- Submissions

Full Text
Research & Development in Material Science
Synthesis and Mechanical Characterization of Al Casting Produced by Slip Casting and Pressure Less Sintering
Muhammad Asif Hussain1*, Muhammad Nouman1, Ehsan Ul Haq1*, Adnan Maqbool1, Muhammad Ramzan Abdul Karim2, Muhammad Zubair1, Muhammad Shoaib Butt3, Furqan Ahmed1 and Muhammad Asif Rafiq1
1Department of Metallurgical and Materials Engineering, University of Engineering and Technology, Lahore, Pakistan
2Department of Materials Science and Engineering, Ghulam Ishaq Khan Institute of Engineering Sciences and Technology, Topi, Pakistan
3School of Chemical and Materials Engineering (SCME), National University of Sciences and Technology (NUST), Islamabad, Pakistan
*Corresponding author: Muhammad Asif Hussain & Ehsan Ul Haq, Department of Metallurgical and Materials Engineering, University of Engineering and Technology, Lahore, Pakistan
Submission: June 08, 2021;Published: June 14, 2021

ISSN: 2576-8840 Volume 15 Issue 3
Abstract
In this work, the feasibility of using slip casting technique on aluminum powder was studied. It has been found that aluminum powder can be slip cast using sodium alginate as a defloculant binder and water as a liquid vehicle. The results showed that morphology of powder particles plays an important role in slip making, and spherical shaped powder showed better results compared to irregular shaped powder. Slips with water-powder (W/P) ratios of 0.538, 0.600 and 0.666, and with minimum viscosities were casted in plaster molds. Castings were sintered in vacuum furnace at 500 ºC, 550 ºC and 580 ºC for 5h. The results showed that sintered densities were increased with increasing sintering temperature. The optical microscopy of the sintered samples revealed that with the increase in sintering temperature, the fusion of powder particles improved leading to better density and reduced porosity. At an optimum sintering temperature of 550 ºC, mechanical properties were found to be increased while due to decreasing porosity and crystal grain growth lower values of mechanical properties were obtained for the samples sintered at 580 °C. The results of this work showed that there is a great potential to use slip casting process for the manufacturing of aluminum metallurgical parts.
Keywords: Aluminum; Slip casting; Powder morphology; Deflocculant; Metallurgical parts
Introduction
The aluminum (Al) metallurgical parts have been used in various industrial applications including but not limited to structural, aerospace and automotive industries etc. owing to their excellent corrosion resistance, light weight, and good mechanical properties especially good specific strength [1-3]. In conventional ingot making, the properties of Al-alloys were improved by changing the composition of alloying elements, optimizing the processing techniques like heat treatments and mechanical treatments. There are various physical and mechanical properties which are not improved through ingot casting technique, and hence there is need to explore new processing techniques which can produce desire properties for complex shaped components. Metal casting is one of the important and widely used routes to shape Al metallurgical parts, however this process is health hazardous and not ecofriendly [4]. The powder metallurgy (P/M) can provide the possible solutions to the aforementioned issues associated with the production of Al metallurgical parts.
The P/M route provides a flexibility to produce Al alloys of the desired compositions and shape through mixing, pressing and sintering processes with enhanced mechanical and physical properties [5]. Currently, the Al powder metallurgical parts have increasing number of industrial applications especially in aerospace and automotive sector as well as structural and power tools because these parts have competitive properties compared to the components produced through ingot casting, extrusion, melt casting, and screw machine processes [5-7].
Despite all the fascinating features and potentials of the P/M route in Al industry, the use of the P/M is limited by the high cost of processing, equipment and handling of the powders (roughly 1.5 to 2 times that of ingot parts) [8]. To counter the issues, arise in the solid-state P/M processing, aluminum (Al) slip casting process has been proposed in the present work. The process of slip casting is being used in ceramics industry for many years [9,10]. However, the idea of using slip casting technique for metal powders processing is relatively new. The powder metallurgical components prepared through slip-casting have many potential applications in defense sector and automotive industry. A broad range of complex porous, hollow and solid parts of various sizes can be fabricated from ecofriendly metallic powder slip casting process, economically. Previously, the use of copper powder has been reported for slip casting and evaluation was based on the physical and mechanical properties obtained from the cast test specimen [11-14]. Moreover, the use of titanium powder for slip casting has also been reported for producing dental prosthetic appliances, rectangular and tubular porous components [15-18]. The slip casting of aluminum/ aluminum alloy powders has not been reported yet. Therefore, it has been performed in this work, and the slip cast samples have been evaluated on the basis of mechanical properties.
Materials and Methods
Aluminum powder
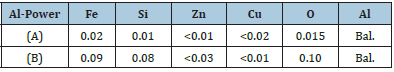
Two types of aluminum powders were used in this study. The powders were purchased from Tanyun Junrong Co., Ltd. China and were used without further processing. Powder (A) had irregular shape and flakes like morphology having average. Powder (B) had spherical shaped particles as it was produced through atomization process (Figure 1). The composition of both powders obtained from the manufacturer is presented in Table 1.
Figure 1: SEM images of Al powders (a) Flake like morphology, (b) Spherical morphology.

Table 1: Size composition of metallurgical slag.
Mold and Al slip preparation, casting and sintering
Plaster mold was made by using slurry prepared by plasterwater ratio of 1.5 and poured into the master mold. It had a cavity of 10mm dia. and 50mm length as shown in Figure 2. After setting of the mixture, the mold was removed from the master mold and dried in oven at about 120 ºC.
Figure 2: Plaster mold of desired casting cavity.

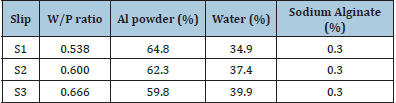
Three different compositions of slips were prepared (i.e. S1, S2 and S3) with different water-powder (W/P) ratios as shown in Table 2. The slips were prepared by dry mixing the Al powder and sodium alginate and then waster was added gradually in this mixture while stirring mechanically. The value of pH of slips was adjusted between 6-10 by adding solution of 2N-NaOH and 2N-HCl. The viscosity of the slips was recorded with the help of a viscometer. After preparation, the slip was poured into the plaster mold and allowed to settle for about 10-20min. The semi dried Al casting was removed from the mold and dried in an oven at 110 ºC for overnight.
Table 2: Composition of slip (mass %).
The sprue and flashes (if any) were removed carefully, and the densities of cylindrical samples, in dried condition, were measured by using mass and volume of the samples. Afterwards, the samples were sintered in a vacuum in Nabertherm furnace (Germany) at 500 ºC, 550 ºC and 580 ºC, and heating rate was kept at 20 ºC/min. Sintering shrinkage was calculated by measuring the volume of the sintered samples before and after sintering. Sintered density of the samples was calculated by using Archimedes method. Relative density was calculated by dividing sintered density with the theoretical density of aluminum (i.e. 2.7g/cc).
Characterization of slip cast samples
The cross-sectional images of the slip cast samples were obtained by optical metallurgical microscope LEICA DM 4000M (Germany) at various magnifications. Scanning electron microscopy (SEM) technique was utilized to examine the powders morphology and particle size of the samples using (SEM, S-3700N, Hitachi, Japan). The phase analysis was achieved by X-ray diffraction machine GNR Explorer (Italy) for Al powder (B), green casting, and sintered samples.
The mechanical properties of all the samples were assessed
by nano hardness tester, (CSM instrument, NHT-MST, 01-0259,
Switzerland). A load of 50mN was applied for 20Sec for each
indentation using Berkovivh indenter (BJ-49) triangular shaped.
The loading unloading curves were obtained for each specimen to
get the hardness, modulus of elasticity and stiffness values (final
readings were obtained by taking the average of various tested
samples).
Results and Discussion
Al powder (A) and powder (B) were selected initially for this study. The SEM images of Al powder (A) and powder (B) are shown Figure (1a & 1b), respectively. Figure 1(a) clearly shows that the powder (A) has irregular flake like morphology while powder (B) shows spherical morphology (Figure 1(b)). Initially, both types of powders were examined by making the slurry and slip casting the samples. It was difficult to prepare the slip of powder (A), and slip casting also came out to be difficult. The slip of powder (A) was sticking with the mold walls, and the cast samples had nonuniform cavities as shown in Figure 3, resulting in low dried density of 0.811g/cc which was around 31.2% of the theoretical density of Aluminum. Also, there was no marked difference between the sintered density (at 550 °C) and the dried density. Whereas, with powder (B), a stable non-sticky slip was formed which produced denser and cavity free cast body. Based on these findings, it was decided to pursue the investigation with spherical shaped Al powder (B). So, in further discussion the term Al powder will be used instead of Al powder (B) for simplification.
Figure 3: Casting of Al powder (A) which is sticking to the walls of mold.

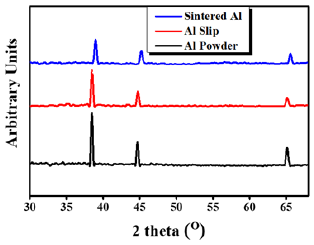
X-ray diffraction was performed on the Al powder (as received), Al dried slurry and sintered samples and the results are shown in Figure 4. Spectrum for as received Al powder and Al dried slurry was found to be same and no phase transformation was observed in the slurry which was dried at constant temperature for a long time. These results encouraged to pursue further investigation for the slip of Al powder in water. Furthermore, the sintered sample (550 °C) did not reveal any peak for aluminum oxide and the spectrum was same as for the as-received Al powder with slight peak shift towards higher 2-theta which might be associated with shrinkage during sintering. These results confirmed that there was no phase change and Al was monolithic after sintering. The acquired spectra showed agreement with the standard JCPDS No. 004‐0787 for aluminum [19,20].
Figure 4: XRD spectra of Al powder, Al-slip and sintered Al.
When fine powder is mixed in water to produce suspension, attractive forces are developed between water molecules and suspended particles in most of the cases which may cause sedimentation. The stability, castability and the green density of the slurry after drying are very important properties and controlled by the shape of particles, stabilizing additives, viscosity and the pH of the slip [18,21]. A stable slip can be made with liquids and dissolved surface-active ions which are adsorbed on the particle surface. Sedimentation and agglomeration is prohibited by repulsion between like charge particles [21]. Al powder was used in the current investigation and a thin oxide layer appeared on particles surface which formed surface OH- groups in suspension or slip. In slip these groups undergo acidic and basic reactions by accepting or releasing hydrogen ions. The resulting surface charge (Zeta Potential) depends upon the concentration of hydrogen ions in the slip [17]. Hence, viscosity of the slip is controlled by resulting surface charge and pH of the slip. Figure 5(a) shows a relationship between pH and the viscosity of slip S1. There is a direct relationship between viscosity and the pH in alkaline and acidic range. A critical point of minimum viscosity of the slip S1 was observed at a pH of 8.4. The viscosity of the slip S1 increased linearly from this critical point towards alkaline or acidic side of pH. A similar trend was observed for the other slips S2 and S3 having higher powder and water ratios (Figure 5(b)). It can be clearly seen in Figure 5(b) that with the increase of water content in slip, viscosity of the liquids decreased, and it is highest in case of slip S1 (water/powder ratio of 0.538). In addition, as water/powder ratio is increased, the slip loses its fluidity and becomes very thick, therefore, slip S1 was taken as the optimum combination of water and Al powder to make the slips for further investigation.
The densities of green castings after drying in an oven at 110 ºC were expected to increase with decrease in W/P ratio. As these two parameters are directly related to set the conditions to form the final casting shape of the slips. With the increase in W/P ratio, the fraction of powder in the unit volume of slip is decreased which ultimately increases the slip fluidity. The water is absorbed by the porous mold via capillary action from the slip casting and makes the powder particles compact. Therefore, an optimum W/P ratio must be achieved for the slip fluidity, and for the good green/ dried density of the casting. Figure 6 shows dried densities of the castings before sintering for three different samples of slips S1, S2 and S3. The dried densities for slips S1, S2 and S3 were found to be 1.82gm/cc, 1.62gm/cc and 1.635gm/cc, respectively. The optimum value for dried density was achieved for slip S1 having 0.538W/P ratio. Therefore, from this point onwards, the effect of sintering temperature on the physical and mechanical properties of the sample S1 (0.538W/P) will be discussed in detail.
Figure 7(a) & 7(b) show the sintering shrinkage and relative densities after sintering of the castings made of slip S1 at temperatures of 500 ºC, 550 ºC and 580 ºC for 5h, respectively (i. e. S1(500), S1(550) and S1(580). After sintering at different temperatures, a small difference was observed in sintering shrinkage and densities of the samples. The shrinkage value varied from 10.41% for S1(500) to 13.10% for S1(580), while the relative density varied from 0.77 to 0.91. Two variables are involved in sintering to tune the density i.e. sintering temperature and time. Here the effect of temperature was observed while keeping the sintering time constant at 5 h. Before sintering, the voids are present between the powder particles in dried casting. During sintering process, the pores are decreased and contact area of the particles is increased that causes densification i.e. decrease in porosity and hence increase in density [22,23].
Figure 8 shows the comparison of apparent, dry, sintered and wrought Al density. The apparent density of Al powder was 1.34g/ cc. After optimization of the slip composition, the green density obtained from the slip cast sample S1 was 1.92g/cc. This green body after sintering S1(580) had the density of 2.34g/cc while the relative density was found to be 0.91. These results show that slip cast profile is almost 13% porous than wrought aluminum. However, it can be improved and the solid body with completely dense structure can be made by adjusting the slip making parameters (i.e. concentration of Al in water, pH and the mixing process).
Figure 9 shows the microstructures of the samples S1(500), S1(550) and S1(580). The microstructure of the sample S1(500) shows two distinct regions i.e. shiny region of sintered Al and dark region of the porosity. It is reflected from the Figure 9(a) that Al particles started to coalesce at 550 °C and porous area was reduced. By increasing the sintering temperature for S1, the porous area was further decreased (Figure 9b & 9c) [20-22]. At a sintering temperature of 550 °C, the microstructure showed higher fraction of shiny area than S1(500) confirming the denser structure, and these results are in conformance with the density data. At a sintering temperature of 580 °C, number of pores were decreased further, and the grain growth was observed because the temperature was approaching near the melting point of Al. Therefore, to avoid grain growth, the samples sintered at 550 °C were selected with the expectation to have better properties than the samples sintered at 580 °C.
The Al powder cast samples after sintering at 500 ºC, 550 ºC and 580 ºC for 5h were subjected to nano-indentation to evaluate their mechanical properties. Figure 10 shows the black, blue, and red curve for the load vs penetration depth for the samples S1(500), S1(550) and S1(580), respectively. The loading curves show that the maximum penetration depth was achieved for the sample S1(500) i.e. about 2100nm and minimum penetration depth for the sample S1(550) i.e. 1200nm. During loading time of 20s, no obvious deformation was observed. The mechanical properties of the subjected specimen were revealed by the unloading curves. A very little bounce back of the material after complete removal of the load shows the plastic nature of the material which is characteristic of metals. Curves showed the higher penetration depth for S1(500) than S1(580).
The higher value penetration depth for sample S1(500) might be attributed to lower densification of the sample at this temperature. The presence of the pores (Figure 9(a)) allowed more penetration which resulted in lower values of hardness for S1(500) as shown in Table 3. Figure 10 (red curve) shows the loading and unloading behavior of the sample sintered at 580 ºC for 5h. The comparison of red and black curves clearly depicts that sample S1(580) has higher penetration depth of 1750nm compared to S1(550), therefore sample S1(580) demonstrated lower values of hardness and modulus of elasticity as shown in Table 3.
Figure 5: (a) pH-viscosity relationship for the slip S1 with W/P ratio 0.538, (b) comparison of pH-viscosity relationship for slips S1, S2 and S3 with increasing W/P ratios.
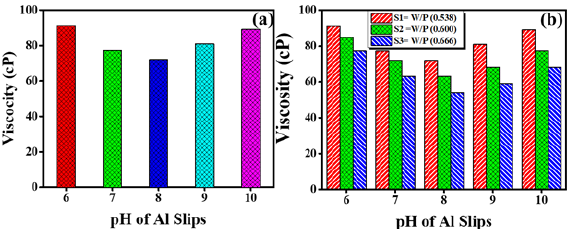
Figure 6: Dried densities of Al-slip castings concentration of Al in water, pH and the mixing process.
Figure 7: (a) Sintering temperature vs shrinkage relationship for casting of slip S1, (b) sintering temperature vs relative density re relationship for casting of slip S1.
Figure 8: Comparison of the densities of Al.

Figure 9: Microstructure of castings made from slip S1 sintered at (a) 500 ºC, (b) 550 ºC, and (c) 580 ºC for 5 hours.

Figure 10: Nano-indentation plots (load vs penetration depth) for sample S1 at different sintering temperature.
Table 3: Nano-indentation data for various slip casted and sintered samples.
The reason for lower hardness for S1(580) might be associated with greater amount of deformation as shown by the sample sintered at higher temperatures, which resulted in higher linear strain and hence lesser modulus of elasticity. Moreover, grain growth was observed in the microstructure of the sample S1(580) which might be the other reason for higher linear strain and lower hardness. Based on above discussion, it can be summarized that sample S1(550) showed best mechanical properties i.e. hardness and modulus of elasticity. Sample S1(500) and S1(580) demonstrated higher values of penetration depth and lower values for hardness, and modulus of elasticity than S1(550). However, the values of mechanical properties were higher for sample S1(580) than S1(500). The higher penetration depth and linear strain of S1(500) can be attributed to less densification and porous structure of the samples, while in case of S1(580) the phenomenon of grain growth is responsible for this behavior.
Conclusion
Aluminum powder metallurgical samples were synthesized by slip casting technique for the first-time using water as a liquid vehicle. Powders morphology played an important role in making a slip. Spherical shaped powder particles were selected to make slip because flaky powder particles produced slip which was sticking with the mold walls and the cast sample had non-uniform cavities. The viscosity of the Al powder slip was controlled by the pH of the solution and stable slip with optimum viscosity was obtained in the acidic range 8.4. In addition to pH, water to powder (W/P) ratio was another controlling factor for slip making and optimum W/P ratio was 0.538. The density of slip cast samples was increased with increasing temperature, however, higher sintering temperature led to grain growth. The modulus of elasticity, hardness and stiffness initially increased with increasing temperature from 500 °C to 550 °C while a more ductile behavior was observed with increasing temperature up to 580 °C due to grain growth.
Acknowledgment
The authors pay their gratitude to University of Engineering and Technology, Lahore, Pakistan for its technical support. Authors also thank PITMAEM, PCSIR Laboratories, Lahore, Pakistan for their assistance in materials characterization.
References
- Kashaev N, Ventzke V, Çam G (2018) Prospects of laser beam welding and friction stir welding processes for aluminum airframe structural applications. Journal of Manufacturing Processes 36: 571-600.
- Bull MJ, Gupta AK, Wheeler MJ (2001) Precipitation-hardened aluminum alloys for automotive structural applications. Google Patents.
- Banhart J (2000) Properties and applications of cast aluminum sponges. Advanced Engineering Materials 2(4): 188-191.
- Neto B, Kroeze C, Hordijk L, Costa C, Pulles T (2009) Strategies to reduce the environmental impact of an aluminium pressure die casting plant: A scenario analysis. Journal of Environmental Management 90(2): 815-830.
- Smith L (2003) A knowledge-based system for powder metallurgy technology. John Wiley & Sons, USA.
- Huo S, Heath B, Ryan D (2009) Applications of powder metallurgy aluminums for automotive valve-trains. SAE International Journal of Materials and Manufacturing 1(1): 511-515.
- Anderson IE, Foley JC (2001) Determining the role of surfaces and interfaces in the powder metallurgy processing of aluminum alloy powders. Surface and Interface Analysis: An International Journal devoted to the development and application of techniques for the analysis of surfaces, interfaces and thin films 31(7): 599-608.
- Hunt WH, Cook CR, Sawtell RR (1991) Cost-effective high-performance P/M aluminum matrix composites for automotive applications.
- King AG (2002) Ceramic technology and processing. Elsevier, Netherlands, pp. 56-113
- Fernandes RS, De Campos E, Santana JGA, Mota RP (2017) Porous ceramics obtained by Slip/Starch Casting Consolidation Method (SSCC). Materials Science Forum 881: 52-57.
- Ru J, Kong B, Zhu H, Shi Z, Zhang D, et al. (2014) Microstructure, capillary performance and gas permeability of biporous copper fabricated by tape casting. Powder Technology 256: 182-187.
- Sulinski HV, Lipson S (1966) Slip-casting of copper powder, modern developments in powder metallurgyed. Springer, Germany, pp. 266-277
- Supriadi S, Putra N, Ariantara B, Rahmanto DD 92016) Fabrication of lotus-type porous copper using slip casting and sintering techniques for heat pipe applications. Applied Mechanics and Materials 819: 601-605
- Le Ferrand H, Bouville F, Niebel TP, Studart AR (2015) Magnetically assisted slip casting of bioinspired heterogeneous composites. Nature Materials 14(11): 1172.
- Xu Q, Gabbitas B, Matthews S (2013) Influence of porosity on mechanical behaviour and gas permeability of Ti compacts prepared by slip casting. Materials Science and Engineering: A 587: 123-131.
- Xu Q, Gabbitas B, Matthews S (2014) Titanium compacts with controllable porosity by slip casting of binary powder mixtures. Powder Technology 266: 396-406.
- Xu Q, Gabbitas B, Matthews S, Zhang D (2013) The development of porous titanium products using slip casting. Journal of Materials Processing Technology 213(8): 1440-1446.
- Xu Q, Gabbitas B, Matthews S, Zhang D (2014) The effect of binder and plasticizer on porous titanium compacts prepared by slip casting. Procedia Materials Science 4: 81-84.
- Mahendiran C, Ganesan R, Gedanken A (2009) Sonoelectrochemical synthesis of metallic aluminum nanoparticles. European Journal of Inorganic Chemistry 2009(14): 2050-2053.
- Wang Z, Gong J, Ma J, Xu J (2014) In situ growth of hierarchical boehmite on 2024 aluminum alloy surface as superhydrophobic materials. RSC Advances 4(28): 14708-14714.
- Ohkawa S, Ishii K, Uo M, Sugawara T, Watari F (2004) Slip casting of titanium powder for dental prosthetic appliances. Materials Transactions 45(4): 1132-1139.
- Hussain MA, Maqbool A, Khalid FA, Farooq MU, Abidi IH, et al. (2015) Improved sinterability of hydroxyapatite functionally graded materials strengthened with SS316L and CNTs fabricated by pressure less sintering. Ceramics International 41(8): 10125-10132.
- Hussain MA, Maqbool A, Khalid FA, Bakhsh N, Hussain A, et al. (2014) Mechanical properties of CNT reinforced hybrid functionally graded materials for bioimplants. Transactions of Nonferrous Metals Society of China 24: s90-s98.
© 2021 Muhammad Asif Hussain & Ehsan Ul Haq. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






