- Submissions

Full Text
Polymer Science: Peer Review Journal
Nano-Organization and Polymers Viscous Flow
Sokolova LV*, Khrustalev AN and Gromin SA
MIREA, Russian Technological University, Russia
*Corresponding author: Sokolova LV, MIREA, Russian Technological University, Russia
Submission: October 21, 2021;Published: October 28, 2021

ISSN: 2770-6613 Volume2 Issue3
Opinion
It is generally believed that the mechanism of viscous flow of polymers on movement of
chains relative to each other due to the movement of segments. However, nana-organization
polymer is formed during synthesis of polymers, and it has a thermodynamic nature. The
structure of the nana-organization includes the different content and size of the ordered
formations, their possible location in the polymer volume, the packing density of the
passing chains in the disordered part, which determines the degree of its ordering [1-3].
The temperature dependence of the viscosity of the homologues of atactic polystyrene in
the coordinate equation of Arrhenius detect an abrupt change in the region of 125-130, and
145-150 °C (Mη=4.104) (PS-1), 165-170 and 200-205 °С (Mη =2.2.105) (PS-2) and 185-190
°С (Mη=8×105) (PS-3). These changes are explained by the manifestation of ρ0- and ρ1-phase
transitions in PS-1, ρ1- and ρ2- in PS-2 and ρ2-transition in PS-3. The activation energy of the
viscous flow of PS-1 decreased from 64 to 50 and to 46kJ/mol, respectively, and from 65 to
40kJ/mol with the manifestation of the ρ1-transition in PS-2, and from 140 to 40kJ/mol in the
case of PS-3. About the existence of change in the structure of the nana-organization of these
homologues is evidenced by the wide angle X-ray diffraction data obtained by heating them.
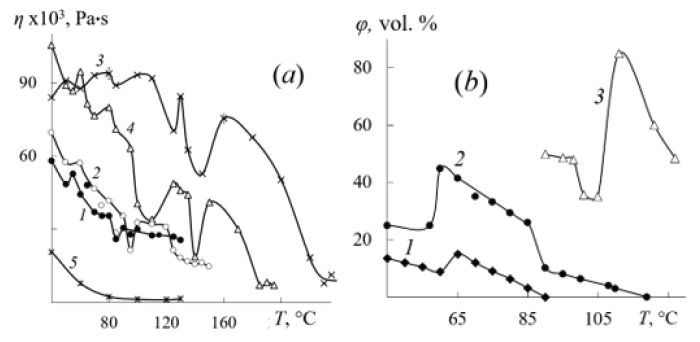
Phase transitions also determine the complex temperature dependences of the dynamic
viscosity of cis-1.4-polyisoprene (97% cis-1,4-units, Mη=9.8×105, Tc−69 °C) (PI) and cis-1.4-
polybutadiene (98.5% cis-1,4-units, Mη=3×105, Tc−105 °C) (PB), butyl rubber (Mη=3×105,
Tc−73 °C) (BR), polyisobutylene (Mη=1.1×106 and Mη= 7.5×104, Tc−73 °C) (PIB-1, PIB-2,
respectively) (Figure 1a). The cohesion energy of all polymers is approximately the same,
with the exception of PB. A sharp decrease in the activation energy of the viscous flow of PB
and PI is due to the complete disintegration of their nana-organization at 90 °C and 118 °C,
respectively according to the data of the diffusion method (ρd-transition) (Figure 1b). The use
of diphenyl guanidine as a penetrant, which is not indifferent to the structure of the PI nanaorganization
(caused a decrease in its viscosity), accompanied by ρd-transition till 118 °C in
case of PI (Figure 1b). The manifestation of the ρ4-phase transition in PI, the ρ6-transition in
PB at 65 °C, and especially the ρ6-transitions in PIB at 120 °C cause increase size and decrease
content of nanostructures and increase of volume content in disorder part of polymer. The
temperature of phase transitions is related to the Tc of polymers by the ratio regardless of
their chemical structure: Ti = {[1.2+0.2 (i-2)]±0.05Tc, where i is the ordinal number of the
phase transition [1]. The decrease in polymer viscosity with an increase in temperature is
explained by an increase in the size of nanostructures while at the same time increasing the
free volume in the disordered part of the nana-organization. Accordingly, the high viscosity of
polymers is a manifestation not so much of the structural characteristics of macromolecules
as of the degree of ordering of the nana-organization. This is most clearly manifested with a
slight deformation (slight stress). The complex temperature dependence degenerates with increasing deformation. The activation energy of the viscous flow
of PB decreased from 12.08 to 1.52kJ/mol, from 11.84 to 1.98kJ/
mol for PI, from 44.57 to 0.43kJ/mol for PIB, as well as from 8.9
to 1.97kJ/mol in BR. The activation energy of the viscous flow of
polymers above the temperature of the ρd-phase transition indicates
that their flow in this case is a consequence of the movement of
segments. In other words, the activation energy of flow above ρd-
phase transition does not depend on the temperature. Based on this,
to observe the transition from state with the non-Newtonian flow
to the Newtonian state of the polymer higher of the temperature
ρd-phase transition.
Figure 1: Temperature dependences of the dynamic viscosity of PB (1), PI (2), PIB-1 (3), PIB-2 (5) and BR (4) (a) and the volume content of nanostructures in them (b).
References
- Sokolova LV (2006) Plast Massy 5(13).
- Sokolova LV (2017) Flexibility of macromolecules and structure formation in amorphous polymers. Polym Sci Ser A 59(4): 483-495.
- Sokolova LV (1994) Polym Sci Ser B 36(10): 1454.
© 2021 Sokolova LV. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






