- Submissions

Full Text
Progress in Petrochemical Science
A Mini-Review on CO2 Reforming of Methane
Mohamad Hassan Amin* and Suresh K Bhargava
Centre for Advanced Materials and Industrial Chemistry, RMIT University, Australia
*Corresponding author:Mohamad Hassan Amin, Centre for Advanced Materials and Industrial Chemistry, School of Science, RMIT University,Melbourne, VIC 3001, Australia
Submission: May 22, 2018;Published: June 07, 2018

ISSN 2637-8035Volume2 Issue2
Abstract
The reforming of methane is considered as one of the industrially important processes for decades, as the process converts natural gas to valuable syngas (a mixture of H2 and CO). There are three major reforming processes, which are classified based on the energetic of the process and reforming agent. Catalytic steam reforming (endothermic reaction), partial oxidation (exothermic reaction) and auto thermal reforming (combined exothermic and endothermic reactions) of methane are commercially available processes for syngas production. Carbon dioxide/dry reforming (endothermic reaction) is another alternative process that has received significant attention in recent years, which demonstrates the environmental benefit. This is a mini review on the development of CO2 reforming of methane.
keywords: CO2 reforming of methane; Dry reforming of methane; Natural gas; syngas
Introduction
Due to increasing anthropogenic CO2 emission problems, the utilization of carbon dioxide as a feed stock has become a topic of significant importance. In addition, energy is the most important issue to modern economies, and it is predicted that fast-rising energy demand will require the US $45 trillion for new infrastructure investment by 2030 [1]. In particular, natural gas-toliquids (GTL) process, which increases the options for the transport and marketability of the remote gas resources has been, received much attention. Fischer-Tropsch (FT) technology is the main technology for GTL process. However, current state-of-the-art, this technology is very expensive due to the high cost of production of the syngas (mixture of H2 and CO) that is the feedstock for the GTL process, when carried out by the presently practiced steam reforming of methane (SRM) process. Therefore, during the past three decades, the conversion of methane to syngas is a hot topic for industrial and academic research [1,2].
The production of syngas from methane can be done through different approaches using an oxidizing agent that will oxidize methane to carbon monoxide while producing hydrogen in a ratio that will vary depending on the type of oxidant used. Carbon dioxide can be considered an oxidizing agent for the oxidation of methane instead of oxygen or water for the production of syngas via a reaction called CO2/dry reforming of methane, DRM. In that case, DRM involves the most reduced form (CH4) combined with its most oxidized form of carbon (CO2). This process was introduced by Fischer and Tropsch for the first time in 1928 [3]. However the extensive investigation on the reforming of methane with carbon dioxide only started in the 1990s when increasing concerns about greenhouse effects were raised by the international scientific community. In comparison to conventional technologies, SRM and POX, this process is suitable for remote natural gas or crude oil fields, where water supplies are limited [4]. DRM converts two cheap greenhouse gases (CH4, CO2) concurrently into valuable syngas (CO + H2), which is a key industrial intermediate. Considering that natural gas deposits contain vast amounts of CO2, its emission to the atmosphere can be avoided if used in DRM, thus lowering the purification costs [5]. Due to its strong endothermic characteristic, it can be environmentally valuable if the necessary reaction heat comes from nuclear or renewable energy. In addition, it produces syngas with an H2: CO molar ratio that is appropriate for products including Fischer-Tropsch fuels and dimethyl ether (DME) [6].
Reaction scheme and thermodynamics
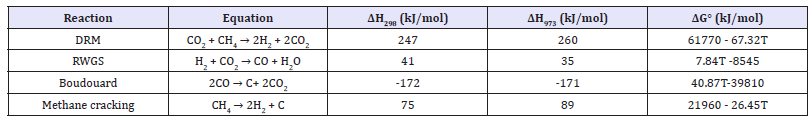
The DRM reaction is favoured by low pressure [7], however, the strong C-H bonds (439kJ mol-1) in methane [8] lead to an endothermic process that requires high temperatures for CH4 conversion. The DRM reaction is accompanied by several side reactions, including methane cracking, reverse water gas shift and the Boudouard reaction. The thermodynamics of the DRM reaction and side reactions at atmospheric pressure are listed in Table 1 & [9,10] (T is the absolute temperature in degrees Kelvin): Assuming ΔG°=0, at atmospheric pressure, methane cracking occurs at temperatures above 557 °C (830K) and DRM can proceed at temperatures above 644 °C (918K). Reverse water-gas shift (RWGS) and Boudouard reactions also take place at temperatures lower than 817 °C (1090K) and 701 °C (974K), respectively. Consequently, the maximum carbon will be deposited at 700°C from both methane cracking and Boudouard reactions. At reaction temperatures above701°C at atmospheric pressure, the Boudouard reaction will be excluded and CH4 cracking can be the only possible source of carbon deposition on the surface of catalysts. At temperatures higher than 817 °C and atmospheric pressure, the RWGS reaction will also be excluded, and consequently, the H2/CO ratio will reach values close to unity.
Table 1: DRM and side reactions at atmospheric pressure.
Kinetics
Although many researchers have conducted investigations on the reforming mechanism, there still are some disputations regarding the details of the accurate reaction mechanism and the rate-determining steps (RDS). For example, the number of RDSs is still argued by some scientists. Some researchers reported that the mechanism of the dry reforming reaction was one single RDS [11-14], while others reported a mechanism with two RDSs [15- 17] for the same reaction. The main reasons for the differences in the reported reforming mechanism can be attributed to two facts: (1) the different supports and promoters employed in the reforming reaction may result in the variation of the reforming mechanism, which has been noticed by some research groups [18-20]; and (2) the mechanism investigation was conducted at different temperatures, which may remarkably affect the reforming mechanism and the RDS. Several different kinetic models have been reported. Most of the kinetic models are based on the reversible dissociative adsorption of CH4 on the catalyst active site to produce H2, or on the reversible and dissociative adsorption of CO2 on the support to yield CO [7].
Catalysts
Based on the above discussion, it appears that the maximum activity and H2/CO ratio and the minimum carbon deposition can be achieved at high temperatures (e.g. higher than 817 °C). On the other hand, when the reaction takes place at high temperatures, high energy consumption makes this process unfeasible for industrial applications. The use of catalytic systems may lead to higher activity at lower temperatures and, as a result, reduction of the energy consumption in the process, which would allow this technology to get closer to an economical process. Thermodynamic calculations showed that the temperature needed for 50% CO2 conversion in DRM without catalyst is 1035°C (1308K) [21], but on the basis of existing literature data and our experiments, very high CH4 and CO2 conversions can be achieved in temperatures as low as 700°C using catalytic systems.
In general, it has been accepted that the catalytic CO2 reforming of methane has a bi-functional mechanism. CH4 can be adsorbed and activated by the active sites of most of the transition metals, mainly groups VIII b of the periodic table, while CO2 is adsorbed and activated by oxides [7]. Therefore, the DRM reaction can be catalyzed by most of the transition metals supported on oxides.
Over the past two decades, intensive efforts have been devoted by numerous research groups to develop catalysts that can achieve high catalytic activity and stability with maximum H2/CO molar ratio and minimum coke formation for DRM at low temperatures [22].
Despite numerous publications and comparison between different catalysts in different reaction conditions in the literature, there still are some disputations regarding the highest active and stable catalyst in DRM. Although, the performances of catalysts can be affected by a variety of factors other than composition, such as content of active components, preparation methods, calcinations ambient, calcinations temperature, reduction and activation procedures, precursors of active components, etc., it is well accepted that nickel has the best catalytic performance among the all tested catalysts except for the noble metals. In the case of noble metal catalysts, some researchers reported that Pd showed the highest activity and stability [23,24] while others reported that Rh and Ru catalysts exhibited the highest activity and stability among the noble metals [25-30]. Likewise, there is an ongoing argument between researchers involving Ni and noble metals to decide which one has the best catalytic behaviour during DRM. Many researchers reported higher catalytic activity for Ni-based catalysts in comparison with metals catalysts [24,26,29,31,32] while some researchers reported that noble metals have better catalytic activity than Ni-based catalysts [26,33]. Admittedly, on a mole for mole basis, Ni is about 10000 times cheaper than Pt and 200 times cheaper than Pd [34]. Therefore, from practical and industrial viewpoints, Ni-based catalysts are the most attractive and promising for DRM [2,35].
Ni-based catalysts, however, have a tendency to undergo deactivation via carbon deposition and sintering and hence significant efforts have been devoted to improving these catalysts. Approaches to improve the activity and stability of Ni-based catalysts for DRM have focussed mostly on the discovery of better supports and suitable promoters/Ni-based catalysts [2,6,36-41]. Researchers reported both positive and negative effects for the addition of a second and/or third metal (bimetallic and tri-metallic) on the performance of catalysts. SiO2 and Al2O3 are two of the most often investigated catalyst support with high melting points and specific surface areas. MgO, CaO, CeO2, La2O3, TiO2 and ZrO2 are also commonly studied. The effects of the precursor, preparation conditions, type of reactor (including plasma reactors, fixed and fluidized bed reactors, membrane reactors) [42], heating method [43], reduction method, feed composition and space velocity on the reforming reaction were investigated. There have been reports showing that the application of plasma may be a good approach to induce high conversions of CO2 and CH4. However, plasma technologies are very expensive, and the utilization of electrical current to produce plasma may be counterproductive to the carbon balance.
Commercial processes
Recently, shale gas resources are growing throughout N. America, the U.K., China and Eastern Europe, a matter which has urged the attention of government, industrial R&D and academics for the development of new processes and technologies for its conversion to fuels and chemicals in competition with conventional routes. These include direct/indirect DME production via DRM and combination of DRM and another process to create innovative economic opportunities [44]. The energy input for DRM requires approximately 20% more energy compared with steam reforming. However this is not a prohibitive extra energy cost for this chemical reaction [45]. Overall the reaction requires +205kJ/mol over the -803.3kJ/mol corresponding to the lower heating value of methane, which is about 25.5% of the energy value of methane. Therefore, a process that would aim at that range of energy consumption for DRM would be competitive to steam reforming, and thus scalable to industrial applications [46]. Importantly, these two reactions give rise to syngas with different H2/CO molar ratios. Both are useful in the formation of syngas for ultimate liquid fuel production [45]. The optimal DME productivity and best material utilization were reported as a H2: CO ratio of 1:1, which can be produced by DRM [47]. This process has been used in a limited number of industrial processes so far. Dry reforming of methane and other hydrocarbons has been applied commercially to produce pure CO from natural gas by the CALCOR process (Calcor process) to produce high purity carbon monoxide [48] or to obtain syngas with a low H2: CO molar ratio as a feedstock for further chemical synthesis by the SPARG process [49].
HTC pure-energy has developed a unique two-stage process for producing hydrogen by the CO2 reforming of methane over Nibased mixed oxide catalyst [46]. HTC technology is feed-flexible and can handle multiple sources of methane including natural gas, sub-quality natural gas, stranded natural gas, flare gas and biogas methane [46]. A patent by Schoedel et al. [50] recently disclosed a method for producing dimethyl ether from methane or natural gas comprising a dry-reforming step, followed by the conversion of carbon monoxide and hydrogen into dimethyl ether. They claim that the dry-reforming step and the synthesis step are carried out at identical pressures or at pressures which do not differ by more than 3bar-preferably by not more than 1 bar [50]. A 3,030 tonnes/ day methanol plant based on DRM was set up in Iran in 2004 [51]. It was reported that the application of DRM results in a very energy efficient plant as the energy consumption is 5-10% less than that of a conventional plant [51].
Conclusion
Although DRM technology is promising from an environmental point of view and there are some reports of using the DRM process in some industrial plants to produce methanol or/and DME, this process is still considered as an uneconomical method. Other than the fact that the DRM is highly endothermic when compared to the steam reforming and ATR, the reaction often involves the production of carbon, which leads to deactivation of the catalyst. In addition, DRM has the downside of requiring large amounts of pure carbon dioxide, which cannot be easily obtained industrially except through a few specific technologies [46]. As an example, isolating carbon dioxide from the air or thermal power plants would be very expensive (even if CO2 has a tipping fee). However, technologies such as gasification or fermentation are technologies that produce significant volumes of high purity CO2 and could be good candidates for the first generation of industrial-scale dry reforming processes [46].
Although DRM alone has not found commercial applications, combined DRM and steam reforming systems have been operational in the industry in the last several years [52]. The combination of DRM and steam reforming has more attractiveness in its application compared to DRM alone because the addition of steam reduces the risk of carbon deposition and gives better control over the syngas ratio [53]. Since their optimum reaction temperature is close, these reactions can be conducted in one step [54]. In addition, the combination of DRM and POX (CDPOX) appears to be a more commercially attractive method to produce syngas because it couples the advantages of DMR and POX and offsets their disadvantages, simultaneously [55]. Compared to POX and DMR, CDPOX is a green process and has the following advantages [55]: 1) effective heat supply due to the combination of endothermic and exothermic reactions, 2) Controllable product ratio of H2/CO according to the need of the post-process, and 3) A safer operating environment due to the reduction of hot spots produced in POX alone [53]. In addition to the bi-reforming processes, recently a synergetic combination of DRM, SRM, and POX (tri-reforming) has been proposed.
References
- Mohamad JP, Sage V, Lee WJ, Periasamy S, Deepa D, et al. (2015) Trireforming of methane for the production of syngas: review on the process, catalysts and kinetic mechanism. Apcche 2015 Congress Incorporating Melbourne.
- Amin MH, Sudarsanam P, Field MR, Patel J, Bhargava SK (2017) Effect of a swelling agent on the performance of Ni/porous silica catalyst for CH4- CO2 reforming. Langmuir 33(40): 10632-10644.
- Tropsch H, Fisher F (1928) Conversion of methane into hydrogen and carbon monoxide. Brennst Chem 3: 39-46.
- Verykios (1995) A stable and active nickel based catalyst for carbon dioxide reforming of methane to synthesis gas. J Chem Soc Chem Commun, pp. 71-72.
- Goeppert MC, Paul JJ, Prakash S, George A (2014) Recycling of carbon dioxide to methanol and derived products-closing the loop. Chemical Society Review 43: 7995-8048.
- Amin MH, Mantri K, Newnham J, Tardio J, Bhargava SK (2012) Highly stable ytterbium promoted Ni/γ-Al2O3 catalysts for carbon dioxide reforming of methane. Applied Catalysis B: Environmental 119-120: 217-226.
- Mun Sing F, Subhash B (2009) Catalytic technology for carbon dioxide reforming of methane to synthesis gas. Chem Cat Chem 1(2): 192-208.
- Aritomo Y, Enrique I (2010) Catalytic activation and reforming of methane on supported palladium clusters. Journal of Catalysis 274(1): 52-63.
- Hendrik DW, Geboers J, Pierre J, Bert S (2011) Design of active and stable NiCeO2ZrO2MgAl2O4 dry reforming catalysts. Applied Catalysis B: Environmental 105(3-4): 263-275.
- Shaobin W, Graeme J (1996) Carbon dioxide reforming of methane to produce synthesis gas over metal-supported catalysts: state of the art. Energy & Fuels 10(4): 896-904.
- Shaobin W, Graeme J (1998) Role of CeO2 in Ni/CeO2-Al2O3 catalysts for carbon dioxide reforming of methane. Applied Catalysis B: Environmental 19(3-4): 267-277.
- Tsipouriari VA, Verykios XE (1999) Carbon and oxygen reaction pathways of CO2 reforming of methane over Ni/La2O3 and Ni/Al2O3 catalysts studied by isotopic tracing techniques. Journal of Catalysis 187(1): 85-94.
- Luo JZ, Ng CF, Au CT (2000) CO2/CH4 reforming over ni-LA2O3/5a an investigation on carbon deposition and reaction steps. Journal of Catalysis 194(2): 198-210
- Junmei W, Enrique I (2004) Isotopic and kinetic assessment of the mechanism of reactions of CH4 with CO2 or H2O to form synthesis gas and carbon on nickel catalysts. Journal of Catalysis 224(2): 370-383.
- Guolin X, Keying S, Ying G, Hengyong X, Yongde W (1999) Studies of reforming natural gas with carbon dioxide to produce synthesis gas: X. The role of CEO2 and MGO promoters. Journal of Molecular Catalysis A: Chemical 147(1-2): 47-54.
- Djaidja SL, Kiennemann A, Barama A (2006) Characterization and activity in dry reforming of methane on NIMG/AL and NI/MGO catalysts. Catalysis Today 113(3-4): 194-200.
- Aparicio LM (1997) Transient isotopic studies and microkinetic modeling of methane reforming over nickel catalysts. Journal of Catalysis 165(2): 262-274.
- Michael MA, Bradford CJ (1996) Catalytic reforming of methane with carbon dioxide over nickel catalysts II. Reaction kinetics, Applied Catalysis A: General 142(1): 97-122.
- Vaso A, Xenophon E (2001) Kinetic study of the catalytic reforming of methane with carbon dioxide to synthesis gas over NI/LA2O3 catalyst. Catalysis Today 64(1-2): 83-90.
- Wang S, Lu GQM (2000) Effects of promoters on catalytic activity and carbon deposition of Ni/γ-Al2O3 catalysts in CO2 reforming of CH4. Journal of Chemical Technology and Biotechnology 75(7): 589-595.
- Sodesawa, Nozaki (1967) Catalytic reaction of methane with carbon dioxide. Reaction Kinetics and Catalysis Letters 12(1): 107-111.
- Ali F, Mansouri SS (2011) Influence of nanocatalyst on oxidative coupling, steam and dry reforming of methane: a short review. Arabian Journal of Chemistry 9(Supp 1): S28-S34.
- Rostrupnielsen, Hansen JH (1993) CO2-reforming of methane over transition metals. Journal of Catalysis 144(1): 38-49.
- Joong K, Kim KH, Kim JK (1994) The reaction of CO2 with CH4 to synthesize H2 and CO over nickel-loaded Y-zeolites. Catalysis Letters 28(1): 41-52.
- Green ML, Claridge JB, Tsang SC (1994) Methane conversion to synthesis gas by partial oxidation and dry reforming over rhenium catalysts. Catalysis Today 21(2-3): 455-460.
- Ferreira A, Rodrõ Âguez R (1998) Comparative study at low and medium reaction temperatures of syngas production by methane reforming with carbon dioxide over silica and alumina supported catalysts. Applied Catalysis A: General 170(1): 177-187.
- Michael CJ, Bradford (1999) The role of metal-support interactions in CO2 reforming of CH4. Catalysis Today 50(1): 87-96.
- Rezaei M, Alavi SM, Sahebdelfar S, Zi Feng Y (2006) Syngas production by methane reforming with carbon dioxide on noble metal catalysts. Journal of Natural Gas Chemistry 15(4): 327-334.
- Zhaoyin H, Heliang F, Xiaoming Z, Tatsuaki Y (2006) Production of synthesis gas via methane reforming with CO2 on noble metals and small amount of noble-(Rh-) promoted Ni catalysts. International Journal of Hydrogen Energy 31(5): 555-561.
- Barama S, Batiot CD, Capron M, Bordes R, Bakhti M (2009) Catalytic properties of Rh, Ni, Pd and Ce supported on Al-pillared montmorillonites in dry reforming of methane. Catalysis Today 141(3-4): 385-392.
- Schuurman Y, Marquez Alvarez C, Kroll VCH, Mirodatos C (1998) Unraveling mechanistic features for the methane reforming by carbon dioxide over different metals and supports by TAP experiments. Catalysis Today 46(2-3): 185-192.
- De Miguel SR, Vilella IMJ, Maina SP, San José Alonso D, Román Martínez MC, et al. (2012) Influence of Pt addition to Ni catalysts on the catalytic performance for long term dry reforming of methane. Applied Catalysis A: General 435-436: 10-18.
- Sang L, Sun B, Tan H, Du C, Wu Y, et al. (2012) Catalytic reforming of methane with CO2 over metal foam based monolithic catalysts. International Journal of Hydrogen Energy 37(17): 13037-13043.
- Tasker SZ, Standley EA, Jamison TF (2014) Recent advances in homogeneous nickel catalysis. Nature 509(7500): 299-309.
- Amin MH, James T, Suresh KB (2018) Insight into the role of the pore structure of supports for nickel nanocatalysts in the CO2 reforming of methane.
- Amin MH, Putla S, Bee Abd Hamid S, Bhargava SK (2015) Understanding the role of lanthanide promoters on the structure-activity of nanosized Ni/γ-Al2O3 catalysts in carbon dioxide reforming of methane. Applied Catalysis A: General 492: 160-168.
- Amin MH, James T, Suresh KB (2013) A comparison study on methane dry reforming with carbon dioxide over Ni catalysts supported on mesoporous SBA-15, MCM-41, KIT-6 and γ-Al2O3. Chemeca 2013: Challenging Tomorrow, Engineers Australia, Chemeca 2013 (41st: 2013: Brisbane, Qld.). Chemeca 2013: Challenging Tomorrow. Barton, ACT: Engineers Australia, pp. 543-548.
- Amin, Hasan Md, James T, Suresh BK (2013) An investigation on the role of lanthanide promoters in promoted gamma-alumina-supported nickel catalysts for dry reforming of methane. Chemeca Challenging Tomorrow.
- Amin MH, Deepa D, Loh Q, Choudahry VR, Selvakannan PR, et al. (2016) Nanocrystalline FeOClx grafted MCM-41 as active mesoporous catalyst for solvent-free multi-condensation reaction, RSC Advances 6(73): 69334-69342.
- Newnham J, Mantri K, Amin MH, Tardio J, Bhargava SK (2012) Highly stable and active Ni-mesoporous alumina catalysts for dry reforming of methane. International Journal of Hydrogen Energy 37(2): 1454-1464.
- Amin MH, Tardio J, Bhargava SK (2013) An investigation on the role of ytterbium in ytterbium promoted γ-alumina-supported nickel catalysts for dry reforming of methane. International Journal of Hydrogen Energy 38(33): 14223-14231.
- Haag S, Burgard M, Ernst B (2007) Beneficial effects of the use of a nickel membrane reactor for the dry reforming of methane: Comparison with thermodynamic predictions. Journal of Catalysis 252(2): 190-204.
- Fidalgo B, Dominguez A, Pis JJ, Menendez JA (2008) Microwave-assisted dry reforming of methane. International Journal of Hydrogen Energy 33(16): 4337-4344.
- Resources TCG (2014) Natural gas conversion vs. syngas routes: A future of convergence. TCGR the Catalysis Group
- Jiang Z, Xiao T, Kuznetsov VL, Edwards PP (2010) Review-Turning carbon dioxide into fuel. Phil Trans R Soc A 368: 3343-3364.
- Lavoie JM (2014) Review on dry reforming of methane, a potentially more environmentally-friendly approach to the increasing natural gas exploitation. Front Chem 2: 81.
- Peng XD, Wang AW, Toseland BA, Tijm PJA (1999) Single-step syngasto- dimethyl ether processes for optimal productivity, minimal emissions and natural gas-derived syngas. Ind Eng Chem Res 38(11): 4381-4388
- Gunardson HH, Abrardo JM (1999) Produce CO-rich synthesis gas: Petrochemical product growth will lead to more demand for carbon monoxide. Hydrocarbon Process 78(4): 87-94.
- Suib SL (2013) New and future developments in catalysis. Elsevier, USA.
- Schoedel N, Schmigalle H, Behens A, Goeke V, Thaller C, et al. (2015) Method for producing Di-methyl ether from methane. Patent Application Publication, USA.
- Kim AP, Nielsen CS, Dybkjær IB, Perregaard J (2010) Large Scale Methanol Production from Natural Gas. Haldoor Topsoe, Denmark
- Preeti G, Kanchi CK, Lou HH (2012) Evaluation of the economic and environmental impact of combining dry reforming with steam reforming of methane. Chemical engineering research and design 90(11): 1956- 1968.
- Wan Daud WMA, Usman Md (2015) Recent advances in the methanol synthesis via methane reforming processes. RSC Advances 5(28): 21945-21972.
- Olah GA, Goeppert A, Surya Prakash GK (2005) Beyond oil and gas: the methanol economy. Angew Chem Int Ed 44(18): 2636-2639.
- Ghoneim SA, El Salamony RA, El Temtamy SA (2016) Review on innovative catalytic reforming of natural gas to syngas. World Journal of Engineering and Technology 4: 116-139.
© 2018 Mohamad Hassan Amin. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






