- Submissions

Full Text
Research & Investigations in Sports Medicine
Urine Osmolarity in Street Runners
Davanzo Gustavo Gastão1, Borghi, Filipy1, Crege Danilo Roberto Xavier de Oliveira1, da Silva Priscila Cristina1, Lazarin Fernanda2, Matsuguma Aline Mika3, Macedo Denise Vaz2 and Grassi-Kassisse Dora Maria1*
1 Department of Structural and Functional Biology, Institute of Biology, Brazil
2 Departamento de Bioquímica, Instituto de Biologia, Brasil
3 Departamento de Bioquímica e Biologia de Tecidual, Instituto de Biologia, Brasil
*Corresponding author: Dora Maria, Laboratory of Stress Study, Department of Structural and Functional Biology, Brazil
Submission: September 22, 2018;Published: December 10, 2018

ISSN 2577-1914 Volume4 Issue3
Abstract
The hydroelectrolytic balance during the run is essential to a good performance and to the athlete’s health, especially for street runner fans who increase annually. Thereby, we aim to evaluate the hydration state by urine osmolarity in street runners. 62 volunteers (49M/13W, over 18 years) provided urine samples before(a) and after(b) the run. There was a decrease in urine osmolarity (UO) (mOsm/L) after the run (601+34a vs. 692+33b). Men exhibited higher UO than women (750+30b vs 476+88b) and a decrease after the run (631+36a), while women presented no alterations (487+90a). 30% of volunteers started the run with diluted urine (334+35, G1) and 70% in concentrated status (839+17, G2). The G1 group kept their UO in diluted state (267+32) even sweating during the run, while G2 group presented a decrease in UO, however it kept the concentrated status (738+27). Women and G1 start in a water loss state, which was maintained thought-out the run, leading to dehydration. Men and G2 started with high ADH levels, producing concentrated urine and, different than expected, they had urine dilution during the run. Considering this, we could identify an excess of hydration, before and during the exercise. This leads to the hydro-electrolytic disturbance, compromising the athlete performance.
Keywords: Street run; Hydration; Osmolarity; Urine; Runner
Introduction
Regular exercise is recommended for people of all age groups, showing beneficial effects in many diseases, such as diabetes, hypertension, coronary heart disease and heart failure [1-3]. The number of streets run fans increases every year, which is evidenced by the increase of the street races competitions around the world [4].
During exercise, muscle contraction increases the heat production, leading to an increase in body temperature and consequently the production of sweat, helping to cool the skin and lower body temperature [5]. The blood redistribution to the muscle leads to reduced blood flow to the kidney, decreasing the glomerular filtration rate, producing less urine. At the same time, there is an increase in blood osmolarity, triggering an increase in the synthesis of antidiuretic hormone (ADH), which will increase the reabsorption of water in the kidneys, especially in the collecting duct, resulting in hyperosmotic urine [6]. That’s why rehydration is so important after any exercise. The mechanisms involved in urine volume changes and its concentration after water intake are already well described in the literature [7].
The urine osmolarity should average twice the plasma osmolarity (278 to 298mOsm/Kg) and its ideal concentration should be around 600mOsm/L [8,9]. In extreme situations, urine may reach a minimum of 50mOsm/L, indicating an ADH inhibition, reflecting in extremely dilute urine. On the other hand, in situations of dehydration, the body is able to concentrate the urine, reaching values up to 1200mOsm/L [10,11]. Variations of 1% in plasma concentration are sufficient to cause thirst, followed by an increase in ADH levels [12]. The sympathetic nervous system tone is increased during exercise in order to improve cardiac function, pulmonary ventilation and redirect blood flow to the muscles and brain. These changes decrease the blood flow to the gastrointestinal tract, thus hindering gastric absorption throughout the sporting practice [13]. Therefore, the aim of the current study was to analyze the hydration status of the street runners before and after a test, using urine osmolarity as a reference.
Methods
Subjects
Sixty-two volunteers (49 men, 13 women) over 18 years signed the free and informed consent, declaring understanding on the procedures that would be performed during the protocol. The study was conducted according to the guidelines laid down in the Declaration of Helsinki and ethical approval was granted from Research Ethics Committee of the School of Medical Sciences/ University of Campinas (CAAE: 0917.0.146.000-11). The present study was conducted during the 2013 “Volta Unicamp” run, at University of Campinas (Unicamp) in October and the volunteers participated in 10km category started at 8 am.
Procedure
All volunteers collected a urine sample one hour before the race. The research was conducted anonymously. At the end of the race, a new urine sample was collected. All samples were kept on ice and then stored at 4 °C. Osmolarity analysis was performed on Fiske Model OS osmometer.
Statistical analysis
Results for urinary osmolarity (mOsm/L) are presented as mean±SD. Normality test was performed by D’Agostino & Pearson test, followed by paired Student t-test when normality data was detected and Mann Whitney or Wilcoxon test when normality was not detected. Statistical analysis and figures were performed using GraphPad Prism software version 7.00 for Windows (GraphPad Software, San Diego, California, USA). The acceptance level of significance was set at p< 0.05.
Results
The urine osmolarity (UO) results of all volunteers showed a significant decrease after the run (Figure 1). The men (M) presented higher OR than women (W) before (b) the race (M: 750+30b vs. W: 476+88b, Figure 2). We did not observe any differences when comparing women after (a) vs. before (b) the run (W: 476+88b vs. W: 487+90a, Figure 2). However, men presented significant decrease in UO after the run (M: 750+30b vs. M: 631+36a, Figure 2).
Figure 1:Urine osmolarity (mOsm/L) of the street runners before and after 10km run, n=62; *p< 0.05
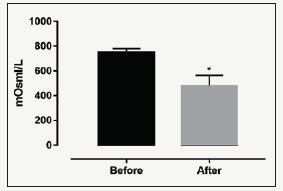
Figure 2:Urine osmolarity (mOsm/L) of street runners for women (n=13) and men (n=49) before and after the 10km run. *p< 0,05: after vs before in the same group; #p< 0.05 between sex.
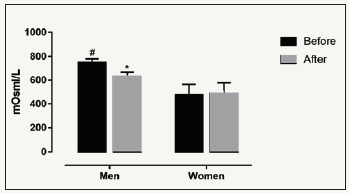
Figure 3:Urine osmolarity (mOsm/L) before and after runners who had their urine diluted at the end of the run, G1 (n=18), and concentrated after the run, G2 (n=44). *p< 0.05: after vs before in the same group; #p< 0,05: G1 vs G2.

We observed that 30% of all volunteers started the run with low UO (< 600mOsm/L, diluted) (334+35, G1) and the majority (70%) in the concentrated state (836+17, G2, Figure 3). After the run, the G1 group maintained a low UO (267+32, Figure 3), while the G2 group presented a significant decrease in the UO values, but maintained a concentrated status (738+27, Figure 3).
Discussion
The main findings of this study are: 1) Urine osmolarity of most volunteers decreased after the run; 2) Men have higher pre-run urine osmolarity values than women. Many studies have shown that there are no changes in plasma osmolarity after exercise, but there was a reduction in body mass due to loss of water and electrolytes during exercise [9,14,15]. If this loss is greater than 2% of the body mass, a dehydration condition ensues and an increase in urine osmolarity is expected [5]. However, the present study introduces that the UO of most volunteers decreased after the run, probably due to fluid intake during the run. The women effort during the run was not enough to cause changes in the osmolarity after the run. A small fraction of the volunteers (30%) started the run with UO less than 600mOsm/L, indicating diluted urine. There are studies in the literature on pre-exercise hyperhydration, such as that of Goulet et al. [16] who found that pre-exercise hyperhydration has beneficial effects, improving the resilience of trained individuals. However, Gigou et al. [17] found no difference in the performance of heattrained runners.
Minor changes in the hydration state can impair performance and physiological function by decreasing stroke volume, thermoregulation and fluid balance in the body [10,18,19]. We observed that most of the runners presented high values of UO at the beginning of the run (G2), which are usually accompanied by higher ADH levels and probable thirst sensation, contributing to the fluid intake during the run. This also corroborates with the UO values presented by men, since the literature indicates that they present higher rates of sweat production and incidence of dehydration after exercise compared to women [20,21]. There are some limitations in this work that should be considered. During the protocol, we did not observe possible changes in the runner’s performance, we did not evaluate ADH and plasma osmolarity before and after the run, as well as the fluid consumption during the protocol. In addition, we did not investigate the urinary flow, which limited the free water clearance analysis. The absence of such data could improve the accuracy of the discussion but did not compromise the results obtained and the interpretation of the data.
Conclusion
This study provides physiological support to improve the knowledge of athlete support staff, such as physiologists, nutritionists and physicians, and to alert athletes’ habits and prescriptions of hydration. It is important that health professionals become aware of the importance of hydration before, during and after runs, as well as properly advise athletes to acquire adequate hydration habits to improve their health and also their performance during the run, taking into account the liquids ingestion and to avoid the state of super hydration observed in our study.
Acknowledgement
The authors wish to thank the volunteers and the support team. The study was funded by CAPES, SAE, Faepex-PRP, CNPq and FAPESP. We state there are no competing financial interests in the work described.
References
- Warburton DE, Nicol CW, Bredin SS (2006) Health benefits of physical activity: the evidence. CMAJ 174(6): 801-809.
- Warburton DE, Bredin SS (2016) Reflections on physical activity and health: What should we recommend? Can J Cardiol 32(4): 495-504.
- Lee IM, Sesso HD, Oguma Y, Paffenbarger RS (2003) Relative intensity of physical activity and risk of coronary heart disease. Circulation 107(8): 1110-1116.
- Meardon SA, Willson JD, Gries SR, Kernozek TW, Derrick TR (2015) Bone stress in runners with tibial stress fracture. Clin Biomech (Bristol, Avon) 30(9): 895-902.
- Sawka MN, Cheuvront SN, Kenefick RW (2015) Hypohydration and human performance: Impact of environment and physiological mechanisms. Sports Med 45(1): S51-S60.
- Sawka MN, Burke LM, Eichner ER, Maughan RJ, Montain SJ, et al. (2007) American college of sports medicine position stand. Exercise and fluid replacement. Med Sci Sports Exerc 39(2): 377-390.
- Perrier E, Demazieres A, Girard N, Pross N, Osbild D, et al. (2013) Circadian variation and responsiveness of hydration biomarkers to changes in daily water intake. Eur J Appl Physiol 113(8): 2143-2151.
- Armstrong LE, Johnson EC, Munoz CX, Swokla B, Le Bellego Let al. (2012) Hydration biomarkers and dietary fluid consumption of women. J Acad Nutr Diet 112(7): 1056-1061.
- Armstrong LE, Maresh CM, Castellani JW, Bergeron MF, Kenefick RW, et al. (1994) Urinary indices of hydration status. Int J Sport Nutr 4(3): 265- 279.
- Perrier ET, Armstrong LE, Daudon M, Kavouras S, Lafontan M, et al. (2014) From state to process: defining hydration. Obes Facts 7(2): 6-12.
- Miles BE, Paton A, de Wardener HE (1954) Maximum urine concentration. Br Med J 2(4893): 901-905.
- Verbalis JG (2003) Disorders of body water homeostasis. Best Pract Res Clin Endocrinol Metab17(4): 471-503.
- Van Nieuwenhoven MA, Brummer RM, Brouns F (2000) Gastrointestinal function during exercise: comparison of water, sports drink, and sports drink with caffeine. J Appl Physiol 89(3): 1079-1085.
- Pereira ER, MT DEA, Mendes TT, Ramos GP, Maia-Lima A, et al. (2017) Evaluation of hydration status by urine, body mass variation and plasma parameters during an official half-marathon. J Sports Med Phys Fitness. 57(11): 1499-1503.
- Francesconi RP, Hubbard RW, Szlyk PC, Schnakenberg D, Carlson D, et al. (1987) Urinary and hematologic indexes of hypohydration. J Appl Physiol 62(3): 1271-1276.
- Goulet ED, Rousseau SF, Lamboley CR, Plante GE, Dionne IJ (2008) Pre-exercise hyperhydration delays dehydration and improves endurance capacity during 2h of cycling in a temperate climate. J Physiol Anthropol 27(5): 263-271
- Gigou PY, Dion T, Asselin A, Berrigan F, Goulet EDB (2012) Pre-exercise hyperhydration-induced bodyweight gain does not alter prolonged treadmill running time-trial performance in warm ambient conditions. Nutrients 4(8): 949-966.
- Casa DJ, Stearns RL, Lopez RM, Ganio MS, McDermott BP, et al. (2010) Influence of hydration on physiological function and performance during trail running in the heat. J Athl Train 45(2): 147-156.
- Cheuvront SN, Carter R, Sawka MN (2003) Fluid balance and endurance exercise performance. Curr Sports Med Rep 2(4): 202-208.
- Sawka MN, Toner MM, Francesconi RP, Pandolf KB (1983) Hypohydration and exercise: effects of heat acclimation, gender, and environment. J Appl Physiol Respir Environ Exerc Physiol 55(4): 1147-1153.
- Bar-Or O (1998) Effects of age and gender on sweating pattern during exercise. Int J Sports Med 19(2): S106-S107.
© 2018 Grassi-Kassisse Dora Maria . This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






