- Submissions

Full Text
Polymer Science: Peer Review Journal
Synthesis and Application of C-Dot - The Material for the Future
Sardar S and Bandyopadhyay A*
Department of Polymer Science and Technology, University of Calcutta, India
*Corresponding author: Abhijit Bandyopadhyay, Department of Polymer Science and Technology, University of Calcutta, Kolkata-700009, India
Submission: June 10, 2021;Published: June 18, 2021

ISSN: 2770-6613 Volume2 Issue1
Abstract
In this mini review, we have tried to highlight various synthetic routes of carbon dot (C-dot) and their applications. We have mentioned both top-down and bottom up approaches and substrate requirement for its synthesis and specific properties that the resultant C-dots is going to display. New age applications like energy harvesting and storage, coloured lighting etc. of C-dots are also stated in this review.
Keywords: C-dot; Carbon; Synthesis; Graphite; Electro-chemical fields; Organic compounds
Introduction
C-dot has been regarded as one of the most useful and interesting nanomaterial among the contemporary nanomaterials because of its unique features such as excellent photo luminescent property, outstanding biocompatibility, long cycle life, light weight, small size, unique physical characteristics, easy aqueous dispersibility and pH sensitivity [1]. The number of scholarly articles published on C-dot is steadily increasing in number covering its expanded field of applications. As of now, C-dots have been explored as agents for bio imaging, sensing, photo catalysis, electro catalysis, fluorescent ink, light emitting diode, and solar cells [2].
Mini Review
Chemical structure of C-dot varies on the route adopted for its synthesis. It typically has a size of a few nanometers and has molecular weight of a few thousand to tens of thousands of times that of bigger particles. The surface often contains a variety of functional groups such as –OH, –COOH and –NH2, which makes them water dispersible and also boosts their ability to polymerize forming various inorganic, organic, or physiologically active molecules [3]. The synthesis of C-dot can be done in a variety of ways. This may be divided into two categories: “top-down” and “bottom-up” procedures. With the aid of arc-discharge, electrochemical synthesis, laser-ablation, and chemical oxidation, the “top-down” approach breaks bulk carbon materials such as carbon nanotubes, graphite, candle soot, and activated carbon into C-dots [2]. However, no definite substrate is required for the bottom-up technique. In this case, it is made from a variety of organic compounds through carbonization using plasma treatment, hydrothermal or acidic oxidation reactions, microwave, and carbonizing organics [4]. All of these aforementioned techniques need complex experimental settings, and they are also difficult techniques for the creation of luminous C-dots as well. As a result, green techniques are critically explored for the manufacture of C-dots particularly for opto-electronic applications. Researchers are now banking on soy milk, chicken eggs, plant leaves, pomelo peel, grass, and other inexpensive or readily accessible natural resources as precursors for the production of C-dots to make the process cheap and more viable. Electro-Luminescence (ECL) features are found to be pronounced in the synthesized C-dots as well. Graphite has also been found to produce C-dots with enhanced Electro-luminescence behaviour. In that case, the C-dots are produced through electrochemical oxidation of graphite. Since the potential cycle is maintained between +1.8 and -1.5V, the resultant C-dots display an enhanced Electroluminescence property having the maximum at 535nm. When C-dot is dispersed in ethanol, the dual ECL signal is often observed (TBAB). Except for certain recent studies on electrochemical abilities in the context of electrosynthesis, electro-catalysis, and sensing of C-dots, only a little study has been done on the electrical characteristics of C-dots so far [5].
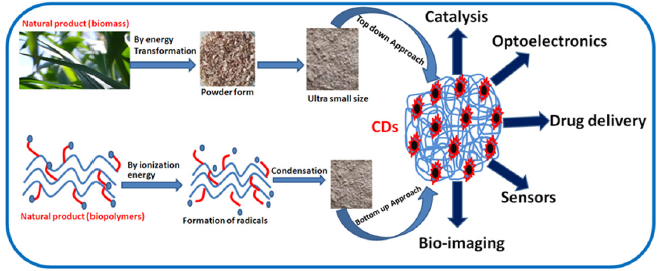
Recently, formation of C-dot based polymer composites has been reported whereby epoxy resin, agarose gel, and iono gels have been used as the matrix and the resultant composites are targeted for diverse luminescent hues and white light generation [6]. Energy production/storage at low cost have been the priority of the current era. Due to the abundance of trap states, C-dots exhibit charge entrapment properties. As a result, they are advantageously employed as an electrical capacitor. Since the practical demonstration of the quantum size impact on photoluminescence of C-dots and the theoretical prediction of quantum confinement, it has received a lot of attention in optoelectrical and photovoltaic technologies in the last few years. They have the potential to still improve the efficiency with which sunlight is converted into energy. C-dots are also attracting a lot of attention as a light-harvesting material for solar cells. Energy harvesting reduces CO2 emissions which in other way lowers the renewable energy cost. (Figure 1) summarizes the synthesis and application fields of C-dots through a schematic [7]. The synthesis procedure and their subsequent application is demonstrated in tabular form (Table 1), [8].
Table 1: The source and their application of C-dots [8].
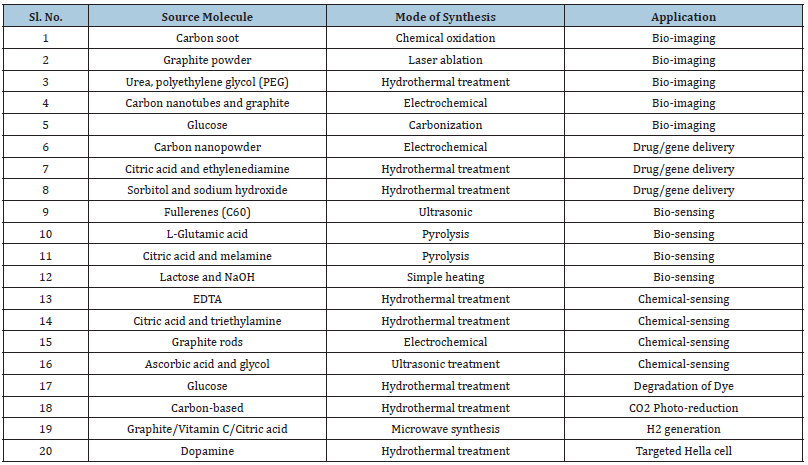
Figure 1:Schematic of synthesis and different applications of C-dots.
Conclusion
In conclusion, it can be said that C-dots are going to be the key material in near future to address the key issues like energy harvesting and storage, low cost lighting, diagnostic etc. either in sole form or in the form of flexible polymer composites. It is thus important to explore its structure-property relationship for extracting more benefits in opto-electrical and electro-chemical fields.
References
- Song Y, Zhu S, Shao J, Yang B (2017) Polymer carbon dots-A highlight reviewing their unique structure, bright emission and probable photoluminescence mechanism. J Polym Sci Part A Polym Chem 55(4): 610-615.
- Jlassi K, Eid K, Sliem MH, Abdullah AM, Chehimi MM, et al. (2020) Rational synthesis, characterization, and application of environmentally friendly (polymer-carbon dot) hybrid composite film for fast and efficient UV-assisted Cd2+ removal from water. Environ Sci Eur 32: 12.
- Yang S, Zhang Y, Xue Y, Lu S, Yang H, et al. (2020) Cross-linked polyamide chains enhanced the fluorescence of polymer carbon dots. ACS Omega 5(14): 8219-8229.
- Woo J, Song Y, Ahn J, Kim H (2020) Green one-pot preparation of carbon dots (CD)-embedded cellulose transparent film for Fe3+ indicator using ionic liquid. Cellulose 27: 4609-4621.
- Aziz SB, Abdullah OG, Brza MA, Azawy AK, Tahir DA (2019) Effect of carbon nano-dots (CNDs) on structural and optical properties of PMMA polymer composite. Results Phys 15: 102776.
- Putro PA, Yudasari N, Isnaeni, Maddu A (2021) Spectroscopy study of polyvinyl alcohol/carbon dots composite films. Walailak J Sci Technol 18: 1-12.
- Ve A, Dergisi D, Dinc S, Kara M (2018) Synthesis and applications of carbon dots from food and natural products: A mini-review. J Apitherapy Nature 1(1): 33-37.
- Sharma A, Das J (2019) Small molecules derived carbon dots: Synthesis and applications in sensing, catalysis, imaging, and biomedicine. J Nanobiotechnology 17: 1-24.
© 2021 Bandyopadhyay A. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






