first years of life, deviations from the physiological developmental trajectories occur. Most of the data correlate with those in the literature and with theoretical assumptions and neuroscientific knowledge; this supports us in thinking that the 0-2 neurodevelopmental screening and monitoring form can represent for pediatricians and childhood specialists an easy-to-use, repeatable and reliable tool for the early identification of conditions of vulnerability and neurodevelopmental risk, but above all for the implementation of differentiated care programs ranging from psycho-educational indications to parents, to specialist consultancy and therapeutic, habilitation/rehabilitation pathways, tailored to the needs and global characteristics of each child, not just addressed at the disorder. All the pediatricians who took part in the study appreciated the possibility of having a tool which in just few minutes could allow for the identification of children at risk and/or with problems. Promptly identifying those at risk and/or with problems allows for early intervention, but also and above all, to refer only those who really need them to specialist services, avoiding unnecessary overloading of the competent structures for the diagnosis and treatment of these disorders.
The future objectives will be: the comparison of the forms administered to the same child, which will make it possible to better define the individual developmental trajectory, therefore the trend of the specific indicators by domain in the different age groups, the possible correlation between indicators of different domains, the impact of the early activation of individualized diagnosis and treatment pathways, the comparison of prevalence in the pre- and post-pandemic period from Sars-CoV-2. This study shows that in the developmental age, in the first two years of life, about 15% of the population examined presents a delay or a lack of acquisition of the fundamental stages of neurodevelopment. Epigenetics has shown how the early identification of these and many other conditions is essential in optimizing the interventions necessary to prevent and/or limit damage which overtime acquires harmfulness and becomes more difficulty to remedy.
Author Contributions
Conceptualization, Magda Di Renzo, Paolo Pace and Elena Vanadia; methodology, Monica Rea; writing original draft preparation Lidia Racinaro; supervision Magda Di Renzo & Alberto Villani. All authors have read and agreed to the published version of the manuscript.”
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki. Ethical review and approval were waived because of the use of secondary data.
Informed Consent Statement
Informed consent was obtained from all pediatricians involved in the study.
Data Availability Statement
Data available on request due to restrictions (privacy). The data presented in this study are available on request from the corresponding author. The data are not publicly available due to because they are collected by pediatricians in their private practices.
Acknowledgment
We thank all the pediatricians and family who participated in this research.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Cioni G, D'Acunto G, Guzzetta A (2011) Perinatal brain damage in children: neuroplasticity, early intervention and molecular mechanisms of recovery. Progress in Brain Research 189: 139-154.
- Robins DL, Casagrande K, Barton M, Chen CMA, Dumont MT, et al. (2014) Validation of the modified checklist for autism in toddlers, revised with follow-up (M-CHAT-R/F). Pediatrics 133(1): 37-45.
- Zubler JM, Wiggins LD, Macias MM, Whitaker TM, Shaw JS, et al. (2022) Evidence-informed milestones for developmental surveillance tools. Pediatrics 149(3): e2021052138.
- Renzo DM, Bianchi DCF, Vanadia E, Petrillo M, Racinaro L, et al. (2016) T.U.L.I.P. Protocol (TCE, UOI, Leiter-R as indicators of predictivity) for the assessment of the developmental potential in children with autism spectrum disorders. Autism Open Access 6(4): 188: 1-7.
- Di Renzo M, Bianchi DCF, Vanadia E, Petrillo M, Racinaro L, et al. (2017) Sensory processing and repetitive behaviors in clinical assessment of preschool children with autism spectrum disorder. Journal of Child and Adolescent Behavior 5(2): 1-8.
- Parenti I, Rabaneda LG, Schoen H, Novarino G (2020) Neurodevelopmental disorders: From genetics to functional pathways. Trends in Neurosciences 43(8): 608-621.
- De Felice A, Ricceri L, Venerosi A, Chiarotti F, Calamandrei G (2015) Multifactorial origin of neurodevelopmental disorders: Approaches to understanding complex etiologies. Toxics 3(1): 89-129.
- Oldenburg KS, O'Shea TM, Fry RC (2020) Genetic and epigenetic factors and early life inflammation as predictors of neurodevelopmental outcomes. Seminars in Fetal and Neonatal Medicine 25(3): 101-115.
- Als H (1982) Toward a synactive theory of development: Promise for the assessment and support of infant individuality. Infant Mental Health Journal 3(4): 229-243.
- World Health Organization (2007) International classification of functioning, disability, and health: Children & youth version: ICF-CY. World Health Organization.
- Kliegman RM, Behrman RE, Jenson HB, Stanton BM (2007) Nelson textbook of pediatrics e-book. Elsevier Health Sciences.
- Dubowitz LM, Dubowitz V, Goldberg C (1970) Clinical assessment of gestational age in the newborn infant. The Journal of Pediatrics 77(1): 1-10.
- Einspieler C, Prechtl HF (2005) Prechtl's assessment of general movements: A diagnostic tool for the functional assessment of the young nervous system. Mental Retardation And Developmental Disabilities Research Reviews 11(1): 61-67.
- Dumont MT, Fein D (2005) Screening for autism in young children: The modified checklist for autism in toddlers (M‐CHAT) and other measures. Mental Retardation And Developmental Disabilities Research Reviews 11(3): 253-262.
- Coelho MME, Bronstein J, Aedo K, Pereira JA, Arraño, et al. (2019) M-CHAT-R/F validation as a screening tool for early detection in children with autism spectrum disorder. Rev Chil Pediatr 90(5): 492-499.
- Green E, Stroud L, Bloomfield S, Cronje J, Foxcroft C, et al. (2016) Griffiths scales of child development. (3rd edn), Hogrefe Ltd.: Oxford, UK.
- Bayley N (2006) Bayley scales of infant and toddler development. (3rd edn), San Antonio: NCS Pearson, USA.
- Zubler J, Whitaker T (2022) CDC's revised developmental milestone checklists. American Family Physician 106(4): 370-371.
- Tierney AL, Nelson CA (2009) Brain development and the role of experience in the early years. Zero Three 30(2): 9-13.
- Clark GMB, Clark DA (2015) Sensory development. Pediatric Clinics North Am 62(2): 367-384.
- Bremner AJ, Lewkowicz DJ, Spence C (2012) Multisensory development. Oxford University Press.
- Carruth BR, Skinner JD (2002) Feeding behaviors and other motor development in healthy children (2-24 months). J Am Coll Nutr 21(2): 88-96.
- WHO Multi centre Growth Reference Study Group (2006) WHO motor development study: Windows of achievement for six gross motor development milestones. Acta Paediatr Suppl 450: 86-95.
- Skinner BF (1965) Science and human behavior. Simon and Schuster, NY, USA.
- Chomsky N (2011) How the world works.
- Bruner J (1985) Child's talk: Learning to use language. Child Language Teaching and Therapy 1(1): 111-114.
- Piaget J, Inhelder B, Cecchini M (1971) Mental imagery in the child: A study of the development of imaginal representation. London: Routledge and Kega Paul Ltd, UK.
- Vygotsky LS (2012) Thought and language. MIT press, Cambridge, UK.
- Camaioni L (2017) The development of intentional communication: A re-analysis. In New perspectives in early communicative development. Routledge, London, UK, pp. 82-96.
- Malik F, Marwaha R (2023 Cognitive Development. In: Stat Pearls. Treasure Island (FL): Stat Pearls Publishing.
- Malik F, Marwaha R (2022) Developmental Stages of Social Emotional Development in Children. In: Stat Pearls. Treasure Island (FL): Stat Pearls Publishing.
- Tronick E (2007) The neurobehavioral and social-emotional development of infants and children. WW Norton & Company, NY, USA.
- Trevarthen C (2012) Embodied human intersubjectivity: Imaginative agency, to share meaning. Cognitive Semiotics 4(1): 6-56.
- Trevarthen C (2014) Modes of perceiving and modes of acting. In Modes of perceiving and processing information. Psychology Press, London, pp. 99-136.
- Feldman R (2009) The development of regulatory functions from birth to 5 years: Insights from premature infants. Child Development 80(2): 544-561.
- Kennedy E, Majnemer A, Farmer JP, Barr RG, Platt RW (2009) Motor development of infants with positional plagiocephaly. Physical & Occupational Therapy In Pediatrics 29(3): 222-235.
- Baldwin DA (1991) Infants' contribution to the achievement of joint reference. Child development 62(5): 875-890.
- Batki A, Baron CS, Wheelwright S, Connellan J, Ahluwalia J (2000) Is there an innate gaze module? Evidence from human neonates. Infant Behavior and Development 23(2): 223-229.
- Parsons JP, Bedford R, Jones EJ, Charman T, Johnson MH, et al. (2019) Gaze following and attention to objects in infants at familial risk for ASD. Frontiers in Psychology 10: 1799.
- Benson JB, Haith MM (2009) Social and emotional development in infancy and early childhood. Academic Press.
- Horta BL, Loret DMC, Victora CG (2015) Breastfeeding and intelligence: A systematic review and meta‐analysis. Acta paediatr 104(467): 14-19.
- Zheng W, Chotipanvithayakul R, Ingviya T, Xia X, Xie L, et al. (2022) Sensory stimulation program improves developments of preterm infants in Southwest China: A randomized controlled trial. Frontiers in Psychology 13: 867529.
- Ogundele MO, Yemula C (2022) Management of sleep disorders among children and adolescents with neurodevelopmental disorders: A practical guide for clinicians. World Journal of Clinical Pediatrics 11(3): 239-252.
- Fabrizi L, Slater R, Worley A, Meek J, Boyd S, et al. (2011) A shift in sensory processing that enables the developing human brain to discriminate touch from pain. Current Biology 21(18): 1552-1558.
- Feldman R (2012) Bio-behavioral synchrony: A model for integrating biological and microsocial behavioral processes in the study of parenting. Parenting 12(2-3): 154-164.
- Deliens G, Leproult R, Schmitz R, Destrebecqz A, Peigneux P (2015) Sleep disturbances in autism spectrum disorders. Review Journal of Autism and Developmental Disorders 2: 343-356.
- Guinchat V, Chamak B, Bonniau B, Bodeau N, Perisse D, et al. (2012) Very early signs of autism reported by parents include many concerns not specific to autism criteria. Research in Autism Spectrum Disorders 6(2): 589-601.
- Gross JJ (2013) Handbook of emotion regulation. Guilford publications.
- Gulsrud AC, Jahromi LB, Kasari C (2010) The co-regulation of emotions between mothers and their children with autism. Journal of Autism and Developmental disorders 40(2): 227-237.
- Cassidy J (1994) Emotion regulation: Influences of attachment relationships. Monogr Soc Res Child Dev 59(2-3): 228-249.
- Winnicott DW (2012) The family and individual development. Routledge, London, UK.
- Winnicott DW (2018) The maturational processes and the facilitating environment: Studies in the theory of emotional development by D. W. Winnicott. International Journal of Group Psychotherapy 16(4).
- Stern DN (2020) The motherhood constellation: A unified view of parent-infant psychotherapy. Routledge, London, UK.
- Cole PM, Dennis TA, Smith SKE, Cohen LH (2009) Preschoolers' emotion regulation strategy understanding: Relations with emotion socialization and child self‐regulation. Social development 18(2): 324-352.
- Thompson RA (1994) The development of emotion regulation: Biological and behavioral considerations. Monographs of the society for research in child development 59(2-3): 25-52.
- Marco EM, Macrì S, Laviola G (2011) Critical age windows for neurodevelopmental psychiatric disorders: Evidence from animal models. Neurotoxicity Research 19(2): 286-307.
- Friedmann N, Rusou (2015) Critical period for first language: The crucial role of language input during the first year of life. Current opinion in neurobiology 35: 27-34.
- Zeanah CH, Carter AS, Cohen J, Egger H, Gleason MM, et al. (2016) Diagnostic classification of mental health and developmental disorders of infancy and early childhood dc: 0-5: Selective reviews from a new nosology for early childhood psychopathology. Infant mental health journal 37(5): 471-475.
- Di Renzo M, Laurenti A, Bianchi DCF, Vanadia E, Petrillo M, et al. (2022) Osteopathic evaluation and positional plagiocephaly: A descriptive study on a population of children with ASD. American Journal of Pediatrics 8(2): 104-110.
- Santiago GS, Santiago CN, Chwa ES, Purnell CA (2023) Positional plagiocephaly and craniosynostosis. Pediatric Annals 52(1): e10-e17.
- Martiniuk L, Vujovich DC, Park M, Yu W, Lucas BR (2017) Plagiocephaly and developmental delay: A systematic review. Journal of Developmental & Behavioral Pediatrics 38(1): 67-78.
- Ballardini E, Sisti M, Basaglia N, Benedetto M, Baldan A, et al. (2018) Prevalence and characteristics of positional plagiocephaly in healthy full-term infants at 8-12 weeks of life. Eur J Pediatr 177(10): 1547-1554.
- Glascoe FP (2000) Early detection of developmental and behavioral problems. Pediatrics in review 21(8): 272-279.
© 2023 Magda Di Renzo. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






