- Submissions

Full Text
Open Journal of Cardiology & Heart Diseases
Adult ADHD Therapy Related Dilated Cardiomyopathy and Thromboembolic Phenomenon
Laura Salado Vega1, Christine Sommerville2, Elissa Williams2 and Seshasayee Narasimhan1,2,3,4*
1Department of Internal Medicine, Manning Base Hospital, Taree, Australia
2Department of the Heart Centre, Taree, Australia
3Department of Cardiology, Manning Base Hospital, Taree, Australia
4Department of Conjoint Senior Lecturer, University of Newcastle, Callaghan, Australia
*Corresponding author: Seshasayee Narasimhan, Department of Internal Medicine, Cardiovascular Division, Manning Base Hospital, Taree, NSW 2430, Australia
Submission: January 29, 2024;Published: February 14, 2024

ISSN 2578-0204Volume4 Issue3
Abstract
Stem cells, with their remarkable potential for self-renewal and differentiation into diverse specialized cell types, constitute a central focus in scientific research and medical exploration. This review delves into the intricate realm of stem cell biology, spotlighting the processes of cell differentiation and development. Stem cells, spanning embryonic, adult, and induced pluripotent varieties, offer unparalleled insights into early embryonic development, and bear the promise of revolutionizing regenerative medicine and disease modeling. The journey of stem cell differentiation, governed by intricate mechanisms, entails signal transduction pathways, gene expression regulation, epigenetic modifications, niche interactions, and asymmetric cell division, culminating in cell fate commitment and lineage-specific maturation. Stem cells play a pivotal role in embryonic development, giving rise to the gamut of tissue-specific cell types, and hold substantial potential in regenerative medicine, disease modeling, and drug discovery. Yet, persistent challenges and ethical dilemmas envelop the field, spanning concerns over embryonic stem cell use, reprogramming techniques, tumor formation, immune rejection, and regulatory complexities. As the field advances, the collaborative endeavors of multidisciplinary teams drive emerging trends, technologies, and potential breakthroughs, offering a glimpse into a future where stem cells may redefine healthcare. The resolution of these challenges and ethical considerations is instrumental in advancing the ethical and responsible application of stem cells in both research and clinical contexts.
Keywords:Stem cells; Biology; IPSCs; Regenerative medicine; Cell differentiation; Tissue engineering
Learning Objective
Institution of appropriate cardiovascular evaluation and close monitoring of adult ADHD medications and anxiety/depression medications with particular attention to interactions and increasing serum levels of dexamphetamine. It is our recommendation that adult attention deficit disorder (ADHD) prior to initiation of therapy, have a detailed cardiovascular assessment including electrocardiogram (ECG), echocardiogram (ECHO) and treadmill stress echo to assess the functional capacity and exclude reversible ischemia. Furthermore, close monitoring of cardiac performance and cardiovascular risk factors should continue during the whole length of treatment, regardless of prior cardiovascular history.
Introduction
This case report calls attention to the importance of appropriate cardiovascular evaluation prior to initiation of adult ADHD treatment to prevent negative cardiac outcomes. Dexamphetamine, the dextro-isomer of amphetamine is a well-known central nervous system (CNS) stimulant [1]. Amphetamines have the potential to cause changes in blood pressure, heart rate as well as structural myocardial changes that can lead to cardiomyopathy and heart failure [2-4]. These have been widely reported in literature in the context of illegal use of amphetamines such as methamphetamine [4] but rarely described in the context of medically prescribed amphetamine. We present a case of a 42-year-old male without previous cardiac history arriving to our emergency department in florid heart failure secondary to severe dilated cardiomyopathy with a 10-year history of dexamphetamine use for ADHD and 1 year history of duloxetine use for the management of depression.
Case Presentation
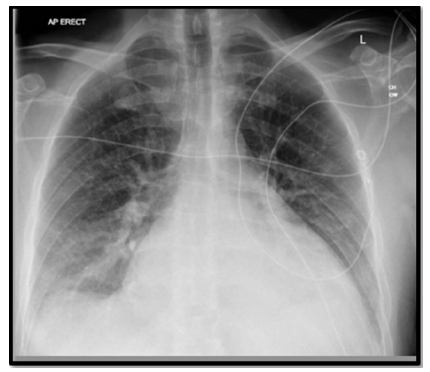
We present a case of a 42-year-old male presenting with acute heart failure and cardiogenic shock as evidenced by hypotension. The patient’s social history includes prior intravenous drug use (IVDU), he is a non-smoker, does not drink alcohol or currently use illicit drugs. He has a background medical history of anxiety/ depression treated with Duloxetine 60mg daily and Diazepam 5mg twice a day for the past year, his adult ADHD was treated with Dexamphetamine Sulphate 10mg three times a day for the last 10 years. The patient presented to our emergency department (ED) with a five-day history of worsening dyspnoea, intermittent pleuritic chest pain and a new onset of epigastric pain. On arrival he was tachypneic, tachycardic and afebrile. His admission chest x-ray (CXR) showed cardiomegaly with features of left heart failure (Figure 1). His ECG showed sinus tachycardia (Figure 2).
Figure 1:Initial ECG showing sinus rhythm with left ventricular hypertrophy.
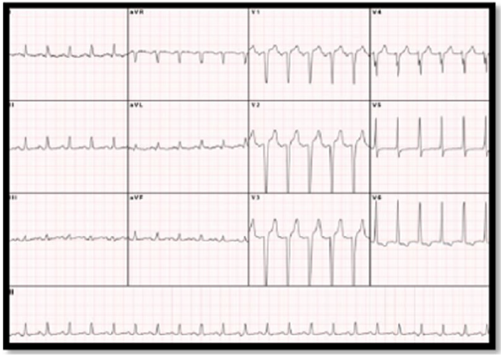
Figure 2:initial chest x-ray showing prominence of upper lobe and perihilar vascular markings noted suggestive of left heart failure, and cardiomegaly.
Given his critical state, he was admitted to the intensive care unit (ICU) for invasive haemodynamic monitoring and IV Metaraminol. CT pulmonary angiogram (CTPA) was completed which showed significant bilateral (B/L) pleural effusions, cardiomegaly, pericardial effusion with no clear pulmonary emboli visualized. His pro-BNP was 4010ng/L. Inpatient transthoracic echocardiogram completed showed dilated left ventricle (LV) with severe systolic dysfunction with a left ventricular ejection fraction (LVEF)=10%, multiple mural thrombi noted (Figures 3-5), right ventricular (RV) systolic dysfunction, mild mitral regurgitation (MR), small pericardial effusion without tamponade physiology & bilateral pleural effusions.
Figure 3:Echocardiogram.
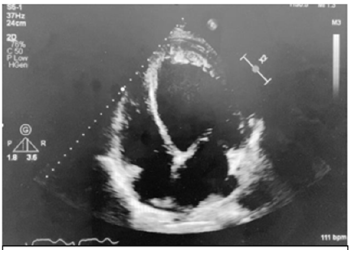
Figure 4:Echocardiogram
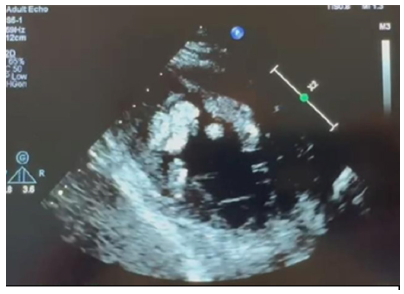
Figure 5:Echocardiogram
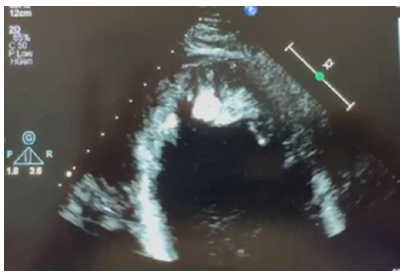
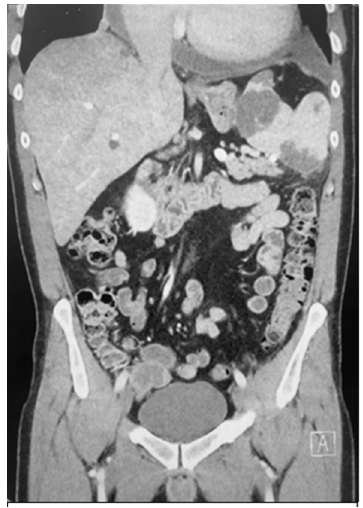
Apixaban 5mg twice a day was commenced for his mural thrombi, Intravenous (IV) Levosomendine for his acute heart failure with reduced ejection fraction (HFrEF) with a plan of introducing bisoprolol fumarate and SGLT2-inhibitor once IV Levosomendine was completed. Due to delicate hemodynamics, Valsartan did not commence. Given the LVEF, his ADHD & anxiety/ depression medications were temporarily ceased. The patient had a long admission and made a slow but steady recovery. On day 11, he reported abdominal pain. Abdominal ultrasound (US) was performed which showed hepatosplenomegaly with possible multifocal splenic infarcts (Figure 6). CT abdomen and pelvis were completed confirming multiple splenic infarcts and bilateral renal infarcts (Figure 7).
Figure 6:Ultrasound of abdomen showing splenic infarcts.
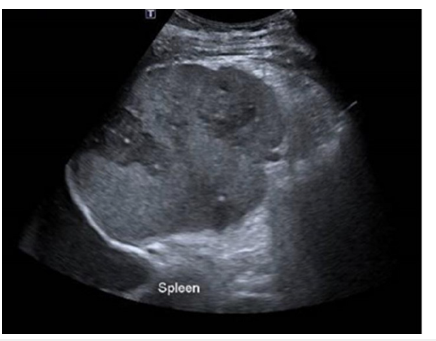
Figure 7:Ct abdomen showing splenic infarcts, and hepatosplenomegaly.
Other investigations completed during hospitalisation were negative viral multiplex including Covid, Hepatitis & HIV. Autoimmune screen (ANA, ANCA-i, DNA Ab and ENA) was negative. His biogenic amines and thyroid function test were normal. His HFrEF therapy included Bisoprolol 5mg daily, Empagliflozin 10mg daily, Furosemide 40mg daily, Ivabradine 7.5mg twice a day and Digoxin 125mcg daily. He was discharged on day 16 with outpatient follow-up with the heart failure nurse and a repeat transthoracic echocardiogram in 3 months to assess the LV mural thrombus. Unfortunately, 1 week post discharge, the patient returned to hospital with transient expressive aphasia. He had an inpatient CT Brain + angiography and MRI Brain which excluded stroke, demyelination, or haemorrhage. The patient was compliant with his Apixaban. He was given a diagnosis of transient ischemic attack (TIA) and was discharged.
The patient was reviewed in the heart failure clinic on a regular basis. His Bisoprolol was maximized to 10mg daily. Valsartan 40mg daily was introduced tolerating well both. His general practitioner was advised to replace Valsartan 40mg daily with Sacubitril/ Valsartan 24/26mg twice a day in a month provided his renal functions and hemodynamics remained stable. The focus from the patient’s perspective was to resume his anxiety/depression and ADHD medications. The patient was advised that this should be supervised by a psychiatrist and a referral was made. The three months repeat transthoracic echocardiogram showed an improvement in the LVEF from 10% to 20%, disappearance of LV mural thrombi and a reduction in the MR. The patient is closely followed up by both cardiology & psychiatry. His community pharmacist has been advised to monitor adult ADHD medications and anxiety/depression medications with particular attention to interactions and increasing serum levels of dexamphetamine.
Discussion
Our case report highlights the importance of cardiovascular evaluation and medication interactions review prior to dexamphetamine initiation for the management of adult ADHD. The prevalence of this diagnosis and the need for medical management is certainly on the rise [5,6] increased awareness is key to prevent cardiovascular complications on a population with possibly a higher risk of developing of cardiovascular risk factors [7]. Amphetamine induced cardiomyopathy and secondary heart failure in adults is becoming more prominent in literature [3], while the majority of reported cases are associated with long term use of methamphetamine and its illegal derivatives, [2,8,9] though rare, some cases have been reported in association with medically prescribed amphetamine in the form of dexamphetamine for the treatment of ADHD and Narcolepsy [6,10]. Dexamphetamine is considered the first-line pharmacological treatment in Australia for people with adult ADHD in which symptoms are causing significant impairment [11]. Dexamphetamine is the dextrorotatory enantiomer of amphetamine and widely used in the treatment of ADHD, it is considered a central nervous system (CNS) stimulant and known to lead to increases in blood pressure (BP) and heart rate (HR) as well as vasospasm, vasoconstriction among others, this thought result from catecholamine signaling and sympathetic stimulation [3]. In addition, reported direct cardiac toxicity linked to amphetamine use could provoke structural changes that might contribute to the development of dilated cardiomyopathy and heart failure [2-4].
The elimination of amphetamines involves both renal excretion and liver metabolism via cytochrome P450 2D6 (CYP2D6), illegal methamphetamine is metabolized to amphetamine, and around 37%-54% is excreted unchanged in the form of methamphetamine, this allows its detection in urine [12,13]. Studies have shown that elevated urinary concentration of methamphetamine are likely to indicate illicit use [12], dexamphetamine on the other hand will be detected in urine as amphetamine, thus urinary screen test that differentiate between methamphetamine and amphetamine concentration could contribute to the assessment of illicit use versus legal use. Duloxetine has the potential to act as cytochrome P450 2D6 (CYP2D6) inhibitor causing a major drug interaction with amphetamine/dexamphetamine [14,15]. We point out the possibility that this interaction has played a role in our case by decreasing the hepatic metabolism of amphetamine and potentiating its cardiovascular effects in our patient. Sinha et al. [6] explained the increasing concern regarding adults with ADHD and cardiac events such myocardial infarction and stroke, pointing out that ADHD medication has been linked to increased heart rate and blood pressure measurements [16]. In their systematic review and meta-analysis in 2017 concluded that amphetamine use for the treatment of ADHD had a small but statistically significant increase of blood pressure and heart rate suggesting that cardiovascular monitoring should be considered Furthermore, A Swedish nationwide population-based cohort study published in World Psychiatry in 2022 depicted that ADHD patients without pre-existing cardiovascular diseases, had a significant associated risk of any cardiovascular disease (HR=2.05, 95% CI: 1.98-2.13), recommending careful monitoring of ADHD patients adjusted to their age and individual characteristics to diminish their cardiovascular risk.
The current Australian Evidence-Based Clinical Practice Guideline for Attention Deficit Hyperactivity Disorder (ADHD) recommends performing a cardiovascular assessment and keep a record of baseline HR and BP pre-treatment, ECG is not recommended unless cardiology opinions are needed, which applies to patients with pre-existing cardiac history or symptoms pointing towards cardiac origin [11]. It is our recommendation that adult ADHD prior to initiation of therapy, have a detailed cardiovascular assessment including ECG, echo and treadmill stress echo to assess the functional capacity and exclude reversible ischemia. Furthermore, close monitoring of cardiac performance and cardiovascular risk factors should continue during the entire treatment period, regardless of prior cardiovascular history.
Conclusion
Adult ADHD is becoming more prevalent with dexamphetamine being one of the preferred prescribed therapeutic agents. We highlight the importance of adequate pre-treatment cardiovascular assessment and monitoring to prevent detrimental cardiovascular outcomes, associated with medically prescribed dexamphetamine. It is interesting to note that the concomitant use of duloxetine and dexamphetamine as done in our patient could have potentiated the clinical picture of left ventricular systolic dysfunction, left ventricular mural thrombus and the thromboembolic phenomenon which has been noted in the abuse of methamphetamine. Urinary screen tests differentiate between methamphetamine and dexamphetamine, and this could shed light to the type of amphetamine used, and in the case of medically prescribed dexamphetamine, provide information about concomitant use of illegal methamphetamine and its risks. Further studies are required to understand the cardiovascular effects of dexamphetamine use and optimise an assessment pathway for these patients. Education regarding dexamphetamine use including its cardiovascular effects and drug interactions is essential to prevent further cardiovascular complications in all patients.
Conflict of Interest
The authors declare that there is no conflict of interest.
References
- National Center for Biotechnology Information (2024) PubChem Compound Summary for CID 5825, Dextroamphetamine Sulfate.
- Chapman L, Khalifa I, Sheriff N, Colwell N (2022) Amphetamine-induced cardiomyopathy complicated by embolic stroke: A case report. European Heart Journal Case Reports 6(2): ytac044.
- Lewars JT, Wiarda KP (2023) Amphetamine-dextroamphetamine-induced cardiomyopathy, an emerging cause of heart failure in young patient populations: A case study involving the study drug. Cureus 15(6): e40942.
- Reddy PKV, Ng TMH, Oh EE, Moady G, Elkayam U (2020) Clinical characteristics and management of methamphetamine-associated cardiomyopathy: State-of-the-art review. Journal of the American Heart Association 9(11): e016704.
- Abdelnour E, Jansen MO, Gold JA (2022) ADHD diagnostic trends: Increased recognition or overdiagnosis? Missouri Medicine 119(5): 467-473.
- Sinha A, Lewis O, Kumar R, Yeruva SL, Curry BH (2016) Adult ADHD medications and their cardiovascular implications. Case Reports in Cardiology 2016: 2343691.
- Li L, Chang Z, Sun J, Garcia Argibay M, Du Rietz E, et al. (2022) Attention-deficit/hyperactivity disorder as a risk factor for cardiovascular diseases: A nationwide population-based cohort study. World Psychiatry 21(3): 452-459.
- Yeo KK, Wijetunga M, Ito H, Efird JT, Tay K, et al. (2007) The association of methamphetamine use and cardiomyopathy in young patients. The American Journal of Medicine 120(2): 165-171.
- Solís Olivares CA, Ramírez García HA (2017) Amphetamine-related dilatated cardiomyopathy: A growing phenomenon. Rev Mex Cardiol 28(1): 35-39.
- Bitetzakis CJ, Mazalewski W, Patel J, Chen A (2018) Prescription amphetamine use associated with acute cardiomyopathy. Journal of Cardiac Failure 24(8): 1071-9164.
- Tamara M, Edwina B, Karina C, Noel C, Evelyn C, et al. (2023) The Australian evidence-based clinical practice guideline for attention deficit hyperactivity disorder. Aust N Z J Psychiatry 57(8): 1101-1116.
- Robert W, Amadeo P, Cameron W, Charles M, Javier V, et al. (2013) Differentiating medicinal from illicit use in positive methamphetamine results in a pain population. Journal of Analytical Toxicology 37(2): 83-89.
- Wagner DJ, Sager JE, Duan H, Isoherranen N, Wang J (2017) Interaction and transport of methamphetamine and its primary metabolites by organic cation and multidrug and toxin extrusion transporters. Drug Metabolism and Disposition 45(7): 770-778.
- Vo K, Neafsey PJ, Lin CA (2015) Concurrent use of amphetamine stimulants and antidepressants by undergraduate students. Patient Preference and Adherence 22(9): 161-172.
- Skinner MH, Kuan HY, Pan A, Sathirakul K, Knadler MP, et al. (2003) Duloxetine is both an inhibitor and a substrate of cytochrome P4502D6 in healthy volunteers. Clinical Pharmacology and Therapeutics 73(3): 170-177.
- Hennissen L, Bakker MJ, Banaschewski T, Carucci S, Coghill D, et al. (2017) Cardiovascular effects of stimulant and non-stimulant medication for children and adolescents with adhd: A systematic review and meta-analysis of trials of methylphenidate, amphetamines and atomoxetine. CNS Drugs 31(3): 199-215.
© 2024 Seshasayee Narasimhan. This is an open access article distributed under the terms of the Creative Commons Attribution License , which permits unrestricted use, distribution, and build upon your work non-commercially.
 a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in
a Creative Commons Attribution 4.0 International License. Based on a work at www.crimsonpublishers.com.
Best viewed in 







.jpg)






























 Editorial Board Registrations
Editorial Board Registrations Submit your Article
Submit your Article Refer a Friend
Refer a Friend Advertise With Us
Advertise With Us
.jpg)






.jpg)














.bmp)
.jpg)
.png)
.jpg)










.jpg)






.png)

.png)



.png)






