- Submissions

Full Text
Orthopedic Research Online Journal
A Case of Acute Compartment Syndrome of Upper Limb in a Dengue Haemorrhagic Fever Patient Secondary to Intravenous Cannula Insertion
Kishan Rao Subramaniam1*, Rajandra Kumar Karupiah1, Jeffry Raj2 and Zamzuri Zakaria1
1 Department of Orthopedic, International Islamic University, Malaysia
2 Department of Orthopedic Hospital Tengku Ampuan Afzan, Malaysia
*Corresponding author:Kishan Rao Subramaniam, Department of Orthopedic, International Islamic University, Malaysia
Submission: April 01, 2019;Published: April 03, 2019

ISSN: 2576-8875 Volume5 Issue3
Abstract
Dengue fever with its haemorrhagic complications is a common viral illness in Malaysia which can sometimes lead to high morbidity and mortality. We present a case report of a 27-year-old male who was admitted to our hospital with dengue haemorrhagic fever. He developed multiple hematomas and bruising in his right forearm and arm due to intravenous cannula insertion. Due to coexisting thrombocytopenia, the limb progressed to acute compartment syndrome and emergency fasciotomy was done and his limb was salvaged. This case illustrates an important potential complication of dengue fever and the importance of timing of surgery in the event of compartment syndrome.
Introduction
Acute compartment syndrome is defined as increased osteo-fascial compartment pressure which leads to reduced perfusion and may lead to irreversible muscle and nerve damage [1]. It is considered an orthopaedic emergency. Common causes of acute compartment syndromes are trauma, crush injuries, burns and bleeding disorders [2]. The treatment is emergent fasciotomy. Here we are reporting a case of an acute compartment syndrome of right upper limb due to intravenous cannula insertion in a patient with dengue haemorrhagic fever.
Case Report
27-year-old male Malay gentleman was admitted to medical ward due to dengue fever. He presented with 3-day history of fever, headache, myalgia, arthralgia and reduced oral intake. He was hemodynamically stable. His initial blood investigations revealed a classic dengue fever picture with white blood cells 2.1x109/L, platelets 32x109 /L, haemoglobin 15.1g/dL, haematocrit 41%, albumin 35g/L and normal coagulation profile. Both dengue non-structural protein-1 (NS1 antigen) and dengue IgM were positive. Renal profile, liver function test and other investigations were normal.
However, on day 8th of admission he was referred to orthopaedic team due to increasing pain and swelling of right upper limb where an intravenous (IV) cannula was inserted. Upon review, noted gross swelling of right forearm and arm with multiple large hematoma and extensive bruising at previous IV cannula and blood withdrawal sites (Figure 1). Compartment was tense but passive stretch test was inconclusive. Blood investigation showed white blood cells 1.7x109/L, platelets 15x109/L, haemoglobin 14.9g/dL, haematocrit 47% with prolonged activated partial thromboplastin time (APTT) at 70 seconds. Subsequently, the pain and swelling worsened after brief period of close observation and patient had numbness at median nerve distribution site as well.
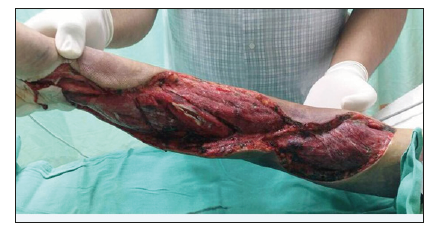
Emergency fasciotomy was planned, repeated platelet counts dropped to 10x109/L. In view of severe thrombocytopenia and prolonged APTT, platelet concentrates, fresh frozen plasma were transfused while preparing patient for surgery. Emergency fasciotomy of forearm and arm with carpal tunnel release was performed under general anaesthesia. Incision was made over the carpal tunnel extending into volar aspect of forearm and lateral aspect of arm releasing the fascia and intermuscular septaes. Intraoperative findings noted large amount of blood clots and intramuscular bleeding at flexor muscle compartment. Blood clots were evacuated and hemostasis were secured. Postoperatively, the muscles were healthy with good distal circulation (Figure 2 & 3).
Figure 1:
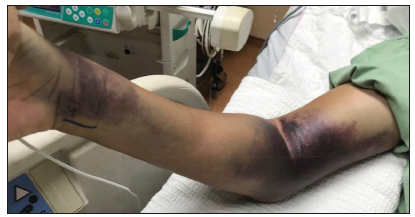
Figure 2:Intraop picture. Noted large amount of blood clots at flexor compartment..

Figure 3:At day 3 of wound inspection. Muscles appear healthy..
Secondary suturing of the wound was done 1 week later. Patient was then sent for regular physiotherapy for grip strengthening and hand function. At 8 weeks post op,patient had good grip strength and good hand function. Patient was able to return to work at 3 months post operation.
Discussion
The first known epidemic of DHF occurred in Manila, Philippines, in 1953 to 1954, but within 20 years the disease in epidemic form had spread throughout Southeast Asia [3]. Currently, dengue fever causes more illness and death than any other arbovirus disease of humans [3]. Each year, an estimated 100 million cases of dengue fever and several hundred thousand cases of DHF occur. DHF is a leading cause of hospitalization and death among children in many Southeast Asian countries [4].
There are four dengue virus serotypes, called DEN-1, DEN-2, DEN-3, and DEN-4 [5]. They belong to the genus Flavivirus, family Flaviviridae. Infection with one dengue serotype provides lifelong immunity to that virus, but there is no cross protective immunity to the other serotypes [3]. Thus, persons living in an area of endemic dengue can be infected with three, and probably four, dengue serotypes during their lifetime.
Humans are infected with dengue viruses by the bite of an infective mosquito. Aedes aegypti [6], the principal vector, is a small, black-and-white, highly domesticated tropical mosquito that prefers to lay its eggs in artificial containers commonly found in and around homes, for example, flower vases, old automobile tires, buckets that collect rainwater, and trash in general.
After a person is bitten by an infective mosquito, the virus undergoes an incubation period of 3 to 14 days (average, 4 to 7 days) [7], after which the person may experience acute onset of fever accompanied by a variety of nonspecific signs and symptoms such as frontal headache, retro-orbital pain, body aches, nausea and vomiting, joint pains, weakness, and rash.
Hemorrhagic manifestations in dengue fever patients are not uncommon and range from mild to severe. Skin hemorrhages, including petechiae and purpura, are the most common, along with gum bleeding, epistaxis, menorrhagia, and gastrointestinal (GI) haemorrhage [5].
The critical stage in DHF is at the time of defervescence, but signs of circulatory failure or hemorrhagic manifestations may occur from about 24 hours before to 24 hours after the temperature falls to normal or below [5]. Blood tests usually show that the patient has thrombocytopenia (platelet count, >100,000/mm3) and hemoconcentration relative to baseline as evidence of a vascular leak syndrome [3]. The tourniquet test, which indicates that the patient has increased capillary fragility, may be diagnostically helpful to the physician [4].
Acute compartment syndrome is defined as increased in osteofascial compartment pressure that may cause irreversible tissue and nerve damage if left untreated [6]. Diagnosis of acute compartment syndrome is primarily clinical, using compartment pressure measurement as an adjunct in evaluation. Because the features of early acute compartment syndrome are non- specific, a high clinical suspicion must be maintained for all at-risk populations [2]. The classic features such as pain, pallor, paresthesias, paralysis, and pulselessness are all late findings of acute compartment syndrome and are associated with irreversible damage [8]. However, pain out of proportion to injury not relieved by regular analgesia and positive passive stretch test of involved muscles are early symptoms that require further attention and monitoring.
The pathophysiology in compartment syndrome is the rise in intra-compartmental pressure increases venous pressure, which in turn reduces the arteriovenous pressure gradient, reducing local tissue perfusion [9]. The reduction in tissue perfusion, coupled with a reduction in venous drainage, causes significant tissue oedema. This change in vascular pressure also causes a reduction in lymphatic drainage, further increasing pressure in the compartment [9]. Finally, the edematous tissue compresses the arterioles leading to end-organ ischemia.
Emergent fasciotomy is paramount to the management of acute compartment syndrome [10]. A low systemic BP should be corrected to hopefully increase the compartment perfusion, and any applied external compressive forces (casts, splints, dressings, eschars) should be removed [10]. Delay in treatment may lead to tissue hypoperfusion causing muscle necrosis, Volkman’s ischemic contractures and loss of function of the affected extremity [11]. Necrotic muscle is a significant risk factor for bacterial superinfection and may quickly be seeded by bacteria, and lead to sepsis. Necrotic muscle may therefore require repeated debridement and even possible extremity amputation.
In our case, we believe that the hematoma formation due to venepuncture could possibly be the cause of increase in intra compartmental pressure leading to acute compartment syndrome. This was further aggravated by presence of thrombocytopenia and was confirmed intraoperatively with presence of large amount of blood clots at the site of previous intravenous cannula insertion. The real dilemma in this case was the timing of performing the surgery. Performing an emergent fasciotomy with low platelet counts may lead to uncontrollable bleeding which may cause severe morbidity and mortality. However, delaying the operation in order to increase the platelet counts may risk losing the limb. In our case, we performed emergent fasciotomy within few hours after being diagnosed and platelet concentrates were transfused pre and intra operatively.
Conclusion
We highlight this case as we believe once clinically compartment syndrome is diagnosed, even though platelet count is low, fasciotomy should still be emergently performed as waiting for platelet counts to rise and normalise would lead to permanent functional loss of patient’s limb and Volkman’s ischemic contracture would set in. We conclude that emergency fasciotomy should be performed with pre-emptive measures of platelet coverage just prior to op and intra operatively, with good anaesthetic support to save the limb. Last
but not least, we must be aware of this different form of dengue complication, where urgent and aggressive intervention may enable us to salvage limbs and prevent mortality.References
- Academy A, Board OS (2018) Management of acute compartment syndrome.
- Frca CF, Frcs MT (2011) Acute limb compartment syndromes. 11(1): 24- 28.
- Gubler DJ (1998) Dengue and Dengue Hemorrhagic Fever. 11(3): 480- 496.
- Officer M, Gastroenterologist C, Hospital A, Consultant ID, Hospital SB, et al. (2016) Dengue fever with compartment syndrome. Pp. 2016-2018.
- Wills BA, Oragui EE, Stephens AC, Daramola OA, Dung NM, et al. (2002) Coagulation abnormalities in dengue hemorrhagic fever: Serial investigations in 167 vietnamese children with dengue shock syndrome. Clin Infect Dis 35(3): 277-285.
- Report C (2013) Acute compressive ulnar neuropathy in a patient of dengue fever: An unusual presentation. 3(2): 25-28.
- Pizarro-torres D (2016) Original dengue with severe plasma leakage: A new monitoring approach. Pp. 115-121.
- Kamath SR, Ranjit S (2006) Clinical features, complications and atypical manifestations of children with severe forms of dengue hemorrhagic fever in south India. 73: 889-895.
- Donaldson J, Haddad B, Khan WS (2014) The pathophysiology, diagnosis and current management of acute compartment syndrome. Open Orthop J 8: 185-193.
- Raza H, Mahapatra A (2015) Acute compartment syndrome in orthopedics: Causes, diagnosis and management. Adv Orthop 2015: 543412.
- Medicine E (2017) Feature 106.
© 2019 Kishan Rao Subramaniam. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)































 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






